Abstract
Three 1-week-old and three 3-month-old Holstein calves that had received colostrum were inoculated endobronchially with bovine adenovirus 3 (BAV-3). The gross and histologic lesions in these six infected calves were localized mainly in the right caudal lobe of the lung and were closely associated with the site of the deposition of the inoculum. The pneumonic lesions were severe necrotizing bronchitis, bronchiolitis, and alveolitis, accompanied by infiltration of inflammatory cells and proliferation of type 2 pneumocytes. Intranuclear inclusion bodies, BAV-3 antigen, and virus particles were detected in the degenerated epithelial cells in the 1-week-old but not the 3-month-old calves. After infection, the total cell count in the bronchoalveolar lavage (BAL) fluid cells was increased. The results of BAV-3 isolation from BAL fluid were correlated with the detection of intranuclear inclusion bodies in the desquamated epithelial cells in the BAL fluid cells from the right caudal lobe but not in cells from the left caudal lobe. CD8+ T lymphocytes in the pneumonic lesion were found only in the 3-month-old infected calves. The difference in the immunopathologic reactions between the 1-week-old and the 3-month-old infected calves may be attributed to differences in immune system development.
Ten serotypes of bovine adenovirus have been isolated from cattle with a wide variety of diseases, including calf pneumonia, enteritis, conjunctivitis, and keratoconjunctivitis.5,7,10,15,21 Bovine adenovirus 3 (BAV-3) has been associated with respiratory infection and with respiratory/enteric diseases of cattle.12 Serologic studies have indicated that the infection rate is high in adult cattle in some herds.15,16 There have been few detailed immunohistopathologic analyses of the development of pneumonic lesions after experimental infection with adenoviruses. Endobronchial (EB) inoculation has not previously been used in the study of BAV-3 infection.
The purpose of this study was to evaluate the effects of EB inoculation of BAV-3 in 1-week-old and 3-month-old calves, with particular emphasis on bronchoalveolar lavage (BAL) fluid as an aid to immunohistochemical and virologic confirmation of BAV-3 infection.
Materials and Methods
Virus
BAV-3 was isolated from a calf in Japan that was exhibiting signs of pneumonia, coughing, and rhinitis. Following plaque purification, the virus was identified as BAV by biophysical characterization and as BAV-3 by a cross-neutralization test. After the virus was propagated in bottle cultures of Madin–Darby bovine kidney (MDBK) cells, aliquots of the virus were kept frozen (−80 C) until they were used as inoculum. Bovine viral diarrhea virus–free cultures were confirmed by methods described previously.22,23
Animals
Eight male Holstein calves were used in this experiment. Three 1-week-old calves (Nos. 1–3) and three 3-month-old calves (Nos. 4–6) were infected, and an additional 1-week-old calf (No. 7) and an additional 3-month-old calf (No. 8) were kept as uninfected controls. All eight calves had received colostrum within a few hours after birth and had neutralizing antibody against BAV-3 (as determined by cytopathic effect inhibition test) ranging from 1:2 to 1:32. The six infected calves were kept in six separate isolation pens throughout the experiment, and the two control animals were kept in two similar pens to prevent cross-infection.
Experimental procedure
Before inoculation with BAV-3, the calves were given atropine hydrochloride (0.02 mg/kg, intramuscularly) and anesthetized with xylazine (0.3 mg/kg, intravenously). Six calves (Nos. 1–6) were given 10 ml (103.0 TCID50/0.1 ml) of viral suspension by EB inoculation using a bronchoscope (Olympus type 10; Olympus, Tokyo, Japan). This instrument was inserted into the right main bronchus of the caudal lung lobe. The procedure was completed within 10 minutes for each calf. One calf from each age group was euthanatized by an intravenous overdose of barbiturate on each of 3 postinoculation days (PIDs 3, 5, and 7). Two control calves (Nos. 7, 8) were given 10 ml of noninfected maintenance medium by EB inoculation and were euthanatized on PID 5.
Clinical signs and body temperature for all calves were recorded at least twice daily during the experimental period.
Cytologic evaluation of BAL fluid
After removal of the lungs at necropsy, a cannula (4 mm diameter) was inserted into each bronchus of the right and left caudal lobes, and 30 ml of sterile phosphate-buffered saline was introduced and immediately withdrawn. The 20 ml of BAL fluid recovered was placed in a sterile bottle, and the number of nucleated cells per microliter of BAL fluid was counted using a Coulter counter. One slide prepared from the centrifuged cells was stained with a Diff-Quik kit (International Reagent Corp., Koube, Japan). Evaluation included a 200-cell differential count and a morphologic description of the cells.
Virus isolation
BAL fluid samples (1 ml) collected separately from the right and left caudal lobes were centrifuged (3,000 rpm), and 1 ml of maintenance (growth) medium without bovine serum was added to each cell pellet. The cells were then stored at −80 C until used. Samples were subjected to ultrasonic disruption for 20 seconds at 4 C, and a 0.1-ml volume of decimal dilution of each sample was inoculated onto confluent cultures of MDBK cells in a 96-well microtiter plate. The cells were examined daily for 1 week for cytopathogenic effect to determine the presence and titer (TCID50) of BAV-3.
Pathologic and immunohistochemical examination
All calves were necropsied. Samples of lungs, trachea, mediastinal lymph node, kidney, liver, thymus, spleen, intestine, brain and trigeminal ganglia were collected and fixed in buffered 10% formalin. A total of 33 lung samples were collected, three from each of the right and left cranial lobes, three from each of the right and left middle lobes, nine from each of the right and left caudal lobes, and three from the accessory lobe. All specimens were embedded in paraffin, and sections were cut and stained with hematoxylin and eosin (HE).
BAV-3 antigens in 33 formalin-fixed, paraffin-embedded lung tissues and CD4+ and CD8+ T lymphocytes in cryostat sections from three parts of the pneumonic lesions from the right caudal lobe were demonstrated by the strept–avidin–biotin complex immunoperoxidase (SAB-IP) method18 using an SAB kit (Nichirei Corp., Tokyo, Japan). Anti–BAV-3 rabbit serum was used as the primary antibody at a dilution of 1:8,000. Anti-bovine CD4 (VMRD, Pullman, WA) and anti-bovine CD8 (VMRD) monoclonal antibodies were used at ratios of 1:200 and 1:1,600, respectively. Samples were counterstained with methyl green. Tissue sections from the uninfected controls (calf Nos. 7, 8) and sera from nonimmunized rabbits and mice were used as controls.
Samples of the pneumonic lesions from the right caudal lobe of each calf were prepared for electron microscopy by postfixing them in 1% osmium tetroxide and then dehydrating and embedding them in an Epon mixture. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with a TEM-100CX (JEOL) electron microscope.
Results
Clinical and macroscopic findings
The two control calves were clinically normal and had no gross lesions at necropsy.
All of the infected calves had mildly elevated rectal temperatures (up to 39.5 C) at 1–3 days after infection. Respiratory signs such as sneezing, coughing, and nasal discharge were not observed in any of the infected calves. The gross pneumonic lesions in the experimental calves (Nos. 1–6) consisted of dark red, slightly depressed, irregular areas of consolidation and were located primarily in the middle to caudal parts of the right caudal lobe (Fig. 1). That area corresponded well with the area used for EB inoculation. The bronchial and mediastinal lymph nodes were always pale, moderately enlarged, and congested.
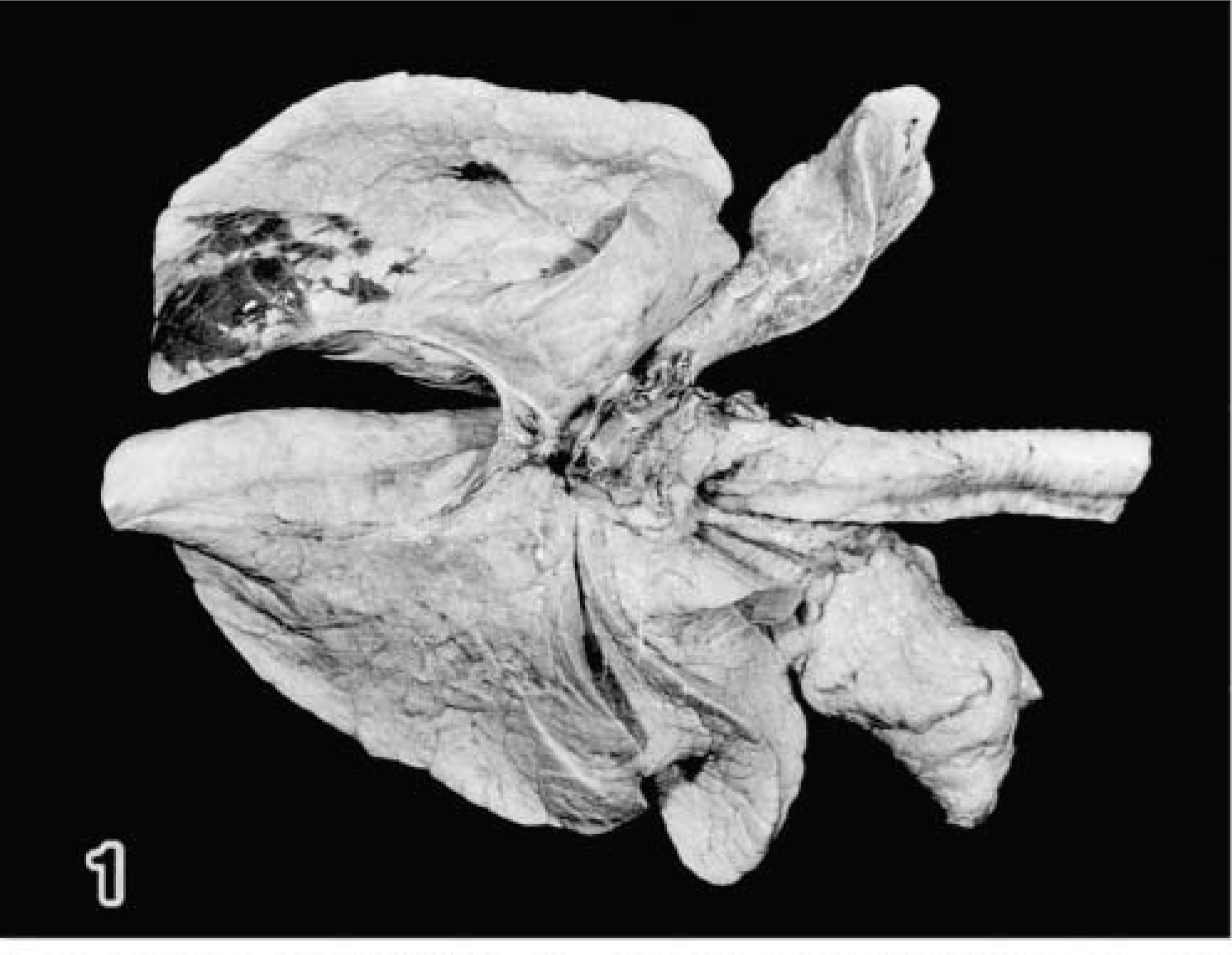
Lung; calf No. 2. Pneumonic lesion is localized in the middle to caudal parts of the right caudal lobe.
BAL fluid examination
Total cells counts in the BAL fluid of the right caudal lobe were higher than those of the left caudal lobe in the infected calves and higher than those in the noninfected control calves (Table 1). Relative to the control calf values, the neutrophil and other cell values for the infected calves, including values for desquamated epithelial cells, were high at all time points after infection. In the three younger calves (Nos. 1–3), many eosinophilic and basophilic intranuclear inclusion bodies were detected in the desquamated epithelial cells of the BAL fluid from the right caudal lobe (Fig. 2) but not from the left caudal lobe. The three older infected calves (Nos. 4–6) had no inclusion bodies in BAL fluid cells from either the left or the right caudal lobe.
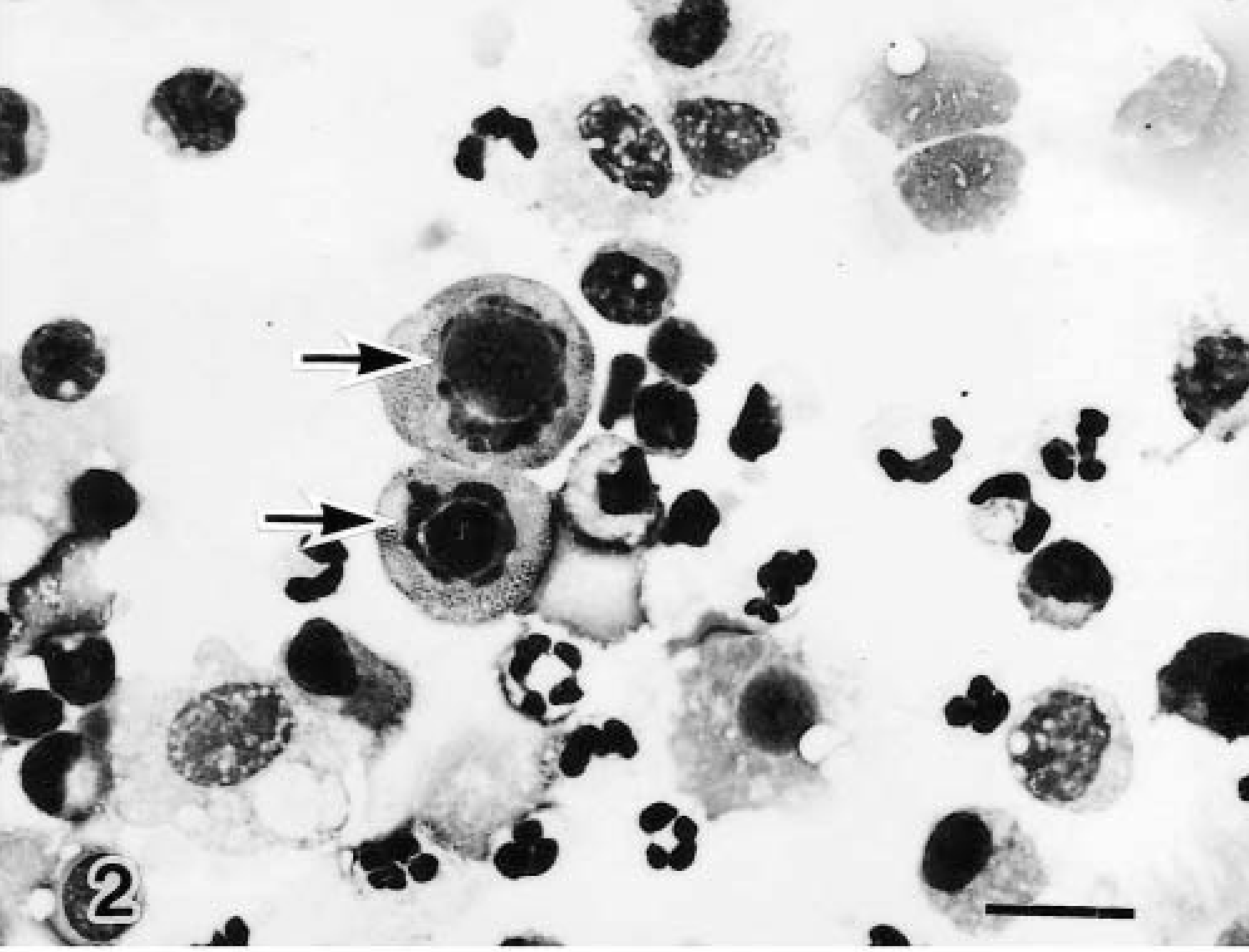
Bronchoalveolar fluid cells, right caudal lobe; calf No. 1. Eosinophilic intranuclear inclusion bodies (arrow) are visible in the desquamated epithelial cells. Diff-Quik. Bar = 15 μm.
Cytologic and virologic findings in bronchoalveolar lavage fluid from calves experimentally infected with BAV-3.
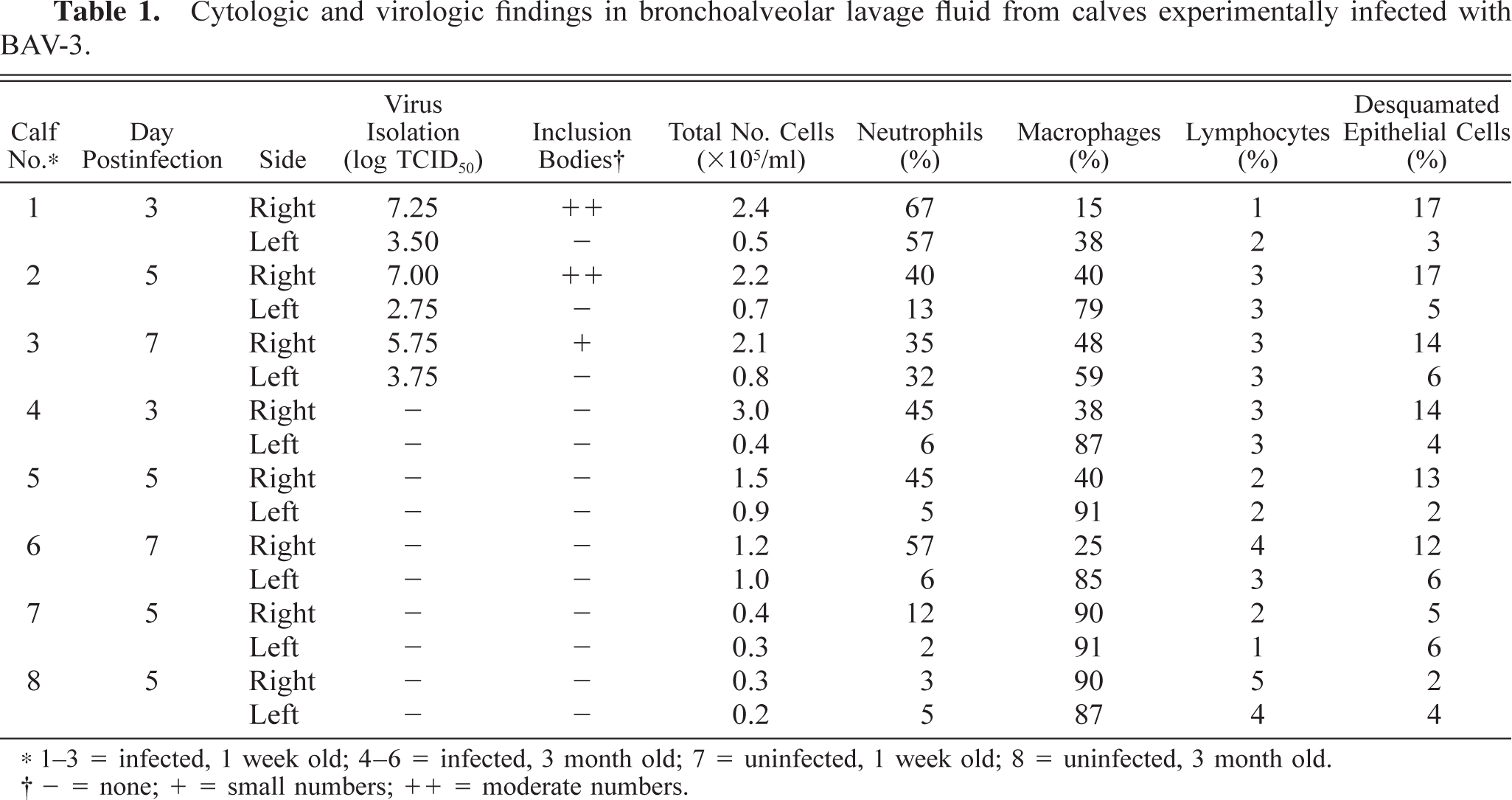
1–3 = infected, 1 week old; 4–6 = infected, 3 month old; 7 = uninfected, 1 week old; 8 = uninfected, 3 month old.
– = none; + = small numbers; ++ = moderate numbers.
BAV-3 was isolated consistently from BAL fluid cells from the right and left caudal lobes of the three 1-week-old calves but not from the three 3-month-old calves. The highest viral titer was 105.75 TCID50/ml (on PID 3; Table 1). No BAV-3 was cultivated and no viral inclusion bodies were demonstrated in the two control calves.
Histopathology
The 1-week-old infected calves had bronchitis, bronchiolitis, and alveolitis characterized by accumulations of neutrophils and necrotic epithelial cells containing intranuclear inclusion bodies.
On PID 3, one infected calf (No. 1) showed moderate to marked changes in the bronchiolar mucosa, including a mild infiltration of neutrophils and multifocal necrosis of individual epithelial cells. Many of the epithelial cells contained eosinophilic viral intranuclear inclusion bodies in the most superficial layers and in the airway lumen (Fig. 3). On PIDs 5 and 7, the epithelium lining of terminal and respiratory bronchioles was completely eroded in two calves (Nos. 2, 3), and the bronchiolar lumen contained necrotic debris, fibrin, macrophages, and neutrophils. There were aggregates of macrophages, lymphocytes, and neutrophils around the bronchioles. In pneumonic areas of two calves (Nos. 2, 3), there were increased numbers of type 2 pneumocytes (Fig. 4), and some alveolar cells contained basophilic intranuclear inclusion bodies (Fig. 5). Collections of macrophages, lymphocytes, plasma cells, and occasional neutrophils were found in alveolar spaces and alveolar septa.
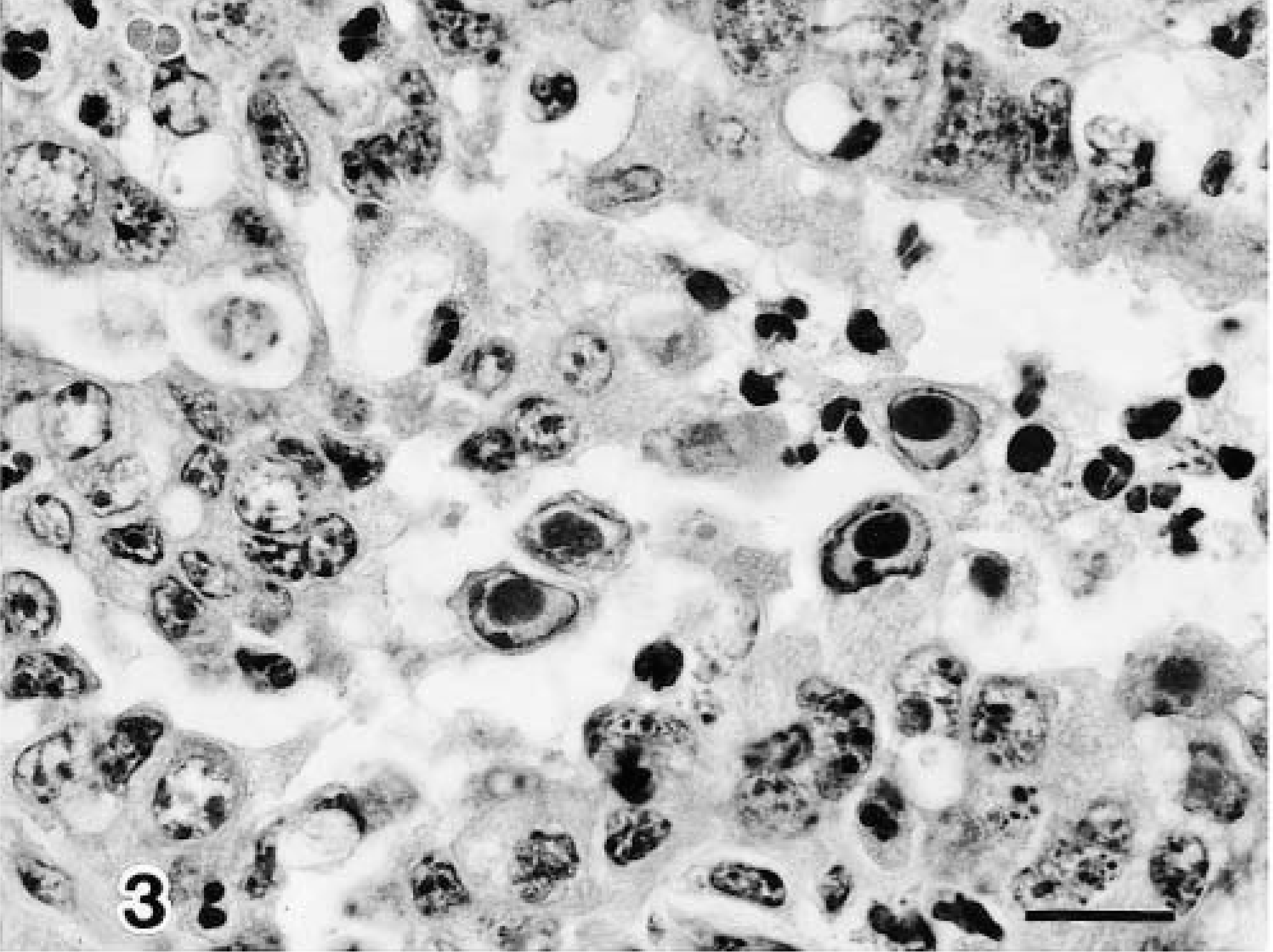
Lung; calf No. 1. Degenerated bronchiolar epithelial cells contain eosinophilic viral inclusion bodies. HE. Bar = 15 μm.
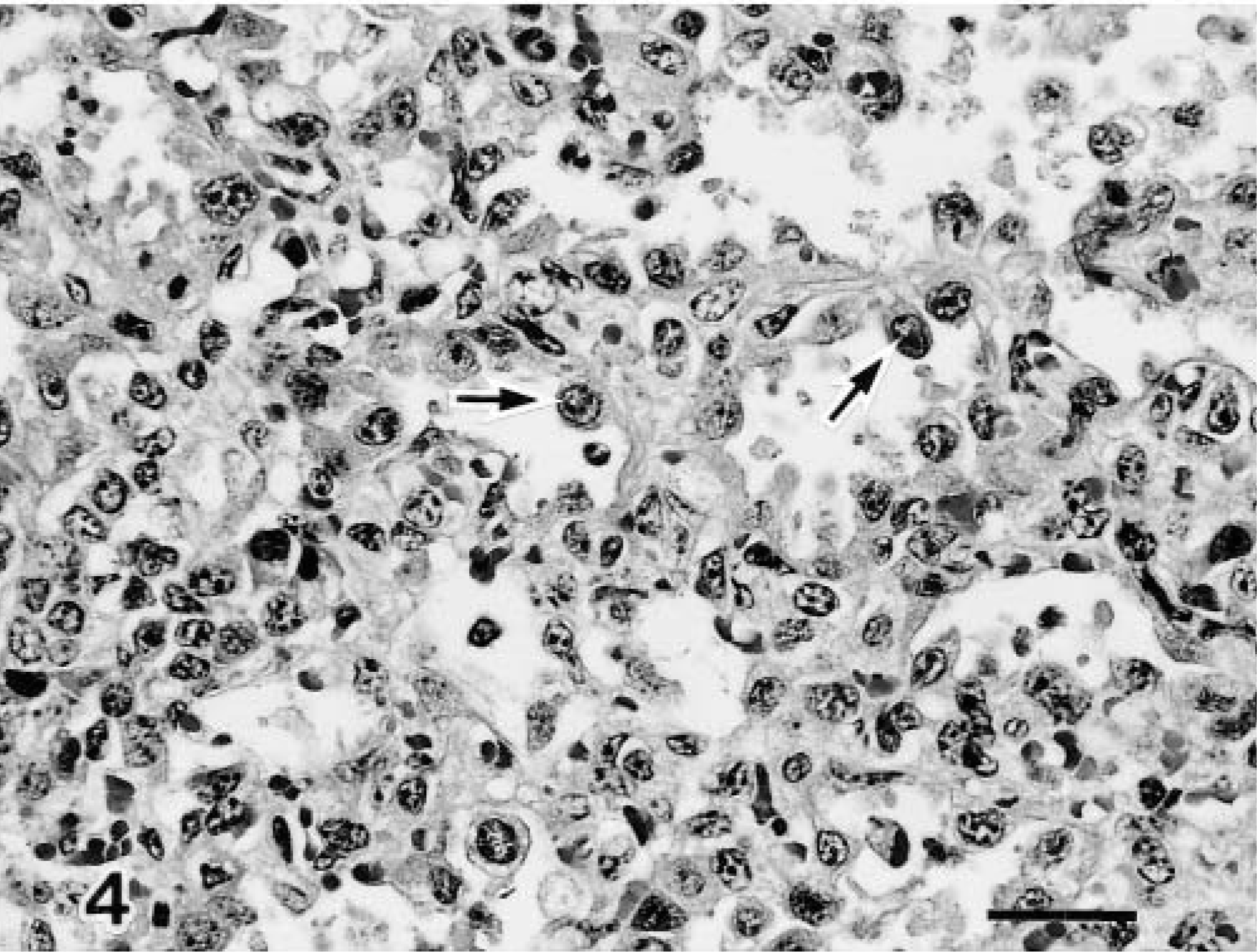
Lung; calf No. 2. Increased numbers of type 2 pneumocytes with lymphocyte infiltration are visible in the pneumonic areas (arrows). HE. Bar = 50 μm.
Lung; calf No. 3. Some alveoli contain basophilic intranuclear inclusion bodies(arrow). HE. Bar = 15 μm.
In the 3-month-old infected calves, both moderate proliferative bronchitis and bronchiolitis occurred, characterized by epithelial hyperplasia and severe infiltration of leukocytes with thickening of the alveolar wall. The bronchiolar epithelium and alveolar epithelium had desquamated in a few sites but did not have intranuclear inclusion bodies.
In addition to pneumonic lesions, most of the infected calves had a mild suppurative lymphadenitis in the bronchial and mediastinal lymph nodes. Neither gross nor histologic lesions were recognized in the nasal cavity, nasal–pharyngeal tonsil, brain, or small intestine of any of the six animals. No significant histologic lesions were observed in the control calves.
Immunohistochemistry
BAV-3 antigen was detected in the bronchial, bronchiolar, and alveolar epithelial cells in the 1-week-old calves (Fig. 6) but not in the 3-month-old calves, and the distribution of this antigen was correlated with the presence of the intranuclear inclusion bodies. BAV-3 antigen in the bronchiolar epithelial cell was decreased in calves examined at PID 7 but was detected in some alveolar cells (Table 2). No viral antigen was detected in any other organs in the six infected or two control calves.
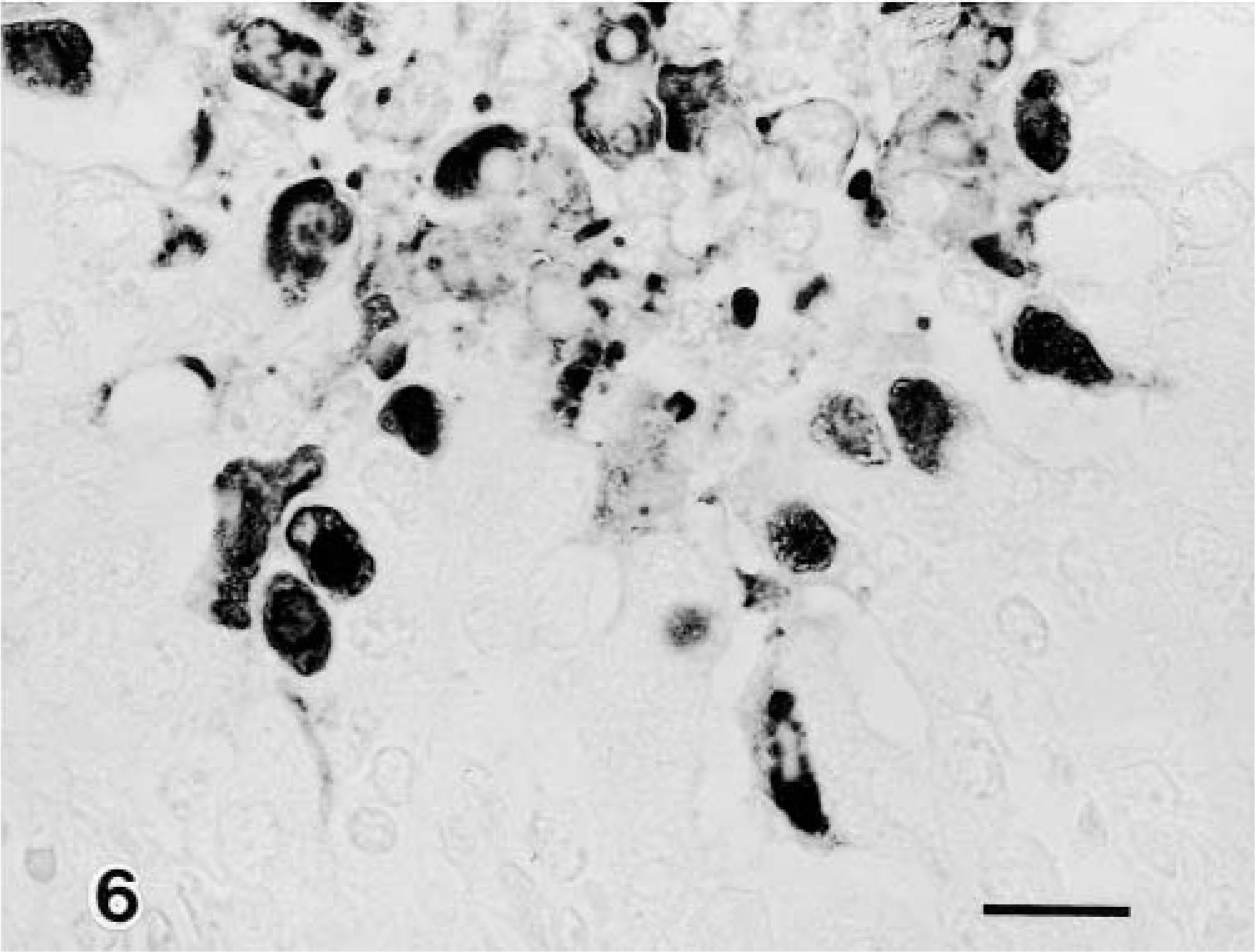
Lung; calf No. 1. BAV-3 antigen is visible in the bronchiolar epithelial cells. SAB-IP. Bar = 25 μm.
Distribution of histologic lesions, BAV-3 antigen, and T lymphocytes in the lungs of calves experimentally infected with BAV-3.
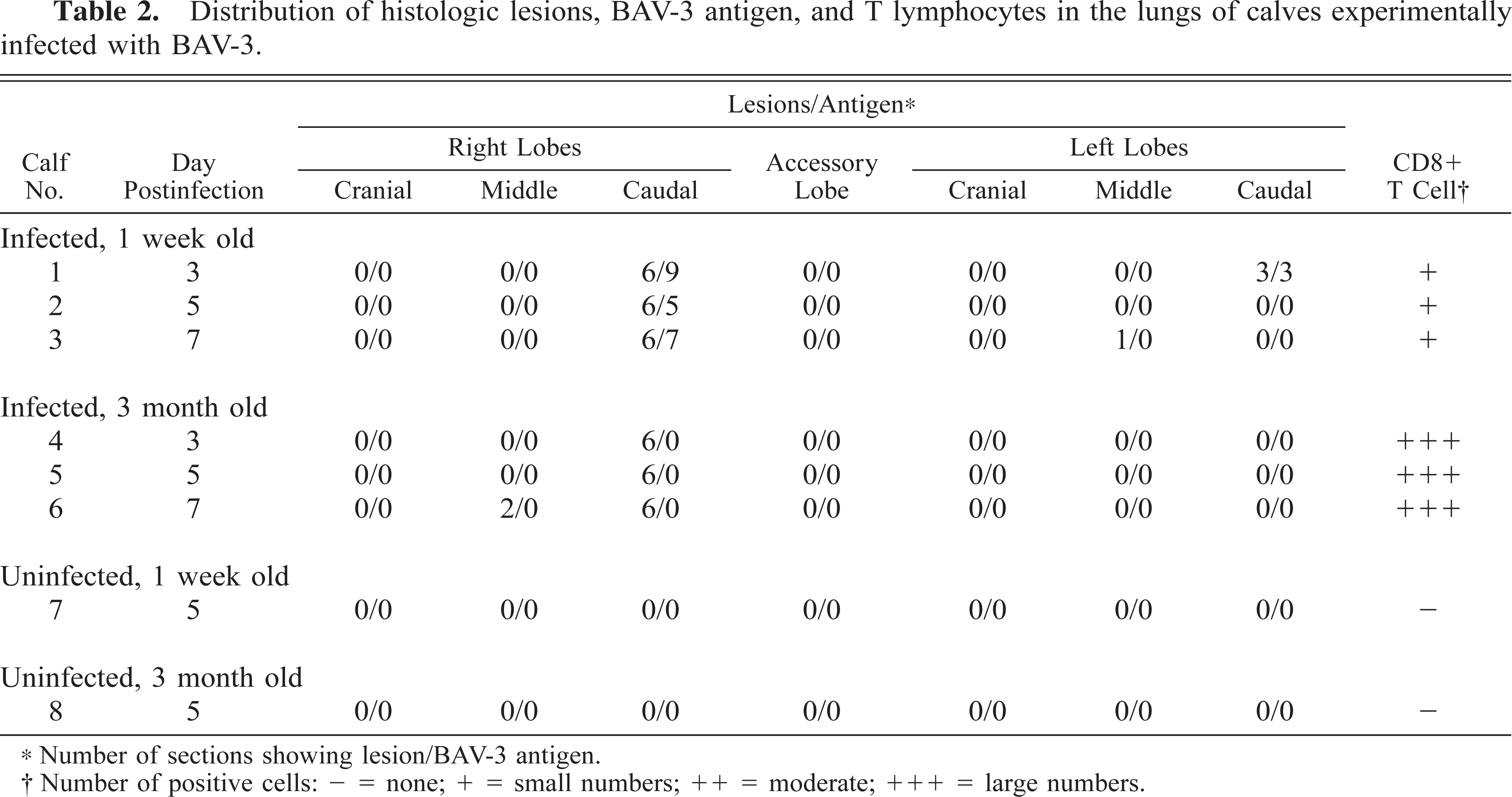
Number of sections showing lesion/BAV-3 antigen.
Number of positive cells: – = none; + = small numbers; ++ = moderate; +++ = large numbers.
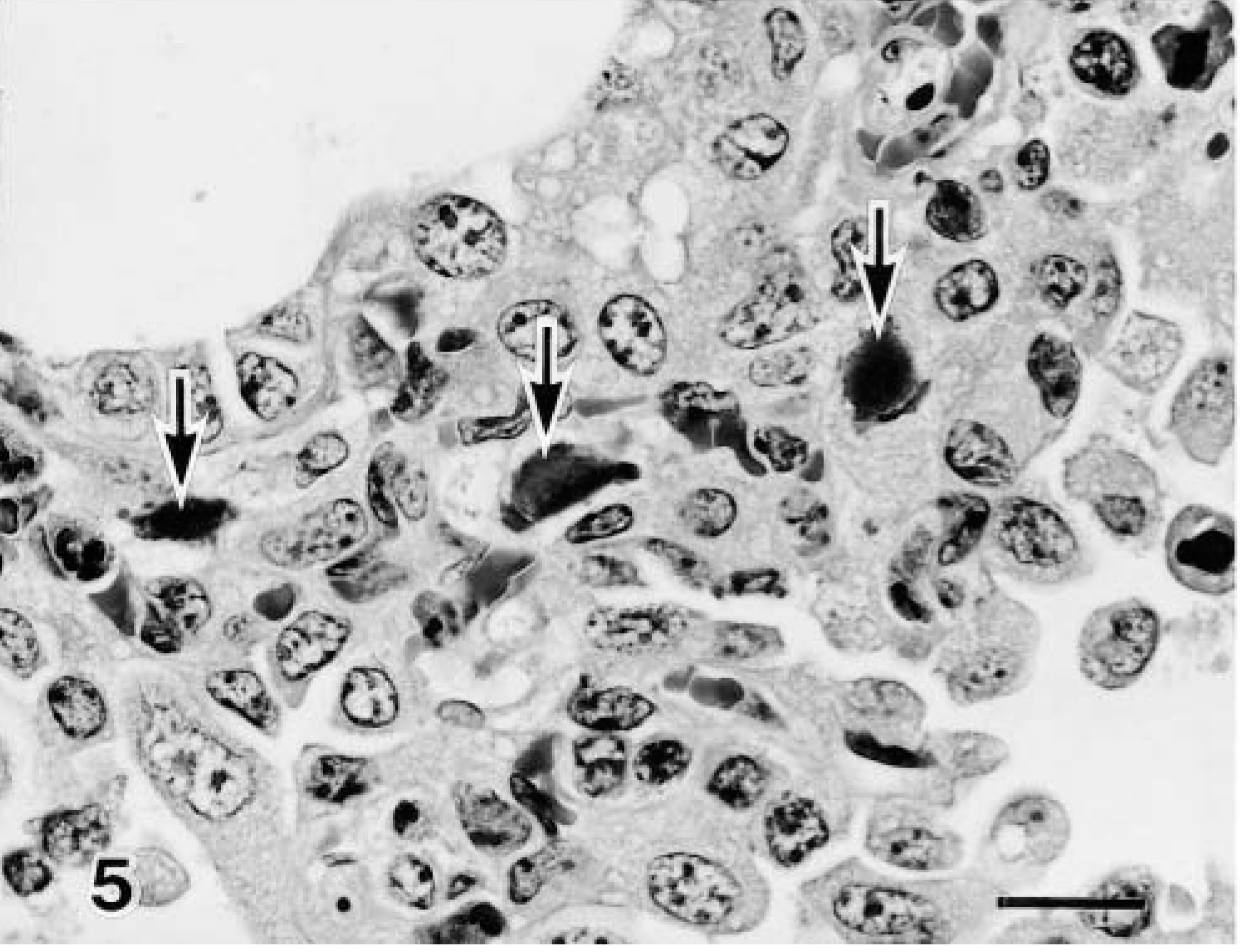
There were more CD8+ T lymphocytes in the pneumonic lesions in the 3-month-old calves than in the 1-week-old calves (Fig. 7). Low numbers of CD4+ T lymphocytes were detected in the pneumonic lesions, but the number of these lymphocytes did not differ between the 1-week-old and the 3-month-old infected calves. Neither CD8+ nor CD4+ T lymphocytes were present in the two noninfected control calves (Table 2).
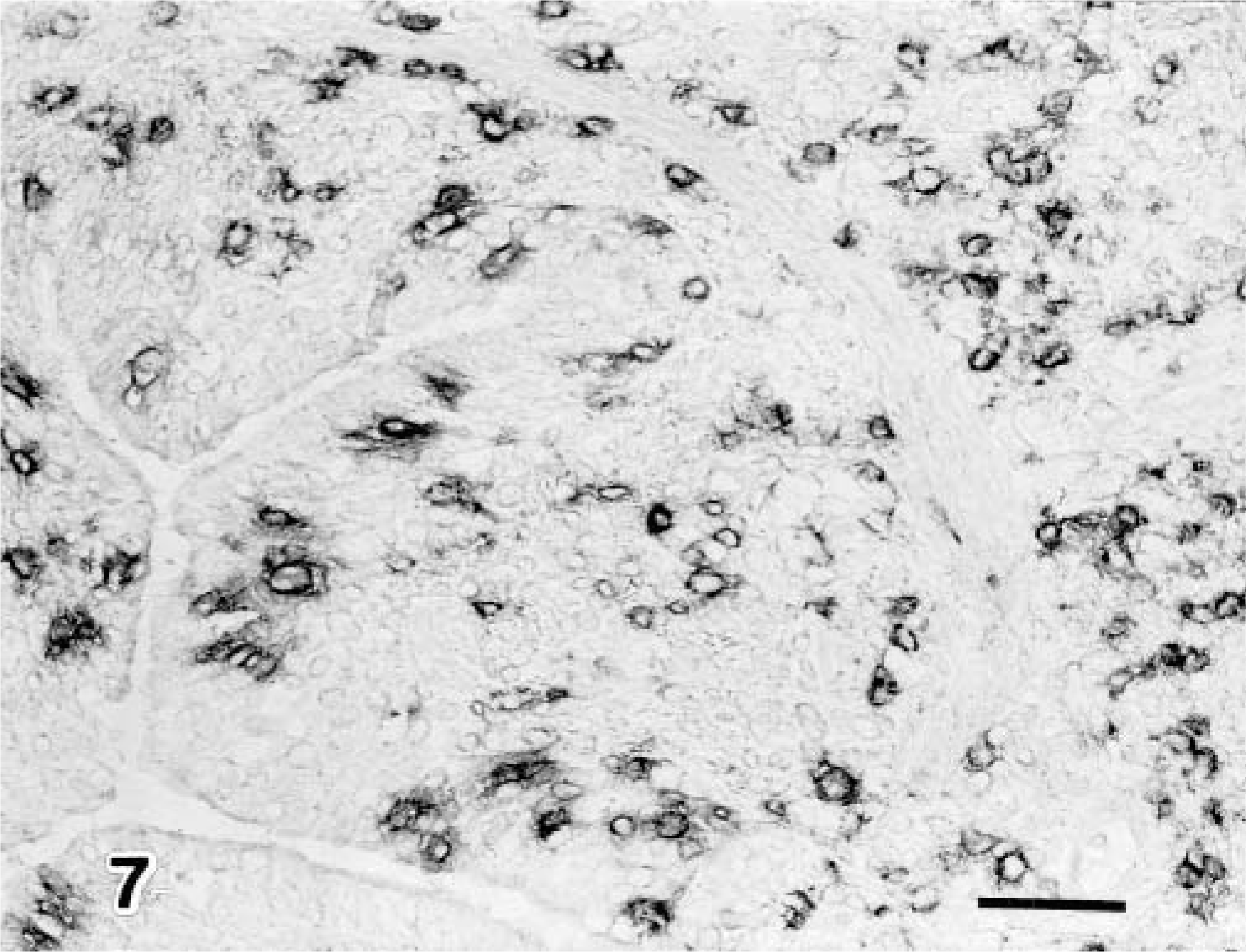
Lung; calf No. 5. Many CD8+ T lymphocytes are visible in the bronchus associated with the pneumonic lesion. SAB-IP. Bar = 50 μm.
Electron microscopy
Viral particles were present on the ciliated bronchiolar epithelial cells and the desquamated ciliated cells (Fig. 8). Viral particles 65 nm in diameter were arranged in the nuclei of epithelial cells in crystalline arrays along with more finely granular material of low electron density (Fig. 9). Viral assembly and release were associated with loss of the nuclear envelope and dispersion of assembled virions into the cytoplasm.
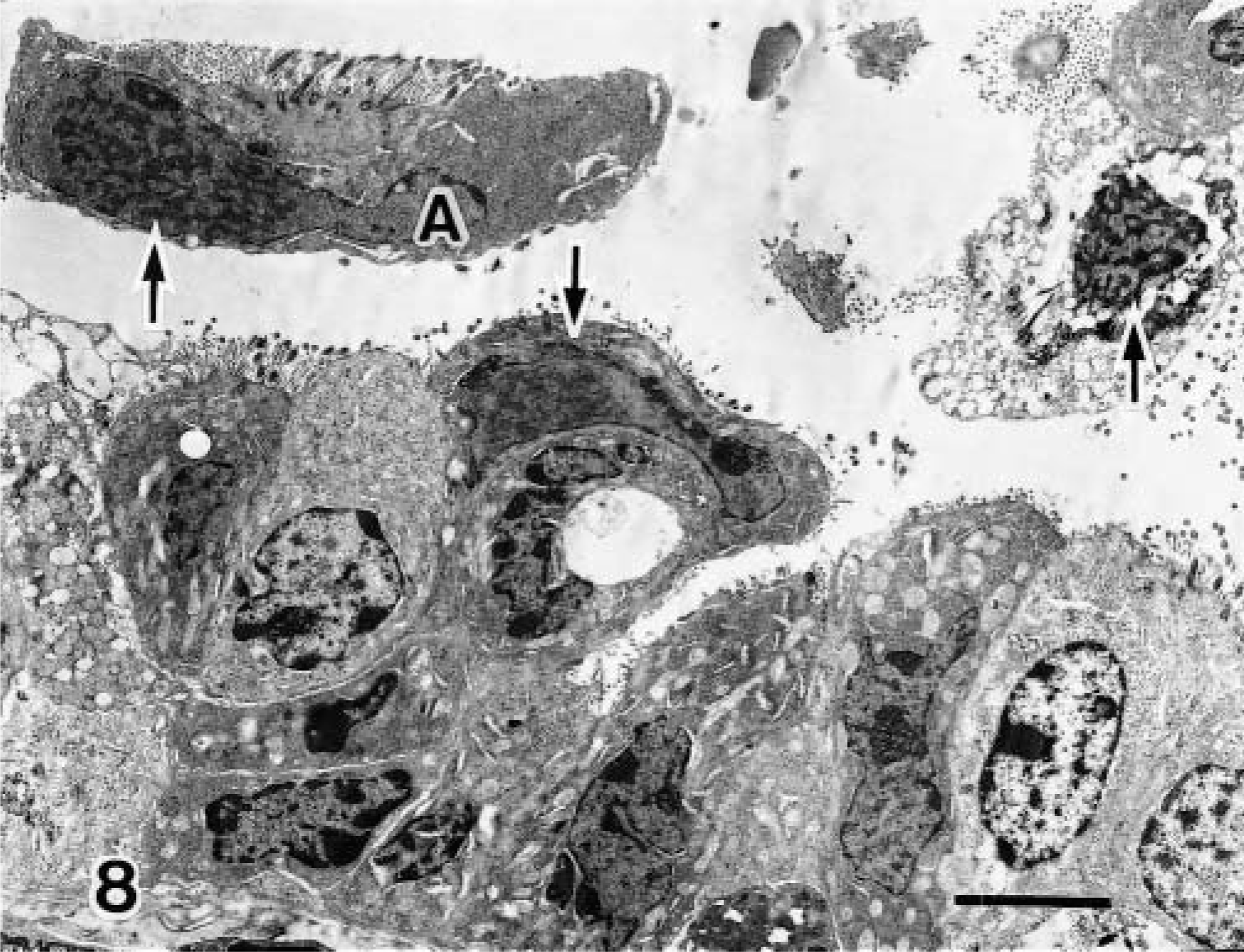
Transmission electron micrograph. Lung; calf No. 1. Viral inclusion bodies (arrows) are visible in the ciliated bronchiolar epithelial cells and desquamated ciliated cells. Bar = 1 μm.
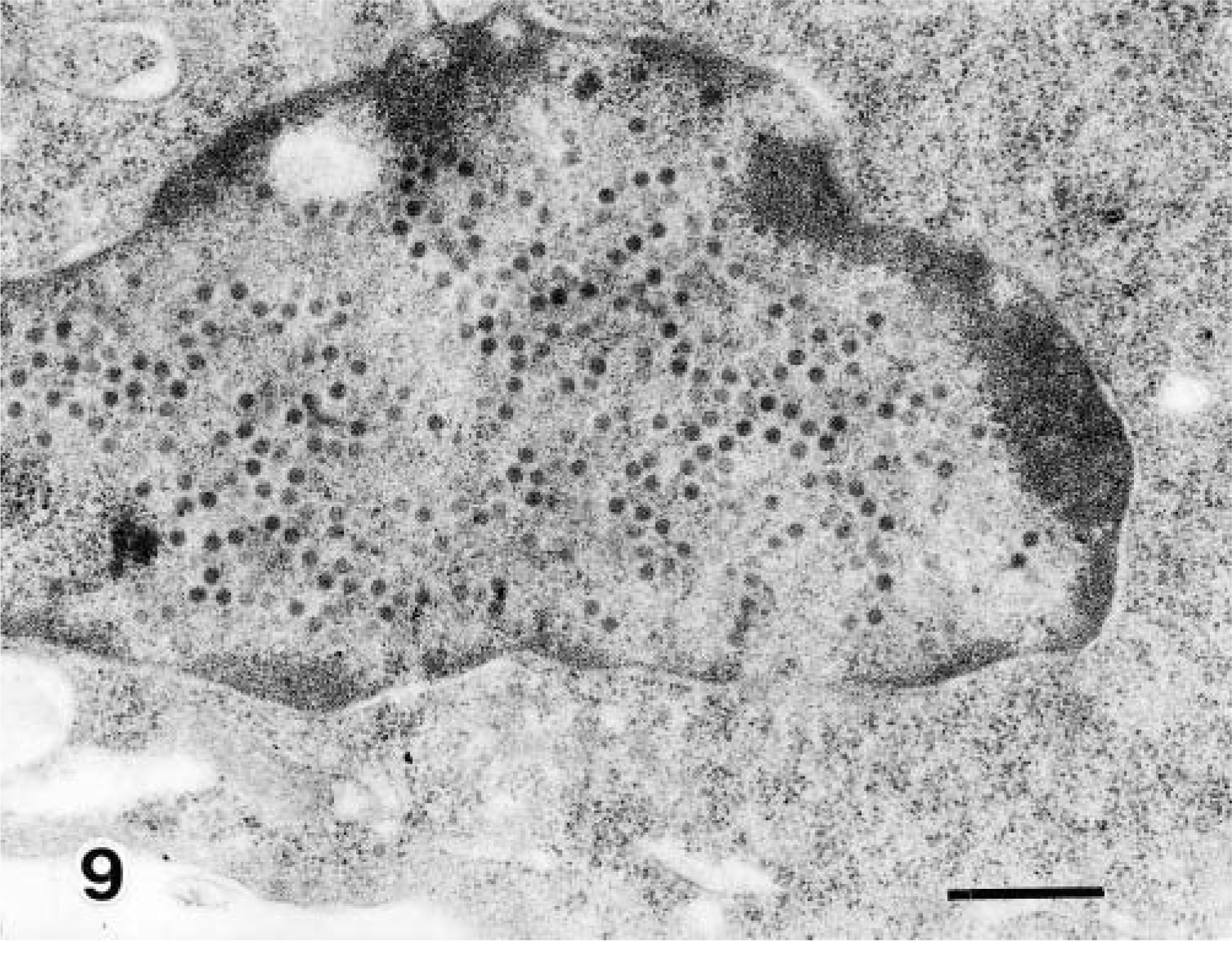
Transmission electron micrograph. Lung; calf No. 1. Higher magnification of cell A in Fig. 8. Viral particles are loosely arranged. Bar = 500 nm.
Discussion
The previous studies, calves 1.5–4 months of age inoculated with BAV developed no demonstrable clinical or histopathologic evidence of infection.5,7,10 Colostrum-deprived calves inoculated intratracheally and/or intranasally on the day of birth with BAV-3 showed mild clinical signs and had moderate pneumonia.6 In the present study, the clinical signs evoked by EB inoculation with BAV-3 were mild in both experimental groups of calves. The pneumonic lesions produced in the lungs of the 1-week-old and 3-month-old calves were localized primarily at the site of inoculation in the right caudal lobe. Macroscopic observation revealed that BAV-3 infection induced pulmonary changes in 1-week-old and 3-month-old calves, even though these calves had received colostrum. Therefore, EB inoculation, which does not mimic the natural route of exposure of the agent, would seem to be an efficient method for studying the pathogenesis of BAV-3 and other respiratory infectious agents.19,20
The lesions of adenoviral respiratory diseases in naturally and experimentally infected cattle5,6,10,12,14–16 and in dogs, goats, and other species2,3,8,11,13,17 have included severe necrotizing bronchitis with necrosis of epithelial cells and intranuclear inclusion bodies. In the present study, the pneumonic lesions in calves infected with BAV-3 included moderate to severe necrotizing bronchitis, bronchiolitis, and alveolitis, characterized by the infiltration of inflammatory cells and proliferation of type 2 epithelial cells. Corresponding to the detection of the intranuclear inclusion bodies in the 1-week-old infected calves, abundant BAV-3 antigen was detected in the degenerating bronchial, bronchiolar, and alveolar epithelial cells in the right caudal lobe. BAV-3 particles were also found in the epithelium of the bronchi and bronchioli in the 1-week-old infected calves but not in the 3-month-old calves. The pathologic pattern these 1-week-old BAV-3–infected calves is similar to the infection patterns observed for other adenoviruses. Moreover, the reaction in the 3-month-old BAV-3–infected calves closely resembled the reactions in colostrum-deprived calves to infection with BAV-1, BAV-2, and trypsin-treated BAV-3.5–7
Examination of BAL fluid can provide information on the pathophysiology of respiratory infectious diseases.1,4,9,19,20 In the present study, the total cell count in BAL fluid from the right caudal lobe increased after BAV-3 infection, and the cells consisted of neutrophils and desquamated epithelial cells containing intranuclear inclusion bodies. Moreover, in the lower respiratory tracts of the 1-week-old calves, the results of BAV-3 isolation from BAL fluid cells were correlated with the detection of intranuclear inclusion bodies in the desquamated epithelial cells in BAL fluid. Thus, examination of BAL fluid cells is valuable for the diagnosis of BAV-3 infection and other respiratory infectious diseases.19,20
CD8+ cells are part of the inflammatory response to infection.11 Adenovirus 7 pneumonia in children is associated with a marked increase in activated peripheral blood CD8+ T cells and interferon-γ–producing CD3+ T cells.13 In the present study, the 3-month-old calves infected with BAV-3 had much higher numbers of CD8+ T lymphocytes in the pneumonic lesions, but their lungs contained no demonstrable BAV-3 antigen. No reaction of T lymphocytes in the lungs was found in the 1-week-old infected calves or in either of the control calves. These results suggest that increased CD8+ T lymphocytes in the pneumonic lesion is an important immunologic parameter for the host defense against BAV-3 infection in calves. Therefore, the different immunopathologic reactions in the 1-week-old and the 3-month-old infected calves may be attributed to the difference in immune system development.
Footnotes
Acknowledgements
We thank M. Kobayashi and M. Shimada for preparing the pathologic sections and Dr. Y. Ando and T. Fujisawa for preparing the photographs.
