Abstract
The expression of cytotoxic molecules in feline intestinal T-cell lymphoma cells was examined immunohistochemically using endoscopic samples of 50 cases. Cases included 14 large-cell lymphomas (LCLs) and 36 small-cell lymphomas (SCLs). Most LCL and some SCL exhibited marked erosion and villous atrophy. Clonal T-cell receptor (TCR) gene rearrangement was detected in 10/14 (71%) LCL cases and 33/36 (92%) SCL cases. No clonal immunoglobulin heavy chain (IgH) gene rearrangement was detected. Immunohistochemically, all cases were positive for CD3 and negative for CD79α, CD30, CD56, and Foxp3. LCLs were positive for CD8 in 13/14 cases (93%), T-cell intracellular antigen 1 (TIA1) in 14/14 cases (100%), and granzyme B in 6/14 cases (43%). SCLs were positive for CD8 in 28/36 cases (78%), TIA1 in 33/36 cases (92%), and granzyme B in 2/36 cases (6%). TIA1- and granzyme B-positive neoplastic lymphocytes were predominantly observed in the mucosal epithelium of 10/50 cases (20%) and 6/50 cases (12%), respectively. No significant differences in survival time were found based on cell size or epitheliotropism. However, cases with TIA1+ and/or granzyme B+ neoplastic lymphocytes predominantly in the mucosal epithelium had significantly shorter survival times (P < .05), suggesting that mucosal epithelium infiltration of neoplastic cells with a cytotoxic immunophenotype is a negative prognostic factor. Therefore, intraepithelial cytotoxic lymphocytes may be associated with mucosal injury and impaired intestinal function, leading to a poor prognosis in cats with intestinal T-cell lymphoma.
Lymphoma is the most common neoplasm of the feline intestines, most cases of which are T-cell lymphoma. 32 In veterinary clinical practice, endoscopic examinations are commonly performed to diagnose intestinal lymphomas, which are classified into large-cell lymphoma (LCL) and small-cell lymphoma (SCL). 32 In a previous study, the median survival time of LCL cases was approximately 1.5 months and that of SCL was 28 months. 32 However, the relationship between the properties of neoplastic lymphocytes and survival times remains unclear in feline intestinal T-cell lymphoma.
In the World Health Organization (WHO) classification, human intestinal T-cell lymphomas are classified into enteropathy-associated T-cell lymphoma (EATL), monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL), and indolent T-cell proliferative disorder of the gastrointestinal tract (ITLPD). 44 Histologically, the neoplastic lymphocytes of EATL are large and polymorphic, MEITL are medium and monomorphic, and ITLPD are small and monomorphic.2,5,7,9,12,29,35,44,46 Both EATL and MEITL exhibit cytotoxicity and epitheliotropism and have poor prognoses.2,5,7,9,12,29,44 ITLPD exhibits scant epitheliotropism and no cytotoxic immunophenotype and has a better prognosis.35,46 Therefore, we hypothesized that epitheliotropism and cytotoxicity are related to the survival times in feline intestinal T-cell lymphoma.
Cytotoxic T-cells express one or more cytotoxic granules, such as T-cell intracellular antigen 1 (TIA1), granzyme B, and perforin. 45 Perforin and granzyme B induce apoptosis of target cells through cell death pathways.18,47 TIA1 regulates expression of genes that are involved in the cell death pathways.1,13,45 Granzyme B is expressed in activated cytotoxic T-cells, whereas TIA1 is expressed in resting and activated T-cells. 19
The aim of this study is to clarify the immunophenotypes of feline intestinal lymphomas and examine the association between the expression of cytotoxic granules in neoplastic lymphocytes and survival times. Furthermore, we discussed the similarities and differences of immunophenotype and its prognostic relevance between feline and human intestinal T-cell lymphomas.
Materials and Methods
Cases
Intestinal tissue specimens endoscopically collected from 50 cats at Japan Small Animal Medical Center (JSAMC) between 2018 and 2020 were examined. All cases were diagnosed as intestinal T-cell lymphoma based on the morphological examination of hematoxylin and eosin (HE) stained sections and immunohistochemical results.26 Endoscopic tissue samples were collected from the proximal duodenum, distal duodenum, and ileum. All cats had histories of long-term gastrointestinal signs such as vomiting, diarrhea, and weight loss. The feline chronic enteropathy activity index (FCEAI) was used to measure enteritic disease activity. 24 Survival data were obtained and cases followed until April 2021, except for case 45. Treatments were relatively homogenous, and chlorambucil and/or prednisolone were administered in most cases in accordance with a previous report.42
Clonality Analysis
In all cases, frozen samples were examined for T-cell receptor (TCR) and immunoglobulin heavy chain (IgH) gene rearrangement as previously described.16,33 Tissue samples were endoscopically collected from the duodenum or ileum and stored at –20°C until use. A clonal population was defined by the presence of distinct peaks of fluorescence intensity at least 5-fold higher than other background peaks. Reproducible monoclonal or oligoclonal peaks detected in either the duodenal or ileal sample were considered positive.
Histopathology
Mucosal tissue samples were placed on filter paper with the villous side upward to make a vertical section. A minimum of 6 tissue specimens were collected per region. Formalin-fixed and paraffin-embedded tissues were sectioned at 3-μm thickness and stained with HE. Tissue specimens used for histopathological evaluation included the entire depth of the intestinal mucosa and at least 3 villi.
Histopathological diagnosis was performed based on the consensus of 3 veterinary pathologists including Japanese College of Veterinary Pathologists (JCVP) diplomates (TI, KU, and JKC). Because all samples in this study were endoscopic samples, transmural spread was impossible to assess, resulting in a default diagnosis of mucosal lymphoma. Lymphomas were classified by their nuclear size: LCL, nucleus larger than twice the diameter of a red blood cell; SCL, nucleus smaller than twice the diameter of a red blood cell. Epitheliotropism in lymphoma was scored by the percentage of lymphocytes infiltrating mucosal epithelium: 0, almost none (0%–5%); 1, sparse infiltration and partial aggregates (5%–80%); 2, extensive infiltration replacing the epithelium (80%–100%). The scoring criteria of mucosal epithelial injury and villous atrophy were listed in Supplemental Table S1.
Immunohistochemistry
The reactivities of CD8, granzyme B, CD30, CD56, and FOXP3 were confirmed in the following articles.4,21,27,32,39 Sequence alignment of human and feline TIA1 was obtained from The Universal Protein Resource (UniProt) (Supplemental Fig. S1). In all cases, serial sections were subjected to immunohistochemistry using the primary antibodies listed in Supplemental Table S2. The antibodies were validated by a positive reaction in feline lymph node, tonsil, and brain tissues and negative reaction by omitting the primary antibody. After reaction with the primary antibodies, the sections were incubated with the Envision polymer (Dako), Optiview DAB detection kit (Roche Diagnostics), or biotinylated rabbit anti-rat IgG (Funakoshi) followed by peroxidase-conjugated streptavidin.
Diffuse or aggregated positivity of neoplastic lymphocytes was considered positive; the result was considered negative when less than 5% of neoplastic lymphocytes were positive. Proliferation activity was measured by counting Ki-67-positive cells per 500 neoplastic lymphocytes.
Western Blotting
Western blotting analysis was performed for TIA1 antibody validation. Total protein was extracted from feline and canine tonsil using RIPA lysis buffer (Santa Cruz Biotechnology). Forty micrograms of protein samples diluted (1:4) with a loading buffer containing dithiothreitol were electrophoresed on a 5%–20% polyacrylamide gel (e-PAGEL; ATTO) at 200 V, 300 mA, for 50 minutes. Protein bands were transferred to a polyvinylidene fluoride membrane (Immobilon-P Transfer Membrane; Millipore) at 25 V, 500 mA, for 25 minutes. After 1% skim milk blocking, the anti-TIA1 antibody was applied. The enhanced chemiluminescence detection system (GE Healthcare) and ChemiDoc XRS+ system (Bio-Rad Laboratories) were used for visualization.
Statistical Analysis
All statistical analyses were conducted with Excel 2016 software. The data distribution was first assessed using the chi-square goodness-of-fit test.
As the distribution of tested data did not follow a normal distribution, the Mann-Whitney U-test was applied. The survival times were compared by cell size, epitheliotropism, and immunophenotype of neoplastic lymphocytes using the log-rank tests. The level of significance was set at P < .05.
Results
Breeds
The breeds of 50 lymphoma cases were mixed breed (n = 28), American shorthair (n = 6), Russian blue (n = 3), Japanese domestic cat (n = 2), American curl (n = 2), Chinchilla Persian (n = 1), Scottish Fold (n = 2), Ocicat (n = 1), Bengal (n = 2), and unknown (n = 3). There were 26 males and 23 females, which all cases were neutered. The mean ages of cats with LCL and SCL were 156.4 months and 141.7 months, respectively. The breed, sex, and age in one case were unknown. All data were summarized in Supplemental Table S3.
Histopathology
Of the 50 cases, 14 were LCL (28%) and 36 were SCL (72%) (Fig. 1a, b). LCL neoplastic lymphocytes had clear abundant cytoplasm and demonstrated anisocytosis and anisokaryosis. The nuclei were medium or large (>2 red cells) and pleomorphic. Chromatin was stained lightly, and the nucleolus was distinct. Most LCL cases exhibited severe mucosal injury (score 0, 1/14 (7.1%); score 1, 5/14 (35.7%); score 2, 8/14 (57.1%)) and villous atrophy (score 0, 0/14 (0%); score 1, 4/14 (28.6%); score 2, 10/14 (71.4%)). Neoplastic lymphocytes infiltrated the lamina propria, and marked epitheliotropism was observed in the villi and/or crypts (score 0, 1/14 (7.1%); score 1, 2/14 (14.3%); score 2, 11/14 (78.6%)) (Fig. 1c–e).
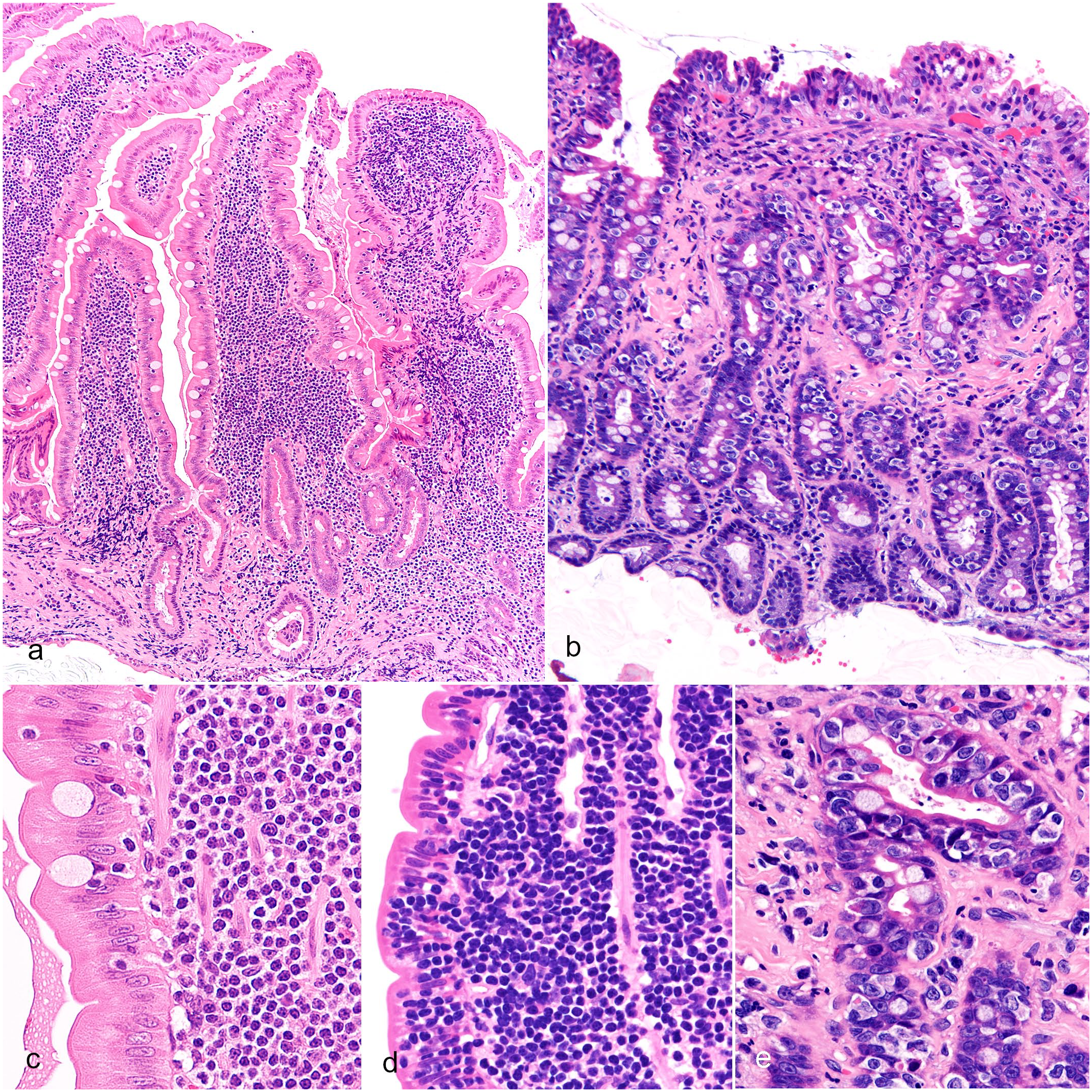
Intestinal T-cell lymphoma, duodenum, cat. Hematoxylin and eosin (HE). Mucosal tissue samples were placed with the villous side upward on filter paper. (a) Small-cell lymphoma (SCL). The villi are preserved, and neoplastic lymphocytes are small, case 1. (b) Large-cell lymphoma (LCL). Marked erosion with mucosal atrophy and infiltration of large neoplastic lymphocytes, case 41. (c) SCL. Epitheliotropism of neoplastic lymphocytes is nearly absent (score 0), case 1. (d) SCL. Neoplastic lymphocytes infiltrate sparsely and are aggregated in the mucosal epithelium (score 1), case 19. (e) LCL. Neoplastic lymphocytes infiltrate extensively and replace the mucosal epithelium (score 2), case 41.
SCL neoplastic lymphocytes were small and monomorphic with scarce cytoplasm. The nuclei were small (<2 red cells) with densely stained chromatin, and the nucleolus was inapparent. Most SCL cases exhibited mild or no mucosal injury (score 0, 23/36 (63.9%); score 1, 11/36 (30.6%); score 2, 2/36 (5.6%)) and varying degrees of villous atrophy (score 0, 4/36 (11.1%); score 1, 26/36 (72.2%); score 2, 6/36 (16.7%)). Neoplastic lymphocytes infiltrated the lamina propria. Varying degrees of epitheliotropism was observed (score 0, 5/36 (13.9%); score 1, 18/36 (50%); score 2, 13/36 (36.1%)) (Fig. 1c–e).
Clonality
Clonal TCR gene rearrangement was detected in 43/50 cases (86%): 10/14 LCL (71.4%) and 33/36 SCL (91.7%). No clonal IgH gene rearrangement was detected.
Immunohistochemistry and Western Blotting
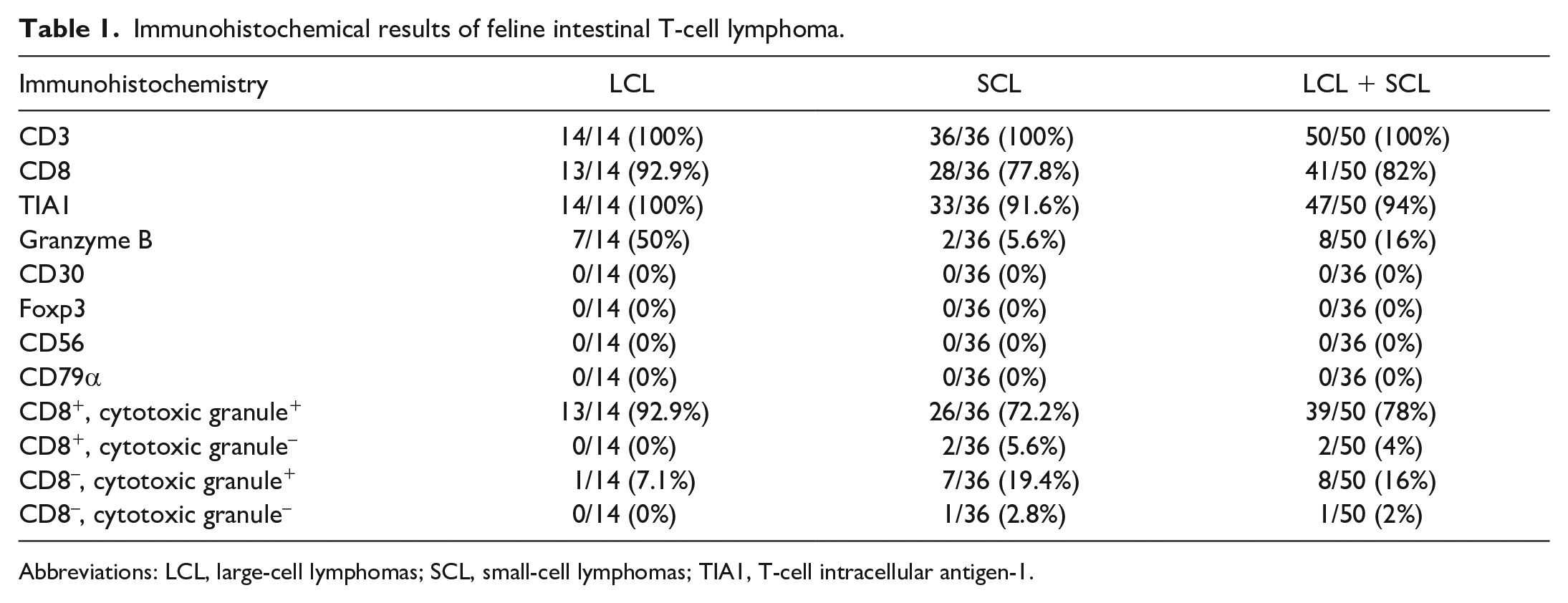
Amino-acid sequences between human TIA1 and feline TIA1 showed 99% identity (Supplemental Fig. S1). By western blotting analysis, anti-TIA1 antibody labeled bands of approximately 15 kDa and 50–75 kDa in tonsil samples from a cat and dog (Supplemental Fig. S2). Immunohistochemistry results are summarized in Table 1. In all cases, neoplastic lymphocytes were positive for CD3 (Fig. 2a, b) and negative for CD79α. Neoplastic lymphocytes were positive for CD8 in 41/50 cases (82%). CD8 positivity was diffuse (18/41, 43.9%) or partial (23/41, 56.1%) (Fig. 3a, b). The percentage of CD8+ LCL (13/14, 92.9%) was higher than that of CD8+ SCL (28/36, 77.8%).
Immunohistochemical results of feline intestinal T-cell lymphoma.
Abbreviations: LCL, large-cell lymphomas; SCL, small-cell lymphomas; TIA1, T-cell intracellular antigen-1.
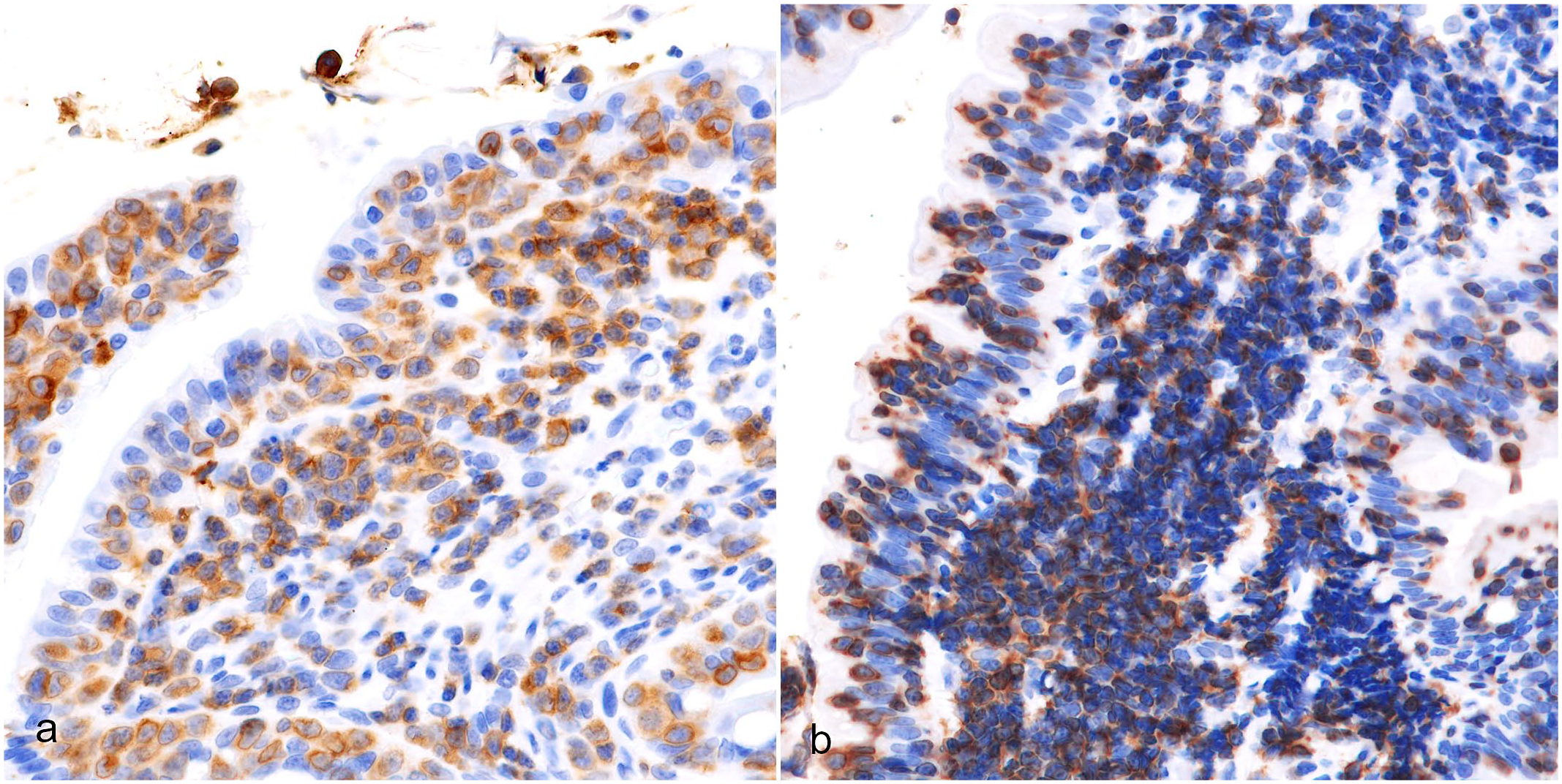
Intestinal T-cell lymphoma, duodenum, cat. Neoplastic lymphocytes are immunolabeled for CD3. Immunohistochemistry. (a) Large-cell lymphoma, case 42. (b) Small-cell lymphoma, case 19.
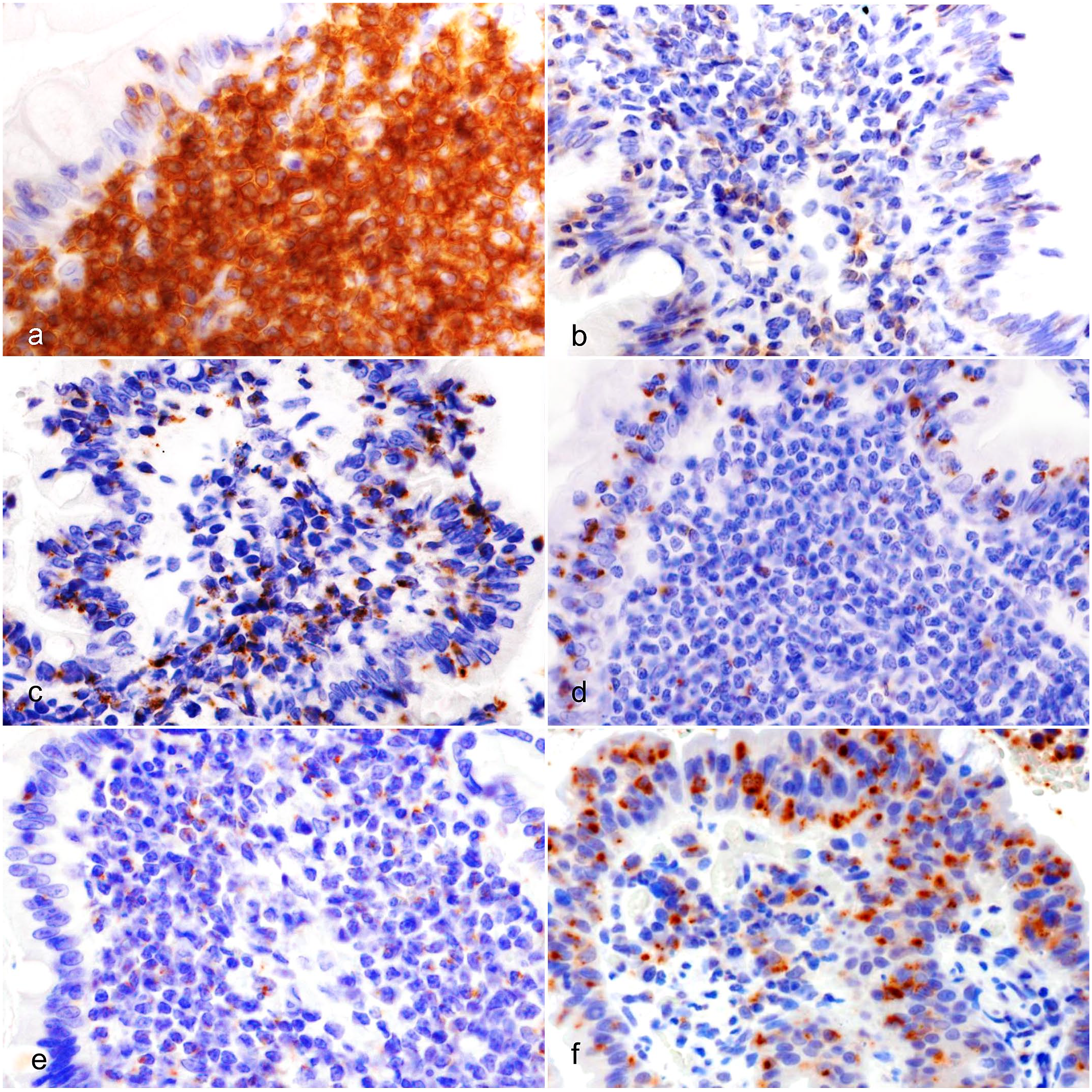
Intestinal T-cell lymphoma, duodenum, cat. (a) Small-cell lymphoma (SCL). Most neoplastic lymphocytes are immunolabeled for CD8, case 14. (b) SCL. Some neoplastic lymphocytes are immunolabeled for CD8, case 15. (c) SCL. Neoplastic lymphocytes are diffusely immunolabeled for TIA1 (T-cell intracellular antigen-1), case 22. (d) SCL. Neoplastic lymphocytes in the mucosal epithelium are predominantly immunolabeled for TIA1, case 3. (e) SCL. Neoplastic lymphocytes in the lamina propria are immunolabeled for TIA1, case 11. (f) Large-cell lymphoma. Neoplastic lymphocytes in the mucosal epithelium are predominantly immunolabeled for granzyme B, case 41.
TIA1 labeling of neoplastic lymphocytes was granular (47/50, 94%). The distribution of TIA1+ neoplastic lymphocytes was diffuse (29/50, 58%), mucosal epithelium predominant (10/50, 20%), or lamina propria predominant (8/50, 16%). The percentage of TIA1+ LCL (14/14, 100%) was higher than that of SCL (33/36, 91.6%). In LCL, the distribution of TIA1+ neoplastic lymphocytes were diffuse (8/14, 57.1%), mucosal epithelium predominant (4/14, 28.6%), or lamina propria predominant (2/14, 14.3%). In SCL, the distribution of TIA1+ neoplastic lymphocytes were diffuse (21/36, 58.8%), mucosal epithelium predominant (6/36, 16.7%), or lamina propria predominant (6/36, 16.7%) (Fig. 3c–e).
Neoplastic lymphocytes had granular labeling of granzyme B (8/50, 16%) (Fig. 3f) in 6/14 LCL (42.9%) and 2/36 SCL (5.6%). The distribution of granzyme B+ neoplastic lymphocytes was mucosal epithelium predominant in the 6 LCL cases and lamina propria predominant in the 2 SCL cases.
Neoplastic lymphocytes were positive for cytotoxic granules (TIA1 and/or granzyme B) in most cases (47/50, 94%). Of these cases, 39 were also positive for CD8. Neoplastic lymphocytes were negative for FOXP3, CD30, and CD56 in all lymphoma cases examined. The mean Ki-67 index of LCL (20.7%) was significantly higher than that of SCL (13.4%) (P = .0299).
Survival Time
Although no significant difference was observed between the 2 groups, LCL cases had a slightly shorter survival time than SCL cases (P = .07). No difference in survival time was observed when cases were analyzed based on their epitheliotropism (P = .13). The median survival time by immunophenotype of neoplastic lymphocytes is shown in Table 2. Cases with mucosal epithelium predominant TIA1+ cells had a shorter survival time than other cases (all cases, P = .0036; LCL, P = .013; SCL, P = .024) (Fig. 4). In addition, cases with mucosal epithelium predominant granzyme B+ cells had a shorter survival time than other cases (Fig. 5) (all cases, P = .00057; LCL, P = .077).
The median survival time by immunohistological characteristics.
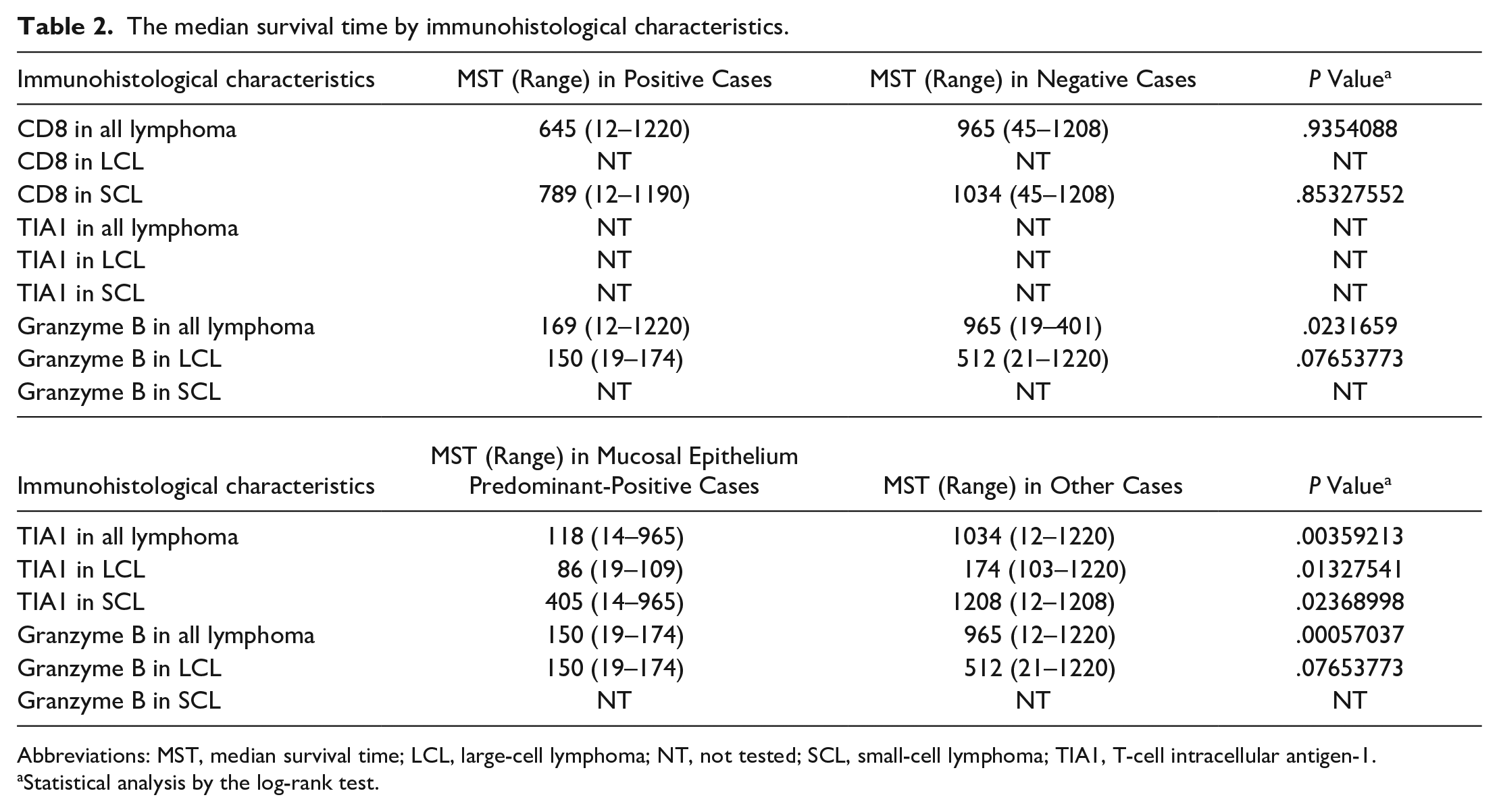
Abbreviations: MST, median survival time; LCL, large-cell lymphoma; NT, not tested; SCL, small-cell lymphoma; TIA1, T-cell intracellular antigen-1.
Statistical analysis by the log-rank test.
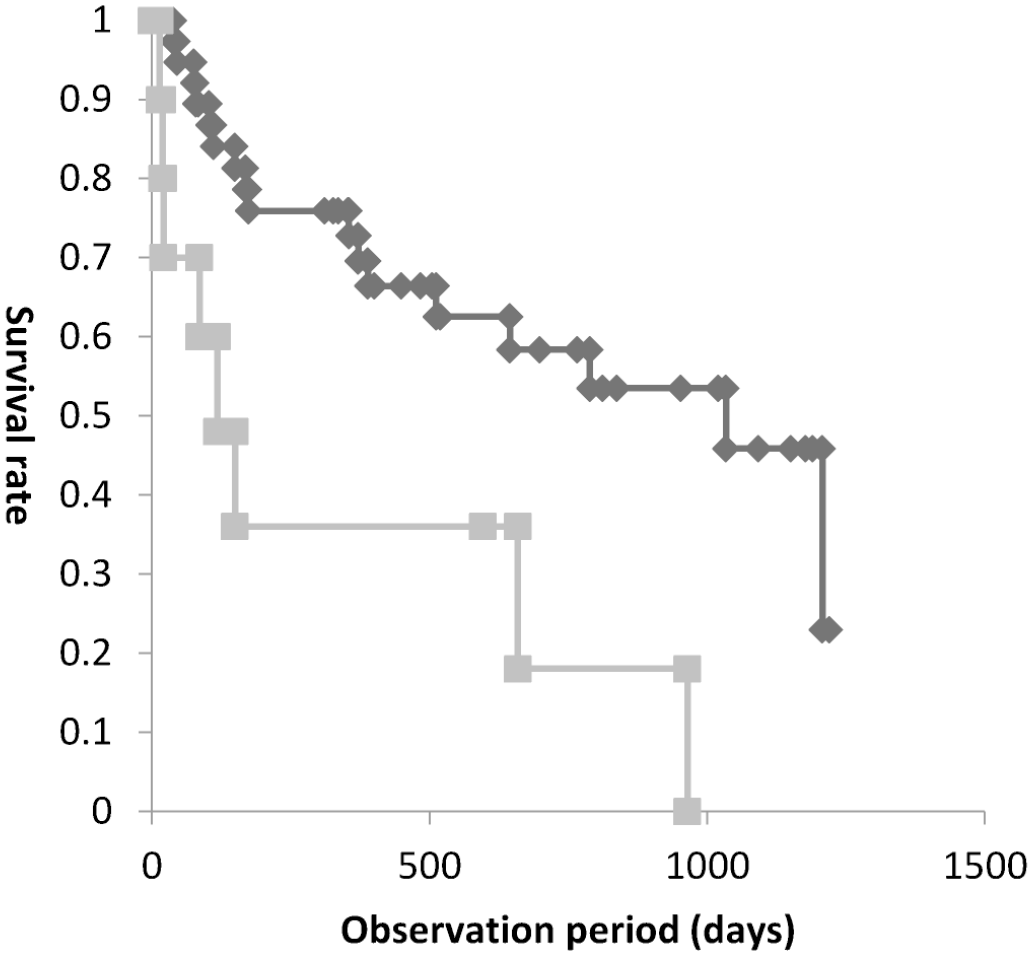
Kaplan-Meier survival graph of lymphoma with mucosal epithelium predominant T-cell intracellular antigen-1 (TIA1)+ neoplastic lymphocytes and other lymphoma cases. The gray line represents lymphomas with TIA1+ neoplastic lymphocytes predominantly located in the mucosal epithelium and the black line represents the lymphomas with TIA1+ neoplastic lymphocytes predominantly located in the lamina propria or with a diffuse distribution (P = .0036).
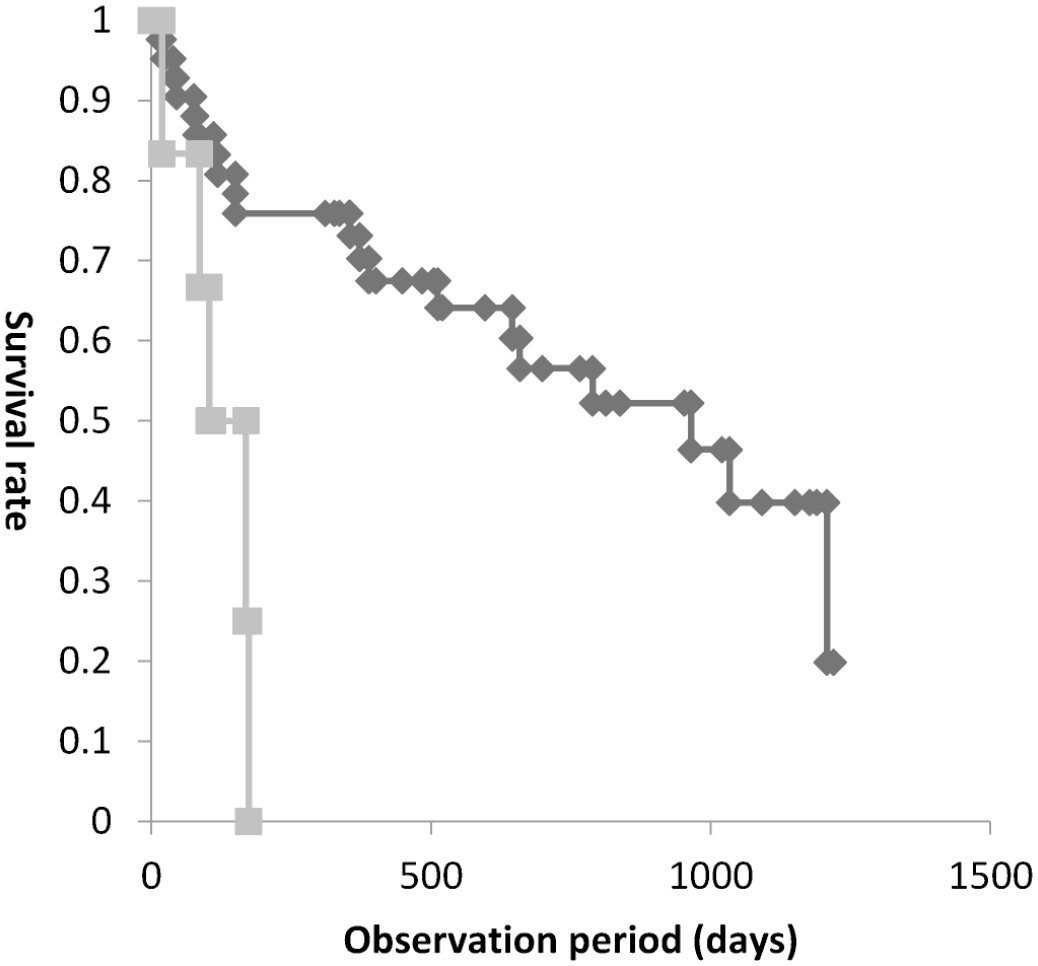
Kaplan-Meier survival graph of lymphoma with mucosal epithelium predominant granzyme B+ neoplastic lymphocytes and other lymphoma cases. The gray line represents lymphomas with granzyme B+ neoplastic lymphocytes predominantly located in the mucosal epithelium and the black line represents the lymphomas with granzyme B+ neoplastic lymphocytes predominantly located in the lamina propria or with a diffuse distribution (P = .00057).
Discussion
In this study, the cell size, epitheliotropism, mucosal injury, cytotoxicity, and survival time of feline intestinal T-cell lymphoma were examined. Consistent with a previous study of feline intestinal T-cell lymphoma, 32 LCL cases also had a shorter survival time than SCL cases in this study. We studied the relations between the neoplastic lymphocytes and survival times, and cases with cytotoxic lymphocytes predominantly in the mucosal epithelium had a significantly shorter survival time than other cases. More aggressive therapy may be required in cases with cytotoxic lymphocytes predominantly in the mucosal epithelium, but we could not confirm it.
TIA1 is one of the cytotoxic granules and is composed of various isoforms including p15-TIA1 and p40-TIA1.13,30 Expression of TIA1 is found in cytotoxic lymphocytes, natural killer (NK) cells, and neutrophils.13,17,31,34,37 In this study, the antihuman TIA1 antibody labeled bands of approximately 15 kDa and 50–75 kDa in tonsil samples from a cat and dog by western blotting. The 50–75 kDa bands may be nonspecific. Amino-acid sequences between human TIA1 and feline TIA1 showed 99% identity. Although a few articles reported that neoplastic lymphocytes of T-cell lymphoma in liver were also positive for TIA1,22,43 this study was the first report that confirmed antihuman TIA1 antibody has cross-reactivity with feline TIA1.
Cytotoxic T-cells are characterized by cytotoxic granules and CD8 expression.8,20,23,28,38 TIA1 and granzyme B are components of cytotoxic granules that induce apoptosis of target cells.13,25,40,43 In this study, almost all LCL and most SCL cases were CD8+ and cytotoxic granule+ (LCL, 13/14, 92.9%; SCL, 28/36, 77.8%). Therefore, neoplastic lymphocytes of most feline intestinal T-cell lymphomas share an immunophenotype with cytotoxic T-cells. There were 2 cases that were CD8+ and cytotoxic granule–, a comparable immunophenotype with inactivated T-cells (naïve T-cells). In addition, 8 cases were CD8– and cytotoxic granule+, a comparable immunophenotype with innate lymphoid cells. 11 Only 1 case was CD8– and cytotoxic granule–. The neoplastic lymphocytes may have been CD4+, but we were unable to perform immunohistochemistry for CD4 due to the lack of an antibody available for formalin-fixed, paraffin-embedded tissue. The neoplastic cells in this study were negative for CD56, which is frequently expressed in NK cells.31,34,37 These results suggest that the neoplastic cells are not likely of NK cell origin.
Both TIA1 and granzyme B are present in cytotoxic granules, but the positive rate for TIA1 was higher than that of granzyme B in both LCL and SCL. This also applies to human intestinal T-cell lymphoma.6,7,41,46 It is considered that TIA1 is expressed in cytotoxic T-cells from an immature stage, while granzyme B is expressed in activated mature cytotoxic T-cells.18,19 The low positive rate for granzyme B may be associated with an immature nature of the neoplastic lymphocytes in feline intestinal T-cell lymphoma. In a previous study on feline intestinal lymphoma, LCL cases had a shorter survival time than SCL cases. 32 In this study, although no significant difference was observed, LCL cases had a slightly shorter survival time than SCL cases. Epitheliotropism of feline intestinal T-cell lymphoma was not associated with survival time. However, cases with TIA1+ and/or granzyme B+ neoplastic lymphocytes predominantly in the mucosal epithelium had a significantly shorter survival time than other cases. This suggests that mucosal epithelium infiltration of neoplastic cells with a cytotoxic immunophenotype is a negative prognostic factor in cats with intestinal lymphoma. Moreover, the 6 cases with granzyme B+ neoplastic lymphocytes in mucosal epithelium exhibited marked mucosal epithelium injury. Infiltration of cytotoxic neoplastic cells may be associated with mucosal injury through apoptosis of the mucosal epithelium and impaired intestinal function.
In this study, 77.8% of SCL cases demonstrated a CD8+ immunophenotype. However, low-grade intestinal T-cell lymphoma showed more heterogenous CD8 phenotypes in a previous report. 14 This may be because the classification for intestinal lymphoma was different between the 2 studies; this study classified intestinal lymphoma by the cell size, while the previous study followed the 2016 revised WHO classification of human lymphoma. In this study, SCL cases showed varying degrees of epitheliotropism, but ITLPD, which corresponds to the low-grade T-cell lymphoma in the previous studies,14,15,36 exhibits scant epitheliotropism. SCL is not always equivalent to low-grade T-cell lymphoma and ITLPD. Therefore, we consider that CD8 phenotype is different from previous studies. Difference of cat breeds between Asia and Europe may also be associated with difference in immunophenotype.
In ITLPD, lymphoid cells do not infiltrate the mucosal epithelium.3,46 ITLPD have heterogenous CD8 and CD4 immunophenotype, and granzyme B is generally negative.3,46 Neoplastic lymphocytes infiltrated the mucosal epithelium in some SCL cases. In this study, most feline SCLs were positive for CD8 and negative for granzyme B.
In human medicine, none of the immunohistochemical markers, including CD8, CD56, CD30, or TIA1, were predictors of survival times.10 In this study, neoplastic lymphocytes were negative for CD56 and CD30. CD8+ or TIA1+ neoplastic lymphocytes were not predictors of survival times in the feline intestinal T-cell lymphoma. However, granzyme B+ neoplastic lymphocytes were significant predictors of survival times. Moreover, we revealed that cases with TIA1+ and/or granzyme B+ neoplastic lymphocytes predominantly in the mucosal epithelium had a significantly shorter survival time in feline intestinal T-cell lymphoma.
In conclusion, this study revealed that intraepithelial neoplastic lymphocytes with a cytotoxic immunophenotype may be associated with a poor prognosis in feline intestinal T-cell lymphoma.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221120010 – Supplemental material for Intraepithelial cytotoxic lymphocytes are associated with a poor prognosis in feline intestinal T-cell lymphoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858221120010 for Intraepithelial cytotoxic lymphocytes are associated with a poor prognosis in feline intestinal T-cell lymphoma by Tatshuhito Ii, James K. Chambers, Ko Nakashima, Yuko Goto-Koshino, Takuya Mizuno and Kazuyuki Uchida in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858221120010 – Supplemental material for Intraepithelial cytotoxic lymphocytes are associated with a poor prognosis in feline intestinal T-cell lymphoma
Supplemental material, sj-xlsx-2-vet-10.1177_03009858221120010 for Intraepithelial cytotoxic lymphocytes are associated with a poor prognosis in feline intestinal T-cell lymphoma by Tatshuhito Ii, James K. Chambers, Ko Nakashima, Yuko Goto-Koshino, Takuya Mizuno and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
