Abstract
Samples from feline invasive mammary carcinomas (FMCs) were used to determine the prognostic significance of the immunohistochemical expression of vascular endothelial growth factor (VEGF) and microvessel density (MVD). Forty-eight queens bearing FMCs were included in a 2-year follow-up study. Mammary tumors were classified according to the World Health Organization system and graded on the basis of histologic criteria. Tumor sections were immunostained using anti-VEGF and anti-von Willebrand factor (vWf) antibodies. VEGF expression was quantified on the basis of the percentage of positive cells. MVD of vWf-positive microvessels was determined by both mean microvessel counts and highest microvessel counts. Normal mammary gland tissues showed an inconspicuous VEGF staining. In FMCs the proportion of VEGF-positive cells was significantly higher in papillary and solid carcinomas than in tubular and papillary cystic tumors. An increased number of cells expressing VEGF was also observed in poorly differentiated FMCS. Sixteen (33.3%) of the queens bearing invasive carcinomas were still alive at the end of the 2-year follow-up period, and 32 (66.7%) had died. The VEGF expression was significantly correlated with the clinical outcome, but no correlation was observed with the invasion of lymphatic vessels. A correlation between the higher percentage of VEGF-positive cells and the unfavorable prognosis was demonstrated by the estimation of curves for overall survival (P = 0.03). Univariate analysis showed that MVD did not correlate with the overall survival. The results of our study demonstrated that VEGF expression, although not associated with increased angiogenesis, is a prognostic indicator in feline mammary tumors. In contrast, there is no support for a role of neovascularization as an indicator of survivability.
Keywords
Introduction
Angiogenesis, the process leading to the formation of new vessels from a preexisting vascular network, is essential for the growth, invasion, and metastasis of solid tumors. Several studies in human oncology indicate that breast cancer is angiogenesis dependent.11 Tumors release angiogenic factors, such as vascular endothelial growth factor (VEGF), which induce the growth of a capillary network surrounding the tumor.9 Recent studies have suggested that VEGF expression is closely associated with the promotion of angiogenesis and with early relapse in primary breast cancer as well as in other tumors.23,29,34 VEGF has been shown to act selectively on endothelial cells by binding to specific Class-III–receptor tyrosine kinases (flt-1 and KDR) and by opening calcium channels to increase intracellular calcium.28 On the basis of these mechanisms, it stimulates angiogenesis by increasing vascular permeability and by acting as endothelial-cell mitogen.8 VEGF secreted from neoplastic tissues may contribute to tumor growth, invasion, and metastasis not only via the autocrine pathway to tumor cells but also via a paracrine pathway to the surrounding microvessels.33
Tumor microvessel density (MVD), a measure of the intensity of angiogenesis, has been evaluated in several neoplasms, including cutaneous melanoma, non–small cell carcinoma of the lung, carcinoma of the prostate, gastric carcinoma, and squamous cell carcinoma of the head and the neck.27 It has been shown that the extent of new vessel formation in human breast carcinoma is correlated with the occurrence of metastasis.36 A good correlation has been demonstrated between intratumoral vascularization and outcome in patients with breast cancer.18 In particular, quantification of immunohistochemically highlighted microvessel “hot spots” has been shown to be a powerful prognostic tool.10,16
In veterinary oncology, studies on the prognostic significance of angiogenesis have involved only canine mammary tumors.14,15,30
Because of the epidemiologic relevance and the aggressive clinical behavior of feline invasive mammary carcinomas (FMCs), the aim of our study is to evaluate the usefulness of the angiogenetic process as a marker of survival in this species. For this purpose we assessed by immunohistochemistry the VEGF expression, and the MVD in 48 feline spontaneously arising invasive mammary carcinomas. We compared these findings with histopathologic parameters and evaluated their prognostic significance in a 2-year follow-up period.
Materials and Methods
The mammary tumors used in this study were retrieved from the archives of the Tumor Registry of the Department of Animal Pathology, University of Pisa, Italy, between January 1998 and December 1999. The samples were surgically obtained by mastectomy or block dissection from 48 female cats of different breeds (36 European Domestic Shorthair cats, 4 Siamese, and 8 Persians) bearing mammary carcinomas. The queens included in the study were examined and surgically treated at the Veterinary Teaching Hospital (Clinic Department, School of Veterinary Medicine, University of Pisa). After anamnesis collection, including age, body size, history of ovariohysterectomy, prevention of oesterus with hormonal treatment, tumor size, adherence to underlying tissues, and skin ulceration, a complete physical examination was done. All of the cats included in the study were followed up for at least 2 years after surgery by referring veterinary surgeons to evaluate the postsurgery course of the disease. Overall survival was defined as the time from the day of diagnosis until the day of death or the last follow-up.
Representative portions of each tumor were fixed in 10% neutral buffered formalin and were routinely embedded in paraffin. Five-micrometer-thick sections were stained with hematoxylin and eosin (HE) for histologic evaluation; additional 5-μm sections were used for immunohistochemical studies. The mammary tumors were classified according to the World Health Organization (WHO) classification,17 and tumors displaying different features were classified according to the most pronounced histologic differentiation. The presence of lymphatic or stromal invasion was also recorded. Histologic tumor grading was performed on HE-stained sections using a previously described classification.7 The mammary carcinomas were classified as well-differentiated carcinoma (WDC, grade I), moderately differentiated carcinoma (MDC, grade II), and poorly differentiated carcinoma (PDC, grade III).
VEGF expression was assessed blind for clinicopathologic data using an anti-VEGF rabbit polyclonal antibody (A-20; Santa Cruz Biotechnology, Inc., CA). Five-micrometer-thick sections were deparaffinized in xylene and rehydrated in alcohol. Endogenous peroxidase activity was blocked by incubating the slides for 5 minutes at 37 C in Endo/Blocker (Bi⊘meda Corp., Foster City, CA) solution diluted 1:5 in methanol. After blocking nonspecific staining with normal horse serum (NHS) diluted 1:10 in Tris buffer saline (TBS: Tris-HCl, pH 7.6), with 2% poly-vinyl-pyrrolidone (PVP) and 1% bovine serum albumin (BSA) (TBS–2% PVP–1% BSA), the sections were incubated with the primary anti-VEGF antibody diluted 1:200 in TBS–2% PVP–1% BSA overnight at room temperature in a moist chamber. Sections were extensively washed in TBS and then incubated with a biotinylated affinity-purified horse panspecific secondary antibody (Vector Laboratories Inc., Burlingame, CA). Sections were again washed in TBS before incubation for 10 minutes in a streptavidin-biotinylated horseradish peroxidase complex (Biospa, Milan, Italy), and the reaction was developed using Nova Red substrate (Vector Laboratories Inc.) for 10 minutes. Finally, the sections were counterstained with hematoxylin, dehydrated, and mounted. Positive controls were included in each staining and consisted of sections from a human breast tumor known to express VEGF. Negative controls were obtained both by omitting the primary antibody and by using an unrelated rabbit-polyclonal antibody.
VEGF expression was assessed using a procedure previously described by Lewis et al.21 After scanning the whole tumor section at 40×, five representative nonadjacent and nonoverlapping fields (most of them VEGF-positive) from each tumor were selected. At 400× magnification the percentage of positive cells per field (at least 100 of the cells evaluated) was considered, and VEGF expression was determined as the mean of the percentages from the five counts.
Tumor vascularity was assessed using an anti–Von Willebrand factor (VWf) rabbit polyclonal antibody (Novocastra Lab., Newcastle upon Tyne, UK). Five-micrometer-thick sections on poly-
The microvessel densities of immunostained sections were assessed, without any knowledge of the subjects' previous investigations or clinical outcome, using a procedure previously described24 and on the basis of a modification of the method by Weidner et al.36 Microvessel quantification was performed by two independent observers, and the results were assessed for interobserver variability. Disagreement was resolved on the conference microscope. The immunostained slides from each tumor were at first scanned at 40× magnification, using a light microscope to select areas with the densest vascularization (hot spots). Normal mammary tissue, large areas of inflammation, granulation tissue, and tumor ulceration through the skin were excluded. After the individuation of the hot spots, three adjacent, nonoverlapping fields from each section were selected using a 200× magnification (field area 0, 27 mm2). The count performed was the field thought to contain the highest number of microvessels found at low magnification, and each subsequent count was the field thought to be the next highest. MVD was quantified as the mean vessel count (MVC), as obtained from the average vascular density of the three fields from each tumor. The highest vessel count (HVC), the number of microvessels in the tumor field with the densest vascularization, within the hot spot selected, was also considered to determine whether those two methodologies may give different prognostic information, as previously suggested.19,24
Statistical analysis was performed using the statistical package SPSS Advanced Statistics 7.5 (SPSS Inc., Chicago, IL). The analysis of variance (ANOVA) was determined, and the difference between groups was evaluated with Bonferroni's test. Linear relationship between variables was measured by Pearson's correlation coefficient, and curves for overall survival were determined by the Kaplan-Meier method.
Results
The mean age of the 48 female cats with invasive mammary tumors examined was 10.9 ± 2.8 (range 4–19 years). Sixteen (33.3%) of these (group A) were alive at the end of the 2-year follow-up period, and 32 (66.7%; group B) had died.
On the basis of the WHO classification,17 papillary carcinoma was the most common (26/48, 54.2%), followed by solid carcinoma (14/48, 29.2%), tubular (6/48, 12.5%), and papillary cystic (2/48, 4.2%). Histologic grading of the mammary carcinomas studied, revealed that 12 (25%) were WDCs (grade I), 28 (58.3%) were MDCs (grade II), and 8 (16.7%) were PDCs (grade III). Presence of lymphatic vessel invasion was detected in 27 (56.2%) of the 48 mammary tumors examined, whereas in the other 21 tumors (43.7%) a local stromal invasion was found to be present.
An inconspicuous VEGF expression was detected in normal mammary tissues (Fig. 1). In tumors, VEGF was commonly expressed in the cytoplasm of neoplastic cells, occasionally in stroma, and weakly in endothelial cells. Some tumor cells showed a granular staining pattern (Fig. 2). The anti-VWf polyclonal antibody showed a strong staining of the microvessels in both normal and neoplastic tissues (Fig. 3) but was also evident in some inflammatory cells and connective tissue, thus making the quantitation of MVD sometimes difficult.
Normal mammary gland; cat. VEGF expression is inconspicuous. Anti-VEGF immunoperoxidase histochemistry and hematoxylin (IPH-H). Bar = 30 μm.
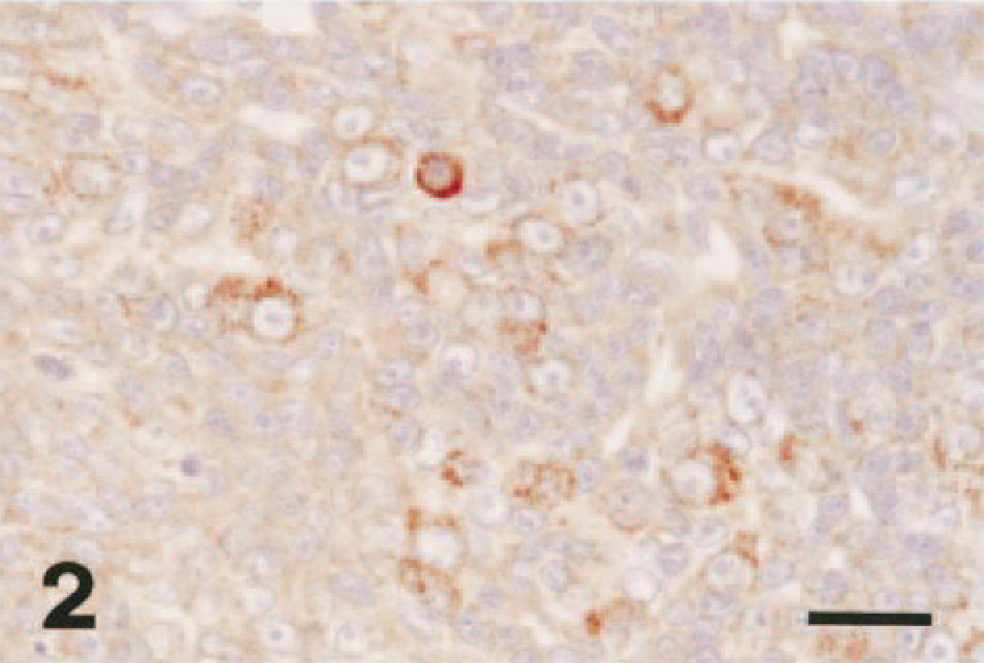
Mammary gland; cat. Invasive mammary carcinoma containing strong immunohistochemical staining for VEGF protein in tumor cells. IPH-H. Bar = 30 μm.
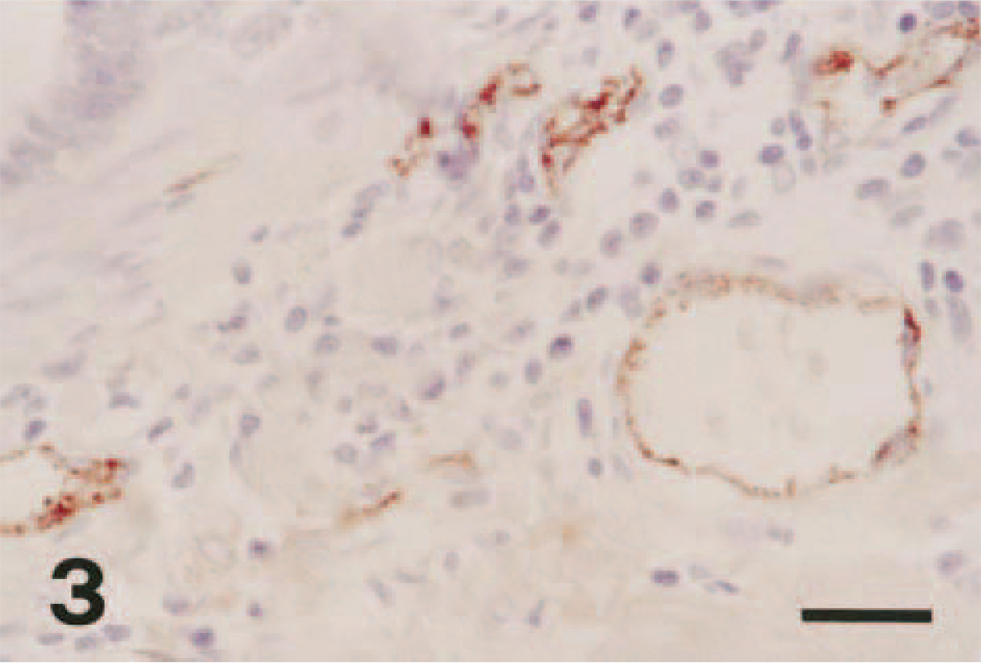
Mammary gland; cat. Invasive mammary carcinoma; endothelial cells of microvessels were strongly stained by the anti-VWf polyclonal antibody. IPH-H. Bar = 30 μm.
The proportion of VEGF-positive cells was significantly higher (P = 0.01) in papillary (mean ± SD, 70.3 ± 22) and solid FMCs (68.9 ± 30) than in tubular carcinomas (39 ± 24.9) and papillary cystic carcinomas (30.6 ± 34.8). When VEGF expression was investigated in FMCs classified according to tumor grading, the percentage of positive cells was significantly higher in PDCs (87 ± 16.3) than in WDCs (56.8 ± 27.9) and in MDCs (61.1 ± 27.1) (P = 0.01 and 0.05, respectively). No significant correlation was found between the percentage of VEGF-positive cells and lymphatic invasion.
Tumor cells of group B queens (72.4 ± 22.6) showed higher (P = 0.003) VEGF values than those of group A subjects (48.1 ± 29.8). The relationship between VEGF values by quartiles and by postsurgical deaths within 2 years is presented in Table 1. The queens with a VEGF value ≥ 45.4 (second–fourth quartiles) had a poor prognosis compared with the queens of the first quartile. The correlation between higher VEGF expression and an unfavorable prognosis was also demonstrated by the estimation of curves for overall survival. There was a significant association between overall survival and higher VEGF values (> median value, 72.1) in the queens with invasive carcinomas investigated (P = 0.03.; Fig. 4). Mean survival time for queens with VEGF expression < 72.1 was 18.4 ± 1.3 months, whereas for subjects with values > 72.1, mean survival time was 14.2 ± 1.2 months.
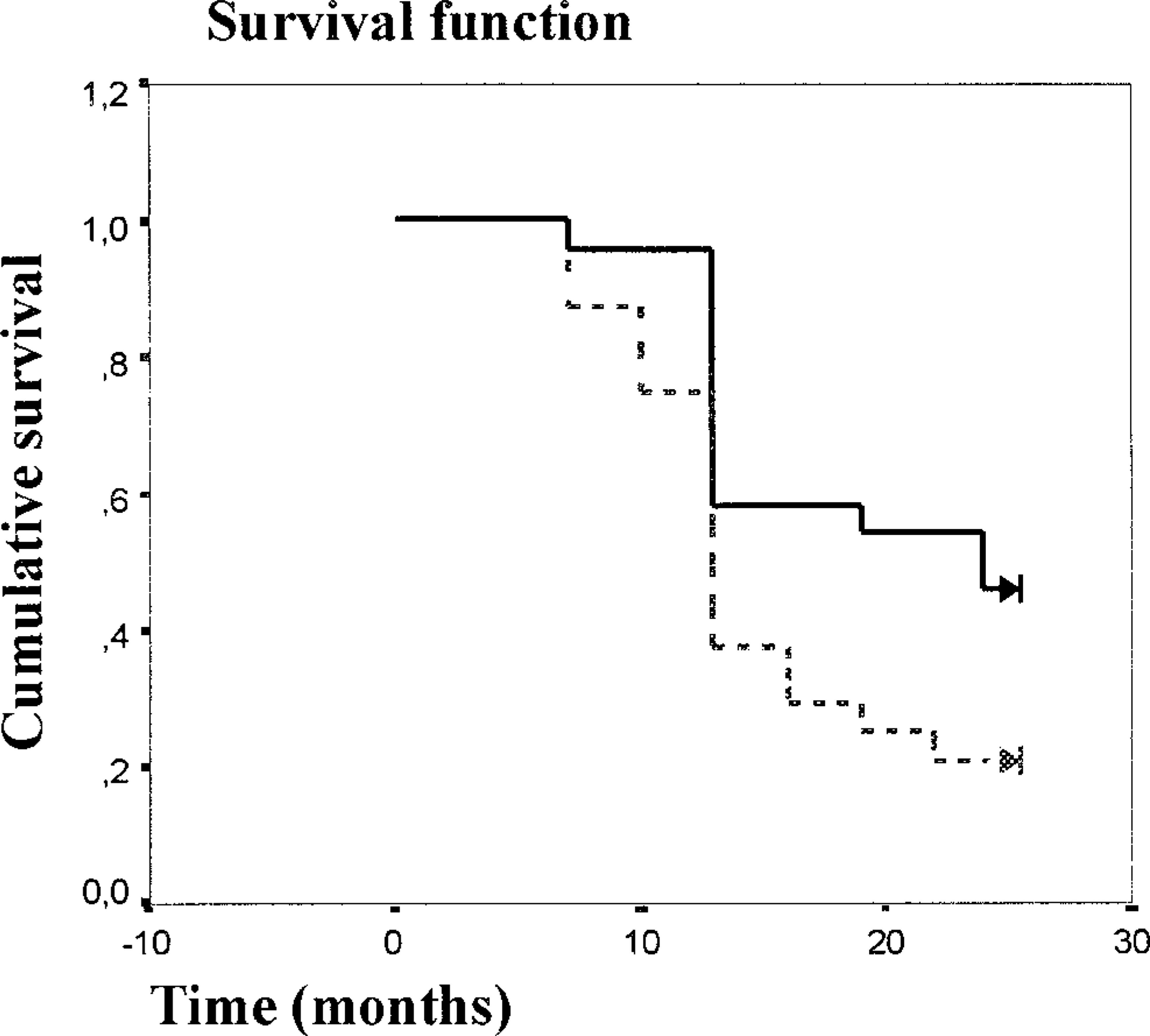
Overall survival curves for the group of 48 queens with invasive mammary carcinomas with low VEGF values (≤ 72.1% —) and high VEGF values (> 72.1% –––) (P = 0.03).
Quartiles of VEGF values and deaths in 48 cats with invasive mammary tumors during the 2 years after surgery.

Regarding the methods used to asses MVD, we found that HVC and MVC values were significantly correlated (r = 0.958; P = 0.001). While correlating the MVD determined by the two count methods with the histologic parameters considered, no significant correlations were found.
MVD in tumors of group B queens (29.7 ± 7.7) was not significantly different from MVD in those of group A subjects (27.9 ± 8.2.). The relationship between MVD determined by HVC by quartiles and postsurgical deaths within 2 years is presented in Table 2. The lack of correlation between higher MVD values and an unfavorable prognosis was also demonstrated by the estimation of curves for overall survival. There was no significant association between overall survival and higher MVD values (> median value, 29.5) in the queens with invasive carcinomas investigated (P > 0.05.; Fig. 5). Even if a significant association between shorter survival times and increased MVD was not evident, the subjects with MVD of > 29.5 had a poor prognosis. In fact, mean survival time for queens with MVD < 29.5 was 18.2 ± 1.4 months, whereas for subjects with values > 29.5, mean survival time was 14.5 ± 1.1 months. VEGF values and MVD determined with either HVCs or MVCs were not statistically correlated (r = 0.259; P > 0.05).
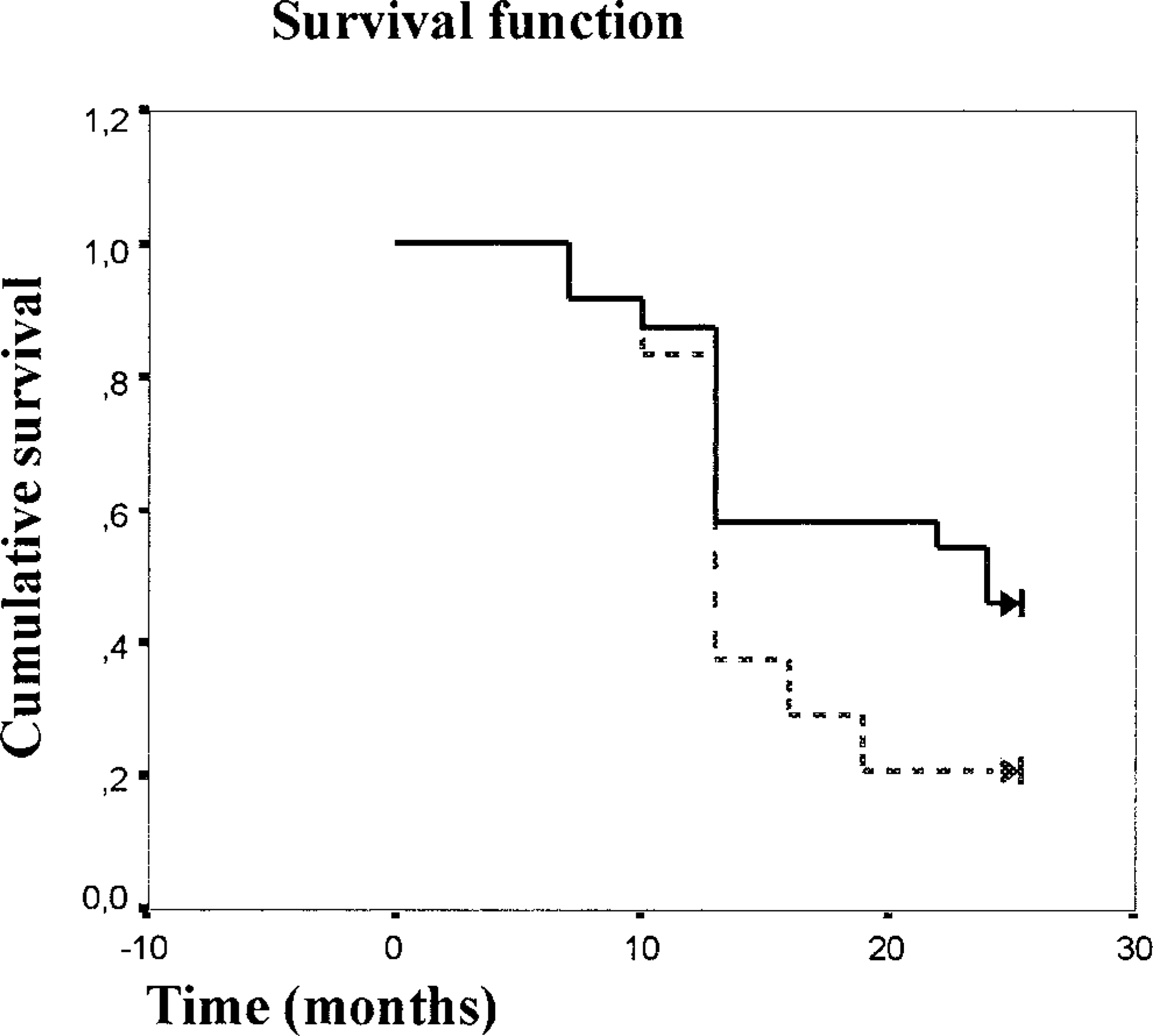
Overall survival curves for the group of 48 queens with invasive mammary carcinomas with low MVD (≤ 29.5% —) and high MVD (> 29.5% –––) (P > 0.05).
Quartiles of MVD determined by highest vessel count and deaths in 48 cats with invasive mammary tumors during the 2 years after surgery.

Discussion
In this study we examined the relationship between angiogenesis with the clinical behavior of FMCs by quantitation of VEGF expression and by determining MVD on sections stained with an anti-VWf polyclonal antibody. The malignancy of FMCs is confirmed by the death rate (66.7%) at the end of the 2-year follow-up period. Previous studies have shown the necessity of a reliable prognostic index in FMCs. Attention has been focused mainly on tumor grading, proliferation markers, hormonal receptor status,2,3,6,26,31 and more recently on p53 mutations and topographic distribution of bcl-2 protein.22,25 Until now, this is the first report on angiogenesis in FMCs and its correlation with the clinical course of the disease. Several studies on human breast carcinomas reported that neoplastic growth and metastatic spread are angiogenesis-dependent processes.8,36 Tumors release angiogenic factors such as VEGF, which induces growth of a capillary network surrounding the tumor,9 and MVD has often proven to affect the prognosis of breast cancer.12,28 In our study VEGF expression was detected in all the carcinomas investigated and was significantly correlated with both histologic type and grading of tumors. The VEGF production, as determined by immunolabeling of the anti-VEGf polyclonal antibody used, was significantly higher in queens with poor prognosis, suggesting its important role in the clinical outcome of FMCs. Many retrospective studies on human breast cancer reported that VEGF expression is inversely correlated to relapse-free survival, overall survival, or both and that high levels of VEGF can identify the subgroups of patients with poor prognosis.11 Our results also demonstrated a correlation between VEGF expression and prognosis, but there was no correlation between MVD parameters and prognosis. This seeming inconsistency may be the result of several factors, as demonstrated by the lack of predictive value of vascular “hot spots” in some human tumor studies.1,10 The ratio of positive correlative studies to negative ones is running about 4 to 1, and this may be affected by several methodological problems. The hypothesis for the lack of correlation between MVD and prognosis should include intra- or interobserver variability, selection of the area of most intense neovascularization, heterogeneity of vascular distribution in neoplastic tissue demanding an excessive number of examined fields for statistical power, or heterogeneous patients' databases.10,28,35 Furthermore, it is possible that tumor malignancy may be related to tumor cell phenotypic expression of VEGF, but this does not translate to increased neoangiogenesis.
With respect to the absence of correlation between MVD and VEGF expression, de Jong et al.5 have suggested that many additional agonists, receptors, and inhibitors other than VEGF may be involved in the formation of new vessels. Especially, the role of VEGF receptors, VEGF-R1-Flt 1 and VEGF-R2-KDR/Flk 1, has been investigated; these results suggest that VEGF might act as a direct autocrine growth factor for tumor cells via VEGF-R1 and that angiogenesis could be promoted in paracrine loops, where VEGF is produced by fibroblasts and tumor cells and then binds to endothelial cells via induced VEGF receptors.4 Recent investigations demonstrated that the molecular biology and function of the VEGF signaling system are virtually identical in humans and canines, both in healthy as well as in disease conditions.32 On the basis of these observations, the role of VEGF receptors should be extensively investigated in veterinary medicine also.
Furthermore, recent studies have suggested the necessity of quantitatively distinguishing between tumor neovascularization and preexisting vessels. For this purpose the use of a monoclonal antibody directed to human CD 105, which preferentially reacts with endothelial cells in angiogenic tissues, has been proposed.20 In our experience anti-VWf was the only antibody tested that cross-reacted with feline mammary tissue endothelium. Attempts to highlight microvessels using anti–CD-31 antibody, a more sensitive method for detecting immature microvessels or single endothelial cells,13 have been unsuccessful (data not shown).
In conclusion, the measure of VEGF expression in paraffin-embedded sections has been demonstrated to be a powerful prognostic indicator for FMCs. This marker should therefore be a useful measurement to assist the veterinary clinician and pathologist in predicting clinical outcome.
