Abstract
Necropsy examination was performed on an 8.5-year-old Finnish ewe euthanatized because of progressive respiratory distress, cachexia, and weakness. Significant postmortem findings included a diffusely enlarged, dark-red friable liver, mild splenomegaly, and mild mesenteric lymphadenopathy. Examination of multiple tissue sections revealed intravascular atypical mononuclear cells in all tissues examined, with a leukemic pattern of infiltration of mesenteric lymph nodes, liver, lung, and spleen. Neoplastic cells were positive for CD79a and negative for CD-3, BLA.36, and CD68 leukocytic markers. The final diagnosis was B-cell leukemia.
Lymphoid neoplasia is not uncommon in sheep, occurring second only to hepatic neoplasia in one survey. 2 Multicentric solid tissue lymphoma is the most common form of lymphoid neoplasia reported in sheep. 2,3,5,6 Persistent lymphocytosis can occur in sheep with lymphoma, and lymphocytosis has been documented to precede detectable solid tissue lymphoma in this species. 6 Persistent lymphocytosis is also a feature of enzootic lymphoma of cattle associated with bovine leukemia virus, 4 and can occur in sporadic bovine lymphoma. 1 True lymphocytic leukemia in the absence of solid tissue lymphoma is rarely reported in sheep. 6,7 Immunophenotypic studies of enzootic bovine lymphoma indicate that most are of B-cell origin. 8 Neoplastic cells in sporadic bovine leukosis can be of either B-cell 1,5,8 or T-cell lineage. 1,5 Immunophenotypic studies of ovine lymphoma report both T-cell and B-cell origin, although most are of B-cell lineage. 5 This is the first report of immunophenotypic studies resulting in a diagnosis of B-cell leukemia in a sheep.
An 8.5-year-old Finnish ewe was presented with a 1.5-month history of progressive respiratory distress, wasting, and weakness. The ewe was severely cachectic and required assistance to stand. Mucous membranes were pale. The ewe was euthanatized with intravenous barbiturate, and necropsy was performed immediately. There was marked muscle atrophy with adequate body fat stores. The lungs were grossly normal, with the exception of a locally extensive area of fibrous adhesions of the right cranial lung lobes to the parietal pleura. The liver was markedly and diffusely enlarged, dark red with a vague cobblestone appearance, and was extremely friable. The spleen was mildly enlarged and blood-filled. There was mild enlargement of scattered mesenteric lymph nodes, but most were grossly normal, as were all other internal and peripheral lymph nodes. Several thread-like nematode parasites were found in the colon and cecum. No other gross abnormalities were found. The gross diagnoses were mild endoparasitism, mild fibrous pleural adhesions because of previous pneumonia, and hepatopathy, splenomegaly, and mesenteric lymphadenopathy of unknown cause.
Samples of lung, spleen, kidney, liver, heart, colon, urinary bladder, mesenteric lymph nodes, small intestine, and abomasum were fixed in 10% buffered formalin, paraffin-embedded, and 6-μm sections were stained with hematoxylin and eosin and examined histologically. Blood vessels in every organ examined contained numerous closely packed relatively homogenous atypical round cells with round nuclei, clumped chromatin, and a thin rim of indistinct amphophilic cytoplasm (Fig. 1). There were approximately 1–2 mitoses per high power field. Neoplastic cells were particularly prominent within pulmonary vasculature, causing diffuse thickening and hypercellularity of alveolar septae (Fig. 2). There was diffuse infiltration of the sinusoids of the liver (Fig. 3) and of the red pulp of the spleen. Mesenteric lymph nodes retained normal architecture, but medullary cords were thickened by sheets of neoplastic cells. The histopathologic diagnosis was leukemia of probable lymphoid origin.
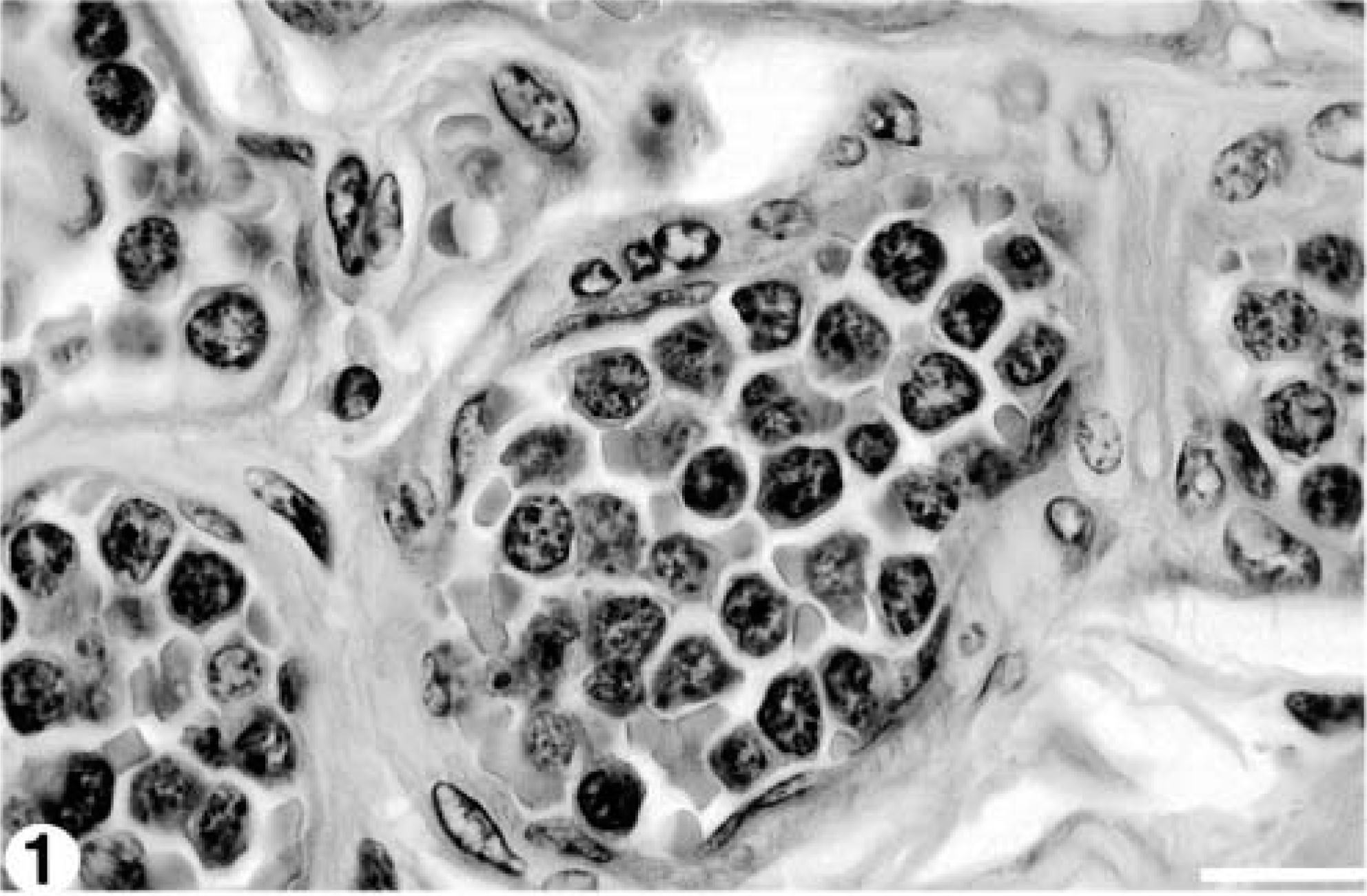
Vasculature; 8.5-year-old Finnish ewe. Numerous intravascular atypical mononuclear cells with clumped chromatin and a thin rim of amphophilic cytoplasm. HE stain. Bar = 20 μm.
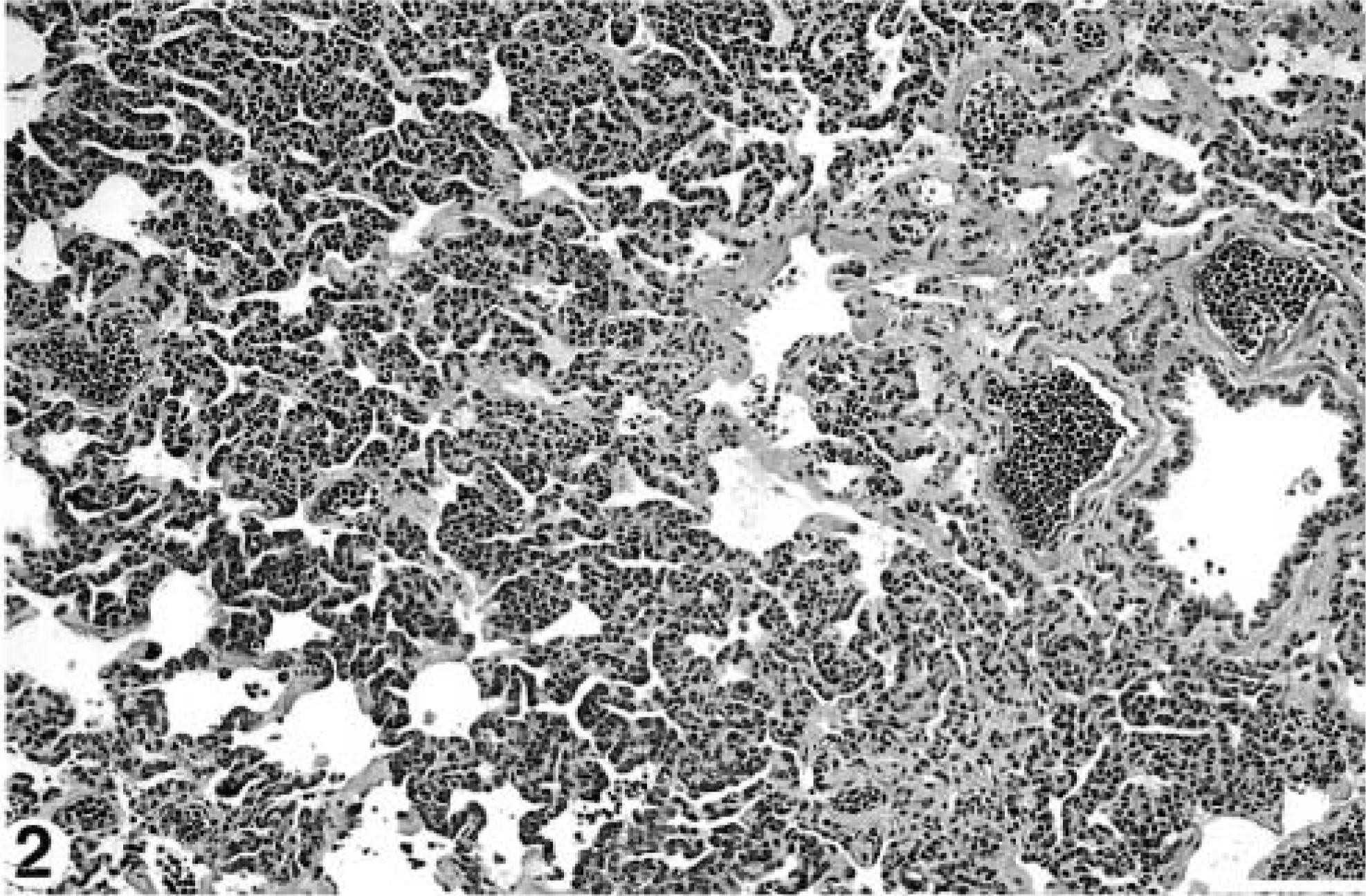
Lung; 8.5-year-old Finnish ewe. Alveolar walls are diffusely thickened and hypercellular because of circulating neoplastic cells. Similar cells are present in veins and arteries. HE stain. Bar = 100 μm.
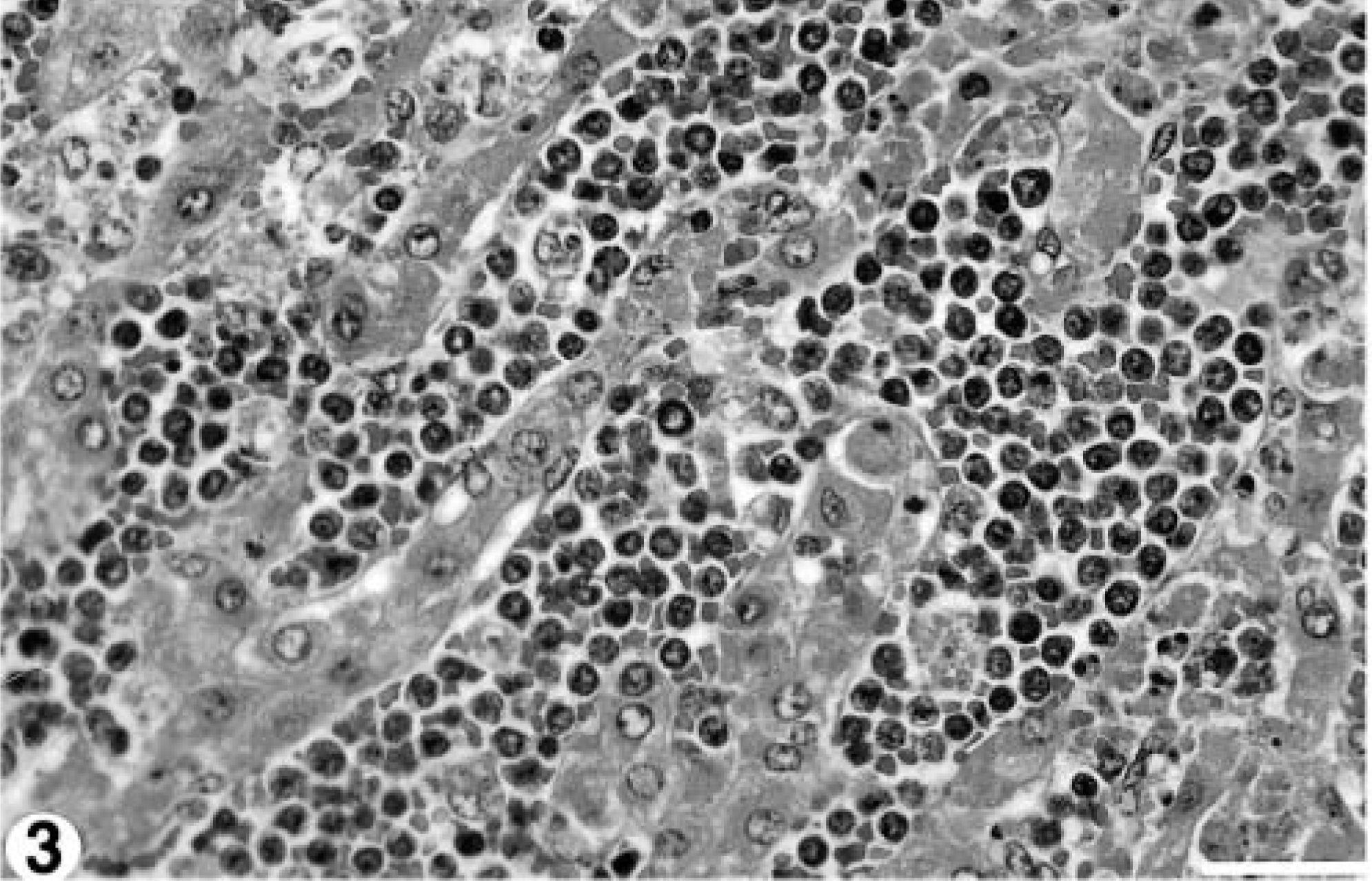
Liver; 8.5-year-old Finnish ewe. Sinusoids are filled with circulating neoplastic cells. HE stain. Bar = 40 μm.
Formalin-fixed, paraffin-embedded sections of liver, heart, colon, urinary bladder, mesenteric lymph node, small intestine, and abomasum were processed for routine immunohistochemistry using antibodies to cell markers known to cross-react with animal tissue (Table 1). A formalin-fixed, paraffin-embedded pulmonary lymph node from a sheep with ovine progressive pneumonia served as a positive control, and nonimmune serum was used as a negative control. The antibodies employed accurately identified B-, T-, and follicular dendritic cells in the control sheep lymph node. Intravascular cells in the leukemic sheep were CD79a-positive (Fig. 4) and negative for CD3, BLA.36, and CD68. Infiltration of CD79a-positive cells within the mesenteric lymph nodes was primarily medullary with few neoplastic cells in the paracortex. The final diagnosis was B-cell leukemia.
Leukemia in a sheep, immunohistochemical procedures and results.∗

∗ mAb = monoclonal antibody; pAb = polyclonal antibody; MW = microwave 20 minutes with 0.01 M citrate buffer, pH 6.0; CSA = catalyzed signal amplification.
† Dako, Carpinteria, CA.
‡ Antibody raised against a human B-cell lymphoma.
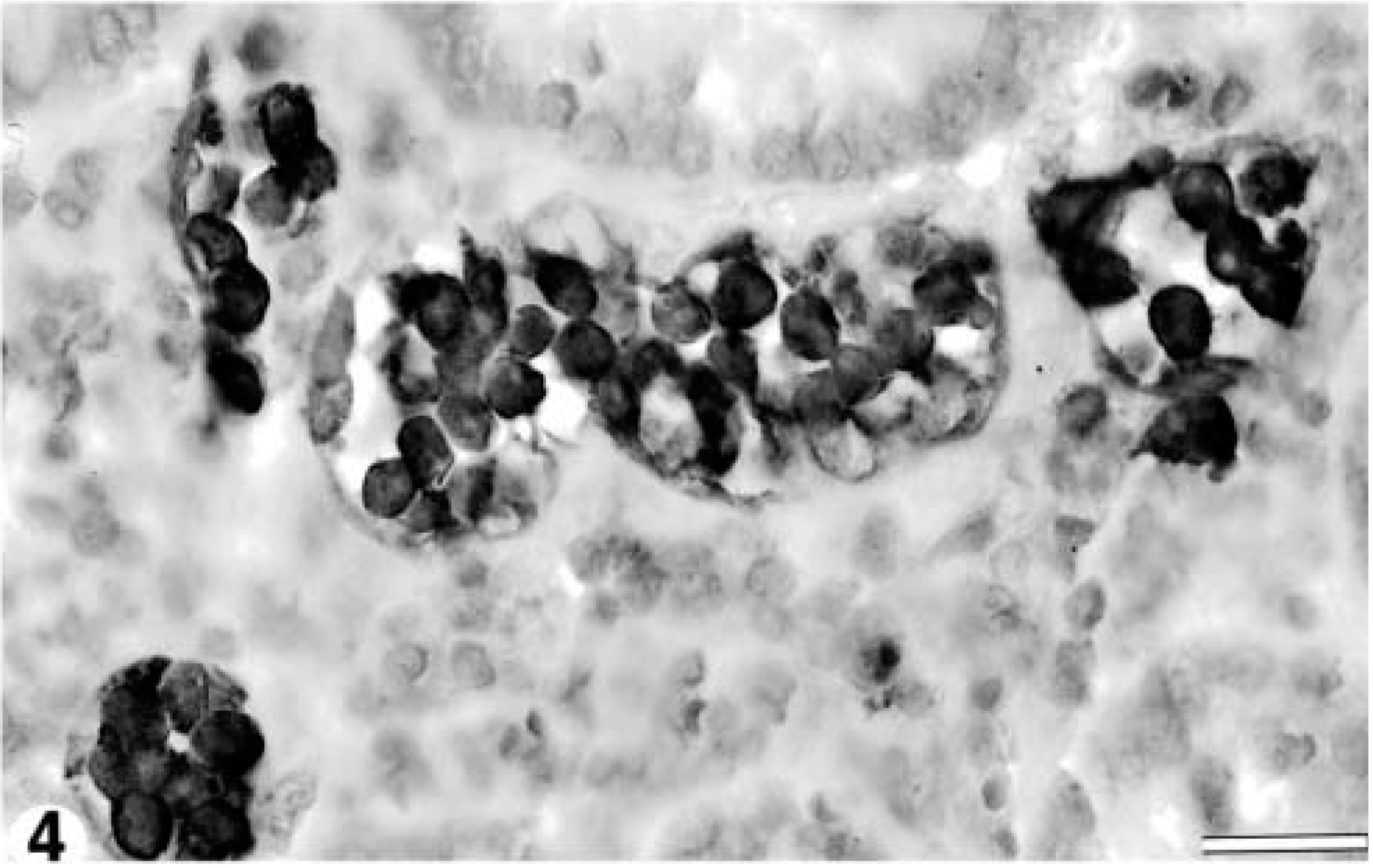
Colon; 8.5-year-old Finnish ewe. Intravascular cells stain positively with anti-CD79a antibody, indicative of B-cell lineage. Anti-CD79a antibody; avidin-biotin–peroxidase complex method; Meyer's hematoxylin counterstain. Bar = 20 μm.
Unfortunately, peripheral blood or bone marrow was not examined in this case. The findings of diffuse intravascular neoplastic B-lymphocytes and the pattern of splenic, hepatic, and nodal infiltration, however, are characteristic of leukemia. 5 Although a leukemic phase can occur in late stages of solid tissue lymphoma, 5 no evidence of multicentric or of solid tissue lymphoma was found.
Both enzootic and sporadic forms of lymphoma occur in sheep. 5 Retrovirus infection has been suggested to be a cause of enzootic lymphoma in sheep, and bovine leukemia virus can be experimentally transmitted to sheep. 7 It is not clear whether bovine leukemia virus or an as yet unidentified endogenous retrovirus is associated with spontaneous development of lymphoma and leukemia in sheep. Reverse transcriptase activity was detected in a previous case of ovine lymphocytic leukemia, although this activity could not be transmitted to cultured cells, and therefore the significance of the findings was considered questionable. 7 Although no viral studies were attempted in this case, no other animals on this farm have been affected with either lymphoma or leukemia, and a sporadic lymphoid leukemia occurring in an aged animal is considered most likely.
Footnotes
Acknowledgements
We thank Dr. Ursula Bechert for translation of reference #6 and Dr. Tony Van Dreumel for literature review assistance.
