Abstract
Melanoma is a devastating disease frequently encountered within both veterinary and human medicine. Molecular changes linked with neoplastic transformation of melanocytes include mutations in genes that encode proteins intrinsic to the regulatory pathways of two tumor suppressor proteins (retinoblastoma protein and p53), proto-oncogene mutation to oncogenes, altered expression of epithelial cadherin and CD44 adhesion molecules, and upregulation of angiogenic factors and other growth factors. Histologic evaluation of the primary mass is the most common means of diagnosis, with cytology used more frequently to document metastasis. Melanoma's highly variable histologic and cytologic patterns can make diagnosis by either method problematic. Adherent epithelioid morphology, including signet ring forms, and nonadherent round and spindle forms are recognized, with pigmentation an inconsistent finding. The site of the tumor, the thickness of the primary tumor or depth of invasion, and the number of mitotic figures per high-power field or per millimeter are used histologically to predict biologic behavior, whereas site and degree of pleomorphism are typically used for cytologic preparations. Diagnosis of amelanotic melanoma can be aided by ancillary diagnostic techniques. Tumor cells are usually positive for vimentin, S100, neuron-specific enolase, and Melan-A, and negative for cytokeratin. Melan-A as a positive marker is not as sensitive as the others are but is likely more specific. Monoclonal antibodies to human melanosome-specific antigens 1 and 5 cross-react with canine antigens for a combined sensitivity rate of 83%. Mouse monoclonal antibody IBF9 specifically recognizes canine melanoma antigen and also has good sensitivity. Serologic markers, including cytokines, cell adhesion molecules, and melanoma-inhibitory activity, are being investigated as potential sentinels of melanoma. Currently, there is no single diagnostic technique capable of differentiating benign from malignant melanocytic neoplasms or predicting survival time.
Introduction
A diagnosis of melanoma in domestic animals typically carries a grave prognosis. It is generally detected at a late stage when excision is rarely curative and metastasis is often already detectable within regional lymph nodes (LN). In dogs it is a common diagnosis, accounting for 7% of all malignant tumors.50 It is the most common malignant neoplasm of the oral cavity and the second most frequent subungual neoplasm.148 In cats it is not as common but carries the same poor prognosis. In gray horses melanoma is so common as to be considered almost inevitable.150 In humans the cutaneous form of melanoma is expected to reach an incidence of 1 in 75 among individuals born in the year 2000.2 Although oral melanoma is not common in humans, accounting for only 1–2% of all melanomas, as in domestic animals, it is generally very advanced when detected and has only a 5% 5-year survival rate.203
The purpose of this review is to provide a reference for diagnosticians and a resource for scientists in the field of melanoma research. Regardless of whether the neoplasm is initiated by chemical carcinogens or ultraviolet (UV) light, melanomas in all species share a similar biology in that they frequently recur and are predisposed to metastasis to regional LN. Animal melanomas are common and provide a useful model for a deadly human disease.145
Terminology for this disease is not consistent in human and veterinary literature. To avoid confusion in terminology in this article and to seek a common ground for both groups of readers, we use the term “melanoma” for all malignant melanocytic tumors, whereas melanocytoma refers to benign forms.
Pathogenesis
Normal melanocytes are dendritic cells derived from neuroectodermal melanoblasts that have migrated during embryogenesis to the epidermis, dermis, and other sites. They are found within the basal layer of the epidermis interspersed between basal keratinocytes. They normally do not form attachments with or even touch each other but rather form adherent and regulatory junctions with five to eight neighboring keratinocytes via epithelial cadherin (E-cadherin) molecules.138 Melanin is not retained within the normal melanocyte. It is packaged in melanosomes and transferred through their dendritic processes to keratinocytes. Conversion of normal melanocytes, that are nonpigmented and isolated from other melanocytes, to pigmented and clustered neoplastic melanocytes is a multistep process, with initiation as the first event, to be followed by promotion, transformation, and metastasis.33,152,249
Virtually nothing is known about initiation of most animal melanomas, but initiation of as many as 65% of cutaneous melanomas in humans and in most of the melanomas diagnosed in Angora goats is thought to occur secondary to mutations generated by both UVA and UVB solar radiation.96,180,218 This conclusion is based on epidemiologic linkage of melanoma with sun exposure by latitude and skin type (fair skin, freckling, and inability to tan). Furthermore, patients with the genetic disorder xeroderma pigmentosum, who have a defect in the enzymes required to repair light-induced DNA mutations (pyrimidine dimers), have a 1,000- to 2,000-fold increase in risk of developing skin cancer, including melanomas, squamous cell carcinomas (SCCs), and basal cell carcinomas.244 The initiators for all other cutaneous and noncutaneous melanomas are not known. Breed and familial clustering in domestic animals suggest that genetic susceptibility, possibly resulting in a greater frequency of spontaneously mutated cells, may be critical to initiation of many of these tumors.48,86,89,99,100
In humans initiation within benign, pigmented precursor lesions contributes to a small percentage of melanoma cases.127 The most important category of precursor lesion is the dysplastic nevus.127 Multiple dysplastic nevi on an individual are associated with an increased risk of melanoma.195 Other precursor lesions in humans include the giant congenital melanocytic nevus,127 but only a single case of malignant transformation of a similar congenital lesion in a golden retriever has been reported.242 Conroy48 described two canine cases of melanoma that arose from junctional or dermal pigmented nevi (hamartomas), although in neither case was metastatic behavior observed. There is a single report of a primary melanoma in the skin of a dog that originated from a subcutaneous melanocytoma.166 Melanoma has been reported to arise from primary acquired melanosis (PAM) in a dog's cornea and conjunctiva.53 In humans approximately 46% of PAMs that demonstrate cellular atypia progress to melanoma.118 In general, however, with the exception of gray horses, malignant transformation of benign lesions is very uncommon in animals, and most melanomas are believed to arise de novo from melanocytes in the epidermis, dermis, ocular epithelium, and oral epithelium.48
The next step in carcinogenesis requires a promoting factor(s). Promoters, possibly acting in an epigenetic fashion by disrupting gap-junctional intercellular communication, stimulate proliferation of the mutated cell, allowing for amplification of the cell population, persistence of the mutation, and opportunities for additional mutations.233 Promoters can include chronic trauma, chemical exposure, burns, hormones, infections, drugs, and other causes for reactive hyperplasia.33,38,119,233 Perhaps mucosal melanoma arises as a result of chronic injury (either mechanical or inflammatory) that results in reactive hyperplasia of the epithelium, disruption of normal keratinocyte-melanocyte interactions, and amplification of cells initiated spontaneously or by unidentified environmental factors.
Genetically or environmentally initiated DNA instability facilitates subsequent neoplastic transformation. Normal suppressors of cell proliferation and activators of apoptosis, e.g., retinoblastoma protein (Rb) and p53, are eventually superceded by unregulated growth factors or growth factor receptors (or both), and inhibitors of apoptosis, e.g., bcl-2.160–162 For example, p53-triggered cell cycle arrest or apoptosis prevents maintenance of DNA mutations in any given cell line. Mutations in p53 (gene for a protein is italicized by convention) can prevent synthesis of p53 or result in abnormal p53 that cannot gain entry to the nucleus, resulting in perpetuation of a DNA mutation. Human melanomas with p53 expression are common, 20–40% among all cases, and may have a different pathogenesis compared with those without p53.251 A canine case of multicentric melanocytoma was demonstrated to lack p53.196 Conversely, overexpression of normal p53 was detected in metastatic melanomas in two horses, suggesting to those authors that p53 is not involved in equine melanoma tumorigenesis.202
Most melanoma cases in humans lack detectable p53 mutations. Mutations in INK4a, INK4b, and Waf-1 are more common.160,231 INK4a and INK4b encode proteins p16INK4a and p15INK4b that are cyclin-dependent kinase inhibitors (CDKI) regulated by Rb. Entry into mitosis is triggered by cyclin-dependent kinases; therefore, INK4a and INK4b mutations ultimately remove the ability to inhibit mitosis and allow uncontrolled proliferation. INK4a also encodes p19ARF, which is an activator of p53, via an alternative exon and reading frame. Waf-1 encodes the protein p21, another CDKI. Expression of Waf-1 is partially regulated by p53. The multicentric melanocytoma case described above lacked functional p21.196 Thus, activities of two important tumor suppressors, p53 and Rb, are essentially sabotaged in the majority of melanoma by mutations in genes that encode proteins intrinsic to these regulatory pathways.231
Expression of the apoptosis suppressor gene bcl-2 was detected in 16 of 18 human cutaneous melanomas examined by Morales-Ducret et al. 1995,162 but the bcl-2 gene product was also found in resting melanocytes in normal skin. On the other hand, atypical bcl-2 overexpression has been detected in human uveal melanoma.28 Suppression of apoptosis complements disruption of the Rb and p53 pathways to favor uncontrolled proliferation.
Proto-oncogene mutation to oncogenes also favors proliferation and tumor development. Oncogenes detected in in vitro and in vivo melanoma studies include c-myc, c-erb-B-2, c-yes, c-kit, and ras. 12,142,160
Autonomous growth is an additional requirement for neoplastic progression. The chief autocrine growth factors linked with melanoma development are basic fibroblast growth factor (bFGF), melanoma growth stimulatory activity or growth regulated proteins, interleukin (IL)-8, IL-10, IL-18, platelet-derived growth factor-A, and α-melanocyte stimulating hormone.115,134,151,245 Late-stage melanomas are also stimulated by factors such as IL-6 and tumor growth factor-β that are normally inhibitory to melanocyte proliferation.134
Neoplastic transformation, resulting from mutations as described above, occurs before metastasis. Metastasis itself is a multistep process starting with detachment from the primary mass, movement to and through endothelium, travel via blood or lymph (or both), adhesion and movement through endothelium, and finally attachment and proliferation within a secondary site. Neoplastic cells must downregulate and then upregulate various adhesion molecules, e.g., Ca2+-dependent adherent molecules (cadherins) and CD44. Many studies have demonstrated both in vitro and in vivo decreases in E-cadherin in melanoma cells, which is supportive of theories that diminished E-cadherin interactions with basal keratinocytes are critical to melanoma tumorigenesis.113,152 But inconsistent decreases in membranous E-cadherin, concurrent with dramatic decreases in membranous placental cadherin and it's associated gamma catenin in late-stage primary and metastatic melanoma tumors have been reported by others.208
The adhesion molecule CD44 most likely plays a role in the facilitation of melanoma metastasis. CD44 is a widely expressed transmembrane glycoprotein that functions in T-cell activation, T-cell extravasation, LN homing, hyaluronate degradation, cell aggregation and migration, angiogenesis, and hematopoiesis.91,219 CD44 interacts with hyaluronic acid to facilitate expansion of proliferating tissues. Reduced expression of standard CD44 and upregulation of CD44 variant number 5 (CD44v5) has been demonstrated to occur in melanoma metastatic to LN.205,215 Melanoma metastasis, but not primary growth, was inhibited by antibody to CD44 in SCID mice injected with the human melanoma cell line SmelanomaU-2.103
At both primary and metastatic locations, angiogenesis is critical to survival and growth of any neoplasm regardless of its derivation. Reports on microvascularity of melanoma and its relationship with tumor aggression are inconsistent but seem to favor the theory that angiogenesis is critical to the metastatic potential of melanoma and correlates with poor outcome.139,165,172 One recent study examined serum concentrations of four angiogenic factors for a possible correlation with tumor progression and survival.240 Serum levels of angiogenin, vascular endothelial growth factor (VEGF), bFGF, and IL-8 were all significantly elevated in melanoma patients compared with healthy controls; however, only elevated levels of VEGF, bFGF, and IL-8, and not angiogenin, had a positive correlation with poor survival and tumor burden.
During this entire process tumor cells must escape immune surveillance, be physically able to survive and move within the circulatory or lymphatic system (or both), and be able to proliferate within a stromal environment different from their origin. One current theory suggests that avoidance of the immune system is facilitated by tumor production of IL-10.115
Other changes required of a melanocyte to become malignant are the subject of considerable research.218 Evaluations of cultured melanoma lines, animal models, and clinical cases are helping in answering questions regarding both general tumor biology and melanoma pathogenesis.
Clinical Features
Canine
Melanoma is relatively common in dogs, accounting for 3% of all neoplasms and up to 7% of all malignant tumors.50 The most frequently affected sites are the oral cavity (56%), lip (23%), skin (11%), and digit (8%), with other sites, including the eye, accounting for only 2%.90
Melanocytic neoplasms that arise in the oral cavity are virtually always considered malignant26 and constitute the most common oral malignant neoplasm (Fig. 1).26,232 They predominantly arise in the gingiva175 and account for 33% of malignant neoplasms in this site,25,31,64,93,108 with fewer cases arising from the lingual, buccal, pharyngeal, tonsillar, and palatine epithelium. Oral melanomas grow rapidly, are invasive, and often recur after surgical resection. Presenting signs include dysphagia, halitosis, ptyalism, bleeding, and occasionally fracture of the mandible.86,190 Seventy to ninety percent metastasize,32,42,232 independent of location within the oral cavity.17 The most common metastatic sites are the regional LN, lung and viscera, including the heart (Figs. 2, 3).48,232

Oral melanoma. Canine. Nonpigmented, exophytic mass protruding from the hard palate. Formalin-fixed tissue.
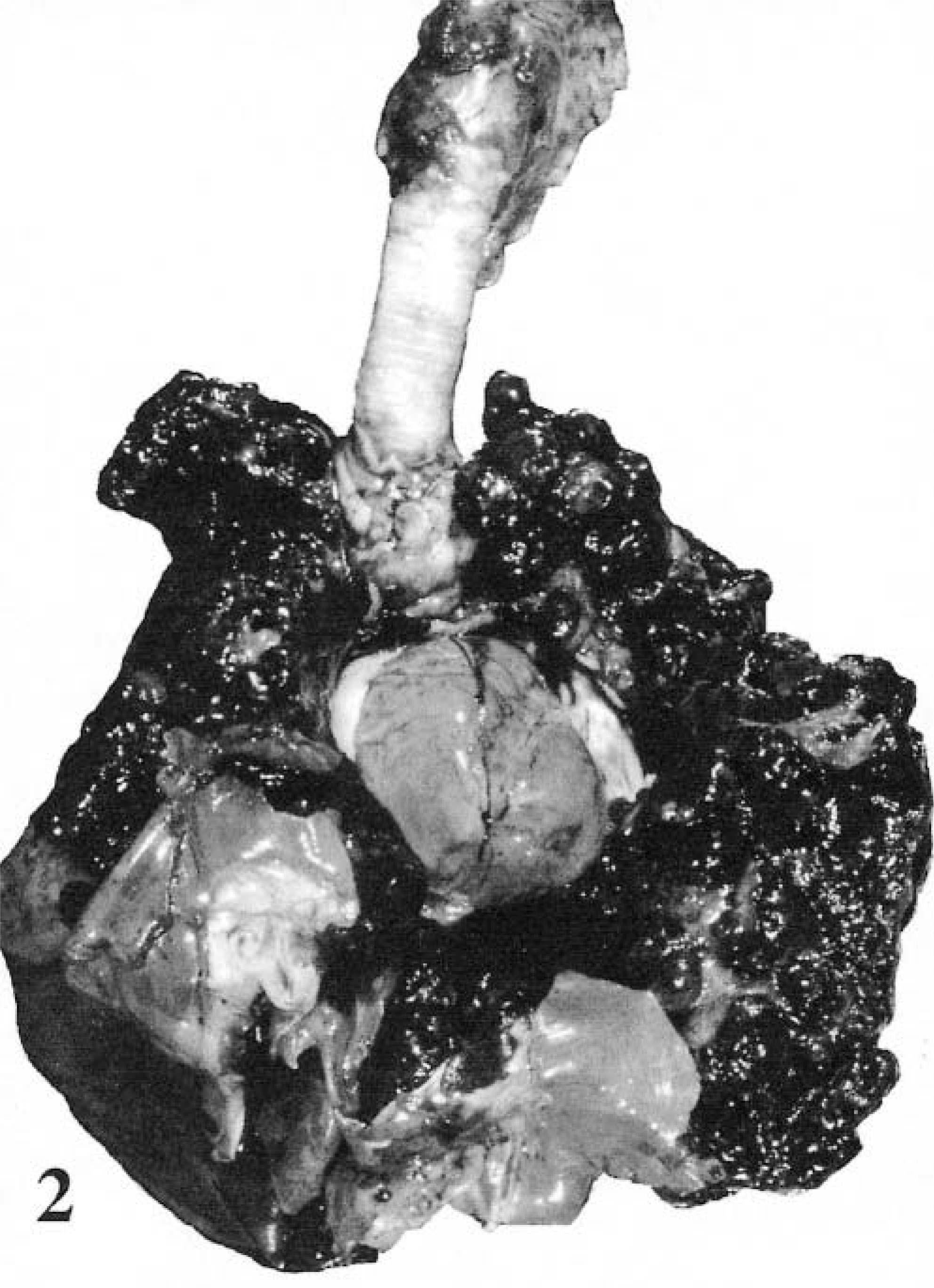
Metastatic melanoma. Canine. Multiple, black, raised, smooth, shiny nodules disseminated throughout the thoracic viscera, effacing the lungs, pericardium, and mediastinal soft tissue.
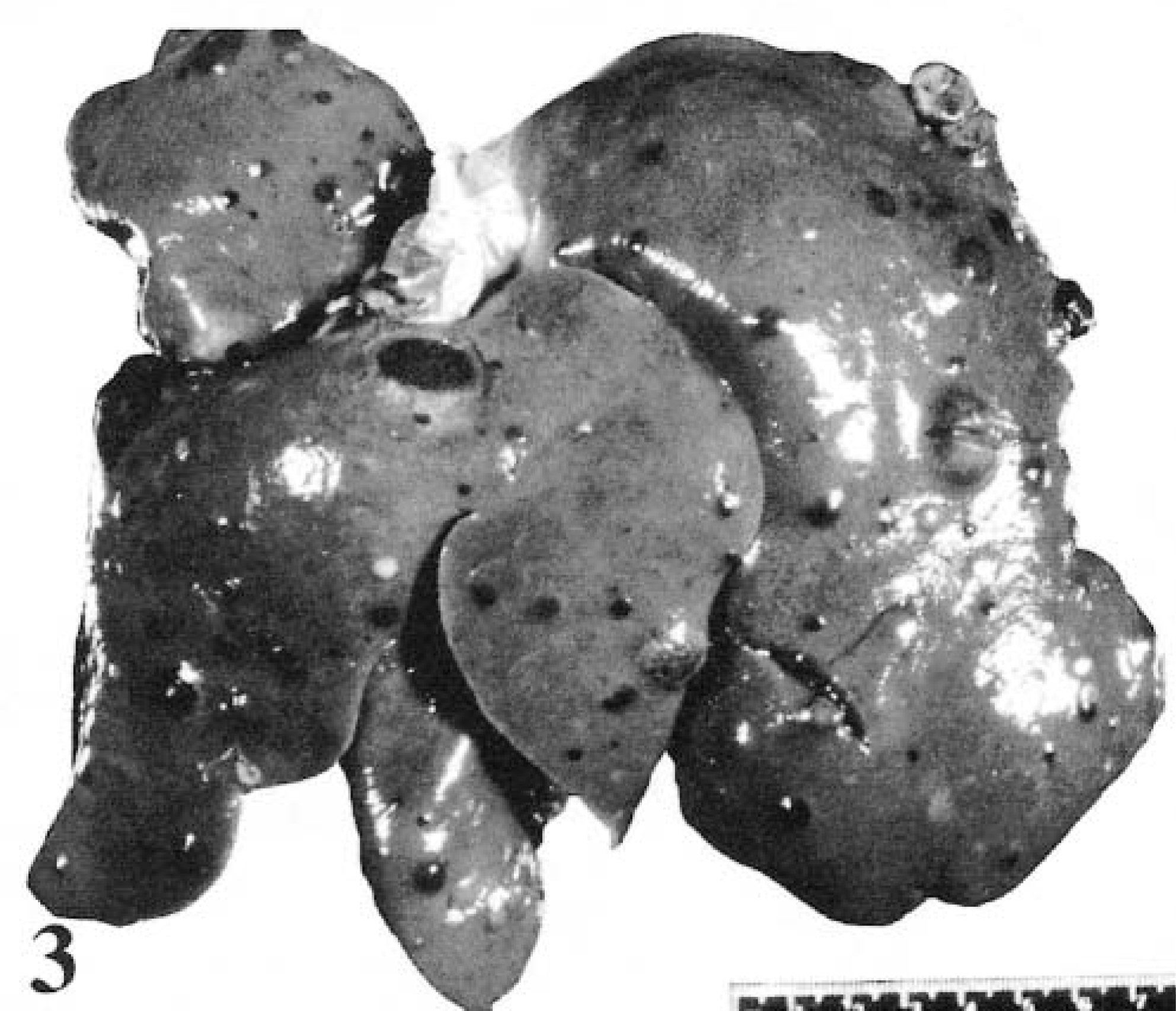
Metastatic melanoma. Canine. Multiple, black, raised, smooth, shiny nodules disseminated throughout the liver.
Collectively, melanocytic neoplasms account for 4–20% of all cutaneous neoplasms, but less than 5% of these tumors are malignant.90,187 Malignant tumors are found most frequently on the head, ventral abdomen, and scrotum.7,25,69,86 Melanomas commonly arise not only at mucocutaneous junctions, especially the lips, but also in the prepuce, vulva, anus, nose, and eyelids.
Subungual melanoma is the second most common digital neoplasm after SCC.148 Digits affected by subungual melanoma often have deformed nails, growth of a mass from the nailbed, or paronychia (Fig. 4). Subungual melanoma can result in destruction of phalangeal bone(s)7 but less commonly (5%) than in cases of subungual SCC (80%).148 Marino et al.148 reported radiographic evidence of metastasis in 58% of digital melanoma, either before or soon after surgery, and a median survival time of 12 months.

Digital melanoma. Canine. Subgross appearance of malignant melanocytes forming an exophytic protrusion, which also effaces and invades underlying phalangeal bone. HE. Bar = 3.5 mm.
Melanocytic tumors are the most common ocular neoplasms, and arise most frequently in the anterior uvea.47,60,66,164,206,211 In one study of 244 dogs with intraocular melanocytic tumors, the anterior uvea was the site for 83% of these tumors, whereas the limbic area and choroid accounted for 12 and 5%, respectively.85 Clinical signs include iris thickening and an irregular pupil, with blindness and ocular pain.65 Secondary symptoms range from keratitis, hyphema and corneal edema to uveitis, unilateral glaucoma, buphthalmos, and retinal separation.35,83,158,206 Most intraocular melanocytic tumors are benign. Reports of histologically documented malignancy range from 15 to 29.8%,85,158,253,254 whereas those with metastatic behavior account for approximately 4%.35,158 All the eight confirmed metastatic ocular melanomas reported between 1981 and 199235,81,158,206,254 involved the anterior uvea, with or without extension to the choroid. In the above mentioned study of 244 dogs,85 only one case of the 56 documented as malignant had metastasis confirmed by a postmortem examination; however, nine dogs had clinical signs suspicious for metastasis, e.g., lymphadenopathy or evidence of pulmonary or abdominal metastatic disease at the time of death (or both). This same study also determined that animals with malignant tumors had significantly shorter survival times after enucleation compared with both the melanocytoma group and control group (dogs with enucleation for other reasons). There was no significant difference in survival between the melanocytoma and control groups.
Melanoma incidence not only varies with site but also with breed. Cutaneous melanoma occurs more commonly in dogs with heavily pigmented skin, with the miniature schnauzer, standard schnauzer, and Scottish terrier at increased risk of developing neoplasm.48,86,90 In addition to the above breeds, Irish setters and golden retrievers are at increased risk of developing subungual melanoma. The Irish Setter, Chihuahua, Golden Retriever, and Cocker Spaniel are at increased risk of developing melanoma of the lip.90 German Shepherd Dogs and Boxers are more prone to developing oral melanoma.175,232 The reason for breed predilection is not entirely clear, but it may reflect an underlying genetic risk or increased pigmentation (or both) in certain breeds. The latter is in contrast to humans where there is no proof of any difference in the relative frequency of oral melanoma in African-Americans and Africans, compared with white cohorts.94 Furthermore, cutaneous melanoma is rare in Africans and other races with heavily pigmented skin.94 Some authors report a predisposition to oral melanoma in Japanese, Indians, and Hispanics, although others have contradicted this observation.24,203
Most dogs with melanoma are more than 10 years old with a range of 1–17 years.7,86,232 An increased frequency has been described in male dogs.64,90,232 This is similar to humans, where the male/female ratio of oral melanoma is 2–3:1.13,203
Equine
Up to 15% of all equine skin tumors are melanocytic.51,120 More than 90% of these tumors are benign at initial presentation, but approximately two-thirds are thought to progress to malignancy and are capable of widespread metastasis.92,120,124,130,198,212,213,224,241 The vast majority appear in gray or white horses, usually at or before the age of 5 years, corresponding to the time in their life when their coat color changes.120,137,150 An early theory of equine melanocytic neoplasia suggested that dermal and visceral melanocytic tumors are a manifestation of a storage disease, rather than malignant neoplasia, and occur as a result of the accumulation of melanin in melanophages during the depigmentation process.137,212 Melanocytic neoplasms also occur in non-gray or non-white horses, such as bays and chestnuts, and may be more likely to be malignant.120 Arab breeds seem to have a predilection for cutaneous melanoma, but statistics may be skewed by the fact that most are gray.120,136,198 A similar overrepresentation has been observed in Lippizaners and Percherons.67
It has been more difficult to attribute any breed or color predisposition to ocular melanomas, which are rare, although such a correlation has been demonstrated in some cases.14,67 The same is true for oral melanomas, which are uncommon, accounting for 17% of all oropharyngeal neoplasms in horses.64
Melanomas are unusual in horses less than 6 years old. They increase in prevalence with age, with 80% of gray horses developing malignant forms by the time they are 15 years old.92,150,241 There have been inconsistent reports regarding sex predilection. Sundberg et al. (1977)224 reported multiple dermal melanoma (otherwise called dermal melanomatosis) more frequently in males, whereas others suggest that there is no sex predilection.120,224,241
Most equine melanomas arise in the skin of the perineal region, the ventral surface of the tail, and external genitalia.8,92,150 More atypical sites have been reported, such as the coronary band,130 foot,110 vertebral region,124,198 and pelvic canal.124 Rarely, the thoracic cavity,167,225 nasal cavity,62 and eye14,56,67,149 have been involved. Clinical signs range from interference with bridle, bits, and saddle to more serious obstructive lesions in the urogenital or gastrointestinal tract, progressing to life-threatening pulmonary or visceral metastasis.120 Neurologic signs have been reported secondary to compression of the spinal cord by metastatic nodules.212 Other more unusual manifestations include Horner's syndrome and unilateral sweating.157,167
Equine metanocytic neoplasms have traditionally been grouped according to one of the three growth patterns. Some grow slowly over many years without metastasizing, whereas others grow slowly initially, with a subsequent increase in the rate of growth after a few years. A third subset grows rapidly and is malignant from the beginning.120,213 Valentine (1995) has suggested that there are actually at least four possible syndromes of equine melanoma, three of which have the potential for metastatic behavior.241 Two of these three categories, dermal melanoma and dermal melanomatosis, are histologically very similar and can only be classified based on clinical features. Dermal melanomas are usually solitary, discrete lesions that are surgically excisable, occurring in a wide age-range of gray horses. Dermal melanomatosis denotes the presence of many lesions, often coalescing and usually occurring in typical locations, such as the genital or perineal region, of gray horses older than 15 years. These are not surgically curable and are much more likely to metastasize internally.120,241 The third category refers to anaplastic melanoma in aged, non-gray horses. Although rare, it is the most aggressive, leading to death within months of diagnosis.241 The fourth category is the melanocytic nevi, which are benign, pigmented lesions predominantly occurring in horses less than 6 years old.120,241
Feline
Melanoma is uncommon in cats, accounting for less than 1% of all feline oral neoplasms90,155,220 and approximately 0.5% of feline skin tumors.32,64,86,182 The ocular and cutaneous forms are generally more common than melanoma of the oral cavity.57,64,73,182 The most common cutaneous sites are head (Fig. 5), tail, distal extremities,144 and lumbar area.57,89,90,243 The prognosis is generally poor because of recurrence and regional metastasis in up to half the cases.90,243 In a study of 23 cats with nonocular melanocytic tumors, approximately half were malignant, including all the three oral tumors included in the study.144 In another study, four of the six reported cases of oral melanocytic tumors were malignant, with metastasis to viscera in one case and local invasion in three.182
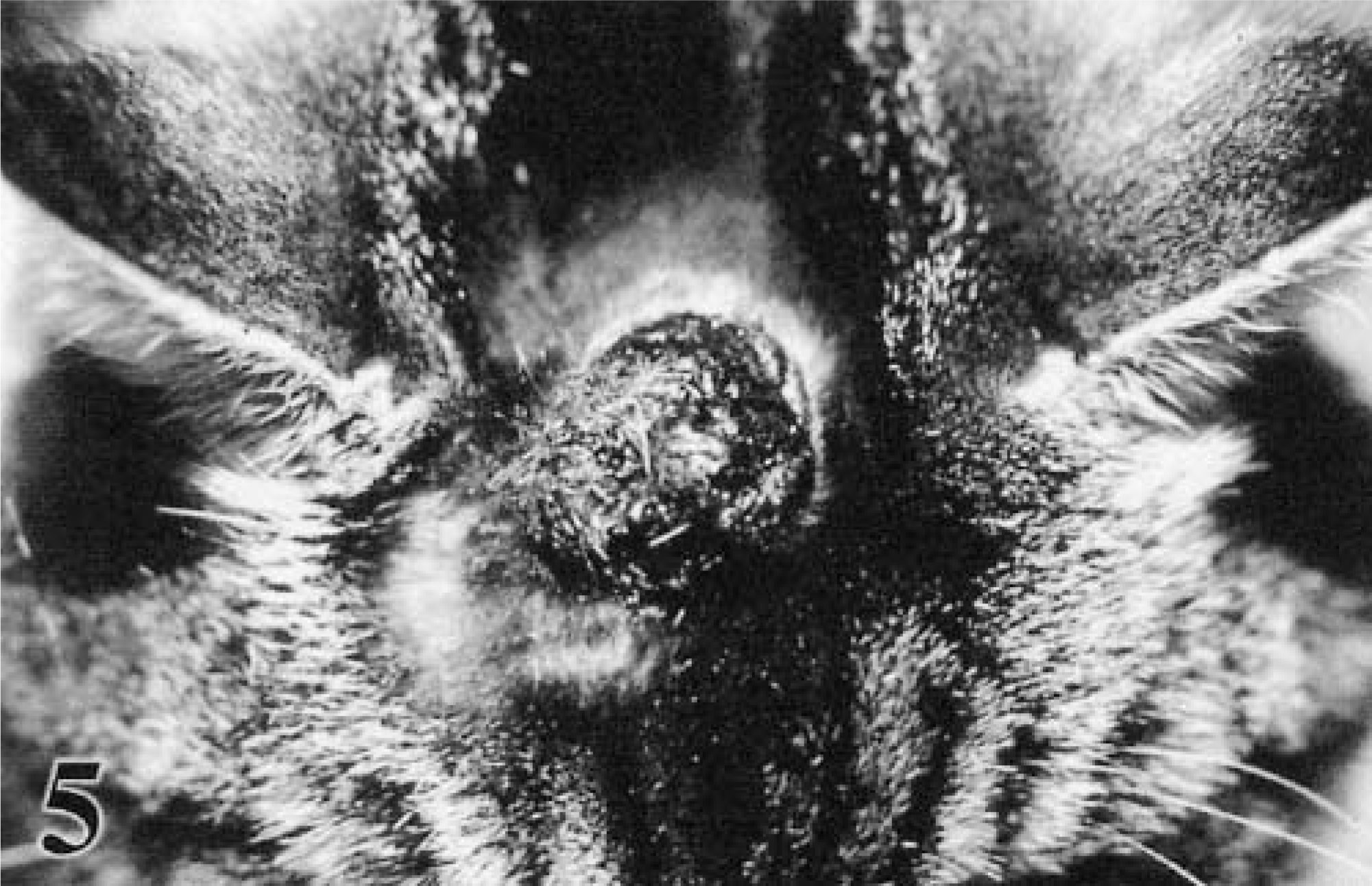
Melanoma. Skin of dorsal skull between ears; feline. Exophytic, crusted, discrete mass elevating the epidermis.
Ocular melanoma tends to occur in the anterior uveal tract, originating within the iris, with one study reporting over 60% of all feline ocular melanocytic tumors at this site.1,57,182 It typically presents as focal to diffuse iris hyperpigmentation rather than a discrete nodule or mass.65,123 Glaucoma is a common sequela.65 Melanoma originating in the limbus, conjunctiva, and nictitating membrane have also been reported, although they are very rare.22,49,199 Harris and Dubielzig (1999)107 have observed an atypical form of feline intraocular melanoma that arises multifocally in parts of the uveal tract other than the iris and tends to invade both the globe and the sclera. Ocular melanomas can be locally infiltrative or may metastasize widely.1,18,182 Some authors have documented a slow growth rate, with a long latent period between detection of primary ocular tumor and occurrence of metastatic disease.1,57,65 Others suggest that metastasis can occur in earlier stages, although this may depend on time of diagnosis and method of treatment.18,182 Melanomas with metastatic behavior have also been described in the palpebral area,182 in the nictitans,199 and, although normally benign, in the limbus.22,57 In a study of 34 cats with diffuse iris melanoma, the overall survival time after enucleation for diffuse iris melanoma was significantly shortened relative to healthy cats and cats with enucleation for other reasons.123 Cats with disease confined to the iris stroma and trabecular meshwork (early disease) had survival times comparable with the controls, but survival times shortened with invasion into the ciliary body (moderate disease) and were shorter still with advanced disease.
The age-range is comparable in several surveys, affecting cats from 2 to 18 years of age but peaking at 8 to 12 years.57,84,86,144,182,243 No sex or breed predilection has been demonstrated.
Other species
Melanocytic neoplasms occurs in other domestic species, including cattle, sheep, alpaca, and swine106,178,192,252 as well as spontaneously in laboratory animals and in birds.5,59 Of these species, swine are probably the most important for two reasons. Firstly, the Sinclair miniature and Duroc breeds have a genetic predisposition for melanomas.178,229 Secondly, the Sinclair miniature pig has served as a model for spontaneous cutaneous melanoma in humans.111,156,178,221 The melanocytic tumors can be congenital and are often multiple.178 In up to 90% of pigs, the tumors usually completely spontaneously regress, a feature that has made this such an attractive model, particularly for studying immunopathogenesis and its potential role in the therapy of melanoma.111,179 On the other hand, 10–15% of affected pigs succumb to metastatic disease with involvement of regional LN, lungs and liver, as well as other viscera.163,178 This model shares many features in common with human cutaneous melanomas, including spontaneous development of tumors, a wide spectrum of melanocytic tumors capable of malignant transformation, correlation between deeply invasive tumors and metastatic disease, and the pattern of metastatic spread and histopathology of tumor regression.111 In humans the regression is usually partial and perhaps a poor prognostic indicator,186 although complete remissions have been reported.27 In both human and swine tumors, regression has been associated with a high number of tumor-infiltrative lymphocytes. These lymphocytes have been identified as CD8+ cytotoxic T cells163 and display granulated lymphocyte morphology.194 These are the lymphocytes that react to Melan-A/MART (melanoma antigen recognized by T cells), a cytoplasmic marker that is weaker or not evident in the population of melanoma cells remaining after partial remission of human melanomas.
Uveitis and vitiligo are associated with the spontaneous regression that occurs in these pigs. Cataracts and a band keratopathy, characterized by deposition of calcium salts beneath the basal corneal epithelial cells, have been less frequently observed.74,135 Such observations have led to a profusion of studies aimed at elucidating the immunopathogenesis of regression. Peripheral blood lymphocytes in swine with regressing melanomas have significant lytic activity against cultured porcine melanocytes; lysis is age-dependent and does not occur in piglets less than 4 weeks old.194 Histologic changes of regression are biphasic, consisting of tumor infiltration by what appear to be pigment-laden macrophages between 4 and 8 weeks of age, followed by a predominantly lymphocytic response over the next few months.98 But the heavily pigmented cells may actually be terminally differentiated melanocytes and not monocyte-derived macrophages.97 The end result is the destruction of normal as well as malignant melanocytes.135 Circulating antibodies specific to pigment cells in pigs with regressing melanomas have been shown to increase with time and correlate with regression and vitiligo.54 More recently, melanoma regression has been associated with loss of telomerase activity, leading eventually to DNA fragmentation and cell death.181 This is an important area of ongoing research, and a complete review of the swine melanoma model is beyond the scope of this article. Other potential animal models for the human disease include the laboratory mouse, the opossum, and the Xiphophorus fish.63
In cattle, melanocytic tumors account for approximately 6% of all tumors, mostly arising in the skin and predominantly affecting cattle with red, gray, or black coats.154 Reports of malignancy are very uncommon, but metastases can occur.213 The majority of melanoma in cattle have been reported from the Indian subcontinent.168,192,193 In contrast, in sheep and goats cutaneous melanocytic neoplasms metastasize frequently.213 Angora goats are predisposed and have been proposed as a potential model of human melanoma because of the probable association with UV light.96,180
Reports in wildlife and exotic species are much less common. There is a single case report of a poorly pigmented cutaneous melanocytoma in a ferret.236 Melanocytic neoplasms are very rare in birds, with only single case reports in the literature.5,129
Gross Pathology
Melanoma can vary considerably in appearance, regardless of the site. Melanoma may be of any color, ranging from gray or brown to black, red, or even dark blue.62,102,153 Pigmentation is not a specific feature because other neoplastic or nonneoplastic lesions can be phenotypically similar. Melanoma vary in size, but most fall within the range of 1–3 cm.48,102 Cutaneous melanoma may be smooth domes, sessile nodules (Fig. 5), polypoid, plaquelike, or even lobulated masses;86,102,166 larger ones are often ulcerated. In the horse they are often flat and firm, may be single or multiple, and can coalesce, creating a cobblestone appearance.120 Alopecia and ulceration are variable features.
Lesions found at necropsy depend on how far the neoplasm has progressed and range from local primary tumor masses to widely disseminated metastases (Figs. 2, 3).57,86,198,254 Individual tumors vary from 1 mm to several centimeters diameter.212 Neoplastic masses can be compressive, although they are usually unencapsulated and infiltrative.187 They can also be poorly defined, effacing normal structures rather than forming discrete masses.110,130 Pigmentation is variable, but often the masses are black.
Regardless of site, melanomas can metastasize via the lymphatic or blood vessels, with regional LN being the usual first target.110 Any of the visceral organs can potentially be infiltrated, but the lungs are most commonly involved.17,21,92,130,158,182,198 Infrequently, extension and metastasis to serosal surfaces, such as pericardium, pleura and peritoneum, occur; associated effusions may be black.157 Melanomas have the potential to invade bones and cause bony lysis in areas such as digit, maxilla, mandible, vertebrae, and ribs.148,182,198
Histopathologic Variants
Histopathologic variants in animals
The term nevus, commonly used in describing pigmented melanocytic lesions of the epidermis and dermis in humans, is not used in veterinary dermatopathology.88 When evaluating melanocytic neoplasms they may be compound, denoting both an epidermal-epithelial and dermal-submucosal component to the neoplasm, or dermal and submucosal with no identifiable epidermal or epithelial component (Fig. 6). Junctional refers to the proliferation of neoplastic melanocytes at the interface between the epidermis and dermis or epithelium and submucosa.88 Pagetoid refers to the presence of either individual or small aggregates of neoplastic cells within the upper levels of the epidermis or epithelium90 (Fig. 7) and was originally used to describe the carcinoma cells (Paget cells) that infiltrate the epidermis overlying mammary ductal carcinomas in situ.52
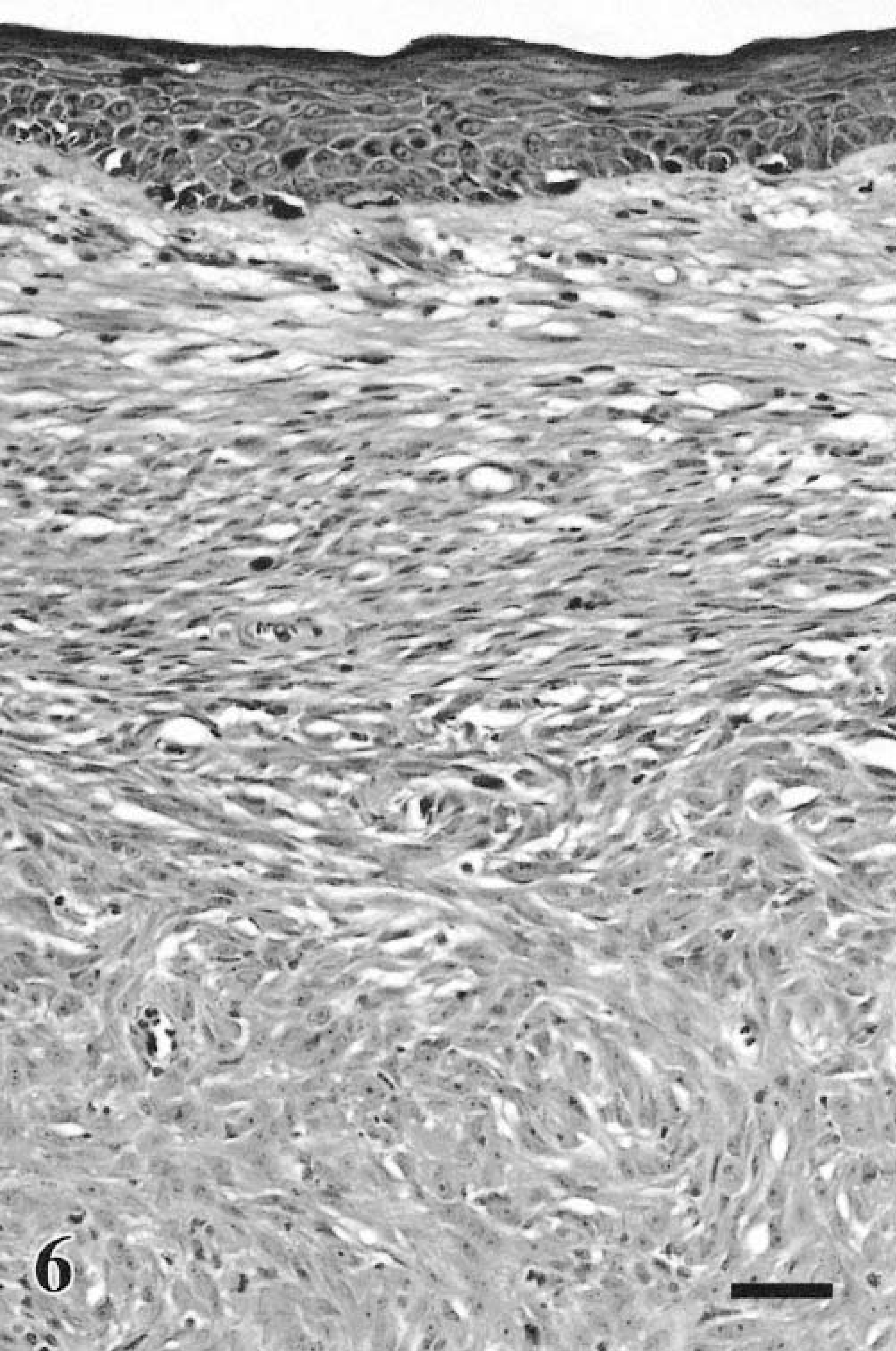
Melanoma. Skin; canine. The dermal neoplastic cells are fusiform and epithelioid. Melanocytic hyperplasia is evident in the epidermis, but it is nonneoplastic. HE. Bar = 40 μm.
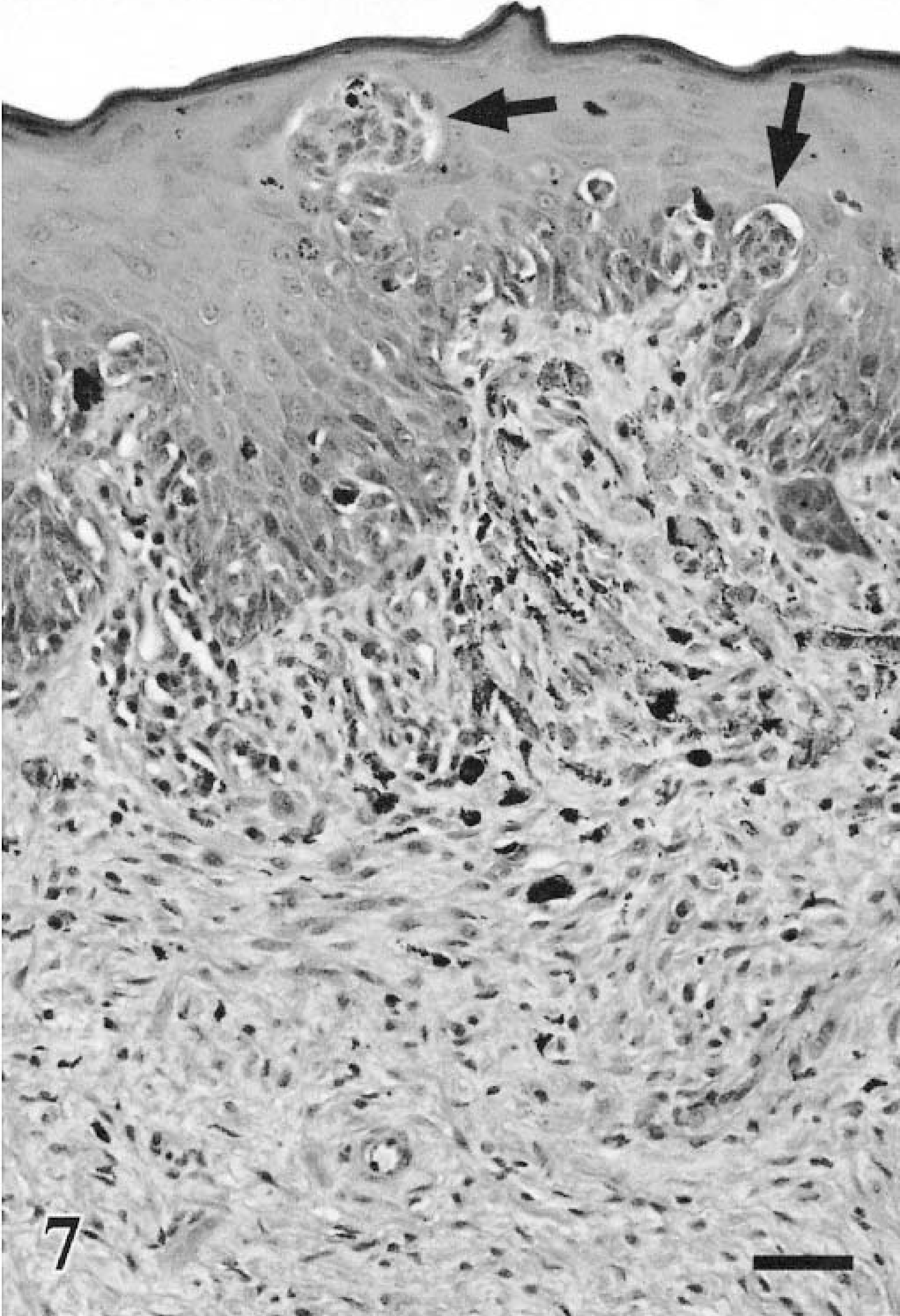
Melanoma. Oral; canine. Pagetoid infiltration of oral mucosa by individual cells and nests of cells (arrows) and neurotization of submucosal neoplasm. HE. Bar = 40 μm.
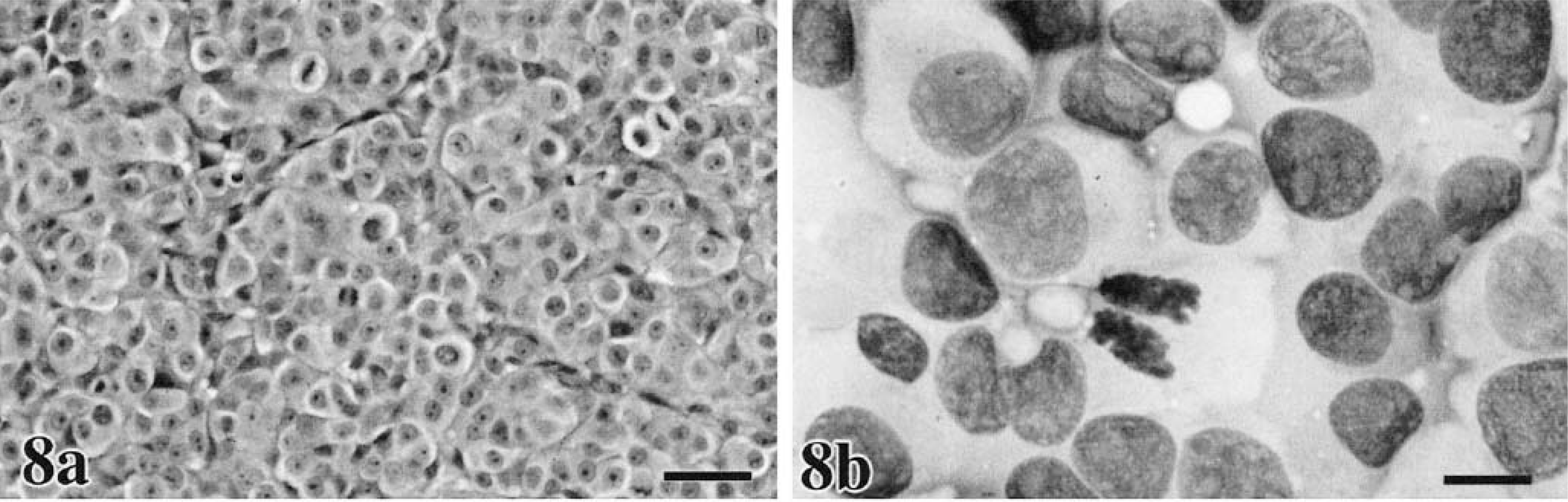
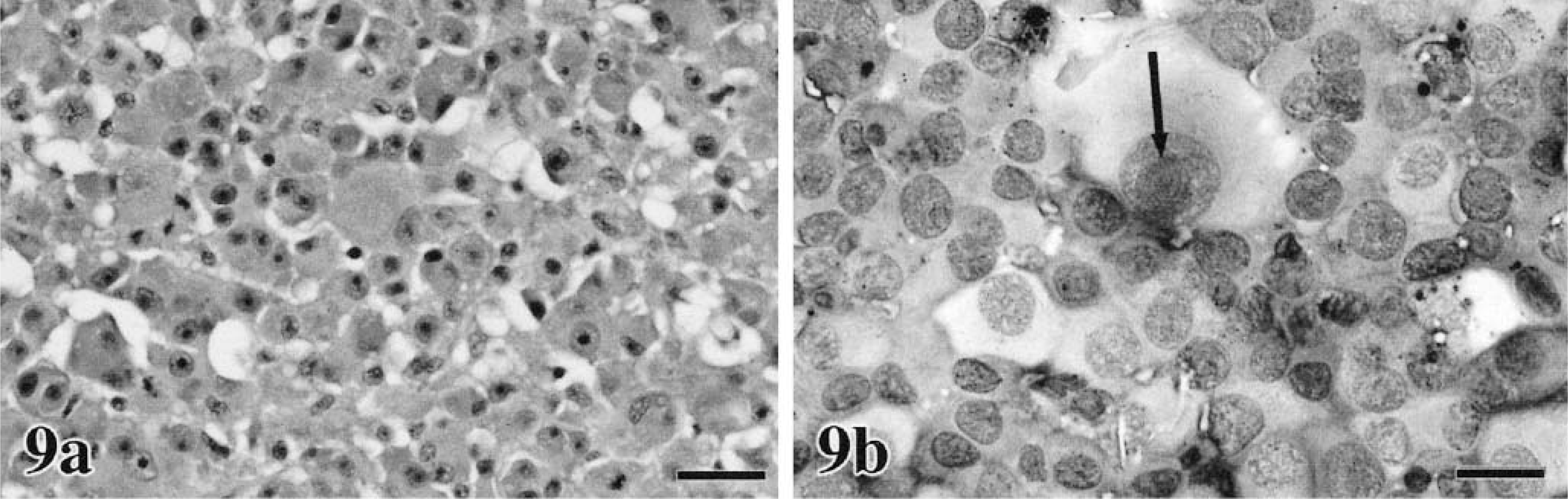
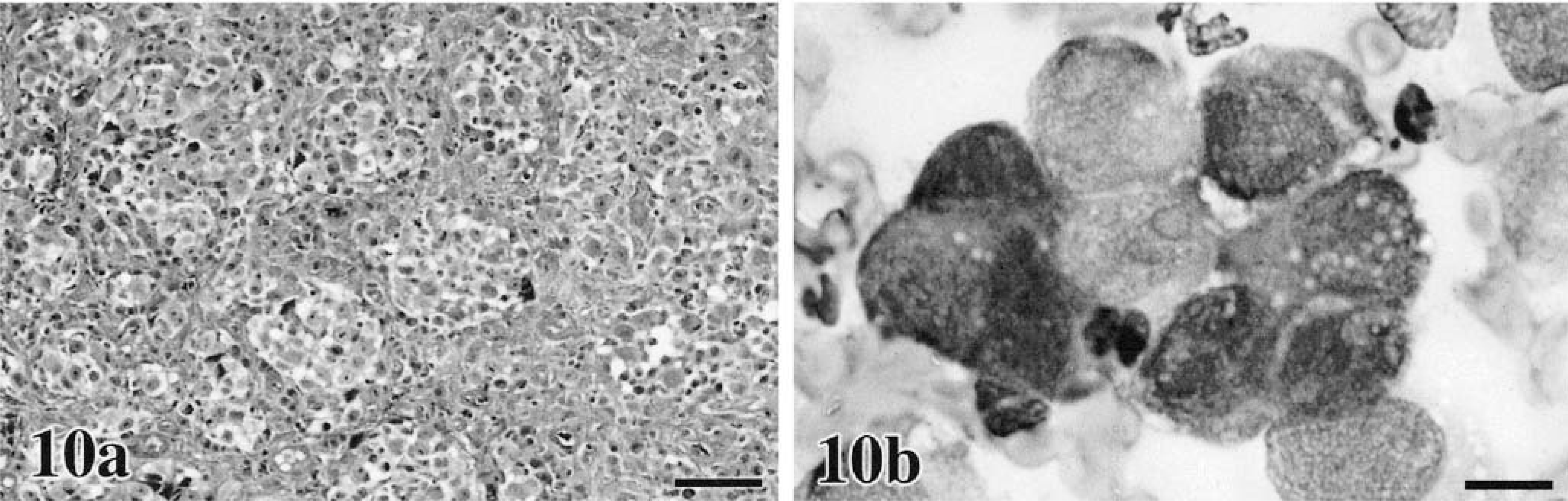
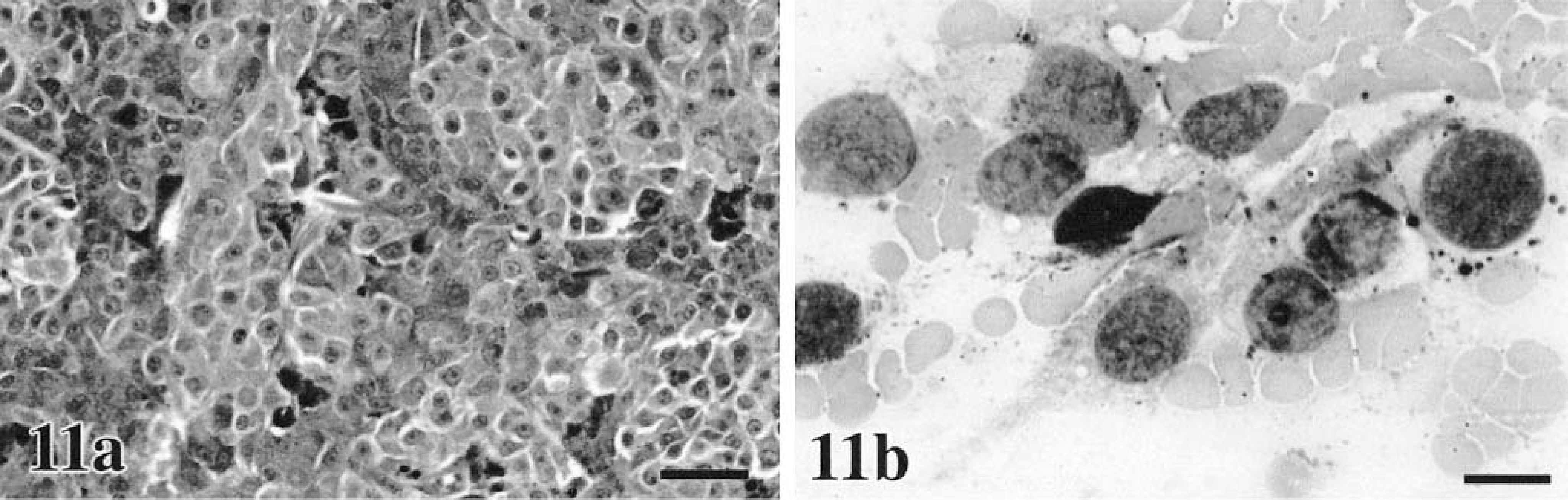
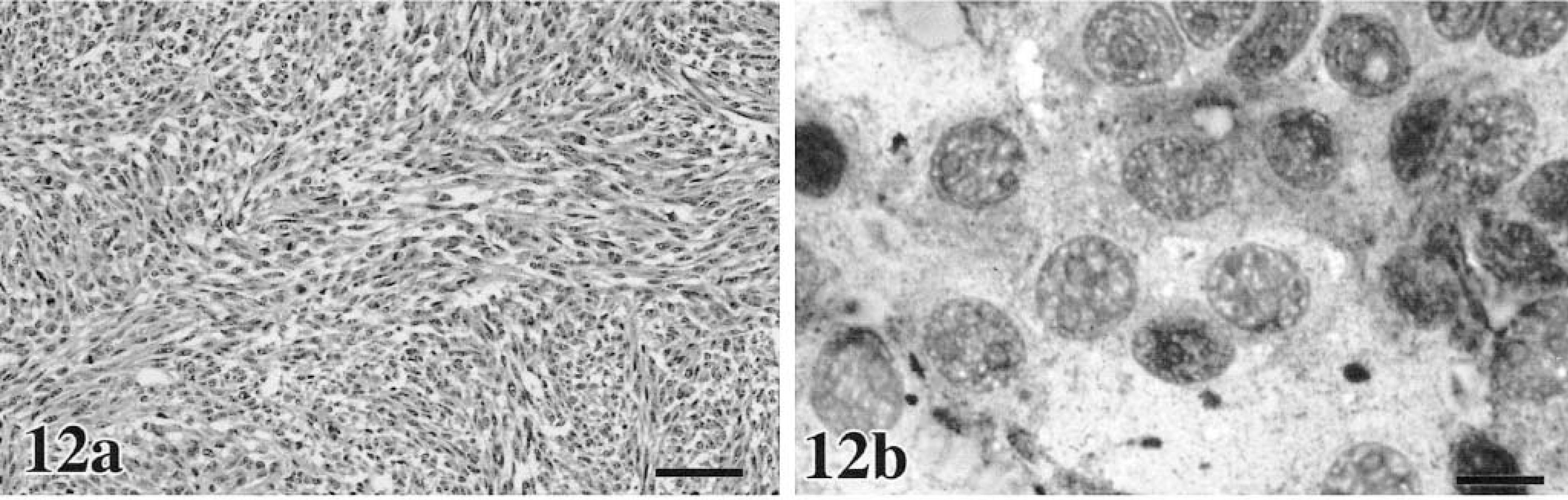
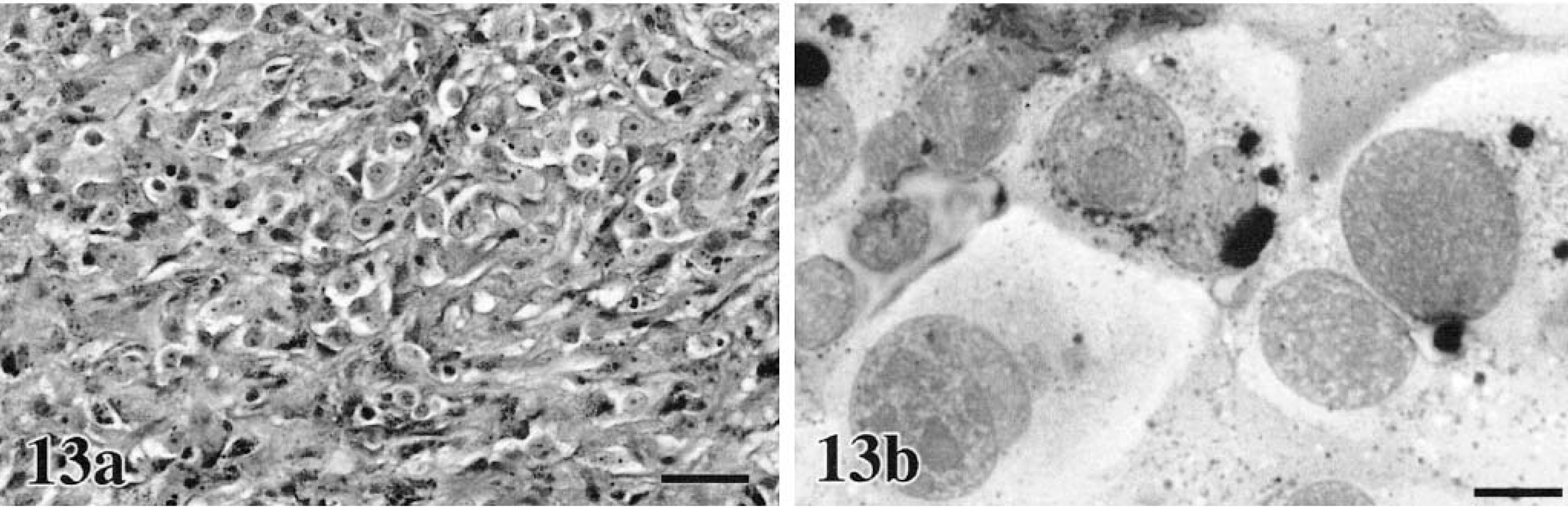
In our domestic species, cutaneous, oral, and ocular melanocytic neoplasms are quite variable in their histologic appearance and in the amount of melanin within the cell cytoplasm. Epithelioid tumors contain round cells, with discrete cell borders, abundant glassy cytoplasm, large nuclei, and prominent nucleoli (Figs. 8a, 9a, 10a, 11a).86 Cells may exhibit “pagetoid” behavior and occur either singly or in small clusters of two to three cells. This feature may be seen within the mucosa adjacent to the submucosal component of the neoplasm. Spindle cell tumors are arranged in streams and interweaving bundles (Fig. 12a) similar to fibrosarcoma or neurofibrosarcoma,102 but the nuclei tend to be larger, nucleoli more prominent, and Pagetoid behavior is not seen. The mixed type consists of both cell morphologies and patterns (Fig. 13a). A fourth type, the whorled or dendritic form occurs only in the skin86,88 and features spindle-shaped cells organized in tightly swirling streams, often with a fingerprint pattern. This last type is usually benign, whereas the others are more typically malignant.
Melanoma. Ear; canine.
Melanoma. Ear; feline.
Melanoma. Lip; canine.
Melanoma. Head; feline.
Metastatic (lungs) and primary (mouth) melanoma. Canine.
Melanoma. Oropharynx; canine.
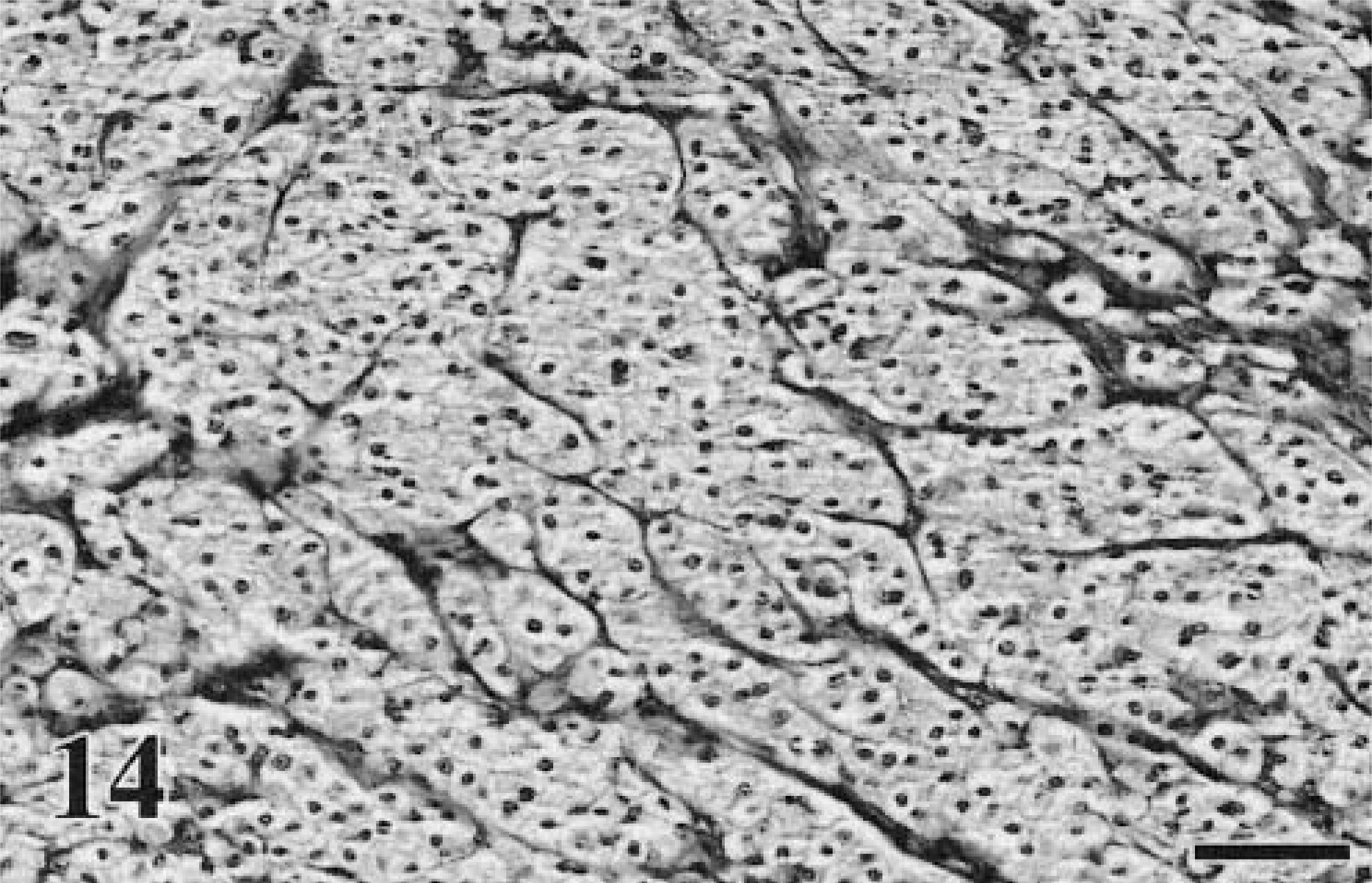
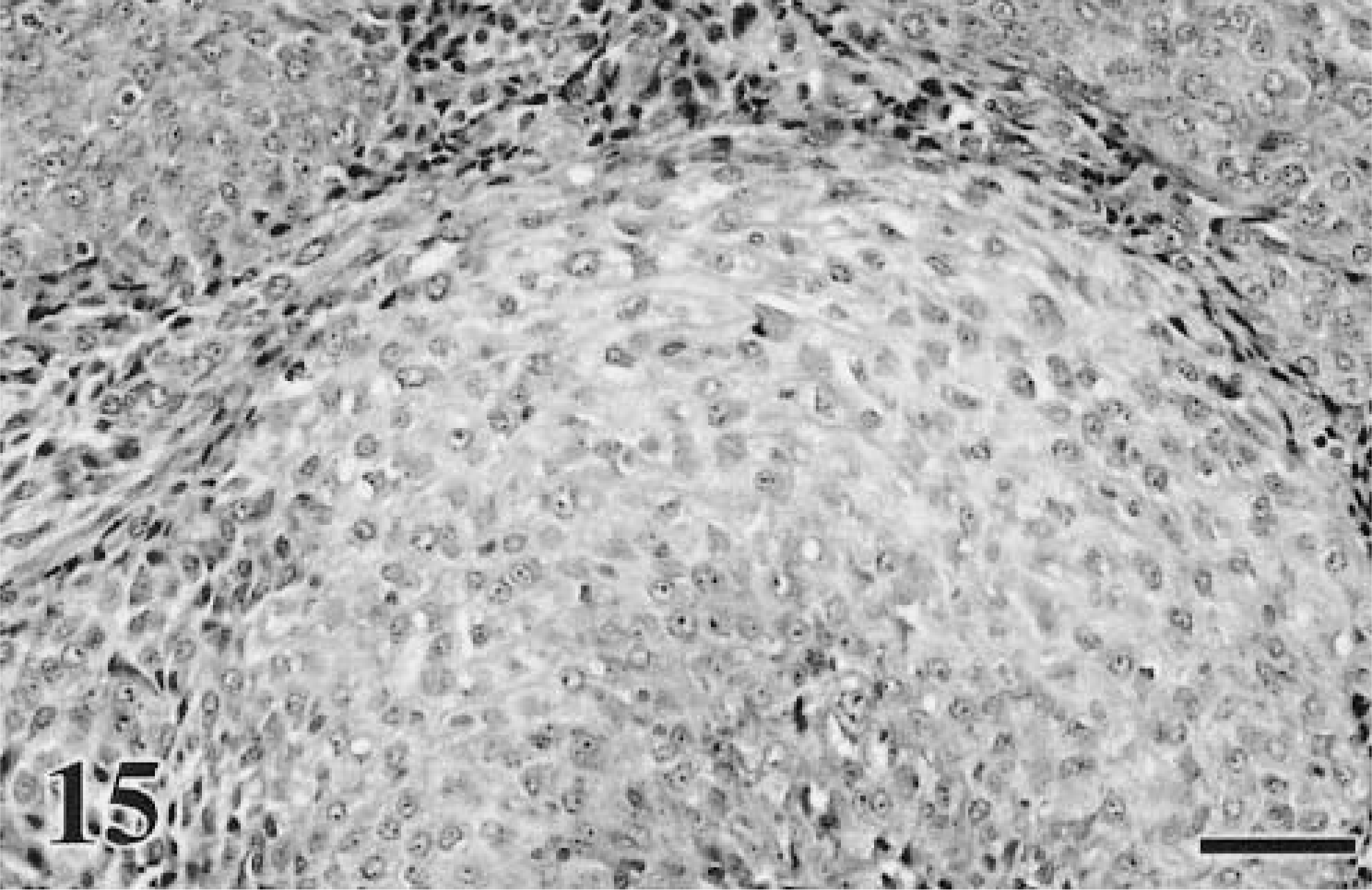
More unusual forms include the clear cell type (also referred to as the large pale or balloon cell type). This occurs in canine skin and oral cavity and feline skin.61,190,243 The neoplastic cells in this subtype have finely granular, palely eosinophilic, and slightly dusty brown cytoplasm. The nuclei are round, with variable anisokaryosis, prominent nucleoli, and marginated chromatin (Fig. 14).61,102 The signet-ring type, which has been documented in the oral cavity of cats, is also amelanotic and consists of pleomorphic cells with flattened, eccentric nuclei. Some cells contain large, pale, intracytoplasmic inclusion bodies, which lie adjacent to the compressed nuclei.243 Ramos-Vara et al.190 recently described an adenoid-papillary–form in canine oral cavity, previously only seen in humans. In this subtype, cells are arranged in acinarlike structures with central lumina.170 Osseous and chondroid metaplasia (Fig. 15) can also occur within melanoma arising at any site, but this is uncommon.42,88,90
Melanoma. Skin of foot; canine. A balloon cell variant. The cells have an abundant amount of pale eosinophilic but slightly dusty brown cytoplasm. There are fine fibrovascular trabeculae of connective tissue forming multiple small lobules. HE. Bar = 60 μm.
Melanoma. Oral cavity–maxilla; canine. There is a transition from cells that are round to more fusiform to cells with a basophilic cytoplasm, indicative of early chondroid differentiation.
Comparison with human melanoma
Generally, cutaneous melanoma in domestic animals does not share the same degree of diversity described in the medical literature.170 In veterinary pathology, because dysplastic nevi are not recognized as a specific entity, the pathologist is not required to differentiate between severely dysplastic nevi, melanoma in situ, and early invasive melanoma, all of which differ in their prognostic significance. Furthermore, most animal melanomas are advanced invasive masses when removed for histopathologic evaluation, corresponding to nodular melanoma in humans, whereas most melanomas in humans are the superficial spreading type, which are most often relatively small, thin tumors that are confined to the epidermis and dermis. Some direct histopathologic correlations can be made between human and canine melanoma in that both forms may be composed primarily of spindle cells, epithelioid or round cells, or a mixture of these cell types.70 There are similarities between oral melanoma in dogs and humans. As in dogs, the prognosis for oral melanoma in humans is very poor regardless of morphology,70 and no link has been found between survival rate and histologic type.72
Cutaneous melanomas in humans are most commonly classified according to their growth pattern variants. The most common types are superficial spreading melanoma, nodular melanoma, lentigo maligna melanoma, and acral lentiginous melanoma.36,222 This classification system is thought to represent subgroups with similar etiologies as well as biologic, clinical, and prognostic features,183,222 although the validity of this theory has been questioned.250 All four have two growth phases of variable duration—radial growth with epidermal involvement only and vertical growth with dermal invasion.222 Superficial spreading melanoma is the most common melanoma seen in Caucasians accounting for 70% of all melanomas.127 This tumor is typically found on the trunk and extremities, with no predilection for chronically sun-damaged skin, and is composed of predominantly large, atypical epithelioid melanocytes. Nodular melanoma is predominantly dermal, with only a minor intraepidermal component. It is generally heavily pigmented, predominantly epithelioid, often ulcerated, and rapidly progressive, with essentially no clinically discernable radial growth phase (RGP).127 It is the second most common melanoma, accounting for 15–30%,127 with tumors generally found in skin that has been chronically sun damaged, e.g., the head, neck, and trunk. Lentigo maligna melanoma originates from lentigo maligna, which is typically seen on sun-exposed skin of elderly patients. The precursor lesion, lentigo maligna, is the early RGP (in situ) and can be present for years before the vertical growth phase (VGP) occurs, at which time the lesion is considered a melanoma. Spindle-shaped cells predominate over epithelioid cells in the VGP for this melanoma. Acral lentiginous melanoma occurs on the hands and feet, including the digits and subungual regions. This is the most common type of melanoma seen in Asians, African-Americans, and Hispanics.222
The variables listed below are used as prognostic indicators in humans with cutaneous superficial spreading and nodular melanomas to estimate 5- and 10-year survival with a 95% confidence level and to stage disease.2,11,36,45,146 Physicians are advised to use these variables for tumor node metastasis (TNM) staging of cutaneous melanomas with other growth patterns, e.g., lentigo maligna melanoma and acral lentiginous melanoma but with the caveat that these other groups may have different etiologies and different prognoses. Variables numbered 1–7 are those used by the Melanoma Staging Committee of the American Joint Committee on Cancer as the basis for the currently accepted staging system for cutaneous melanomas in people11 and have been found to be independent predictors of survival. The remaining variables are not incorporated into the TNM system because they are difficult to objectively relate to survival curves, their status is controversial, or because they are not clearly independent of other factors, e.g., tumor thickness. Recording of these values for individual patients is recommended. It is interesting to note that morphologic atypia is not included in this list.
Modified Breslow29 thickness of primary tumor: measured from stratum granulosum to deepest portion of neoplasm; considered the most significant.146 Using the TNM staging protocol11 assignments are as follows: T1 = <1.0 mm; T2 = 1.01–2.0 mm; T3 = 2.01–4.0 mm; T4 = >4.0 mm. Ulceration: defined as microscopically detected absence of intact epidermis overlying a major portion of the primary melanoma that is not artifactual or secondary to trauma. Ulceration signifies locally advanced disease with higher risk of metastasis compared with nonulcerated tumors of like thickness. Level of invasion: under new guidelines, Clark's levels46 are now only used for thin melanomas (T1)11
Level I—confined to the epidermis (melanoma in situ) Level II—Extend from epidermis into papillary dermis, but papillary dermis not filled or expanded Level III—Expand and fill the papillary dermis Level IV—Infiltration of the reticular dermis Level V—Infiltration of the subcutaneous fat The number of metastatic LN and whether or not the metastasis is detected clinically or microscopically: although both indicate a poorer prognosis than no metastasis, macroscopic (clinical) detection is worse than microscopic detection (clinically occult). Intralymphatic invasion: either satellite metastasis around the primary or in-transit metastasis between the primary and the regional LN; both sites have an equally poor prognostic significance that is about equivalent to detection of metastasis in multiple regional LN. Site(s) of distant metastasis: melanomas metastatic to skin, subcutaneous tissue, and distant nodal sites have a better prognosis compared with lung metastasis, which is intermediate compared with other visceral sites, which have the poorest prognosis. Elevated lactate dehydrogenase (LDH): elevated LDH is associated with diminished survival; a high LDH must be confirmed by two elevated LDH values determined more than 24 hours apart and not because of other factors, e.g., hemolysis or liver disease. Number of metastatic sites: this variable was not incorporated into the TNM system because of the lack of standardization of diagnostic strategies used to search for metastatic sites. Site: a prognostic advantage is seen in patients with nondistal appendicular tumors, as opposed to axial (trunk, head, neck), subungual, palms, or soles,114,227 but this disappears when tumors are controlled for thickness, age, and sex.146
Mitotic count: number per millimeter2: high counts are indicators of a worse prognosis. Tumor infiltrating lymphocytes (TILs): brisk, nonbrisk, or absent; nonbrisk or lack of TILs is considered a high-risk attribute. Age: poorer prognosis if > 60 years. Growth phase: RGP versus VGP.218
Sex: there is a slight survival advantage for women. Regression: absent or present; regression being defined as evidence of loss of continuity of the lesion and fibrosis, which appears to correlate with high risk of metastasis, as opposed to actual regression of disease; surviving cells after regression represent a subpopulation that is more likely to migrate into lymphatics. The prognostic value of this trait is still uncertain.
Criteria of malignancy
Melanoma may be one of the few neoplasms in animals for which location is an important prognostic indicator in its own right. Melanocytic neoplasias involving the oral cavity, subungual region, and mucocutaneous junctions are considered malignant, regardless of any other single feature.7 Interestingly, this also seems to be the case with oral melanoma in humans. There is no evident relationship between histologic characteristics, including mitotic index and pigmentation, and survival rate.70,190 The prognosis for human melanoma at mucocutaneous junctions and mucosal surfaces is also grave.203
In animal skin and eye melanocytic neoplasms, the most reliable histologic feature for distinguishing malignant from benign is the mitotic index.25,26,35,65,158,254 In the World Health Organization's Histologic Classification of Epithelial and Melanocytic Skin Tumors of Domestic Animals,88 three or more mitotic figures per 10 high-power fields indicate malignancy. The identification of mitotic figures in sections must be undertaken with great care to ensure that only true mitoses are counted. Bleached sections should be used for pigmented neoplasms to avoid confusing mitoses with pyknotic nuclei and the small hyperchromatic nuclei of the spindle cells of the supporting stroma. The mitotic count will vary from area to area within the neoplasm, and the count should be performed in areas with the greatest concentration of mitotic figures. Where metastases have been confirmed, the mitotic rate in intraocular melanoma is usually greater than four per 10 high-power fields; less than two mitotic figures per 10 high-power fields is consistent with a melanocytoma.35,254 In cats the value of mitotic activity to prediction of outcome is less clear, particularly with regard to ocular melanoma.21,22,65 Some authors, however, report a link between high mitotic index and increased risk of metastatic disease.57,68
Neoplastic cell morphology is also a useful discriminating feature.25,69,88 Cytologic features of malignancy include the presence of a large nucleus, variation in nuclear size and shape, hyperchromasia, abnormal chromatin clumping, one or more nucleoli, and atypical mitotic figures.65,86 Additional features favoring malignancy are the presence of neoplastic cells, individually or in nests, within the upper layers of the epidermis (Fig. 7),65,86,88 although ulceration and necrosis may prevent evaluation of this feature. The presence or absence of junctional activity is not specific to melanoma and often occurs in melanocytomas.
The gold standard is, of course, lymphatic or vascular invasion.88 Pathologists are often asked to evaluate mandibular LN for evidence of metastatic disease in dogs with oral melanoma. These nodes may be pigmented because of metastatic melanoma or the accumulation of melanophages within the medullary sinuses. The latter occurs when melanophages are transported to the regional LN after inflammation of the oral mucosa, particularly in dogs with pigmented oral epithelium. The cytologic features described above, when used to evaluate bleached sections, usually suffice to distinguish melanophages from melanoma.
In cats the following histologic features have been proposed to be of significance in identifying cutaneous melanoma: nuclear atypia (neoplasms with more extensive atypia are more likely to be malignant), mitotic activity (a trend of greater malignancy with increased numbers of mitoses), and tumor cell type (with epithelioid more likely to be malignant).89 The relationship with cell type is not firmly established in the literature, in that two studies indicate a less precise relationship between cell type and malignant potential.144,155 Furthermore, histologic determination of malignancy does not consistently correlate with clinical behavior.144
The Callender system has been used historically to determine malignant potential in canine anterior uveal melanocytic neoplasms.158,254 This system was designed to predict the behavior of ocular melanoma in humans,37 which led to criticisms of its use in the veterinary literature for two main reasons. Firstly, it was designed for malignant tumors in humans, and most canine intraocular melanocytic tumors are benign.234,254 Secondly, the morphology of the canine neoplastic melanocytes does not match the cell descriptions in the Callender classification.35,158,254 Several authors favor a simpler system, dividing intraocular melanocytic tumors into benign and malignant using well-recognized cytologic features of malignancy as a basis for this classification.158,254 Despite these controversies, some useful morphologic features can be used to ascribe malignancy, especially with canine anterior uveal melanoma. A greater risk of metastasis tends to occur in mixed and epithelioid cell types.206 Melanocytic neoplasms containing narrow spindle-shaped cells with small nuclei and no mitotic figures, or plump cells with large amounts of melanin and small round nuclei, are more likely to be benign.81 This is in contrast to the cat where cellular pleomorphism, degree of pigmentation, nuclear to cytoplasmic ratio, and number of nucleoli bear no relationship to the metastatic behavior of uveal melanocytic tumors.68
Tumor size,26,80,105,200 degree of pigmentation, intensity of proliferating cell nuclear antigen (PCNA)–staining,200 the presence of necrosis, ulceration, or inflammation,26,80,254 and p53 expression201 are of limited prognostic value in animals. Depth of tumor invasion into the skin is an important criterion in human medicine but is felt to be too impractical in animals.69 More sophisticated techniques, such as flow cytometry to detect chromosomal anomalies, offer no real advantage over histology in predicting tumor behavior.25,112 The value of determination of a proliferative index using MIB-1, a monoclonal antibody to Ki-67, which identifies proliferating cells, has been investigated. One study examined 27 canine and feline melanocytic tumors, eight benign and 19 malignant, to correlate Ki-67 and PCNA activity with 6-month survival.200 Ki-67 activity correlated very well with poor survival but so did the presence of invasive growth and classification based on cytologic criteria. PCNA was significantly higher in malignant neoplasms, but levels did not demonstrate a strong correlation with poor survival. In a separate study, MIB-1 was applied to 68 cutaneous canine melanocytic neoplasms to determine if the level of immunoreactivity would correlate with 2-year survival. Eighteen of 68 tumors were classed as malignant histologically. The predictive value of the Ki-67 index for 2-year survival was 97%, which was only slightly higher than the predictive value associated with histologic evaluation (91%).133
Differential diagnosis
Other tumors can look clinically very similar to melanoma, particularly those arising from the skin. These include melanocytoma as well as pigmented lesions of the epidermis and adnexa, e.g., feline basal cell tumors and carcinomas, trichoblastoma, trichoepithelioma, pilomatricoma, sebaceous, and apocrine neoplasms. Ceruminous cysts in the feline may be mistaken for multicentric melanoma of the pinna. Melanocytic hyperplasia (lentigo simplex) on the lips, eyelids, nose, and gingiva of orange, cream, and silver cats appear as pigmented macules at these sites. Hyperpigmented macular lesions clinically resembling melanoma may occur in the skin in dogs, primarily the abdomen and nipple. Similarly, epidermal hamartomas (pigmented epidermal nevi, canine seborrheic keratosis), and dermal hemangioma and hemangiosarcoma can appear as pigmented cutaneous tumors.87,88
Nonmelanocytic neoplasms that commonly arise in the oral cavity and eye seldom present clinically as pigmented tumors; therefore, pigmentation is not a distraction.
Cytopathologic Variants
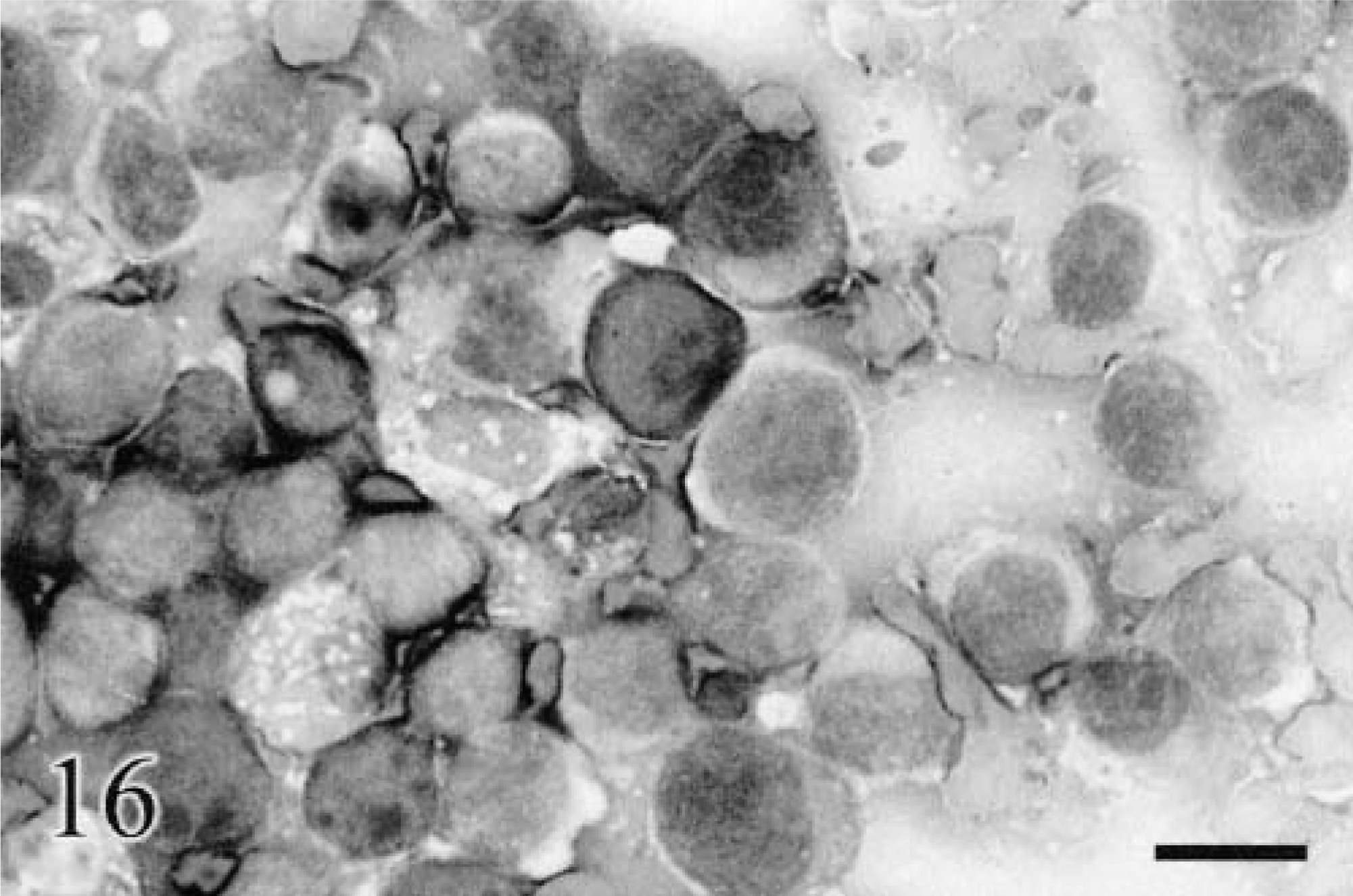
Cytomorphology tends to reflect histologic findings, in that cells can be typed as epithelioid (Figs. 8b, 9b, 10b, 12b), round (Fig. 16), or spindle form (Figs. 11b, 17a–d),9,79,104,238,255 with agreement reported to be 78% in one study.41 Although, just as in histologic classification, cytomorphology is highly variable, a few generalizations can be made.76,239 Nuclei tend to be central to eccentrically located, round to oval, with moderately dense to very fine chromatin and prominent round, oval, or angular nucleoli. Giant nucleoli are a common finding. Nuclei are usually solitary, but occasional binucleated and multinucleated forms are possible. Although not unique to melanoma, longitudinal folding of the nuclear membrane has been reported as a feature in oval nuclear forms stained with Papanicolaou.204 Folding has not been reported in veterinary literature; however, Papanicolaou staining is rarely used in veterinary clinical laboratories. Nuclear indentation or clefting can be seen. Nuclear to cytoplasmic ratios are moderately high to very high. Melanomas do not usually incite a vigorous acute inflammatory response, such as is seen with SCC, unless there is superficial ulceration of the tumor. It is more typical to detect chronic inflammation characterized by few to moderate numbers of melanophages. In cases with visceral metastasis, melanophages may also be observed in the peripheral blood and malignant effusions.228
Cytology. Metastatic melanoma. Mandibular LN; canine. The nonadherent cells are round to very slightly spindle form, with very high nuclear to cytoplasmic ratios, round nuclei, multiple medium-sized nucleoli, and a small amount of light gray to gray-blue cytoplasm. Very rare fine blue-black pigment granules were occasionally detected. The determination of melanocytic derivation was helped by knowledge of the previous biopsy report of an oral melanoma and current physical examination findings, which revealed mandibular lymphadenopathy on same side. Wright-Giemsa. Bar = 20 μm.
The epithelioid and round cell variants tend to be round or irregularly round in contour, with variable cell to cell adhesion observable in epithelioid tumors.78 Spindle-form cells can be plump and only slightly fusiform or display long wispy cytoplasmic processes. Cytoplasm tends to stain very light to moderately dark gray to gray-blue with routine hematology stains. Cytoplasmic borders, particularly in the spindle and epithelioid forms, can be indistinct. Cells may or may not contain uniform noniron pigment granules, which are black, blue-black, or green-black, independent of staining technique, or even if unstained.58 In a sampling of 22 melanomas with cytologic evaluation done at our institution, there were 11 spindle, five epithelioid, and six round cell variants. Only two (18%) of the 11 spindle cell melanomas in our group of 22 lacked pigment, whereas two (33%) of the six round forms and three (60%) of the five epithelioid tumors were amelanotic. Pigment granules are usually very fine, punctate, spherical, or slightly elongated, but not rod-shaped, as can be observed in pigmented keratinocytes. Very finely pigmented cells may display a dusty gray appearance rather than discrete granulation.9 Degree of pigmentation is highly variable, even within a single smear. A very careful search of tumors classed as amelanotic by histologic evaluation usually results in detection of at least few very fine pigment granules in rare cells.238 Fine red granules or vacuoles (or both) may also occasionally be observed within melanocyte cytoplasm of some tumors. When present, they are usually observed consistently in all cells.
Just as in histologic evaluations, distinguishing benign from malignant can be difficult. Using the number of mitotic figures per high-power field does not work well for cytologic specimens because mitotic figures may not preserve well enough to identify, and the number of cells per high-power field is not uniform. Identification of atypical mitotic figures is also difficult unless they are very bizarre because smearing can create chromosomal artifacts that mimic atypia. Generally, the standard cytologic criteria for malignancy is applied, i.e., marked anisocytosis and anisokaryosis, large to giant nucleoli, atypical nucleoli, and marked pleomorphism. Cytologically benign tumors should be assessed histologically to determine malignant potential, unless of course the cells are detected in an inappropriate location, e.g., LN.
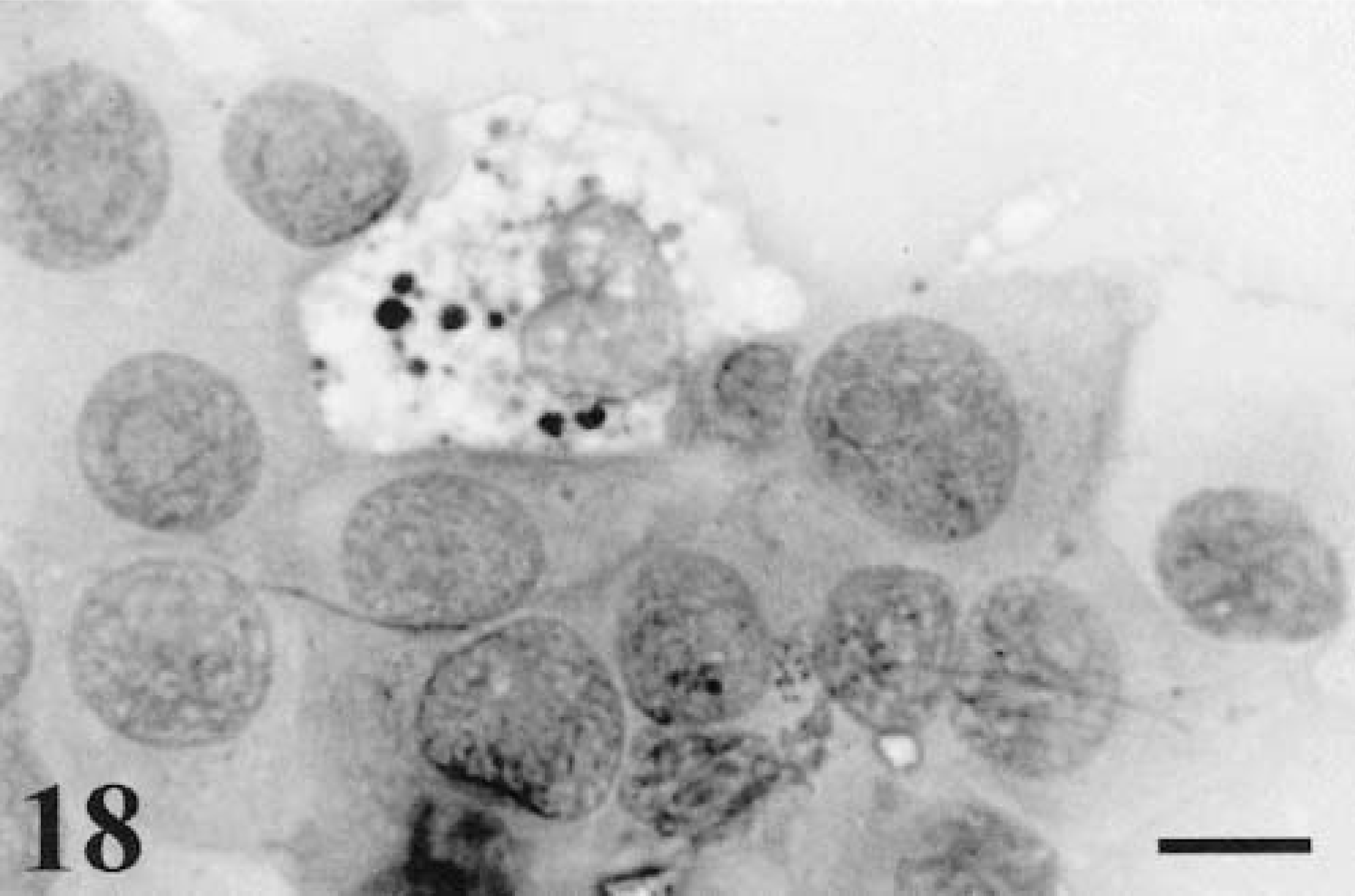
Cytologic determination of specific melanocytic derivation can be problematic in amelanotic or poorly pigmented tumors because of the tendency to mimic other types of neoplasms. Cell size and general morphologic features of the round cell variant can be similar to benign and malignant histiocytic neoplasms, plasmacytomas, poorly differentiated mast cell tumors, and some lymphosarcomas. Lymphoglandular bodies are used as a guide to identification of lymphoid tissue and lymphoid malignancies, but these globular cytoplasmic fragments can occasionally be seen with other neoplasms, including melanomas,75 although usually at a much lower frequency. Spindle-form melanomas containing pigment are easily identified, but amelanotic forms can be confused with reactive tissue,44 hemangiopericytoma, histiocytic sarcoma, or other nonpigmented mesenchymal tumors. Pigmented epithelioid forms can be mistaken for pigmented epithelial cell tumors, e.g., thyroid adenomas of follicular origin128 and thyroid carcinoma.10,216,217 Amelanotic epithelioid melanoma can be very similar to carcinoma; however, careful inspection of smears usually results in detection of a greater range of pleomorphism, which will often include all three forms: round, spindle, and epithelioid. Carcinomas may be markedly variable in size, but, unlike epithelioid melanomas, they usually display a consistent overall shape, with few nonadherent spindle or round forms detected. Careful inspection of cytoplasm for rare black granules and the concurrent presence of melanophages in a markedly pleomorphic neoplasm helps in the identification of melanoma. Melanophages within both melanotic and amelanotic tumors tend to contain few to abundant variably sized pigmented lysosomal granules (compound melanosomes) and can be vacuolated.238 The coarse nature of these granules and the smaller macrophage nuclei and nucleoli help distinguish these cells from neoplastic cells, although this distinction is not always clear (Fig. 18).79 Melanin pigment in both melanocytes and melanophages can be confused with hemosiderin, but hemosiderin is easily distinguished by positive staining using the cytochemical Perls Prussian blue technique.76 Fontana stain, another cytochemical stain,78 and immunocytochemical stains4,6 may also be of assistance but are not in common use for routine cytopathologic evaluation of pigmented tumors.189
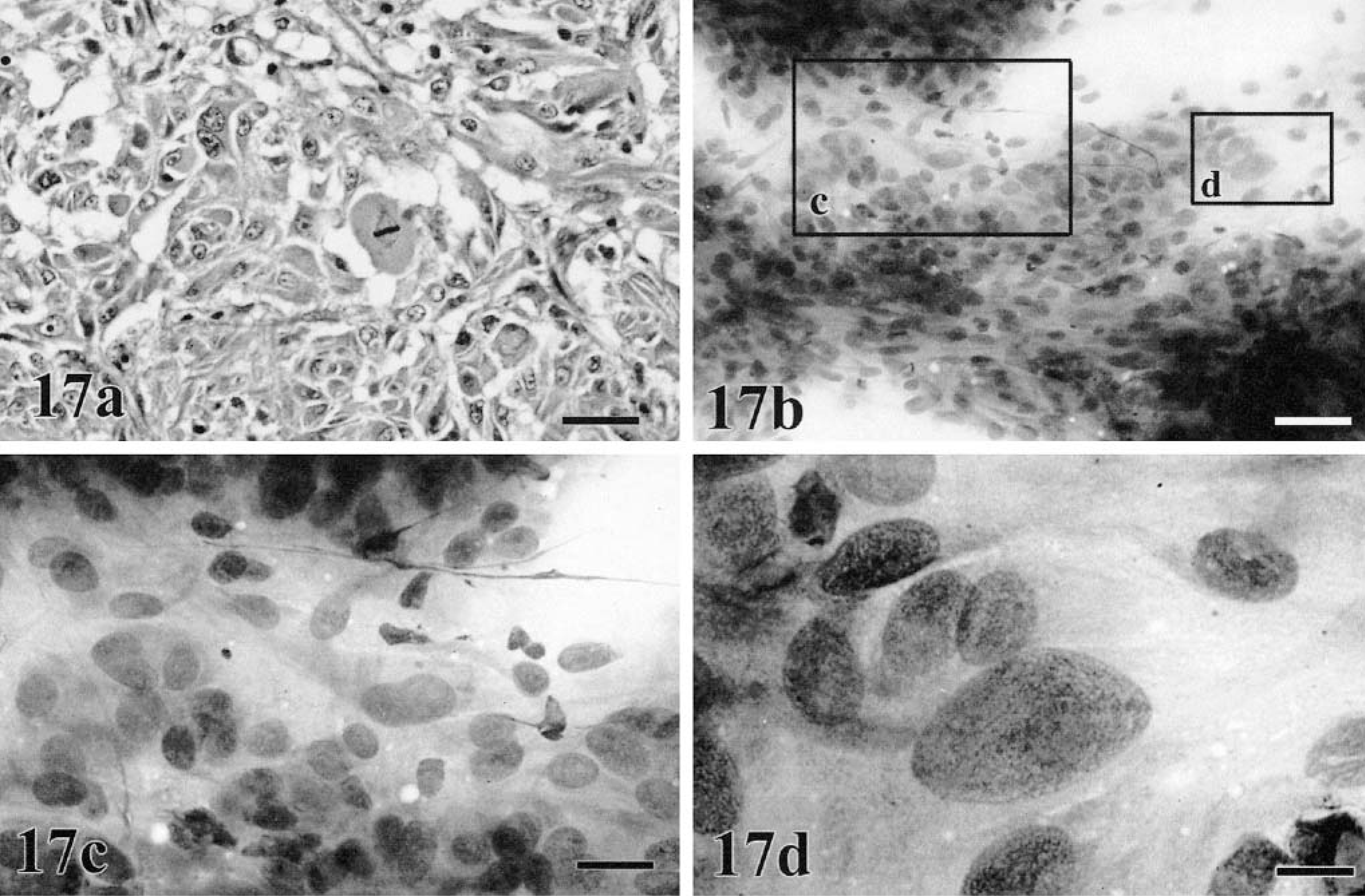
Metastatic spindle cell melanoma. (a) Liver; canine and (b–d) jugular furrow; canine. The primary melanoma was oral.
Melanoma. Ear; feline (same tissue as seen in Fig. 9b). The single melanophage is distinguished from surrounding melanocytes by its coarse pigment granules, cytoplasmic vacuolization, and distinctive nuclear features. Wright-Giemsa. Bar = 10 μm.
The initial definitive diagnosis of melanoma is usually done by histopathologic evaluation, with cytopathology used as a screen before biopsy or as an adjunct to biopsy101 or other diagnostic modalities.237 On the other hand, cytopathology is used more frequently than biopsies for monitoring of metastasis in both human patients and animals.15,23,39,40,197,247 Although there are few large-scale studies in veterinary literature regarding specificity and sensitivity of fine needle aspiration cytology (FNAC) for detection of metastatic neoplasia, it is in common use, particularly for melanoma, carcinomas, and mast cell tumors.132 Morphology of melanoma in the metastatic site is frequently similar to the primary, but not always (Fig. 12a, b). Distant sites are frequently less pigmented. Several reports suggest that even if morphology has changed, the immunocytochemical profile can help confirm the derivation of the tumor.125,131,143,159,171
Regional LN are the most common sites aspirated when monitoring for metastasis. A veterinary study that compared cytologic and histologic findings in FNAC of LN for detection of tumor metastasis found an overall sensitivity and specificity of 100 and 96%, respectively;132 however, the study included only two melanomas, which had negative nodes by both biopsy and cytology. An earlier study to determine the degree of correlation between cytologic and histologic evaluations of skin masses and LN aspirates mentions successful detection of metastatic melanoma in LN for two cases that matched the histologic findings.101 The literature on humans contains many reports, which consistently demonstrate high sensitivity and specificity for FNAC as a means to detect melanoma metastasis in LN.116,141,184,185 Morphology of metastatic melanoma is just as variable as the primary.79 Round (Fig. 19), spindle, (Fig. 20) and epithelioid (Fig. 21) forms can be found in local LN. These cells can be found scattered throughout the smear at a low frequency, focal within regions of a smear, or may completely efface the node, resulting in an aspirate containing no lymphoid tissue. It is often useful to have smears of the primary to compare with cells detected at a low frequency in the LN.
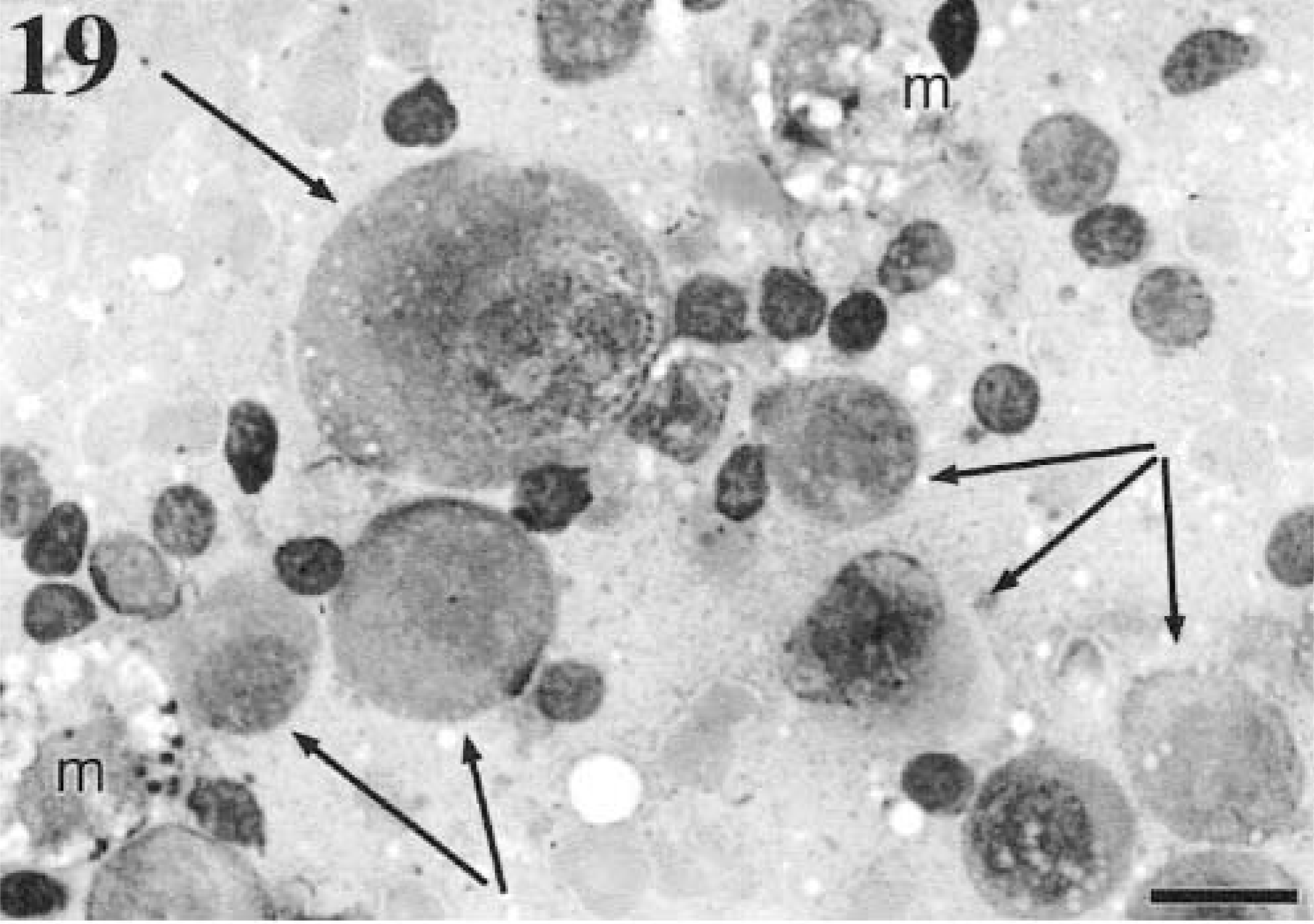
Cytology. Metastatic melanoma. Mandibular LN; canine. The arrows indicate moderately large to giant round, nonadherent melanoblasts, with giant abnormal nucleoli in the largest cell. Most of the small cells are normal lymphocytes. The primary was oral. m = macrophages. Wright-Giemsa. Bar = 20 μm.
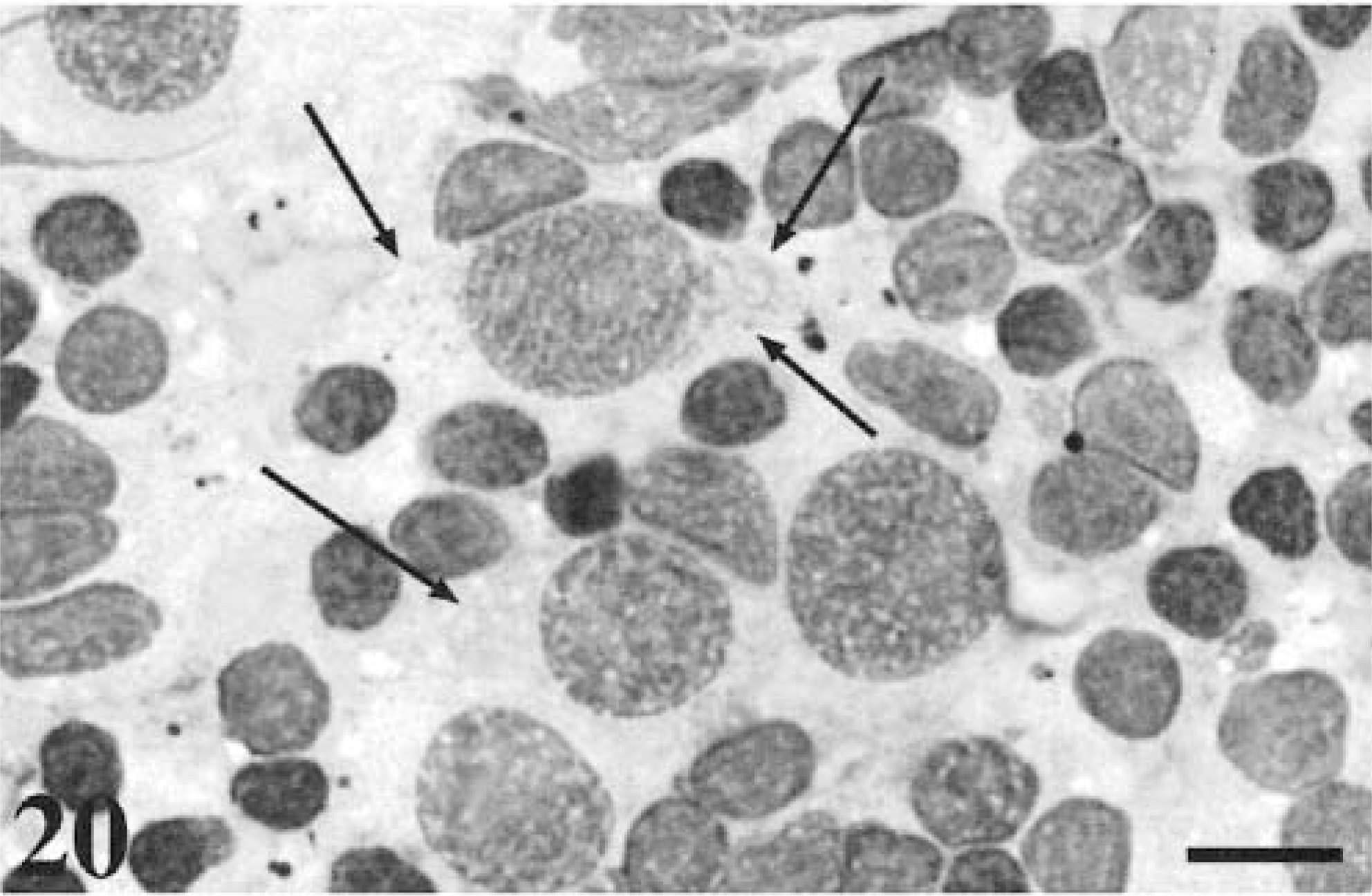
Cytology. Metastatic melanoma. Mandibular LN; feline. The arrows indicate the cytoplasmic limits of the spindle-shaped melanoblasts present in this LN aspirate. The primary was facial. Wright-Giemsa. Bar = 10 μm.
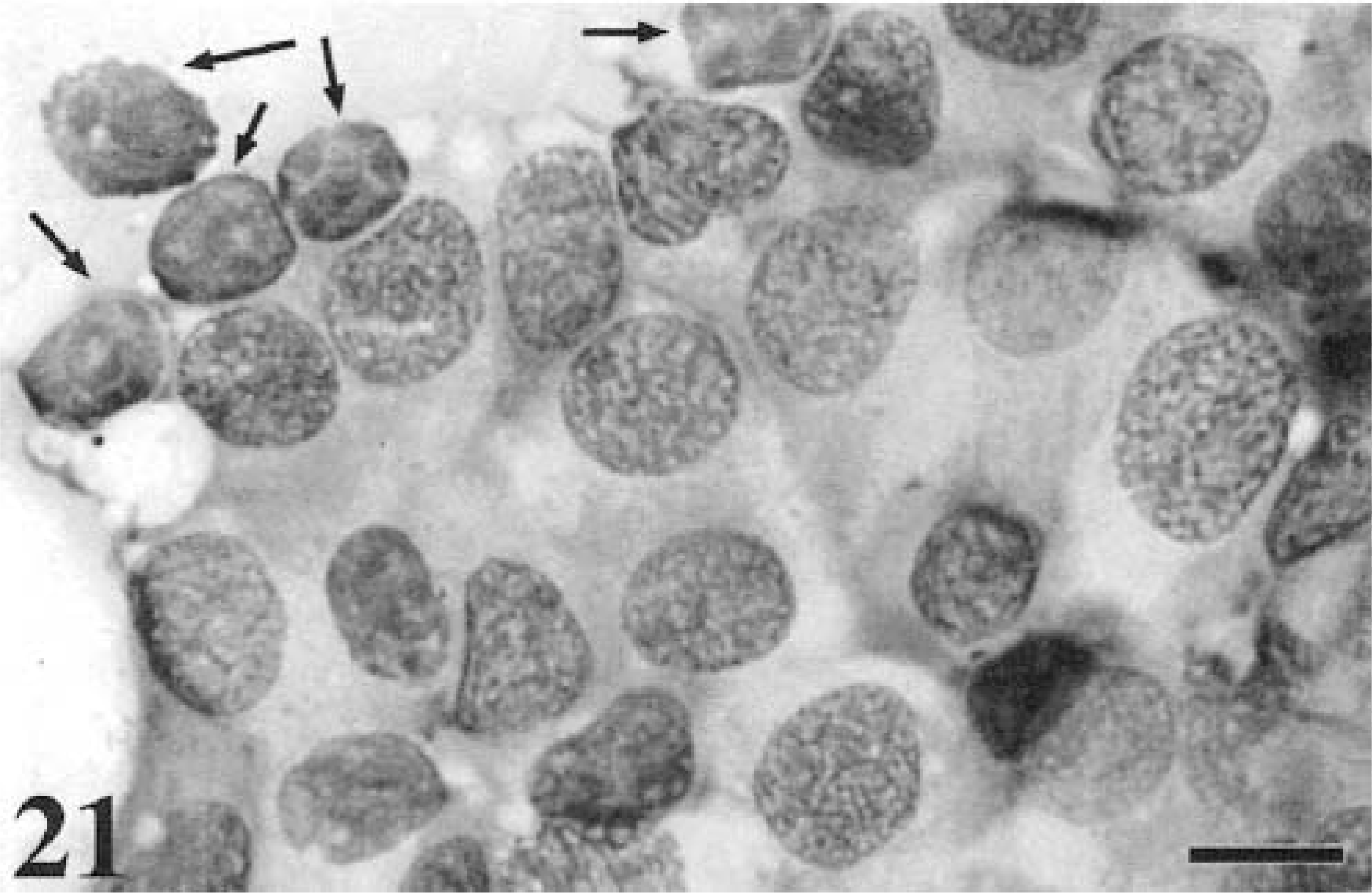
Cytology. Metastatic melanoma. Mandibular LN; canine. The epithelioid melanoblasts almost completely replace normal lymphoid tissue, although occasional small lymphocytes can be seen (arrows). The primary was oral. Wright-Giemsa. Bar = 10 μm.
Evaluation of a LN for metastasis can be complicated by the presence of many melanophages within the LN.79 LN melanosis is not specific to melanoma metastasis, in that it can be detected in a LN draining any region containing hyperpigmentation, including but not limited to melanoma. The pigment may be heavy and obscure identification of neoplastic cells. Also, the melanophages themselves may be mistaken for melanoblasts. Distinguishing features are described above.
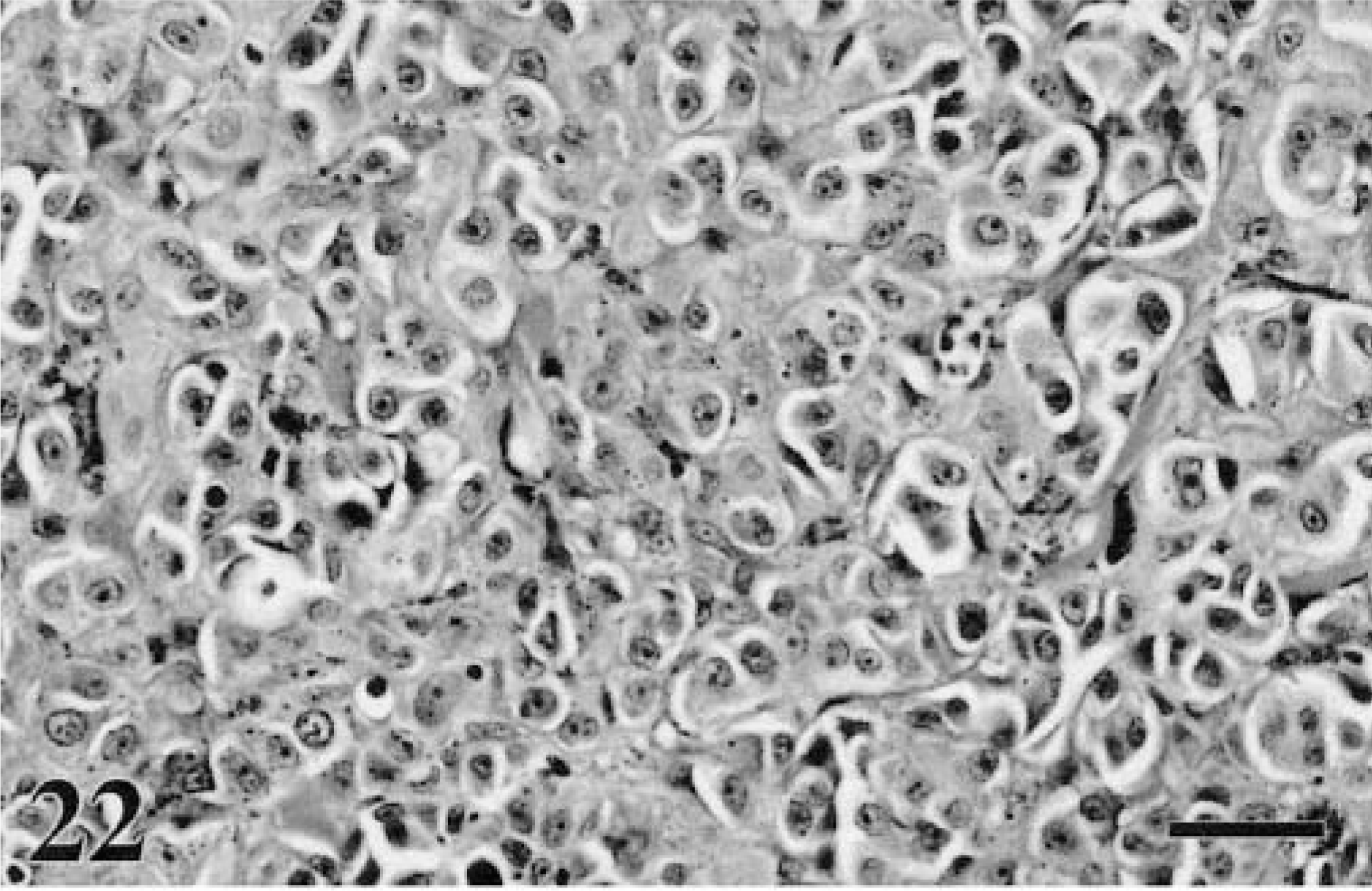
Melanoma-induced peritoneal or pleural effusions frequently contain both the malignant cells and melanophages.6,77,117,228,248 Morphology of the melanoma cells is usually round to epithelioid (Figs. 22–24), with signet ring and balloon cell morphology possible.16,82,121,143,173,174,214,235,248 If spindle form, the cells may display short, blunt cytoplasmic processes (Fig. 25) rather than the longer processes typical of the solid tumors. Scant pigmentation may preclude a definitive interpretation of melanoma, but usually a history accompanies these submissions, enabling a guess as to derivation. Fluid counts are usually consistent with either a modified transudate or inflammatory exudate. There are insufficient reports in the literature regarding melanoma-induced effusions to state the frequency of false negatives, but for neoplasia in general, about 40% will be nondiagnostic.109
Melanoma. Eyelid; canine. Most canine melanomas of the eyelid are benign, but this neoplasm displayed marked pleomorphism. Neoplastic cells extended to the surgical margins. HE. Bar = 30 μm.
Additional Diagnostic Techniques
Amelanotic and poorly differentiated melanomas often elude definitive diagnosis and a number of neoplasms may mimic melanoma microscopically. For these reasons, numerous ancillary diagnostic methods have been developed in an effort to reach a more precise diagnosis. More traditional histochemical techniques are taking second place to immunohistochemistry (IHC), incorporating the use of monoclonal and polyclonal antibodies.183 Other methods include electron microscopy (EM) and, more recently, in situ hybridization (ISH).243
Histochemistry
Bleaching melanomas with hypochlorite is the simplest traditional technique; melanin pigment is extracted to avoid its masking effect of cell morphology and mitotic activity.230 Histochemical stains, such as the Masson-Fontana silver stain, employ the argentaffin reaction to reduce silver in ammoniacal solutions. Melanin acts as the reducing agent in these reactions. The main disadvantage of this technique is the lack of specificity—substances such as lipofuscin can cross-react with these stains.20 More specific to melanin-producing cells is the enzyme tyrosinase, or dihydroxyphenylalanine oxidase (DOPA), which is responsible for converting tyrosine to melanin. In the DOPA test, tyrosine is replaced by dihydroxyphenylalanine, which is in turn converted by DOPA oxidase to a substance indistinguishable from melanin.256 For practical purposes, this test is more applicable to nonpigmented melanomas, where the above chemical reaction results in the formation of brown/black pigment in the cytoplasm of neoplastic cells, confirming the presence of DOPA oxidase (tyrosinase) in those cells. The major disadvantage of this test is that it requires fresh tissue. These traditional methods are now being superseded by IHC.
Electron microscopy
Melanocytes, as well as many other nonmelanocytic tumor cells, contain melanosomes, demonstrable ultrastructurally by EM55,71 Melanosomes indicate that the cell in which they occur is actively producing melanin pigment,43,55 but they must be differentiated from compound melanosomes, which are really secondary lysosomes extruded by melanocytes and taken up by keratinocytes and macrophages.55 True melanosomes usually have cross-striations, whereas secondary melanosomes do not.55 Although melanomas can be recognized ultrastructurally by the presence of aberrant melanosomes, few of these structures are present in amelanotic forms of melanoma. Instead, abnormal or immature premelanosomes are a feature in the Golgi complex. There are a number of drawbacks associated with EM. Firstly, suitable tissue should be collected for reliable diagnosis although often melanosomes are sturdy enough to withstand formalin fixation and paraffin embedding. Secondly, the cells in which the premelanosomes are detected have to be representative neoplastic cells from a pertinent neoplasm. Melanosomes and premelanosomes are not entirely specific to melanomas because they are also found in other tumors originating from the neural crest.70 Melanosomes have been demonstrated in normal and neoplastic keratinocytes71 and in some neuroendocrine neoplasms, particularly those of thyroid parafollicular C-cells (medullary thyroid carcinomas).19,147 Thirdly, detection of these structures requires prolonged examination of multiple sections at high magnification.71
Immunohistochemistry
Specific IHC to identify amelanotic melanoma is needed. Most, if not all, melanomas are vimentin positive and cytokeratin negative in humans, dogs, and cats,183,188,209,243 but other neoplasms can have a similar staining pattern with these antibodies, particularly sarcomas.188 Another moderately useful marker is the antibody against S100, an intracellular and intranuclear acidic, calcium-binding protein.71,209,210,243 Most melanomas are reported to be S100 positive, but the same is true of many other neurogenic and nonneurogenic tumors, including carcinoids.122,169,190,200 Normal S100 cells are also widely distributed, particularly in epithelial and lymphoid tissue.209 Antibody to neuron-specific enolase (NSE) is also frequently used for identification of melanocytic neoplasms, but this marker is also not specific, in that NSE may be found in a variety of tissues, including smooth muscle and neuroendocrine tissue.190
Melan-A, a protein of unknown function that elicits a cytotoxic T-cell response, has a narrow tissue distribution and is generally strongly positive in melanocyte cytoplasm. It may be the most specific IHC test in use at this time. Ramos-Vara et al.190 did an immunohistochemical review of 122 canine oral melanomas plus seven metastatic melanomas with unknown primary sites. Tumors were 92% Melan-A positive, 100% vimentin positive, 98% S100 positive, and 89.1% NSE positive. The authors did not state the proportion of amelanotic melanomas that were Melan-A positive but because 32% of the total number of tumors was nonpigmented it must be assumed that a significant proportion of amelanotic tumors reacted to the antibody. Furthermore, the distribution of Melan-A in a melanoma did not simply duplicate the distribution of melanin because melanophages were negative for Melan-A. Finally, two of the three clear cell melanomas in this study were Melan-A positive.190 Only four (2.45%) of the 163 nonmelanocytic tumors were positive for Melan-A and only weakly so. These included two salivary carcinomas and two transitional cell carcinomas, which were easily distinguished from melanomas by morphology alone. They concluded that Melan-A is a useful marker for melanocytic derivation in tumors of uncertain lineage.
A slightly more recent but smaller study126 of 29 canine melanocytic tumors, which included 10 melanomas, 11 melanocytomas, and eight uncertain, found that Melan-A (designated Melan-A/MART-1) was positive in 90% of pigmented tumors but failed to stain amelanotic tumors. Ten tumors were amelanotic. Seven of these were classed as malignant, whereas the remaining three were uncertain. In their hands, Melan-A was infrequently positive in malignant tumors, with only three of the 10 melanomas staining positive for Melan-A (the three that were pigmented). On the other hand, all the 11 melanocytomas were positive, with 10 of these classed as moderate to strongly positive (all benign tumors were pigmented). These authors suggested that Melan-A staining may correlate with biologic behavior, with benign tumors staining moderately or strongly positive and malignant tumors showing weak or negative staining, but also seemed to show that Melan-A mostly duplicated the distribution of pigmentation.126 The study outlined in the previous paragraph was larger (129 versus 10 malignant tumors), had 92% positive results (versus 30% in the smaller study), and found that distribution did not exclusively coincide with pigmentation. Therefore, the larger study offers stronger support for Melan-A as a good marker for tumors of melanocytic derivation, independent of pigmentation. Seven of the 122 oral tumors had evaluation of metastatic sites. Two were negative for Melan-A at both the primary and the metastatic sites. Five were positive at both the primary and the metastatic sites, but Melan-A staining of metastatic cells was weaker. So, although advancing disease resulted in a weakening of positivity, the strong staining pattern of the primary did not predict nonaggressive disease for these cases. In humans, the loss of Melan-A positivity sometimes seen in melanomas is thought to reflect selection for a Melan-A negative variant after partial regression secondary to tumor infiltration by cytotoxic T-lymphocytes.207
It remains unproven that Melan-A expression is a predictor of less aggressive behavior, but it may be a worthwhile marker for identification of tumors of uncertain lineage, especially when used in combination with S100. A study of 48 feline melanomas found 67% were positive for Melan-A, and 87.5% were positive for S100.191 The conclusion offered by this article is that Melan-A in cat tumors is not as sensitive as S100 but more specific and could be used to distinguish heavily pigmented melanocytic tumors from pigmented basal cell tumors. On the other hand, basal cell tumors are cytokeratin positive, whereas melanomas are not. The authors advise the use of both Melan-A and S100 in diagnosing melanomas in cats.
The advent of murine antibodies specific for melanoma-associated antigens, particularly melanosomes,20 has helped improve the value of IHC in the diagnosis of melanoma in human medicine. Often a panel of appropriate antibodies is advocated,3,95,246 an approach also favored in veterinary literature.20 Monoclonal antibodies to human melanosome–specific antigens (HMSA) 1 and 5 are typical examples, reacting with 60 and 69% of canine melanomas, respectively, increasing to 83% when used in combination. This also demonstrates the heterogeneity of antigen expression and the subsequent value of using multiple antibodies at the same time.20 Although monoclonal antibody for HMB-45 has proven to be a sensitive marker for certain subtypes of human melanomas, HMB-45 is generally considered less useful in animals.20,71,177 But the utility of HMB-45 for animal tumors may be improved by a process that includes pretreatment of formalin-fixed tissues with KMnO4-oxalic acid. One study reports that 88% of melanomas stained positive for HMB-45 after KMnO4-oxalic acid pretreatment, whereas only 6% of nonmelanocytic tumors were positive.223
IBF9 is the first murine monoclonal antibody specifically created to recognize canine melanoma antigen176 and is highly sensitive (greater than 80%). Although it does cross-react with other types of neoplasia, such as basal cell tumors and lymphosarcomas, these neoplasms can easily be ruled out using additional immunohistochemical testing and, on the whole, are not likely to be confused morphologically in the first instance.
In situ hybridization
Although requiring more technical skills and equipment, ISH, using a complementary DNA probe for tyrosinase-specific mRNA, is a highly specific and sensitive tool and can be performed on formalin-fixed, paraffin-embedded tissues.34 Because amelanotic and pigmented melanomas contain similar levels of tyrosinase-specific mRNA, ISH is potentially a valuable tool for the diagnosis of poorly differentiated melanomas.226 It has been used to confirm balloon cell and signet ring type melanomas in cats, although it does suffer from certain technical flaws such as degradation of mRNA with tissue handling and formalin fixation.243
Future Diagnostics
Research into the pathogenesis of melanoma is creating a source of additional information for development of future diagnostic methods. Much work is currently underway in human medicine to try and identify specific tumor markers, i.e., proteins that are associated with malignancy.140 Applicable detection techniques encompass IHC, ISH, polymerase chain reaction (PCR), and reverse transcriptase-PCR, as well as fluorescent IHC. Using these methods, protein or DNA mutations specific to a particular malignant neoplasm may be shown circulating in the blood, for example.140 Serological markers, including cytokines, cell adhesion molecules, and melanoma-inhibitory activity, have also been investigated as potential sentinels of melanoma. Generally, all these potential markers are not yet considered useful for early detection of disease but may be valuable in terms of prognosis or for selecting patients for more aggressive therapeutic strategies.30
As is obvious from the above, there is no single diagnostic technique capable of differentiating benign from malignant melanocytic neoplasms or of predicting survival time. Even the most recent molecular techniques now being applied to the diagnosis, staging, and therapy of malignant neoplasia in humans have their limitations. All the ancillary tests developed to date should be considered just that—ancillary to the well-established and more traditional examination routines used in diagnostic pathology.
