Abstract
A 9-year-old Labrador Retriever was diagnosed with ganglioradiculitis (sensory neuronopathy). This idiopathic disease of mature dogs is characterized by a profound loss of sensory nerve function due to mononuclear inflammatory infiltration of peripheral ganglia and spinal nerve roots, with destruction of sensory neurons. Immunohistochemistry demonstrates that the infiltrating cells are primarily T lymphocytes and that immunoglobulins are not present on the cell membranes of affected neurons. The pathogenesis of ganglioradiculitis remains unclear, but the evidence points to a cell-mediated immune mechanism.
Ganglioradiculitis (sensory neuronopathy) is a rare idiopathic disease of dogs characterized by inflammation of peripheral ganglia and spinal nerve roots.2,3,13,14,16 The disease affects adult dogs without evidence of sex or breed predilection. Clinical signs are typically abrupt in onset and slowly progressive, consisting of sensory ataxia, hypermetria, depression or loss of spinal reflexes, reduced postural reactions, facial hypalgesia, dysphagia, and megaesophagus. Pathologic findings consist of mononuclear inflammatory cell infiltration of both spinal ganglia and autonomic ganglia, with axonal degeneration and secondary demyelination in the spinal cord and peripheral nerves. Here, we describe a case of ganglioradiculitis in a dog, characterizing for the first time the inflammatory infiltrate using immunohistochemistry.
A 9-year-old spayed female Labrador Retriever was admitted to the Cornell University Hospital for Animals with a complaint of hind limb ataxia, excessive salivation, and dysphagia. The signs were sudden in onset and slowly progressive over several months. Two weeks prior to admission, the dog was treated with anti-inflammatory doses of corticosteroids without improvement. On presentation, the dog exhibited a significant and unusual ataxia with awkward, abrupt, exaggerated movements of all four limbs. During limb protraction, both overextensor and overflexor movements occurred, but overextension predominated. The ataxia resembled a cerebellar ataxia but without any obvious balance loss and less overflexion. The dog displayed a marked base-wide stance, had mild swaying of the hind limbs when standing, and would occasionally collapse. Postural reactions were mildly decreased in all four limbs. The patellar reflex was absent bilaterally. Bilateral facial and nasal hypalgesia and mild atrophy of the muscles of mastication were present. The dog continuously salivated. The owner declined further workup, including cerebrospinal fluid analysis and electrophysiologic studies, and was offered a presumptive diagnosis of ganglioradiculitis. The dog was treated for 1 month with cyclosporin. One month later, because of worsening condition, the dog was euthanatized at the owner's request.
At necropsy, gross lesions were primarily confined to the nervous system. The most prominent change was in the dorsal funiculi of the spinal cord, which were uniformly and strikingly white throughout their entire length (Fig. 1). The spinal ganglia of the C7–T1 spinal nerves were slightly increased in size. In the medulla oblongata, the spinal tract of the trigeminal nerve was indistinct. A small dark-green cyst was present adjacent to the pituitary gland.
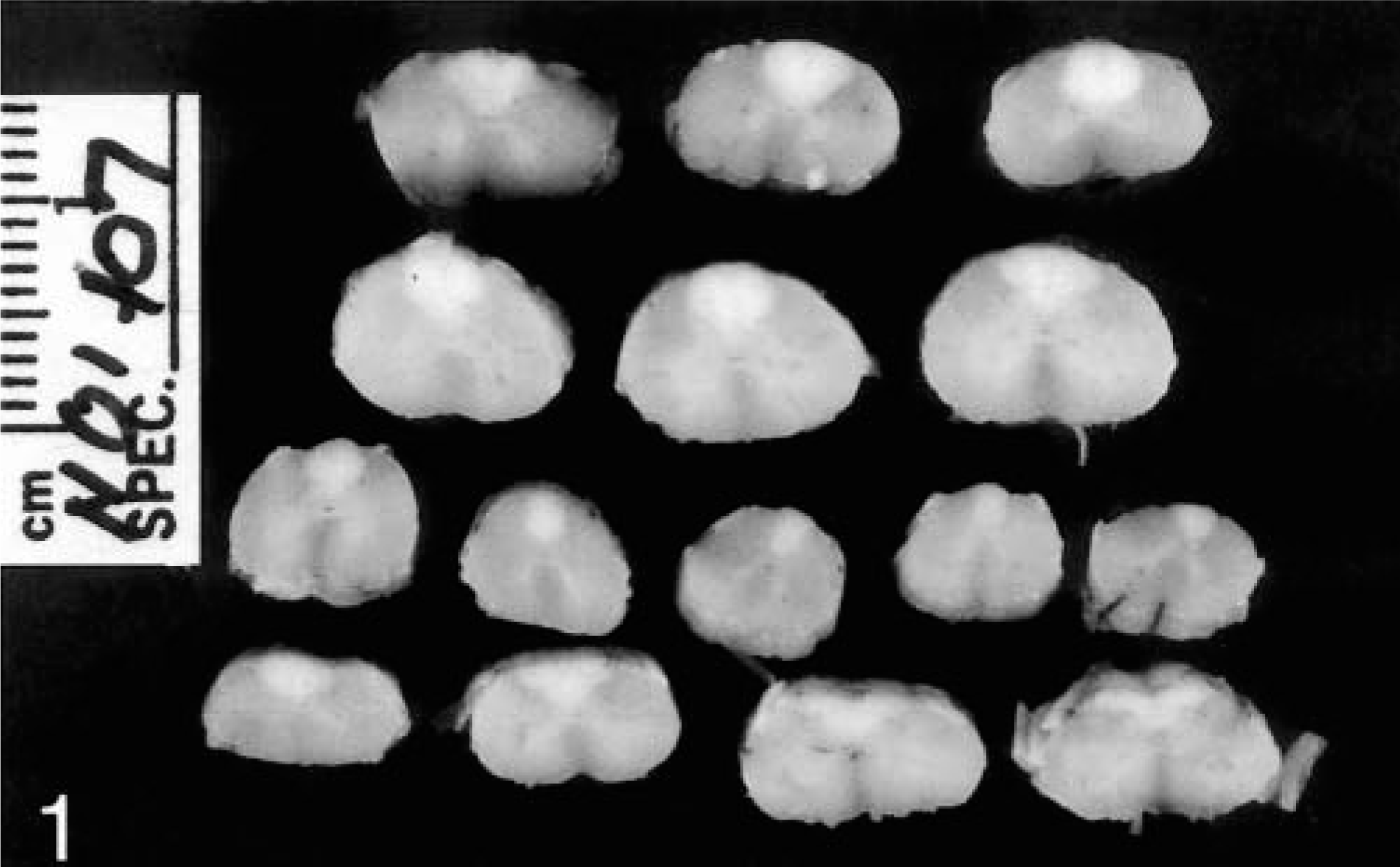
Spinal cord; dog. Wallerian degeneration is present in the dorsal funiculi throughout the length of the cord, resulting in a well-demarcated zone of pallor of the affected tissue.
Representative tissue samples were collected and fixed in 10% neutral buffered formalin. Tissues were processed routinely, sectioned at 5 μm, stained with hematoxylin and eosin (HE), and examined by light microscopy. Selected sections were also stained with Luxol fast blue and Bielschowsky stains. Immunohistochemical staining of formalin-fixed, paraffin-embedded tissue sections from selected ganglia and adrenal gland was performed using a streptavidin peroxidase method according to the manufacturer's instructions (Histostain SP Kit; Zymed Laboratories, San Francisco, CA). The primary antibodies used are summarized in Table 1. The chromagen was 3,3′-diaminobenzidine, and the slides were lightly counterstained with Gill's hematoxylin. Stains for IgG, IgM, IgA, and C3 were repeated using the same streptavidin procedure but with a fluorescein-labeled streptavidin (fluorescein streptavidin; Vector Laboratories, Burlingame, CA). IgG and C3 staining also was done with a nonstreptavidin procedure using a fluorescein-labeled secondary antibody (DAKO, Carpinteria, CA). Positive controls consisted of normal canine spleen, lymph node, and skin. Negative technique controls were performed by replacing the primary antibody with phosphate-buffered saline, normal rabbit serum, or an isotype-matched irrelevant mouse monoclonal antibody.
Immunohistochemistry protocol for evaluation of canine tissues.
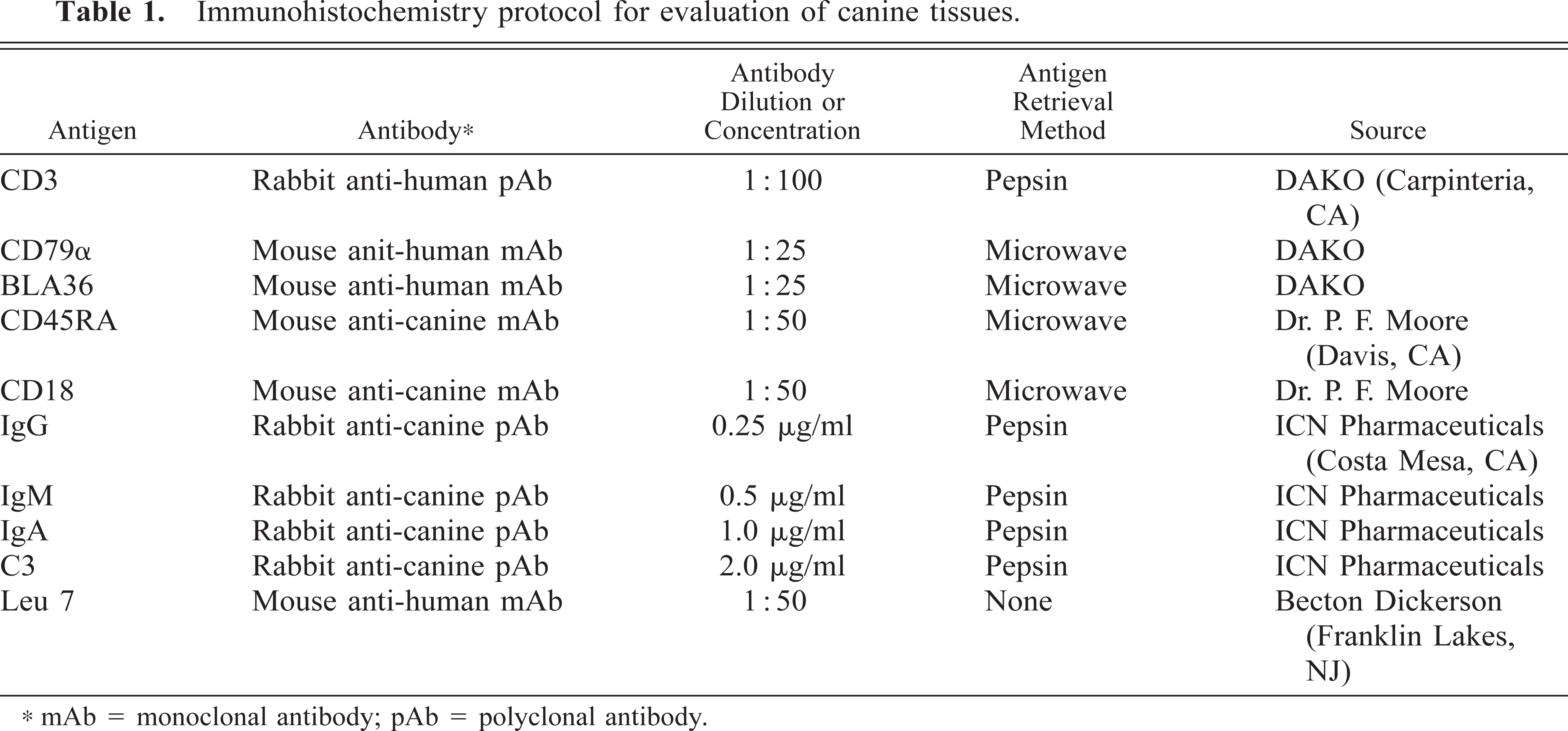
mAb = monoclonal antibody; pAb = polyclonal antibody.
Histologically, there was moderate to severe nonsuppurative inflammation within most peripheral ganglia. In the spinal ganglia, there were moderate to large multifocal accumulations of lymphocytes (Fig. 2). The lymphocytes were between and often surrounding neurons, occasionally forming small, dense, nodular aggregates. Fewer numbers of plasma cells and macrophages were present. Rare degenerate neurons were present and were characterized by decreased size and loss of cellular detail. Leukocytic infiltrates were often found around blood vessels in severely affected areas, and blood vessels were often prominent, with plump, reactive endothelial cells. Inflammation was often most severe peripherally within a ganglion. Between the neurons, there was marked proliferation of Schwann cells within nerve fascicles (Bängner's bands), and numerous vacuolated myelin sheaths (myelin ellipsoids) were present. Similar changes were present within the trigeminal ganglion, cervicothoracic ganglion, cranial cervical ganglion, and distal ganglion of cranial nerve X. Decalcified sections of the petrosal bone revealed no abnormalities in the vestibular or spiral ganglia, but mild lymphocytic infiltrates were noted in the geniculate ganglion of cranial nerve VII and the proximal ganglia of cranial nerves IX and X. In the intestinal tract, submucosal and myenteric ganglia were infiltrated by minimal to small numbers of lymphocytes, without evidence of neuronal degeneration. The adrenal medulla was infiltrated by small numbers of scattered lymphocytes.
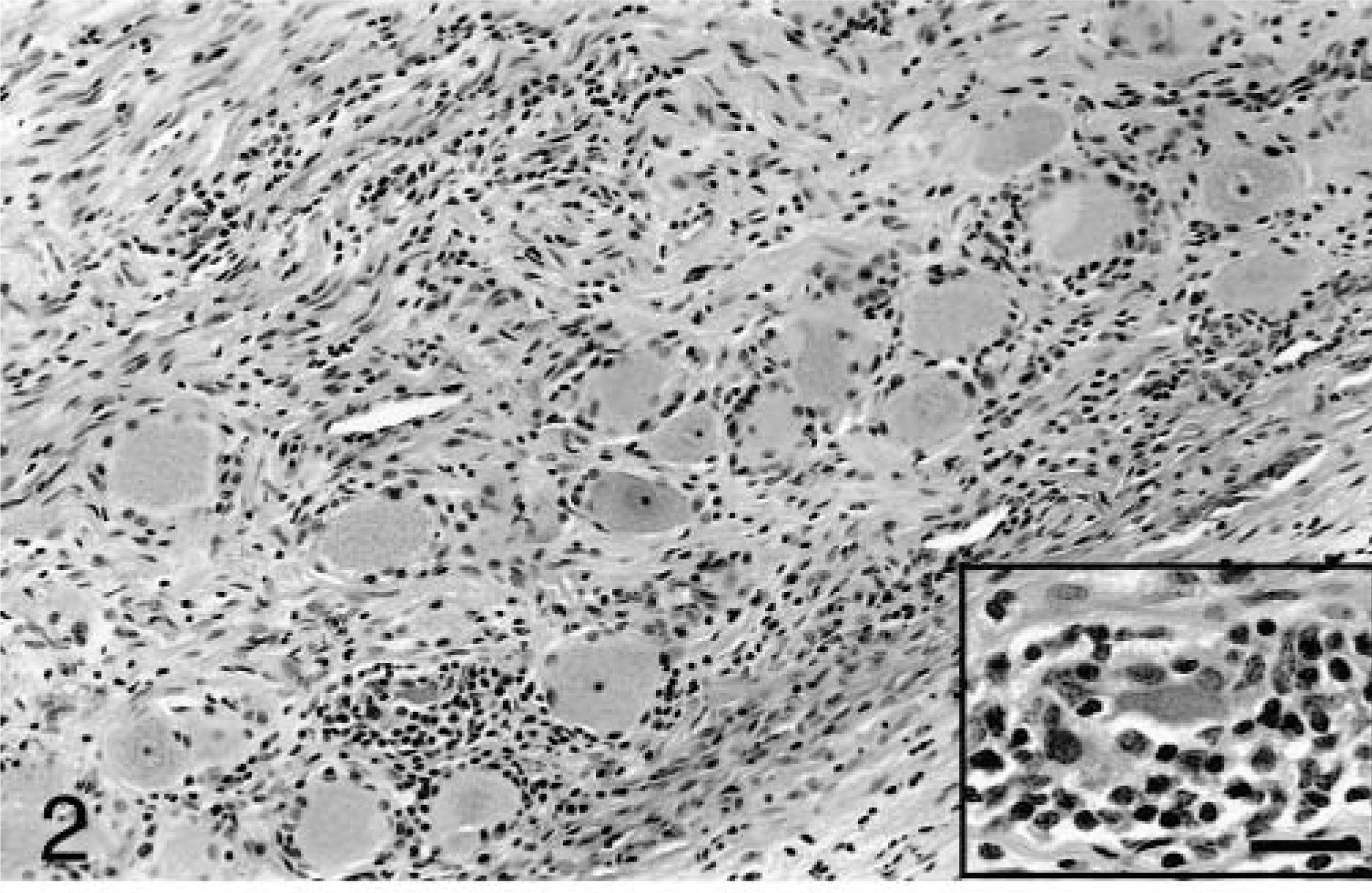
Spinal ganglion (C5–C7); dog. The ganglion is diffusely infiltrated by small to moderate numbers of mononuclear inflammatory cells, primarily lymphocytes. Inset: Neurons are often encircled by lymphocytes and are occasionally degenerate. HE. Bar = 29 μm.
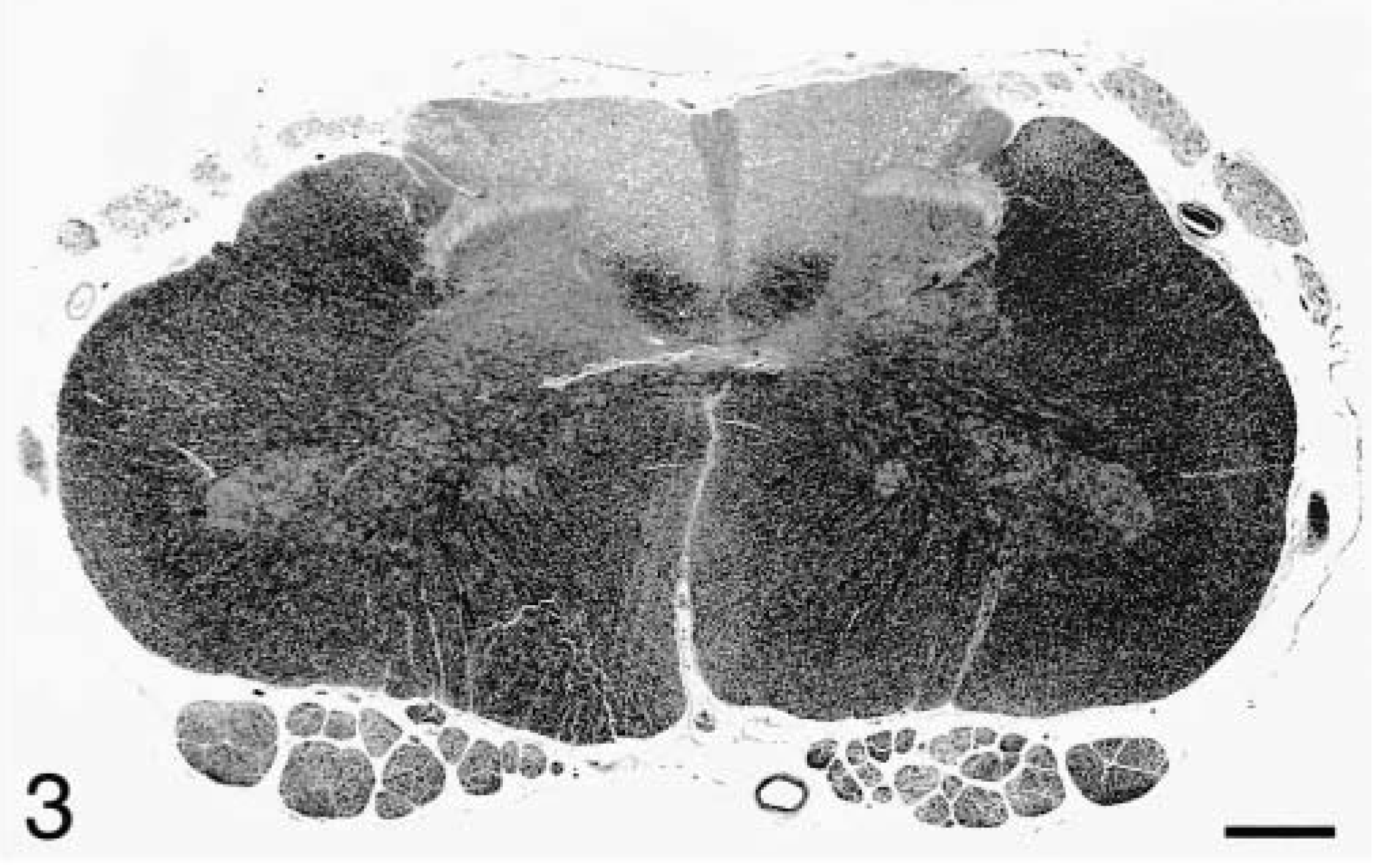
Transverse sections taken at all levels of the spinal cord revealed diffuse vacuolation characteristic of Wallerian degeneration within the fasciculus cuneatus and fasciculus gracilis. The medial aspect of the fasciculus gracilis was less severely affected. Luxol fast blue stains revealed marked myelin loss in the dorsal funiculi (Fig. 3), and Bielschowsky staining revealed fragmentation and loss of axons. On longitudinal sections, there were prominent rows of myelin ellipsoids in the dorsal funiculi. Within the pons and medulla, changes were limited to the spinal tract of the trigeminal nerve, in which small numbers of ellipsoids were present. Lesions were not present within the midbrain, cerebellum, diencephalon, or cerebrum. The pituitary cyst was closely associated with the pars distalis and was classified as a hypophyseal (Rathke's) pouch cyst. It was lined by epithelium that ranged from simple cuboidal to pseudostratified columnar with goblet cells and contained amorphous, pale eosinophilic material.
Lumbosacral spinal cord; dog. Marked myelin loss is evident in the dorsal funiculi and dorsal roots. Luxol fast blue. Bar = 890 μm.
In peripheral nerves, there were changes consistent with Wallerian degeneration, including ellipsoid formation and Bängner's bands. These changes were most severe in the dorsal spinal nerve roots, in which there were moderate to large numbers of ellipsoids, numerous Bängner's bands, and small multifocal accumulations of lymphocytes and fewer plasma cells. Lipid-laden macrophages were commonly seen in association with ellipsoids. Ventral nerve roots appeared normal. With both HE and Luxol fast blue stains, the difference in appearance between dorsal nerve roots and ventral nerve roots was striking (Figs. 3, 4). Axonal loss in dorsal nerve roots was confirmed with Bielschowsky staining. The sciatic nerve exhibited moderate numbers of Bängner's bands and small numbers of ellipsoids. In the femoral, median, and ulnar nerves, there were comparable numbers of ellipsoids, but Bängner's bands were relatively less prominent. Minimal numbers of scattered lymphocytes were present within sciatic, femoral, median, and ulnar nerves.
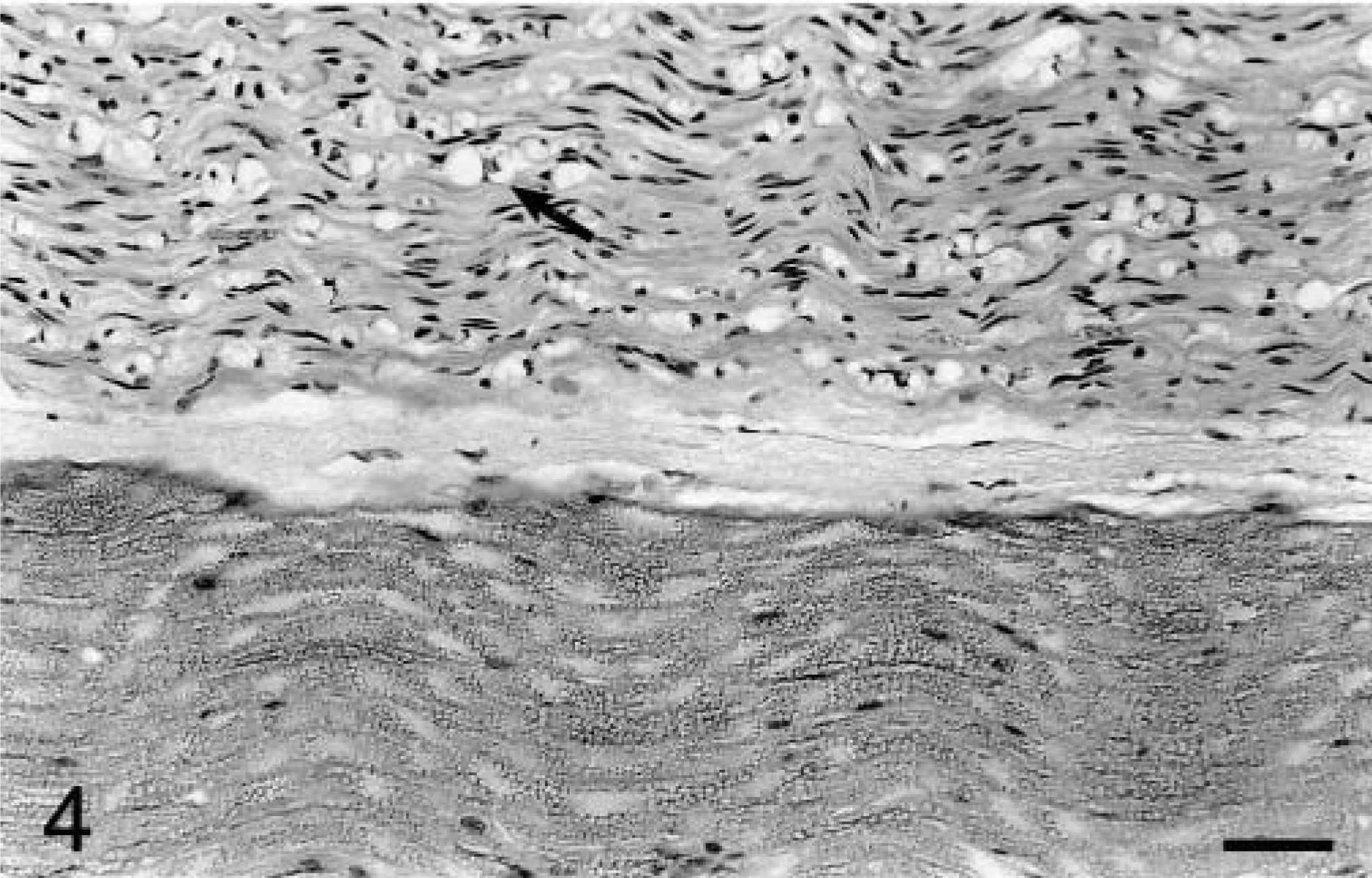
Lumbosacral dorsal and ventral spinal nerve roots; dog. Extensive Wallerian degeneration within the dorsal root contrasts with the adjacent normal ventral root. In the dorsal root, numerous myelin ellipsoids (arrow) and proliferated Schwann cells are present. HE. Bar = 114 μm.
The majority of inflammatory cells within affected ganglia were strongly positive for CD3 via immunohistochemistry and were interpreted as T lymphocytes. CD3-positive cells were scattered randomly or formed small cuffs around neurons, and were closely abutting the plasma membrane of affected neurons (Fig. 5). Occasional intense nodular accumulations of CD3-positive cells were present, without evidence of an associated neuron. Perivascular cuffs of CD3-positive lymphocytes were occasionally present. Small numbers of CD3-positive cells were scattered throughout the spinal nerve root. Scattered cells within ganglia stained positively for BLA36 and CD18, and a few of these cells were closely associated with neuronal cell bodies. CD3-positive lymphocytes were also found in low numbers throughout the adrenal medulla and in the subcapsular area of the adrenal cortex. A relatively greater number of mononuclear cells in the adrenal medulla stained positively with BLA36 and CD18. The BLA36- and CD18-positive cells in both ganglia and adrenal gland were interpreted as dendritic cells. Rare mononuclear cells in ganglia stained positively with CD79α and CD45RA, consistent with B lymphocytes. The inflammatory cells did not stain with Leu 7. Neuronal cell membranes did not stain with antibodies to C3, IgG, IgM, or IgA with the peroxidase or the fluorescent methods. A few intralesional leukocytes, presumably plasma cells, were strongly fluorescent with IgG, IgM, and IgA.
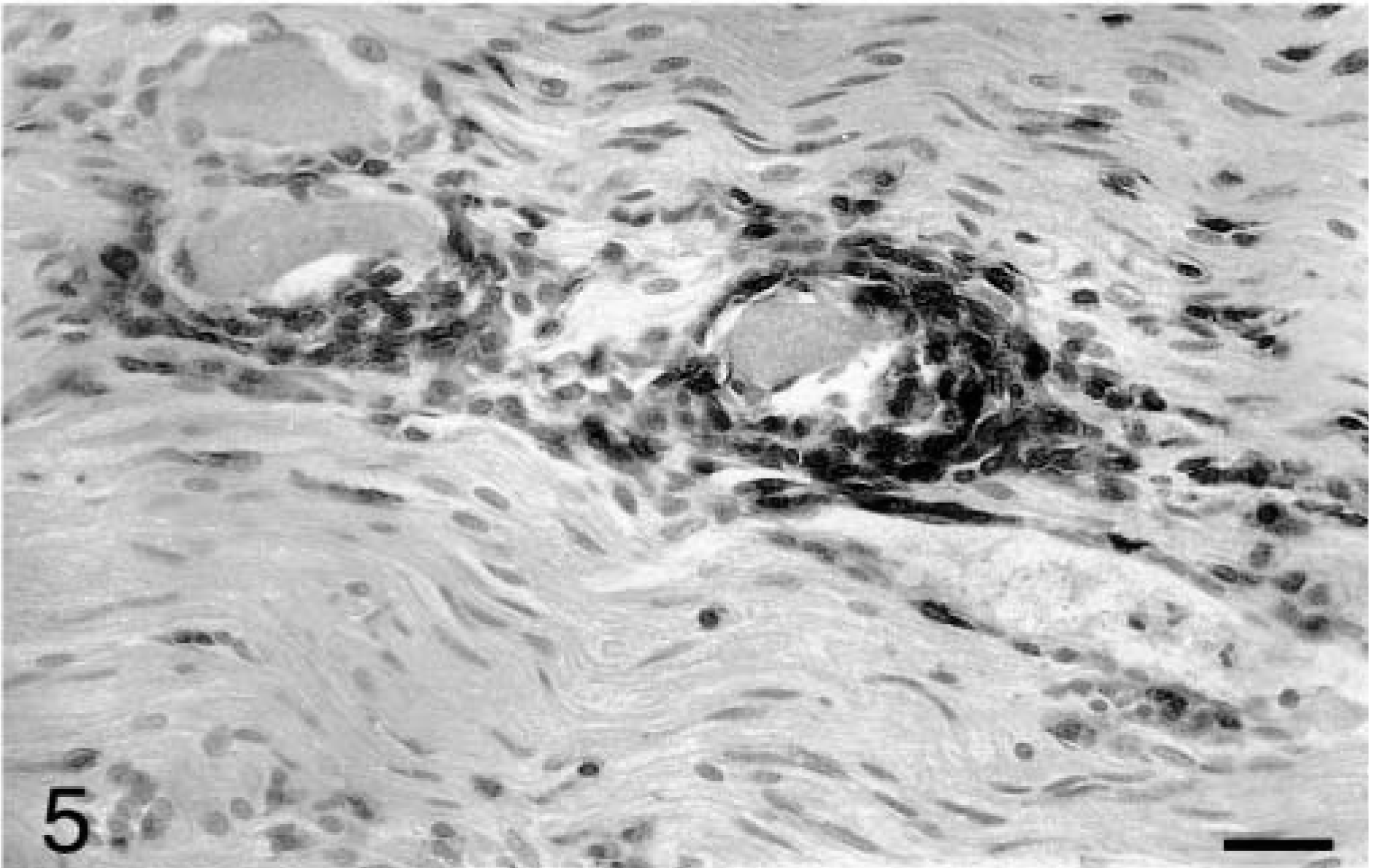
Right cranial cervical ganglion; dog. The majority of infiltrating lymphocytes stain strongly for CD3. Streptavidin peroxidase method, Gill's hematoxylin counterstain. Bar = 29 μm.
The clinical and pathologic findings in this case are similar to those described in previous reports of the disease.2,3,13,16 Inflammation appears to target neurons in craniospinal sensory ganglia, resulting in loss of large-diameter sensory fibers. The clinical signs largely reflect loss of proprioceptive impulses from muscle spindles and Golgi tendon organs. Clinically, the marked proprioceptive deficit resembles a cerebellar ataxia; however, these patients do not manifest classic cerebellar hypermetria with overflexion of joints nor do they show other signs of cerebellar impairment. A primary loss of sensory function results in hypalgesia and the loss of patellar reflexes with preservation of muscle tone and motor function. The dysphagia in this dog was likely caused by damage to the distal ganglia of cranial nerves IX and X. Although there was no clinical evidence of gastrointestinal malfunction, enteric ganglia exhibited mild inflammation. In only one previous report was inflammation within enteric ganglia described.3 The dog of that report had megaesophagus and moderate numbers of inflammatory cells within myenteric ganglia of the abdominal esophagus and gastric cardia.
The distribution and nature of the lesions in this dog are most suggestive of an immune-mediated pathogenesis. Potential immune mechanisms for neuronal destruction in ganglioradiculitis include type IV cell-mediated cytotoxicity involving CD8-positive T cells or antibody-dependent type II mechanisms, including those involving complement or natural killer (NK) cells. Of these proposals, direct T cell–mediated cytotoxicity seems most plausible. The inflammatory infiltrate was dominated by lymphocytes, and the lymphocytes were in close association with degenerate neurons. Most of the infiltrating cells were CD3-positive T lymphocytes. Neurons are capable of expressing class I major histocompatibility complex antigens, and neurons are capable of serving as targets of cytotoxic T cells via the Fas/FasL apoptotic pathway.11 Peripheral nervous system neurons are more susceptible to T cell–mediated lysis than are central nervous system neurons,9 perhaps explaining the restriction of neuronal loss to peripheral ganglia. Immunohistochemical staining for IgG, IgM, IgA, and C3 was negative, further supporting an antibody-independent mechanism. Antibody involvement cannot be completely excluded, however, because many autoimmune diseases have both cell-mediated and humoral components.4
In idiopathic sensory neuronopathy (ISN), a similar disease in humans, there is evidence for both cell-mediated and humoral mechanisms. Immunohistochemical staining has shown that the infiltrating cells are T cells, with some of the lymphocytes identified as CD8 positive and others considered probably CD4 positive.7 Antibodies against spinal ganglion neurons have been demonstrated, and serum from ISN patients inhibits neurite outgrowth in rat spinal ganglion cell cultures.15 It was unclear in that study, however, whether the antibodies were the primary cause of the disease or merely a response to neuronal damage. Other sensory neuronopathies in humans have been associated with neoplasia5 and Sjögren's syndrome.6 Thorough postmortem examination of this dog did not reveal neoplasia. Although lacrimal and salivary glands were not examined histologically, there was no clinical evidence of Sjögren's syndrome, and dogs with Sjögren's syndrome have not shown clinical evidence of ganglioradiculitis.12
Although the infiltrating CD3-positive cells were most likely T cells, CD3 expression may not be restricted to T cells. The CD3 complex is composed of a group of membrane-associated polypeptide chains, γ, δ, ε, and ζ, linked with the antigen-specific heterodimers of the T cell receptor (TCR) complex, either TCR-αβ or TCR-γδ. CD3 components have both extracellular and intracellular domains, and the CD3 antibody used in our study specifically labels the intracytoplasmic domain of the ε chain (manufacturer's specifications). Human NK cells have been shown to express the CD3ε component in the cytoplasm,10,17 suggesting the possibility that some of the observed CD3-positive cells in this dog were NK cells. Murine studies have shown that NK cells are capable of autoimmune neuronal destruction.1,8 Immunostaining for Leu 7 was negative, and although Leu 7 is considered a reliable marker for NK cells in humans,7 its specificity in dogs is unclear. There is currently a lack of specific markers for NK cells in dogs.
The pathogenesis of ganglioradiculitis in the dog remains uncertain, but the findings in this case support a cell-mediated mechanism. In future cases, analysis of serum for antineuronal immunoglobulins and immunohistochemical staining of fresh frozen tissue using a greater variety of canine leukocyte markers may allow a better understanding of the pathogenesis.
Footnotes
Acknowledgements
We thank Dr. Brian Summers for his thoughtful review of the manuscript, Pat Fisher for her expertise with immunohistochemistry, and Alexis Wenski-Roberts for assistance with photography.
