Abstract
The expression of 5 markers associated with angiogenesis was studied in canine squamous cell carcinomas (SCCs) (n = 19) and canine trichoepitheliomas (TCPs) (n = 24). SCCs were assigned histologic grades, and tissue sections from both tumor types were immunohistochemially stained for the expression of vascular endothelial growth factor (VEGF), vascular endothelial growth factor receptor-2 (VEGFR-2), as well as intratumoral microvessel density (iMVD), tumor proliferation index (PI), and tumor apoptotic index (AI), using antibodies against VEGF, VEGFR-2, von Willebrand's factor, Ki-67 antigen, and the terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate endlabeling method (TUNEL), respectively. VEGF and VEGFR-2 were detected in 17/19 (89.4%) and 19/19 (100%) SCCs and in 17/24 (70.8%) and 20/24 (83.3%) TCPs, respectively. In SCCs, there was substantial correlation between histologic grade and PI (r = 0.51); and moderate correlation between VEGF and histologic grade (r = 0.43), VEGFR-2 and histologic grade (r = 0.47), VEGF and PI (r = 0.47), and VEGFR-2 and PI (r = 0.47) (Spearman rank correlation coefficient). In TCPs, there was substantial correlation between VEGF and PI (r = 0.51) and a moderate correlation between VEGFR-2 and iMVD (r = 0.36). The median iMVD of SCCs (15.5) was significantly higher than the median iMVD of TCPs (9.05) (P value < .05). It was concluded that VEGF and VEGFR-2 may promote tumor cell proliferation in TCPs and SCCs. An autocrine pathway for VEGF probably operates in canine SCCs and TCPs, as VEGF and VEGFR-2 expression was found in most tumors and was associated with evidence for tumor cell proliferation.
Keywords
Angiogenesis, the formation of microvasculature, is crucial for the growth and metastasis of malignant tumors. 12 Angiogenesis is promoted by factors produced by tumor cells or their surrounding stroma. 21 Among these factors, vascular endothelial growth factor (VEGF, also known as VEGF-A) is thought to play a key role in tumor angiogenesis. 19 Four VEGF isoforms exist as a result of alternative patterns of splicing: VEGF 121, VEGF165, VEGF189, and VEGF206. 20 VEGF mediates angiogenesis by binding to 2 receptors on endothelial cells known as vascular endothelial growth factor receptor-1 (VEGFR-1) and vascular endothelial growth factor receptor-2 (VEGFR-2). 10 While both receptors bind to VEGF with high affinity, 5, 39 only VEGFR-2 is capable of mediating angiogenesis. 9 VEGFR-1 is thought to play a negative regulatory role by decreasing the availability of VEGF to VEGFR-2. 9, 13 VEGF and VEGFR-2 may be responsible for increased tumor cell proliferation. 7, 23, 29 The simultaneous expression of VEGF and VEGFR-2 on tumor cells may indicate that VEGF has an autocrine function. 6 VEGF may also decrease tumor apoptosis. 22 The increased expression of VEGF has been correlated with the histologic grade of malignancy in many human tumors. 8, 40 Moreover, the relationship between the degree of angiogenesis, often expressed as intratumoral microvessel density (iMVD), and many parameters, including tumor proliferation index (PI), an expression of tumor cell proliferation, and tumor apoptotic index (AI), an expression of tumor cell death by apoptosis, has been extensively studied in human cancer. 22
In the veterinary literature, the association among VEGF, VEGFR-2, iMVD, PI, and AI has been evaluated in a few canine tumors. 26– 28, 32– 36 Therefore, the role of VEGF and VEGFR-2 in angiogenesis and tumor cell proliferation in canine cancer is still unclear, and studying the relationship between these markers may aid in understanding the role VEGF and VEGFR-2 play in cancer progression in dogs.
The purpose of this study was to evaluate and compare the expression of these markers in a malignant and a benign canine skin tumor type, cutaneous squamous cell carcinoma (SCC) and trichoepithelioma (TCP), respectively. Both tumor types are common in dogs and arise from squamous epithelium.
Materials and Methods
Tissue selection and histologic grading
Nineteen and 24 tissue sections of cutaneous SCC and benign TCP, respectively, which were submitted between 1999 and 2004 to Prairie Diagnostic Services at the Western College of Veterinary Medicine, Saskatoon, Saskatchewan, Canada, were selected if at least 1 cm 3 of tissue was available for evaluation. SCC tissues were given histologic grades from 1 to 3, according to previously established criteria. 15 Briefly, well-differentiated tumors were given a grade of 1 (Fig. 1) if they had abundant eosinophilic cytoplasm, prominent intercellular bridges, and many concentric laminated masses of keratin (keratin pearls). Nuclear pleomorphism and mitotic activity were minimal. Moderately differentiated tumors of grades 2 and 3 in the reported classification scheme were grouped together and given a grade of 2. Tumor cells in this category had less eosinophilic cytoplasm, more pleomorphic nuclei, more numerous mitotic figures with fewer keratin pearls, and more prominent dermal invasion (Fig. 2). Grade 3 was given to poorly differentiated tumors that showed little squamous differentiation, marked mitotic activity, and deep infiltration. Tumor cells had amphophilic cytoplasm and often appeared as single cells or in small groups of cells (Fig. 3). Histologic sections from each tumor were graded independently by 2 of the authors (ANA and BAK). Tumors receiving different histologic grades were re-evaluated, and a consensus was established. The histologic features of TCPs were previously described. 14
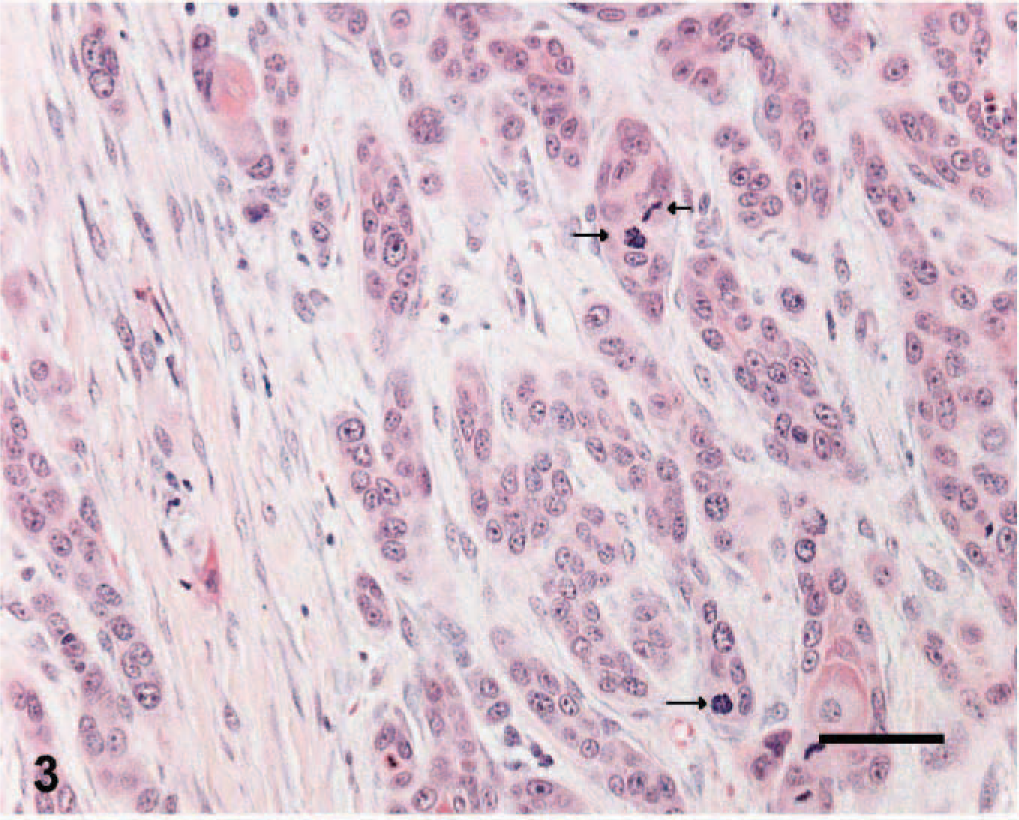
Skin; canine squamous cell carcinoma, grade 3. Note the arrangement of cells in small groups and the marked mitotic activity (arrows). HE. Bar = 25 μm.
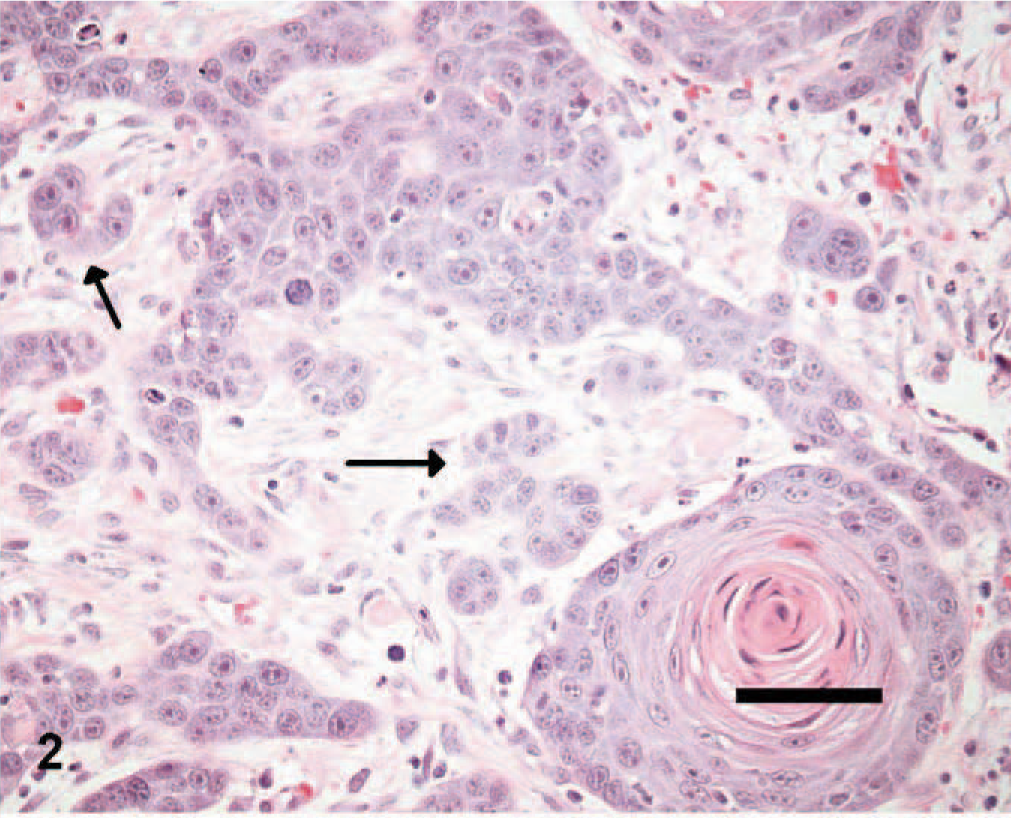
Skin; canine squamous cell carcinoma, grade 2. Note the arrangement of some tumor cells into small nests (arrows), fewer keratin pearls, and more pleomorphic nuclei than in grade 1. HE. Bar = 50 μm.
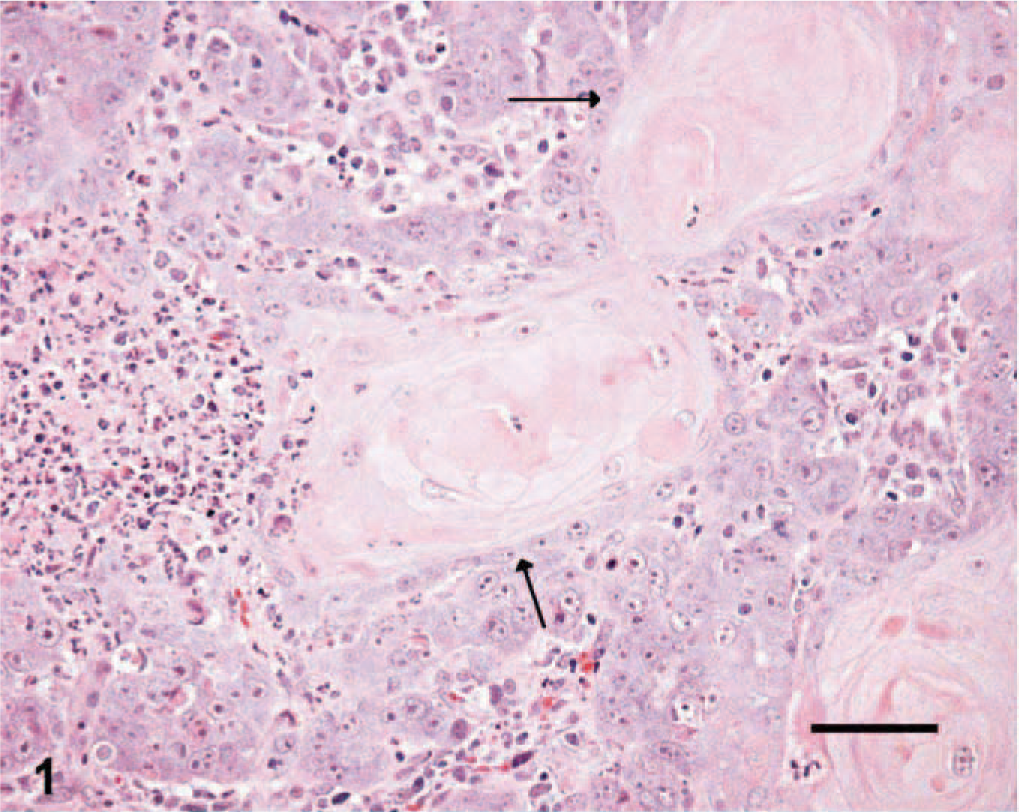
Skin; canine squamous cell carcinoma, grade 1. Note the prominent keratin pearl formation (arrows). HE. Bar = 250 μm.
Immunohistochemical staining for VEGF, VEGFR-2, von Willebrand's factor, and Ki-67
An avidin-biotin-complex (ABC) immunohistochemical staining method 17 was used on formalin-fixed paraffin-embedded tissues. From each paraffin block, consecutive 5-μm tissue sections were cut, mounted, and dried on glass slides. Tissues were deparaffinized in Citrosolv (Fisher Scientific, Ottawa, Ontario, Canada), followed by dehydration in graded alcohol solutions. Endogenous peroxidase activity was blocked using 3% hydrogen peroxide in absolute methanol for 12 minutes at room temperature. Antigen retrieval was achieved by steaming the tissues in a vegetable steamer for 30 minutes using 0.01 M sodium citrate solution at a pH of 6.8. Nonspecific protein binding was saturated using 4% horse serum (Invitrogen, Burlington, Ontario, Canada) in phosphate buffer saline (Invitrogen) for 20 minutes at 42°C, except when staining for von Willebrand's factor (vWF), where 4% goat serum was used. Primary antibodies consisted of the following: monoclonal mouse anti-human VEGF121, 1/20 (NeoMarkers, Fremont, CA); monoclonal mouse anti-human VEGFR-2, 1/1,000 (Santa Cruz Biotechnology, Santa Cruz, CA); polyclonal rabbit anti-human vWF, 1/2000 (DakoCytomation, Mississauga, Ontario, Canada); and prediluted monoclonal mouse anti-human Ki-67 (Zymed Laboratories Inc., San Francisco, CA). The diluted primary antibodies were applied to tissue sections, and the slides incubated 14–18 hours at 4°C. A biotinylated polyclonal secondary horse anti-mouse antiserum (Vector Laboratories, Burlington, Ontario, Canada) was applied to tissues for VEGF, VEGFR-2, and Ki-67 staining at a dilution of 1/400 for 20 minutes at 42°C. A 1/400 dilution of biotinylated polyclonal sheep anti-rabbit secondary antiserum was used to stain tissues for vWF. ABC reagent prepared according to the manufacturer's directions (Vectastain Elite ABC kit, Vector Laboratories) was applied for 35 minutes at 42°C. Color development was achieved by applying a 3-3′-diaminobenzidine-tetrahydrochloride reagent (Sigma, Oakville, Ontario, Canada) for 5 minutes. Tissues were counterstained with hematoxylin (Sigma), rehydrated in graded alcohol, and mounted with coverslips.
When staining tissues for VEGF and VEGFR-2, we also stained serial sections of tissues with omission of the primary antibody and with an isotype-matched irrelevant antibody (NeoMarkers). Tissue from a canine splenic hemangiosarcoma was used as a positive control for VEGF. Tissue from a normal canine lymph node was used a positive control for Ki-67. Normal vessels in the surrounding tissue of each tumor were used as a positive control for vWF.
Terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate end-labeling assay for apoptosis
Apoptosis was detected using the ApopTag Peroxidase in Situ detection kit (Chemicon International Inc., Temecula, CA). Briefly, 5-μm tissue sections were deparaffinized and rehydrated as described above. Protein digestion was achieved using 20 μg/ml proteinase K solution (Invitrogen/VWR, Saskatoon, Saskatchewan, Canada) for 15 minutes at room temperature. Endogenous peroxidase activity was blocked using 3.0% hydrogen peroxide. An equilibration buffer supplied with the kit was applied for 15 seconds, followed by working-strength terminal deoxynucleotidyl transferase enzyme for 1 hour at 37°C. The slides were covered with an antidigoxigenin conjugate and incubated in a humidified chamber for 30 minutes at room temperature. Color development and counterstaining were as described above for immunohistochemical stains.
Immunohistochemical scoring for VEGF and VEGFR-2
In each of the SCC sections, the percentage of positive tumor cells and the darkness of the brown staining were estimated. A score ranging from 0 to 4 was assigned to each tumor. SCCs were given a score of 0 if they showed no staining, a score of 1 if less than 50% of cells were positive with pale staining, a score of 2 if less than 50% of cells were positive with dark staining, a score of 3 if more than 50% of cells were positive with pale staining, and finally a score of 4 if more than 50% of cells were positive with dark staining. TCPs were given a score of 0 or 1 if they were negative or positive, respectively. All cases were evaluated independently by 2 of the authors (ANA and BAK). In instances of discordant scores, a score was established by consensus.
Microvessel density, Ki-67, and apoptosis evaluation
iMVD was evaluated with antiserum detecting vWF (Fig. 4), using the “hot-spot method.” 18 Slides were examined at low power (40×) to identify 10 areas with high vessel density followed by re-examination of each area at high power (400×) and counting of all positively staining structures, irrespective of whether a lumen was identified. Continuous vessels were counted as 1 vessel. The average of the 10 fields was determined and was expressed as count per high-power field in each tumor.
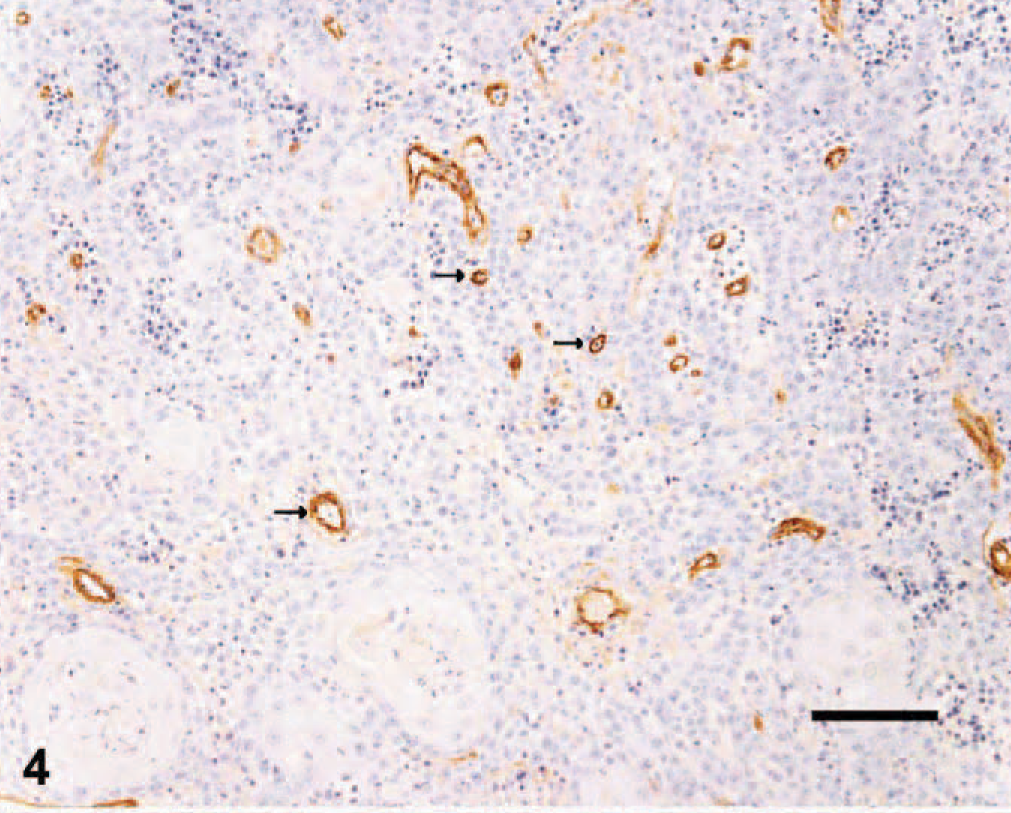
Skin; canine squamous cell carcinoma. Immunohistochemical staining of endothelial cells for von Willebrand factor antigen. Note the density of labeled vessels (arrows). ABC diaminobenzidine method, hematoxylin counterstain, Bar = 50 μm.
A similar method was used to evaluate sections stained with Ki-67 monoclonal antibody to demonstrate dividing tumor cells. The tissue sections were first examined at low power (40×) to identify 5 areas with high numbers of positive cells. Within each area, a high-power field (400×) was selected and 200 cells were counted. A total of 1,000 cells were counted in 5 high-power fields. The average number of positive cells in the 5 fields represented the PI. The AI was similarly determined by counting 200 cells in 5 selected areas and averaging numbers of positive cells using the terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate end-labeling (TUNEL) method assay in five 400× fields.
Statistical analysis
Statistical analysis was performed using Statistix 7 for Windows 2000 (Analytical Software, Tallahassee, FL). The strength of the association between the 6 variables evaluated (histologic grade, VEGF expression, VEGFR-2 expression, iMVD, PI, and AI) was assessed by the Spearman rank correlation coefficient. Correlation coefficients were interpreted as follows: 0 ≥ r, no correlation; 0 < r < 0.1, trivial correlation; 0.1 ≤ r < 0.3, slight correlation; 0.3 ≤ r < 0.5, moderate correlation; 0.5 ≤ r < 0.7, substantial or large correlation; and r ≥ 0.7, very large correlation. 4
The Kruskal-Wallis test was used to test for differences in median iMVD, AI, and PI among VEGF expression, VEGFR-2 expression, or histologic grade group of SCCs. This test was also used to evaluate the differences in VEGF or VEGFR-2 expression levels among the different histologic grades of SCCs.
The Wilcoxon rank sum test was used to compare median iMVD, PI, and AI between SCCs and TCPs. Finally, the Wilcoxon rank sum test was also used in TCPs to compare the median iMVD, PI, and AI between VEGF- or VEGFR-2-positive and negative tumor tissues. A P value calculated from Kruskal-Wallis and Wilcoxon rank sum tests less than .05 was considered significant.
Results
SCCs
Of the 19 SCCs, 8 (42.1%) were assigned a histologic grade of 1; 8 (42.1%), grade 2; and 3 (15.8%), grade 3. The results for each of the parameters evaluated are shown in Table 1. There was a substantial correlation between histologic grade and PI (r = 0.51); a moderate correlation between VEGF and histologic grade (r = 0.43), VEGFR-2 and histologic grade (r = 0.47), VEGF and PI (r = 0.47), and VEGFR-2 and PI (r = 0.47); and a slight correlation between VEGF and VEGFR-2 (r = 0.20) (Spearman rank correlation test). There were no significant differences in median iMVD, PI, or AI among the different histologic grades or among the different expression levels of VEGF or VEGFR-2 (Kruskal-Wallis test).
Squamous cell carcinomas, VEGF, VEGFR-2, iMVD, PI, AI, and histologic grade.∗
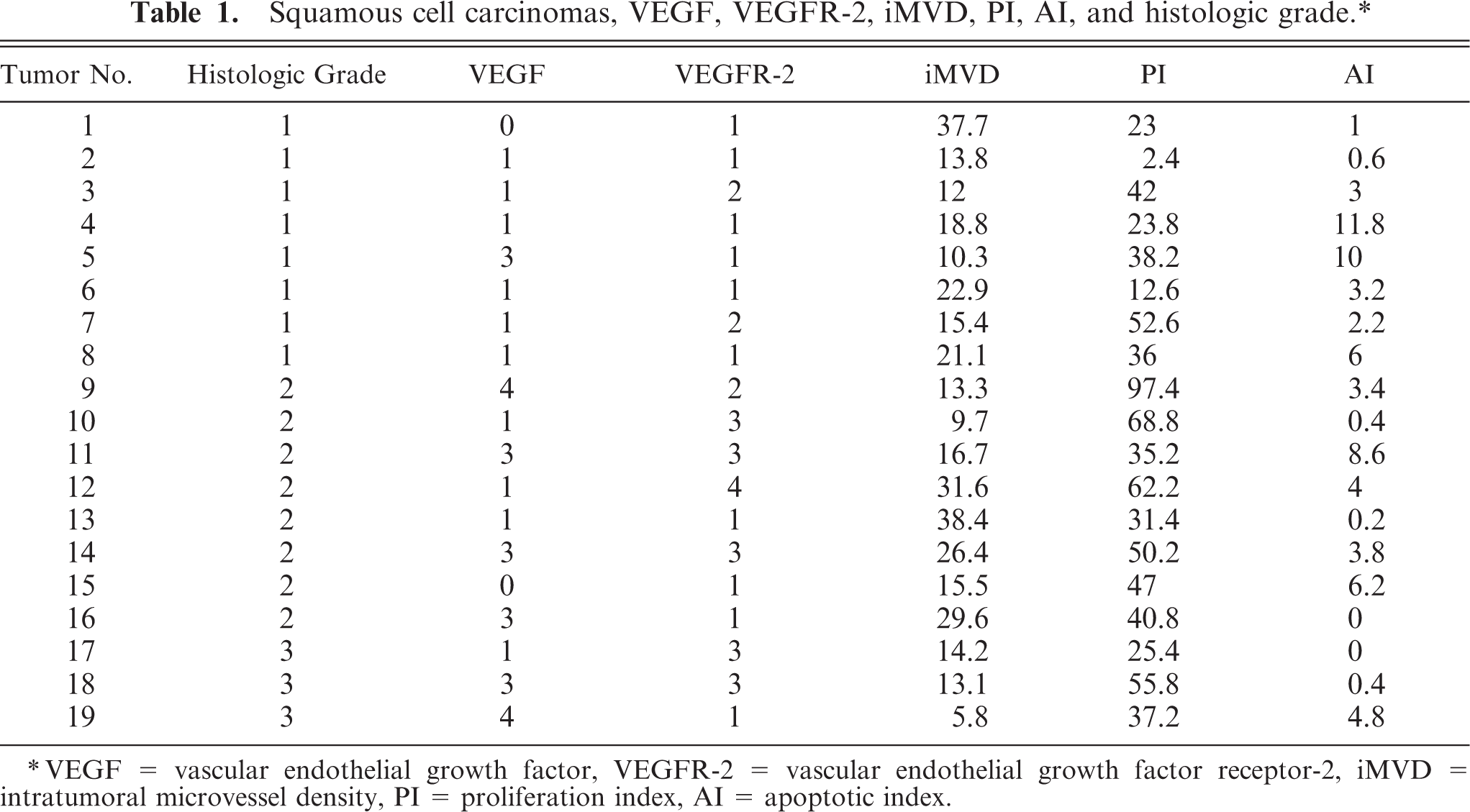
∗VEGF = vascular endothelial growth factor, VEGFR-2 = vascular endothelial growth factor receptor-2, iMVD = intratumoral microvessel density, PI = proliferation index, AI = apoptotic index.
TCPs
The results of testing canine TCPs are shown in Table 2. There was a substantial correlation between VEGF expression and PI (r
Trichoepitheliomas: VEGF, VEGFR-2, iMVD, PI, and AI.∗
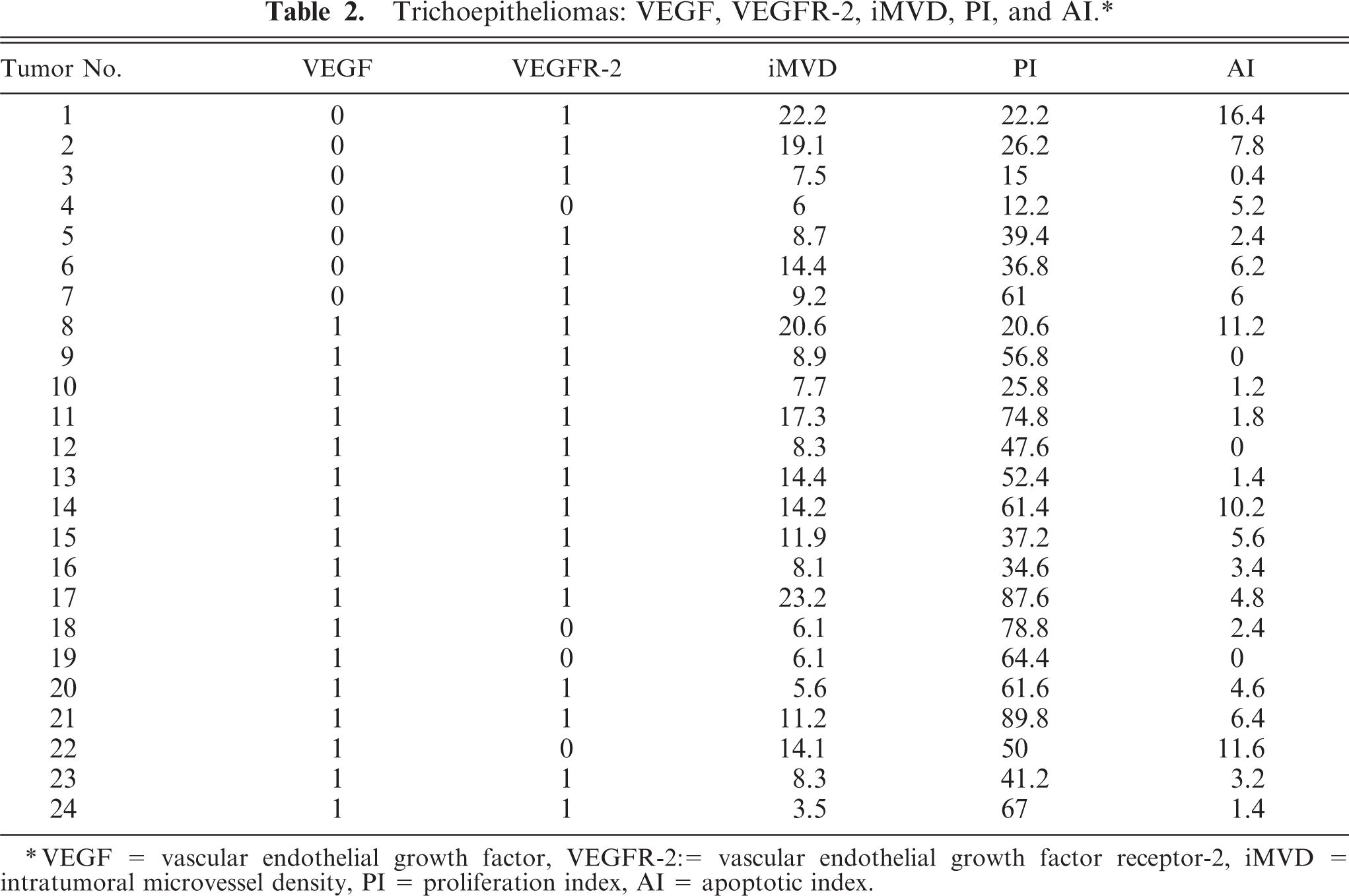
∗VEGF = vascular endothelial growth factor, VEGFR-2: = vascular endothelial growth factor receptor-2, iMVD = intratumoral microvessel density, PI = proliferation index, AI = apoptotic index.
Comparison between SCCs and TCPs
VEGF expression (Fig. 5) was detected in 17/19 (89.4%) SCCs compared with 17/24 (70.8%) TCPs. VEGFR-2 expression (Fig. 6) was detected in all of the 19 SCCs (100%) compared with 20/24 (83.3%) TCPs. Results of iMVD, PI, and AI comparisons between the SCCs and TCPs are summarized in Table 3. Median iMVD was significantly higher in SCCs than in TCPs.
Median iMVD, PI, and AI: comparison between SCCs and TCPs.∗
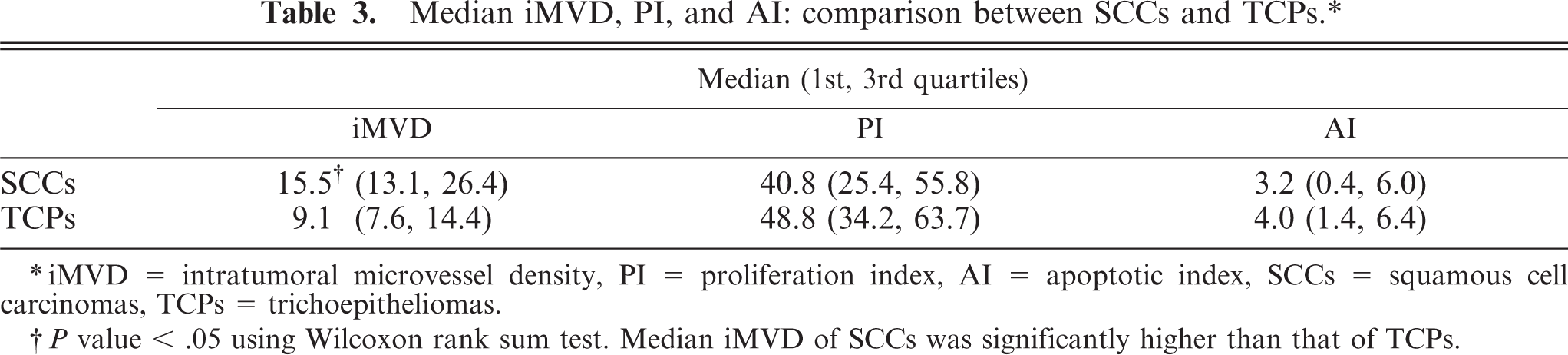
∗iMVD = intratumoral microvessel density, PI = proliferation index, AI = apoptotic index, SCCs = squamous cell carcinomas, TCPs = trichoepitheliomas.
†P value < .05 using Wilcoxon rank sum test. Median iMVD of SCCs was significantly higher than that of TCPs.
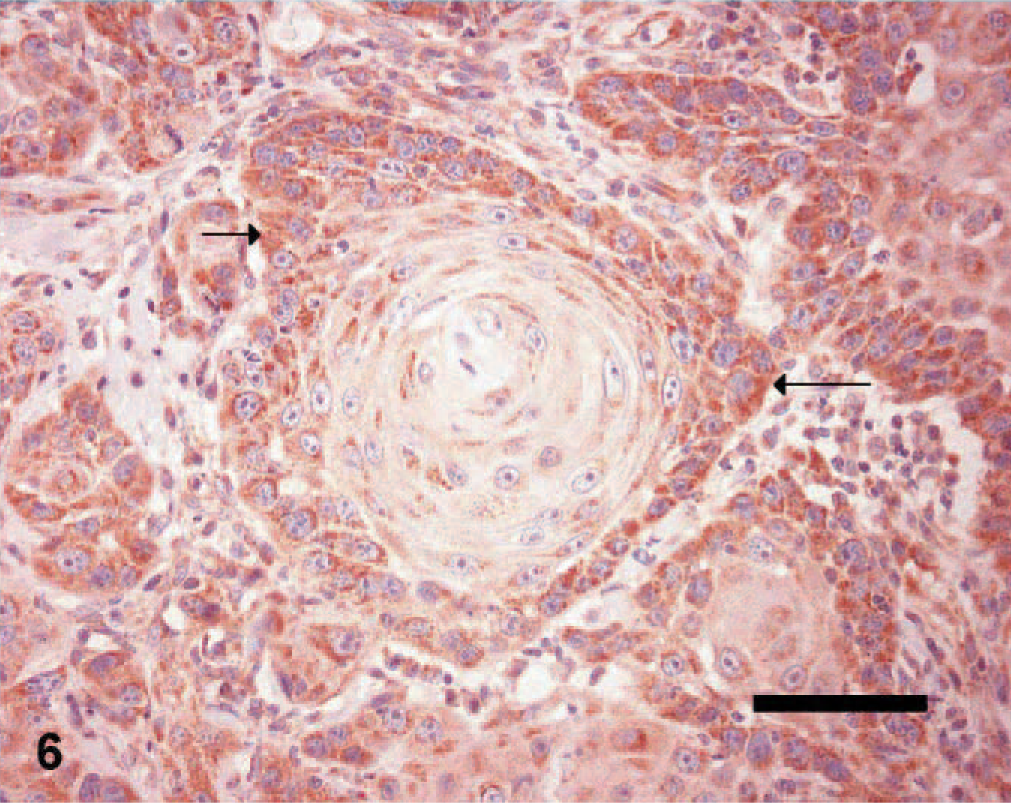
Skin; canine squamous cell carcinoma, grade 2 (same tumor as shown in Fig. 5). Immunohistochemical staining of tumor cells for VEGFR-2 antigen. Note the granular staining in the cytoplasm of tumor cells (arrows). ABC diaminobenzidine method, hematoxylin counterstain, Bar = 50 μm.
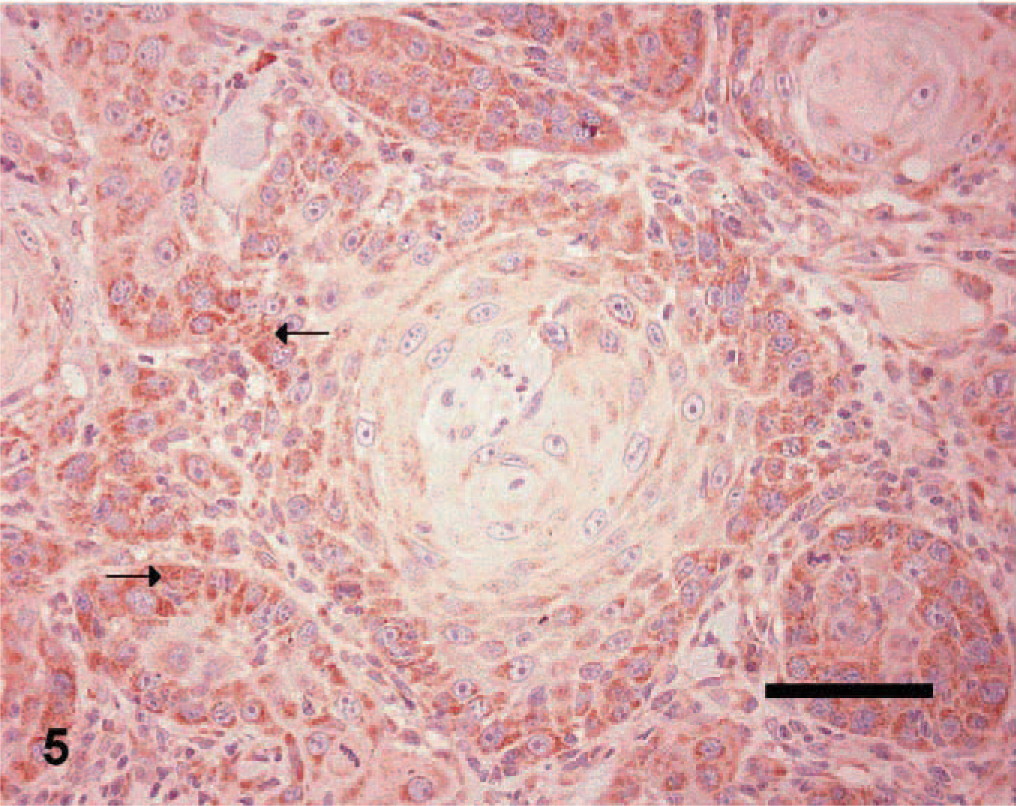
Skin; canine squamous cell carcinoma, grade 2. Immunohistochemical staining of tumor cells for VEGF antigen. Note the granular staining in the cytoplasm of tumor cells (arrows). ABC diaminobenzidine method, hematoxylin counterstain, Bar = 50 μm.
Discussion
The present study demonstrated for the first time the expression of VEGFR-2 in canine SCCs, although the protein has been detected in canine melanomas 32 and mammary tumors. 33 In the present study, VEGF was detected on 89% of canine SCCs, while in a previous report, 100% of canine SCCs were VEGF-positive. 26 The difference in percentages could be due to differences in the methodology used to detect VEGF antigen, or there may be an absence of VEGF expression in a low number of canine SCCs. There was a moderate correlation between the histologic grade of SCC and both VEGF and VEGFR-2 expression; however, in agreement with the previous study, 26 the present study failed to demonstrate differences in VEGF expression that related to histologic grade. The absence of differences in VEGF expression (and, in the present study, VEGFR-2) among the different histologic grades could be due to the small sample sizes in both studies.
Ki-67, a nuclear-associated protein expressed in proliferating cells, has been evaluated in many canine tumors. 16, 25, 28, 30, 31, 37, 38, 44 Ki-67 expression is a useful prognostic indicator in canine mast cell and mammary tumors. 30, 38 In canine lung cancer, adenosquamous and squamous cell carcinomas differed from the other histologic types in having significantly higher proliferation indices. 16 In the present study, there was a correlation between VEGF and VEGFR-2 expression and PI in SCCs and between VEGF expression and PI in TCPs. These findings suggest that VEGF may play a role in stimulating tumor cell division. Such a role for VEGF has been recently demonstrated in human breast cancer, where treatment of a breast cancer cell line with exogenous VEGF resulted in increased cancer cell proliferation and increased expression of BCL-2, an antiapoptotic protein. 24 The lack of a significant difference in median PI that related to the level of VEGF or VEGFR-2 expression in canine SCCs could be due to the small sample size. TCPs had a higher PI than SCCs, although the difference was not statistically significant. Although the difference in the AI is not statistically significant between the 2 tumor types, the trend points in the right direction to counteract the higher PI noted in TCPs. The association of VEGF expression and iMVD in SCCs is uncertain. Some studies report that iMVD correlates with VEGF expression in human esophageal SCCs, 22 while others 1, 41 failed to find such a correlation. In the present study, no correlation was found between iMVD and either VEGF expression or histologic grade. This is in contrast to other studies of canine SCC 27 that reported a significant difference in iMVD among histologic grades of tumors. The disparity with the current study could be due to the differences in iMVD evaluation methods. The present study used expression of vWF to detect endothelial cells, while the previous report utilized expression of CD31; the latter may be a superior marker for endothelial cells in canine tissues. 11
SCCs had higher iMVD than TCPs. The higher iMVD in malignant tumors may reflect their ability to proliferate and metastasize by promoting angiogenesis. The inflammatory neoangiogenesis associated with the presence of free keratin in the dermis in SCCs could also be responsible for the high iMVD. The finding of a significantly higher median iMVD in TCPs expressing VEGFR-2, but not VEGF, may indicate that VEGFR-2 plays a role in vascular proliferation through a pathway that is independent of VEGF. Other members of the VEGF family (i.e., VEGF-C) can stimulate VEGFR-2 and may contribute to angiogenesis. 3 The expression of VEGF and VEGFR-2 in TCPs was high compared with the 15% (3/20) reported in basal cell tumors. 26
The detection of VEGF and VEGFR-2 in canine SCCs and TCPs and the correlation between the expression of these molecules and PI suggest an autocrine function of VEGF in these tumor cells. Autocrine function of VEGF has been documented in human breast and pancreatic cancer. 42, 43 In pancreatic cancer, VEGF treatment resulted in phosphorylation of the mitogen-activated protein kinase and growth of VEGFR-2 expressing pancreatic adenocarcinoma cell lines. 42 The AI did not correlate with the studied parameters of angiogenesis.
In conclusion, VEGF and VEGFR-2 expression was detected in neoplastic cells of TCPs and SCCs. The expression of both molecules was associated with increased PI, which suggests that they may stimulate tumor cell growth through an autocrine pathway. Since most SCCs were found to express VEGF and VEGFR-2, these tumors may respond to therapy targeting these molecules.
Footnotes
Acknowledgements
We thank Ms. B. Trask and Dr. C. Waldner for their technical and statistical assistance, respectively. This study was funded by a grant from the Western College of Veterinary Medicine Companion Animal Health Fund, Saskatoon, Saskatchewan, Canada.
