Abstract
There is a need to develop simple, rapid, and accurate methods for assessing health in fish populations. In this study we demonstrate that use of fluorescein, a nontoxic fluorescent dye, can rapidly and easily detect the presence of skin ulcers in all fish tested, including rainbow trout (Oncorhynchus mykiss), channel catfish (Ictalurus punctatus), goldfish (Carassius auratus), and hybrid striped bass (Morone saxatilis male X M. chrysops female). Exposure of fish to as little as 0.10 mg fluorescein per milliliter of water for 3 minutes was sufficient to identify experimentally induced lesions, even pinpoint ulcerations. Such lesions were not visible to the naked eye but were clearly demarcated with fluorescein treatment. Examination of fish that appeared clinically normal often revealed the presence of focal ulcerations, which might have been a consequence of damage during capture, but it also might suggest that skin ulceration may be common even in “clinically normal” fish. Exposure of either nonulcerated or experimentally ulcerated hybrid striped bass to an excessively high concentration of fluorescein had no apparent effect on health or survival. Our studies suggest that fluorescein may be a highly useful tool for rapid health screening in fish populations.
Skin ulcers are one of the most common maladies affecting both cultured and wild fish.11 Many classical pollutants have either been associated with or have been experimentally proven to cause skin ulcers in fish, including crude petroleum, municipal sewage, industrial waste, pulp mill effluent, organochlorines, polycyclic aromatic hydrocarbons, and heavy metals (copper, zinc, lead). In addition, many other environmental factors have been associated with the development of skin ulcers, including low or high pH, ultraviolet radiation, temperature extremes, low environmental oxygen, and hormonal changes.6
Whereas advanced skin damage is often grossly visible and thus easily identified, fish skin can become ulcerated without displaying any obvious signs of damage. For example, fish that are experimentally exposed to the toxic alga Pfiesteria may not show any gross evidence of skin damage, even when virtually their entire epidermal epithelium is gone.8 There is a need to rapidly and efficiently assess where the initial signs of skin damage occur because intervention (e.g., treatment with antibiotics or other therapeutics) is much more successful when initiated at the earliest time possible during an outbreak.3 Thus, a method for detecting skin damage before it is grossly evident would be a very valuable clinical tool.
Fluorescein (3′,6′-dihydrospiro[isobenzofuran-1(3H),9′-[9H]xanthen]-3-one) sodium (“fluorescein”) is a yellow, relatively nontoxic, vital, hydroxyxanthene dye that produces an intense green fluorescence in slightly acid to alkaline (pH > 5) solutions. Fluorescein has commonly been used to detect ophthalmic lesions, such as corneal ulceration, in humans and animals. It has also been used as a tracer in clinical studies of ocular blood flow (angiography),1 reflecting its low toxicity. Fluorescein exhibits a high degree of ionization at physiologic pH; thus, it neither penetrates intact epithelium nor forms a firm bond with (i.e., stains) vital tissue. But when there is a break in the epithelial barrier, fluorescein can rapidly penetrate.1 When exposed to light, fluorescein absorbs light in the blue range of the visible spectrum, with absorption peaking at 480–500 nm. It emits light from 500 to 600 nm, with a maximum intensity at 520–530 nm.2
Except for a few intertidal species, the skin of all fish is not keratinized. Thus, it does not have a dead, horny layer of epithelial cells on its surface; rather, it is similar to the corneal epithelium, in that living cells are present throughout.4 Thus, we hypothesized that fluorescein might be a safe and effective means of detecting skin ulceration in fish. In this study, we report that fluorescein can be used as a highly rapid, efficient, and sensitive indicator of skin damage in fish.
Materials and Methods
Experimental fish
Clinically healthy fish were held in freshwater at their optimal temperature range, including rainbow trout (Oncorhynchus mykiss; 50–60 mm total length [TL]; 14 C), channel catfish (Ictalurus punctatus; 120–150 mm TL; 23–25 C), goldfish (Carassius auratus; 110–130 mm TL; 23–25 C), and hybrid striped bass (Morone saxatilis male X M. chrysops female; 60–80 mm TL; 27 C). Fish were fed a commercial pelleted feed (Ziegler Brothers, Gardners, PA). Water quality in all culture systems was monitored regularly for ammonia, nitrite, and pH, and remained within acceptable limits during the course of the study. Fluorescein-positive areas were confirmed as ulcerated in representative individuals by fixing the skin in 10% neutral-buffered formalin, processing routinely for histology, and staining tissues with hematoxylin and eosin.7 For all experiments, four to six fish were tested; each experiment was replicated at least twice.
Detection of skin ulcers
To determine the ability of fluorescein to detect skin ulceration, test fish were anesthetized with 50 mg/liter of tricaine (MS-222, Argent Chemical Laboratories, Redmond, WA), and then the skin was removed on the flank by scraping with a #10 scalpel blade. Channel catfish, goldfish, rainbow trout, and hybrid striped bass were then placed in a solution of 0.20 mg fluorescein (AK-Fluor®, 10% fluorescein sodium injection, 100 mg/ml, Akorn, Inc., Decatur, IL) per milliliter of water for 6 minutes; fish were then removed from the fluorescein solution and immediately rinsed with fresh water for 3 minutes by placing them in two to three changes of clean water over a period of 2–3 minutes. The fish were then euthanatized with tricaine and immediately examined for skin damage under ultraviolet light. Protective eyeware was worn to shield out ultraviolet radiation during all experiments. In a separate experiment, the skin of a group of channel catfish was punctured with a 19-ga needle and treated with fluorescein as described above.
In a separate experiment, a group of hybrid striped bass was acutely confined, inducing rapid loss of epithelium from the fins, using procedures described previously.7 Briefly, 60–80 mm hybrid striped bass were acclimated to a temperature of 27 C for 2 weeks in a 1,500-liter tank. Fish were then kept in a small (3.5 × 14 × 12 cm) net enclosure for 120 minutes, after which they were stained with fluorescein. A control group was treated similarly, except that they were not exposed to the acute confinement stress. This acute confinement stress typically causes severe fin ulceration in hybrid striped bass.7 Ulceration was confirmed in fluorescein-positive areas by fixing tissues for histology as described above.
We also examined the skin of five apparently healthy channel catfish, sampled from an aquarium containing 450 liters of freshwater, using the same procedure as described above. Fluorescein-positive areas were fixed for histology as described above and were examined for ulceration.
Safety of fluorescein to fish
Hybrid striped bass (45–55 mm TL) were acclimated to replicate aquaria containing 40 liters of 18 C freshwater and having active biofilters (5 fish per aquarium × two replicates per treatment). After acclimation for 7 days, fish were netted one at a time from each aquarium and anesthetized with 50 mg/ml tricaine buffered with 100 mg/ml sodium bicarbonate. Using a scalpel, five to six scales were removed from each fish; the fish was then placed in a high concentration of dye (0.5 mg fluorescein per milliliter for 12 minutes). A separate group of 10 fish was treated with fluorescein but did not have their scales removed. Fish were fed ad lib with flake food and observed for 28 days.
Photography
Photographs were taken in complete darkness under ultraviolet light, with Ektachrome 200 film (Kodak, Rochester, NY) at F4 to F5.6 (selection with the best depth of focus) using a Nikon camera with a 105-mm macro lens. An ultraviolet light source (Mineralight model UVGL-58, Upland, CA) was held at an angle of 45° to the fish, at a distance that was as close as possible to the fish (to get optimum fluorescence while not being so close as to interfere with the photograph). Photographs were taken using either the long (365λ) or short (254λ) wavelength setting. Because the absorption and emission spectra of fluorescein are very close to each other, a barrier filter was needed to eliminate autofluorescence (i.e., blue background).2 Photographs were taken using either a yellow barrier filter (Kodak Wratten #21 yellow) to filter out the blue autofluorescence, or with no filter. Photographs under white light were taken without a barrier filter.
Results and Discussion
Detection of ulcers using fluorescein
All ulcers that had been experimentally induced by scraping the flank with a scalpel were clearly visible when examined under ultraviolet light, even though these highly extensive lesions were not very evident under normal white light (Figs. 1–10). In real life, the appearance of all fish exposed to fluorescein was completely dark (unstained), except for the bright, apple-green fluorescence of ulcerated areas (e.g., Figs. 3, 5, 6). During photography, use of the long wavelength setting of the ultraviolet light, combined with the yellow barrier photographic filter, was needed to reproduce this effect. When fish were photographed without the yellow barrier filter, areas of the fish that did not bind fluorescein autofluoresced a pale blue; however, this made the outline of the fish much clearer in the photograph (Figs. 2, 8, 10).

Channel catfish with experimentally induced skin ulcer (flank scraped by a scalpel) that has been treated with fluorescein. Photographed under white light. Bar = 2 cm.

Same fish as in Fig. 1, photographed under short-wavelength ultraviolet light with no filter. Note the bright fluorescence. Bar = 2 cm.
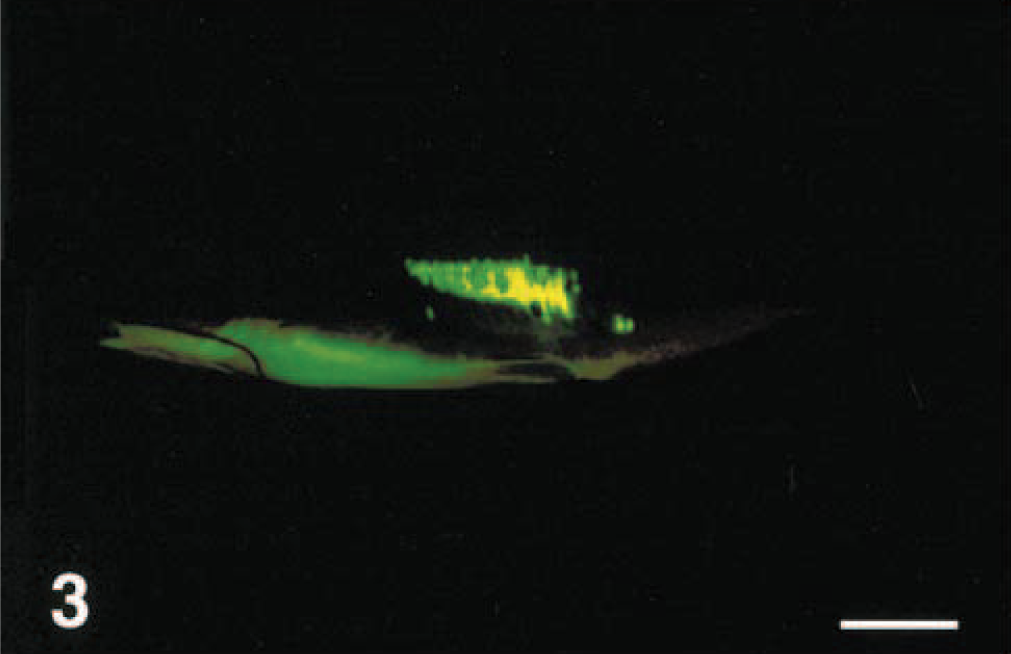
Same fish as in Fig. 1, photographed under long-wavelength ultraviolet light with a yellow filter. Bar = 2 cm. &emsp

Channel catfish with flank punctured with a 19-ga needle and photographed under white light. Bar = 2 cm.
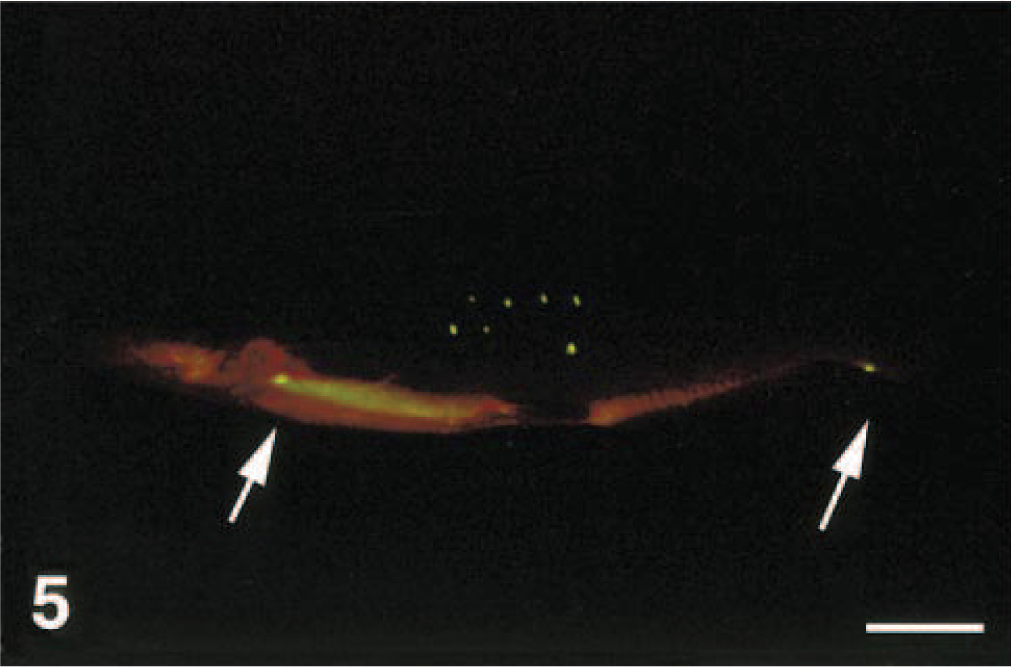
Same fish as in Fig. 4, photographed under long-wavelength ultraviolet light with a yellow filter. Note the seven closely spaced, pinpoint ulcers on the flank (needle marks). Also note the two small areas of fluorescence (spontaneous ulcers) at the bases of the caudal and pectoral fins (arrows). Bar = 2 cm.
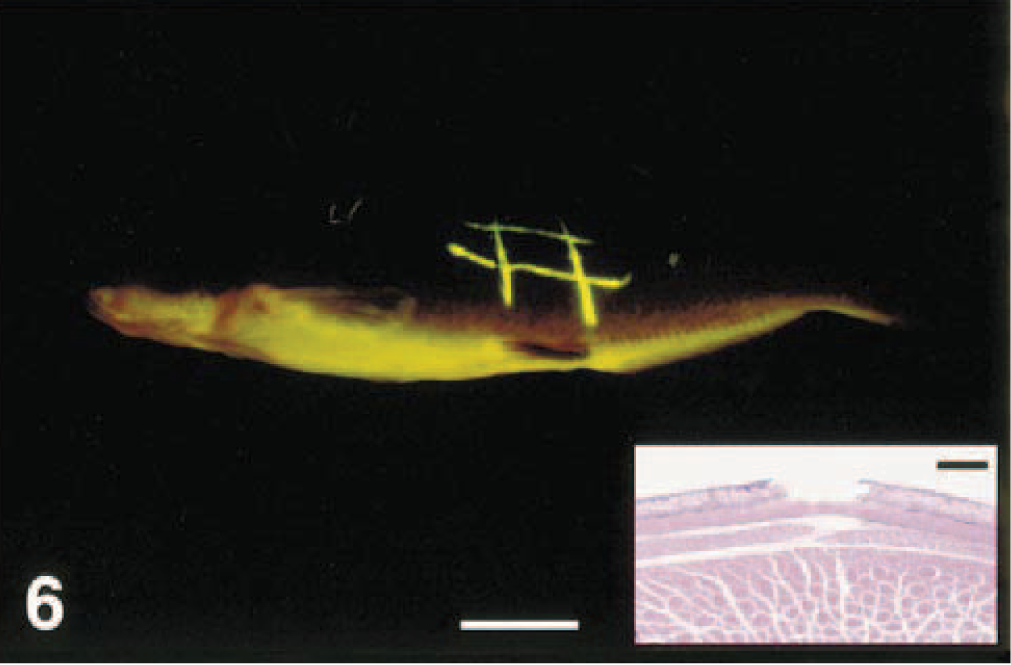
Channel catfish with long, thin ulcers created with the tip of a scalpel blade; photographed under long-wavelength ultraviolet light with a yellow filter. Bar = 2 cm. Inset: Histologic section showing discrete ulceration of the fluorescing area. HE. Bar = 30 μm.

Rainbow trout with experimentally induced skin ulcers (flank scraped by a scalpel) that have been treated with fluorescein. Photographed under white light. Bar = 2 cm.
Same fish as in Fig. 7, photographed under short-wavelength ultraviolet light with no filter. Bar = 2 cm.

Goldfish with experimentally induced skin ulcers (flank scraped by a scalpel) that have been treated with fluorescein. Photographed under white light. Bar = 2 cm.
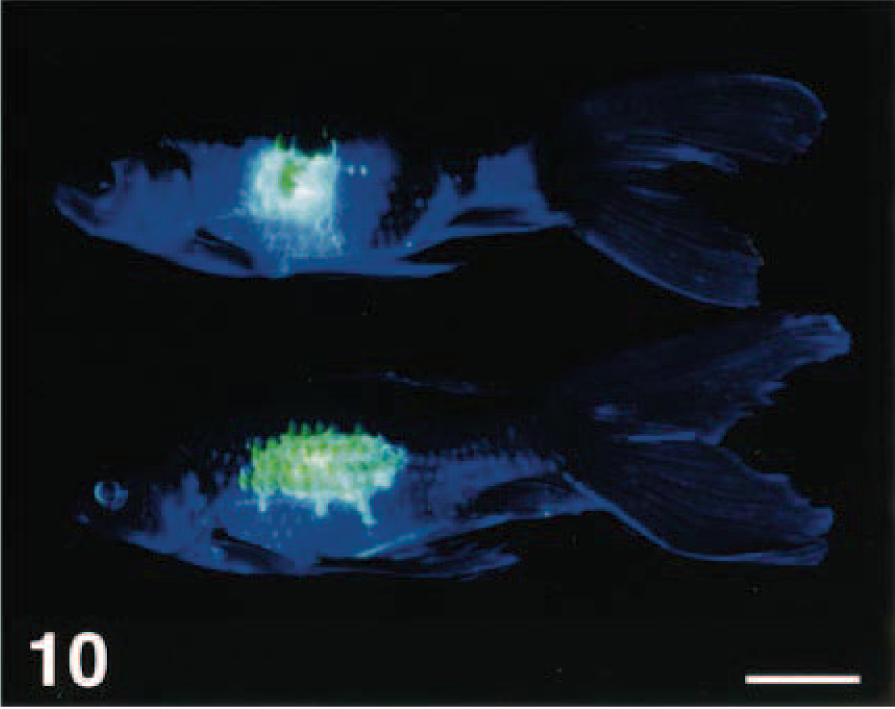
Same fish as in Fig. 9, photographed under short-wavelength ultraviolet light with no filter. Bar = 2 cm.
In initial studies, we tested a range of fluorescein concentrations (0.10–0.60 mg/ml) and exposure times (1–20 minutes) to optimize the conditions for most rapid and sensitive detection of skin ulceration (E. Noga and P. Udomkusonsri, unpublished). Exposing fish to a lower than optimal concentration of fluorescein resulted in weaker staining of the ulcers. Conversely, fish could be left in the fluorescein solution for at least 10 minutes longer than that needed for optimal staining without causing nonspecific staining (i.e., there was no evidence that nonulcerated areas became stained with this prolonged exposure to fluorescein). Thus, longer than needed exposures did not cause a false-positive response. Even experimentally induced, pinpoint lesions (Fig. 4) were readily visible (Fig. 5). Experimentally induced lesions were confirmed to be ulcerated through histology (Fig. 6).
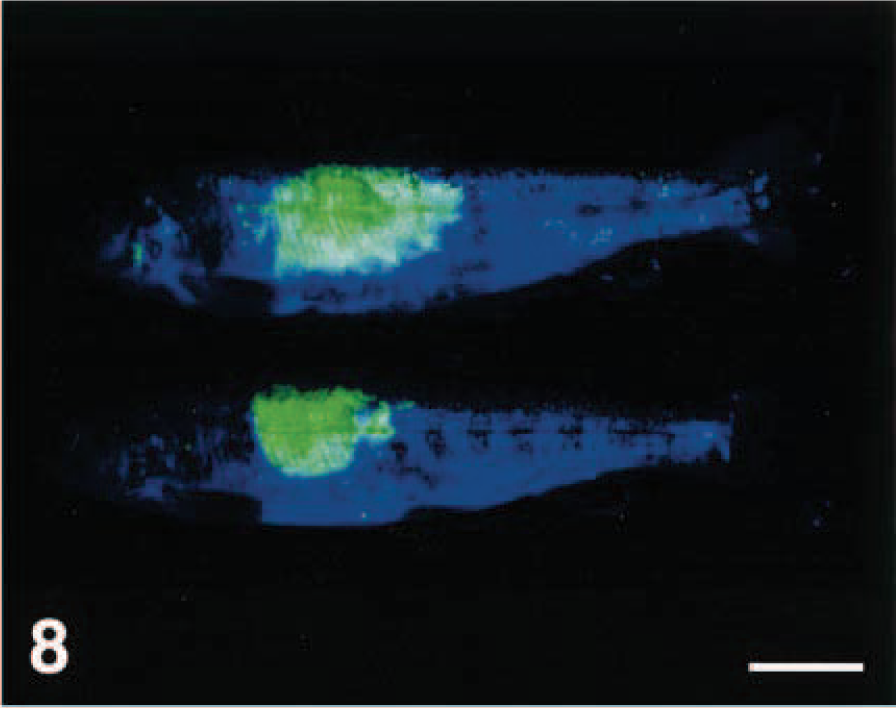
Hybrid striped bass in which fin ulceration was induced by acute confinement displayed severe ulceration on their fins (Figs. 11, 12), which was consistent with our previous findings that exposure of hybrid striped bass to acute stress causes severe ulceration on the fins.7 This occurs rapidly (within minutes to less than 2 hours) when the stress is sufficient. This phenomenon may be a widespread response to acute stress in fish and may explain why many skin diseases can develop so rapidly in fish.

Hybrid striped bass with fin ulceration caused by acute confinement; photographed under short-wavelength ultraviolet light with no filter. Intense green fluorescence in several areas is due to ulcers on the fins (arrow) and cornea (arrowhead). Bar = 1 cm.

Control (unstressed) hybrid striped bass. Photographed under same conditions as fish in Fig. 11. Bar = 1 cm.
In some fish, we observed fluorescence in areas other than those in which we experimentally induced skin ulcers (e.g., Fig. 5). When such lesions were examined using histology on five apparently normal channel catfish, all were confirmed to be ulcerated. Thus, the additional sites of fluorescence that we observed at times in fish, such as those shown in Fig. 5, were apparently due to skin damage that was not visible to the naked eye. This coincides with our observation that the skin of all test fish was extremely delicate and was easily damaged by simple manipulations such as handling. It was very easy to induce ulcers iatrogenically (E. Noga and P. Udomkusonsri, unpublished). Interestingly, recent studies have provided evidence that clinically normal fish may have a significant amount of inapparent skin damage even under presumably nonstressful, routine culture conditions.5
All the 20 hybrid striped bass that were treated with fluorescein and observed for 28 days remained healthy. These findings were expected because excessively higher doses of fluorescein are required to induce toxicity in fish. For example, Pouliquen et al.9 found that exposure of turbot (Scophthalmus maximus) to 700 mg fluorescein per liter for 96 hours was not lethal.
Possible uses of the fluorescein test
The rapid, simple, and apparently highly sensitive nature of the fluorescein test suggests that it may find widespread application in fish health assessment. Because the early stage of skin ulceration is not easily visible to the naked eye, fluorescein can be used to determine the extent of damage to the skin after stressful events, allowing the farmer, pet store retailer, or other culturist to determine the degree of stress. Thus, it could warn the owner if the fishes needed to be given supportive therapy before they become obviously sick. Whereas many ulcers are easily detectable with the naked eye and do not need special techniques to identify them, the power of our test is that it can identify ulcers before they are visible to the naked eye and become obviously infected.
We anticipate that this test may be applicable to health monitoring in a number of situations. These include:
Determining health status—Fluorescein is a nontoxic dye that is used extensively in natural waters to measure water flows.9,10 Its wide use in fisheries applications suggests that it might be easily put to use in diagnosing ulcers on food fish. Nonetheless, because only a sample of fish would probably normally be examined for routine health screenings, even if it were not approved for food fish use, this would probably not hinder its application. At a retail cost of about US$150/kg (Argent Chemical Laboratories, Redmond, WA), it costs less than $0.08 to make 1 liter of a 0.20 mg/ml solution; this is enough to test a large number of small fish. General health screening—Early stages of skin ulceration in fish are at present evident only when using specialized techniques (i.e., histopathology or microscopic examination of fresh fin biopsies). Histopathology is expensive and most importantly time consuming (takes several days) and requires considerable technical skill; because only small tissue pieces can be evaluated, it may not give an accurate indication of the amount of damage present. The use of fresh fin biopsies is limited to evaluation of the fins, not the body, and again only a small area of skin can be easily evaluated. The fluorescein test allows rapid, real-time evaluation of skin health; hence, it could be readily incorporated into routine health screens for both scaled (e.g., trout, goldfish, bass) and scaleless (e.g., catfish) fish because, in all fish, the epithelium forms the outermost layer of the skin. The sensitivity of this test is exemplified by the skin damage detected in clinically normal fish. Indicator of health after a known stressful event—It is well known that certain types of procedures (e.g., shipping, grading, vaccinating, etc.) are stressful and may sometimes lead to an infectious disease epidemic. But it is difficult to predict when such events may lead to epidemic disease. If skin damage were involved in the epidemic, this test would give the farmer more time to respond appropriately to mitigate any adverse effects of the stress. This would save time and money and may possibly reduce the need for medication. Environmental monitor—This test may be useful to routinely assess the health of wild fish populations in both freshwater and marine environments, where skin ulcers are one of the most common indicators of polluted environments.6,11
Aid to bacterial diagnosis—The importance of detecting skin damage early in bacterial infections is exemplified by the studies of Elliott and Shotts,3 who found that Aeromonas salmonicida, the primary bacterial pathogen of ulcer disease of goldfish, was only present in the earliest stages of the disease (i.e., small lesions). Being able to identify and thus culture the earliest lesions, which may not even be visible to the naked eye, could improve the ability to identify important pathogens.
In conclusion, fluorescein may be a highly versatile method for rapidly assessing skin damage in most, if not all, fish.
Footnotes
Acknowledgements
We thank the staff of the NCSU-CVM Biomedical Communications Center, especially D. Wagner and W. Savage, for excellent assistance with these studies. This work was supported by the USDA NRI Competitive Grant Program (Project #97-35204-7722), Grant #NA86-RG-0036 from the National Sea Grant College Program, National Oceanic and Atmospheric Administration, to the North Carolina Sea Grant College Program, and Binational US-Israel Agricultural Research and Development Project #US-3030-98. P. Udomkusonsri is a Fellow of the Royal Thai government.
