Abstract
Equine herpesvirus 1 (EHV-1) uses equine major histocompatibility complex class I (MHC class I) as an entry receptor. Exogenous expression of equine MHC class I genes in murine cell lines confers susceptibility to EHV-1 infection. To examine the in vivo role of equine MHC class I as an entry receptor for EHV-1, we generated transgenic (Tg) mice expressing equine MHC class I under the control of the CAG promoter. Equine MHC class I protein was expressed in the liver, spleen, lung, and brain of Tg mice, which was confirmed by Western blot. However, equine MHC class I antigen was only detected in bronchiolar epithelium and not in other tissues, using the immunofluorescence method employed in this study. Both Tg and wild-type (WT) mice developed pneumonia 3 days after intranasal infection with EHV-1. The bronchiolar epithelial cells of Tg mice showed more severe necrosis, compared with those in WT mice. In addition, the number of virus antigen-positive cells in the lungs was higher in Tg mice than in WT mice. These results suggest that exogenous expression of equine MHC class I renders mice more susceptible to EHV-1 infection.
Keywords
Equine herpesvirus 1 (EHV-1) causes respiratory disease, abortion, and encephalomyelitis. In horses, following oronasal transmission, EHV-1 infection occurs in respiratory epithelial cells and local lymph nodes. 9,12 This results in a leukocyte-associated viremia, followed by infection of endothelial cells in the pregnant uterus and central nervous system (CNS). 13 The pathologic consequences of endothelial infection in the uterus and CNS are vasculitis, thrombosis, and secondary ischemia that cause abortion 22 and neurological signs, 4 respectively. EHV-1 establishes latent infection in T lymphocytes 25 and/or in trigeminal ganglia. 21
Large amounts of experimental data have been generated from infection studies using horses; 10,11,27 however, these studies are labor intensive. In addition, there are difficulties in obtaining large numbers of horses for experiments. Mice have also been used as models of EHV-1 infection. EHV-1–infected mice are reported to show pneumonia, viremia, 2 abortion, 1 and viral latency. 5 But, neither viral replication in endothelial cells nor the subsequent formation of CNS lesions has been reported in the murine model. Equine brain microvascular endothelial cells (EBMECs) are susceptible to EHV-1 infection, whereas their murine counterparts (MEMECs) are EHV-1 resistant, reflecting the inability of the virus to enter mouse cells. 7
Equine major histocompatibility class I (equine MHC class I) is reported to be an entry receptor for EHV-1, 17,18 while EHV-1 is unlikely to use mouse MHC class I as a receptor for entry into murine cells. Mouse NIH3T3 cells, which express mouse MHC class I molecules (haplotype H2q) 8,20 on their surface, are not susceptible to EHV-1 infection. However, exogenous expression of equine MHC class I genes in NIH3T3 cells confers susceptibility to EHV-1 infection. 18 These results indicate that MHC class I is one of the determinants underlying species-related differences in the susceptibility to EHV-1.
In the current study, we generated transgenic mice (Tg mice) expressing equine MHC class I to verify its ability to act as an entry receptor for EHV-1 in vivo and make mice more susceptible to EHV-1 infection. After inoculation with EHV-1, we compared the histopathological changes in Tg mice with those in their wild-type (WT) littermates to evaluate the effect of equine MHC class I expression on EHV-1 pathogenicity.
Materials and Methods
Virus
The EHV-1 strain Ab4 15 was kindly provided by Dr. Hideto Fukushi (Gifu University, Gifu, Japan). Stock viruses were cultured in rabbit kidney (RK13) cells and titrated by plaque formation assays on RK13 cells.
Plasmid
The plasmid vector pCXSN was generated as previously described. 18 The plasmid pCAGGS-MCS(KS), which was kindly provided by Dr. Naomi Ohnishi (Japanese Foundation for Cancer Research, Tokyo, Japan), was generated from pCAGGS 14 by replacing the cloning site with KpnI, XhoI, EcoRI, and SacI endonuclease recognition sequences.
Transgenic Mice
DNA fragments of equine MHC class I heavy chain A68 (GenBank/EMBL/DDBJ entry AB525079) excised from pCXSN-A68-HA 18 were cloned into the XhoI-HindIII site of a pSP73 vector (Promega, Madison, WI) to generate vector pSP-A68. Construction of pSP-V5 68 was done by the insertion of a V5 epitope tag following the putative signal alignment of A68 into the pSP-A68 vector using inverse polymerase chain reaction (PCR). The region of DNA containing the equine β2-microglobulin (β2m) was amplified by PCR from plasmid pCXSN-equine β2m 18 and cloned into the HindIII-EcoRI site of pSP-V5 68 (pSP-V68-B2M). To induce bicistronic expression of the equine MHC class I heavy chain A68 and equine β2m on the cell surface, 2A oligopeptide (ATNFSLLKQAGDVEENPGP) 24 was inserted between the C terminus of A68 and the N terminus of β2M in pSP-V68-B2M using inverse PCR (pSP-V68-2A-B2M). The V68-2A-B2M fragment was then excised as an XhoI-EcoRI fragment and cloned into vector pCAGGS-MCS(KS) to generate the vector pCAGGS-V68-2A-B2M. The equine MHC class I heavy chain A68 with equine β2m excised from pCAGGS-V68-2A-B2M as a SnaBI-HindIII fragment was used as a transgene (Fig. 1). The transgene was microinjected into fertilized eggs of Slc:BDF1 mice (Japan SLC, Shizuoka, Japan). The transgenic founders carrying the A68 gene were backcrossed with BALB/cAJcl (CLEA Japan, Tokyo, Japan). The mouse line designated BALB/cAJcl;BDF1-Tg (CAG-Eq-MHC-I-B2M) 30LCP/A68 was used in this study as Tg mice.
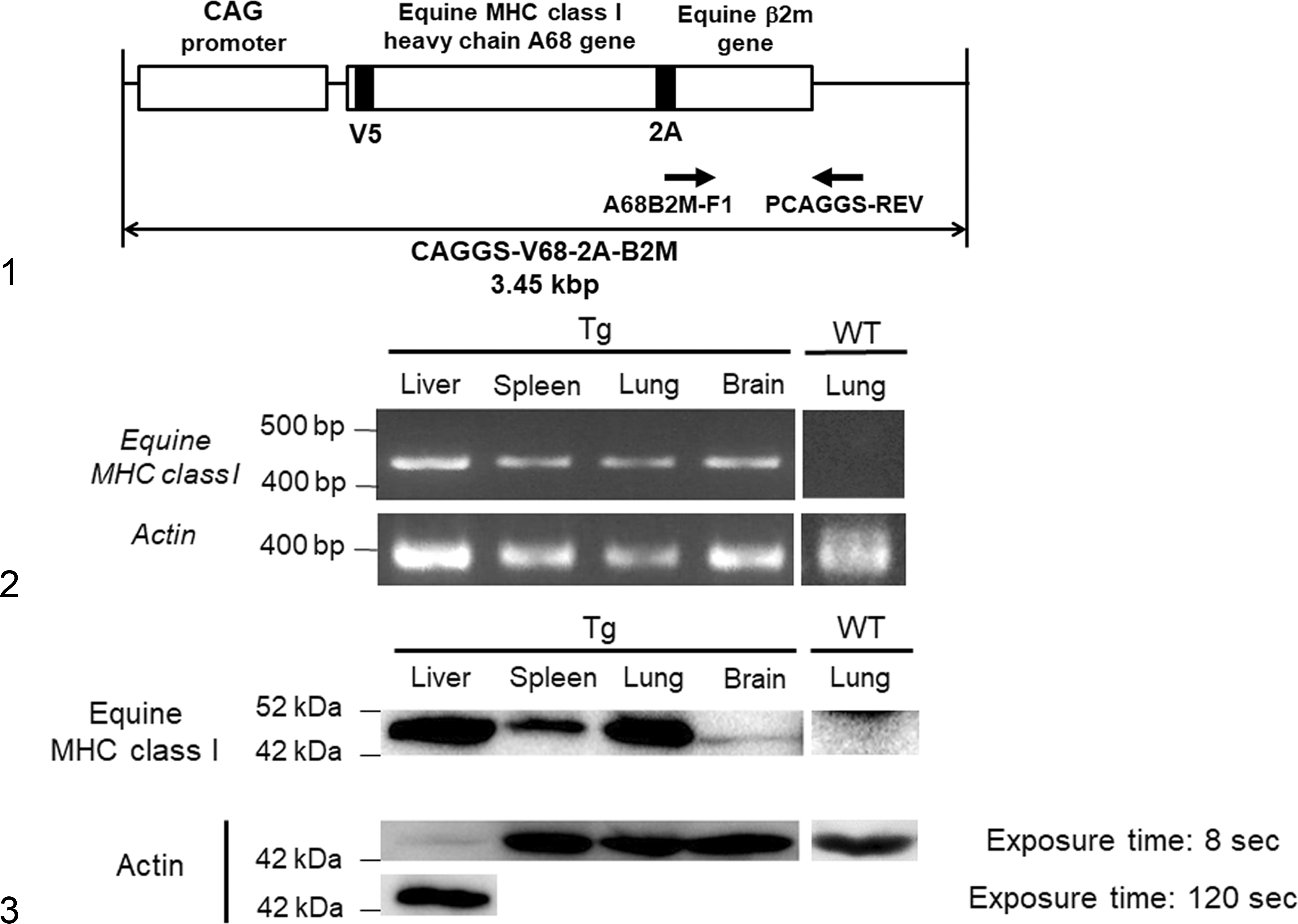
Genotyping of Tg Mice
The founder and its progeny were genotyped by PCR. Mouse tail–derived DNA was used as template. PCR was performed with 2 × Quick Taq HS DyeMix (TOYOBO, Japan), 0.2 μM each of the primers, and 200 ng DNA. Details of the PCR primers used for amplification are shown in Table 1. For amplification of the A68 gene, primers A68-B2M-F1 and PCAGGS-REV were used; for amplification of mouse actin, primers mouse actin F and mouse actin R were used. The PCR cycling profile was 94°C for 2 minutes for denaturation, followed by 35 sequential cycles of 94°C for 30 seconds, 63°C for 30 seconds, and 72°C for 1 minute. The PCR products were electrophoretically analyzed on 1.5% agarose gels.
List of Primers Used for Genotyping.

Abbreviation: bp, base pair.
Reverse Transcription–PCR
The liver, spleen, kidney, and lung of Tg mice were collected. Total RNA was extracted using TRIZOL reagent (Thermo Fisher Scientific, Massachusetts, USA). The RNA samples were treated with DNase I, Amplification Grade (Thermo Fisher Scientific) to digest contaminating DNA. Synthesis of complementary DNA (cDNA) was carried out using the SuperScript III First-Strand Synthesis System (Thermo Fisher Scientific). PCR was performed with 0.2 mM dNTP, 10 × PCR buffer, 1.5 mM MgCl2, 1.25 units of HotStarTaq DNA Polymerase (Qiagen, Hilden, Germany), 0.3 μM of each primer, and 1 μl cDNA in a total volume of 12.5 μl. The PCR primers used for amplification were the same as those used in genotyping of Tg mice. The PCR cycling profile was 95°C for 15 minutes for initial denaturation, followed by 40 sequential cycles of 94°C for 30 seconds, 63°C for 1 minute, and 72°C for 1 minute and final extension at 72°C for 10 minutes. The PCR products were electrophoretically analyzed on 1.5% agarose gels.
Western Blot
The liver, spleen, lung, and brain of Tg mice or WT mice were collected. The tissues were lysed in lysis buffer (10 mM Tris-HCl, pH 7.5; 150 mM NaCl2; 5 mM EDTA; 10% glycerol; and 1% Triton X-100) supplemented with complete protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO). Lysed proteins were homogenized and sonicated. Following sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), proteins were transferred onto Immobilon-P transfer membranes (Merck Millipore, Massachusetts, USA) and labeled with anti-V5 antibody (Thermo Fisher Scientific) or antiactin antibody clone C4 (Merck Millipore).
Immunofluorescence Staining
The lungs of Tg mice or WT mice were collected, and the tissues were fixed in 4% paraformaldehyde/phosphate-buffered saline (PBS). The tissues were paraffin embedded, and sections were prepared using standard methods. Indirect immunofluorescence staining was performed using Tyramide SuperBoost kits with Alexa Fluor Tyramides (Thermo Fisher Scientific). Briefly, the sections were deparaffinized, heated with 0.01 M citric acid buffer (pH 6.0) using a microwave for antigen retrieval, treated with 0.3% hydrogen peroxide in methanol, and blocked with 10% normal rabbit serum. A goat anti-V5-tag polyclonal antibody (ab95038; Abcam, Cambridge, England ) was added, and the sections were incubated overnight at 4°C. The sections were then washed with PBS and incubated with donkey anti-goat horseradish peroxidase (HRP)–conjugated IgG (Santa Cruz Biotechnology, Santa Cruz, CA) for 20 minutes at room temperature. After further washing with PBS, the sections were incubated with Alexa Fluor 488 Tyramide Reagent for 10 minutes at room temperature. Nuclei were stained with Hoechst 33258 (Sigma-Aldrich). As a negative control, sections were stained without primary antibody. The fluorescent-stained sections were examined using an LSM 700 confocal microscope (Zeiss, Oberkochen, Germany).
Experimental Infection
Heterozygous Tg mice and wild-type (WT) littermate mice were used at 6 weeks of age. Seven Tg mice and 3 WT mice were anesthetized with isoflurane and intranasally inoculated with 2 × 106 plaque-forming units (PFU) of EHV-1 Ab4. All animal experiments were authorized by the Institutional Animal Care and Use Committee of the Graduate School of Veterinary Medicine, Hokkaido University (approval number: 13-0092), and all experiments were performed according to the guidelines of this committee.
Histopathology and Immunohistochemistry
The mice inoculated with EHV-1 were euthanized and necropsied at 3 days postinoculation (p.i.). The liver, spleen, kidneys, heart, lung, and brain were collected. All organs were fixed with 4% paraformaldehyde/PBS. The tissues were paraffin-embedded, sectioned at 2 to 4 μm, and stained with hematoxylin and eosin (HE). To calculate the number of necrotic bronchiolar epithelial cells in the lungs, total number of bronchiolar epithelial cells showing necrotic morphology in each section and the total area of the sections were measured using a CellSens Dimension microscope (Olympus, Tokyo, Japan). The number of necrotic cells was divided by the total area of each section. The mean number of necrotic cells in the bronchioles of Tg mice was compared with those of WT mice using the Welch 2-sample t test.
Indirect immunohistochemistry staining was carried out using the labeled streptavidin-biotin (SAB) technique (Histofine SAB-PO Kit; Nichirei, Tokyo, Japan). Briefly, the sections were deparaffinized, heated with 0.01 M citric acid buffer (pH 6.0) using a microwave for antigen retrieval, treated with 0.3% hydrogen peroxide in methanol, and blocked with 10% normal rabbit serum. Goat anti-equine rhinopneumonitis virus/equine herpesvirus type 1 polyclonal antiserum (VMRD, Washington, USA) or polyclonal rabbit anti-human CD3 antibody (Agilent, California, USA) was added, and the sections were incubated overnight at 4°C. The sections were then washed with PBS and incubated with a secondary antibody labeled with biotin for 20 minutes at room temperature. After additional washing with PBS, the sections were incubated with peroxidase-conjugated streptavidin for 10 minutes at room temperature. The bound peroxidase was detected with 3,3′-diaminobenzidine (DAB). The sections were counterstained with hematoxylin. As a negative control, sections were stained without primary antibody.
To calculate the number of cells in the lungs that were positive for viral antigen, the total number of EHV-1–positive cells was counted in each section of the tissue and the total area of the sections was measured using a CellSens Dimension microscope. The number of positive cells was divided by the total area of each section. The mean number of EHV-1–positive cells in the lungs of Tg mice was compared with the number in the lungs of WT mice using the Welch 2-sample t test.
To calculate the number of T cells in the lung, CD3-positive cells were counted in each tissue section using ImageJ. 19 The number of positive cells was divided by the total area of each section. The mean number of CD3-positive cells was statistically compared in Tg and WT mice by using the Mann-Whitney U test.
Results
Tg Mice Expressing Equine MHC Class I
Tg mice expressing equine MHC class I were generated as described in the Materials and Methods. Because endogenous MHC class I is expressed on most somatic cells, 3 we used the CAG promoter, which can induce ubiquitous gene expression (Fig. 1). 14 Expression of equine MHC class I heavy chain messenger RNA (mRNA) and protein was detected in the heterozygous Tg mice but not in the WT littermate mice (Figs. 2, 3). The level of equine MHC class I protein in the brain was lower than in the liver, spleen, and lung (Fig. 3). Immunofluorescence staining revealed the expression of equine MHC class I on the apical surface of bronchiolar epithelial cells of the Tg but not of the WT mice (Figs. 4, 5). Expression of equine MHC class I was not detected by immunofluorescence staining in the liver, spleen, kidney, heart, or brain of either Tg or WT mice.
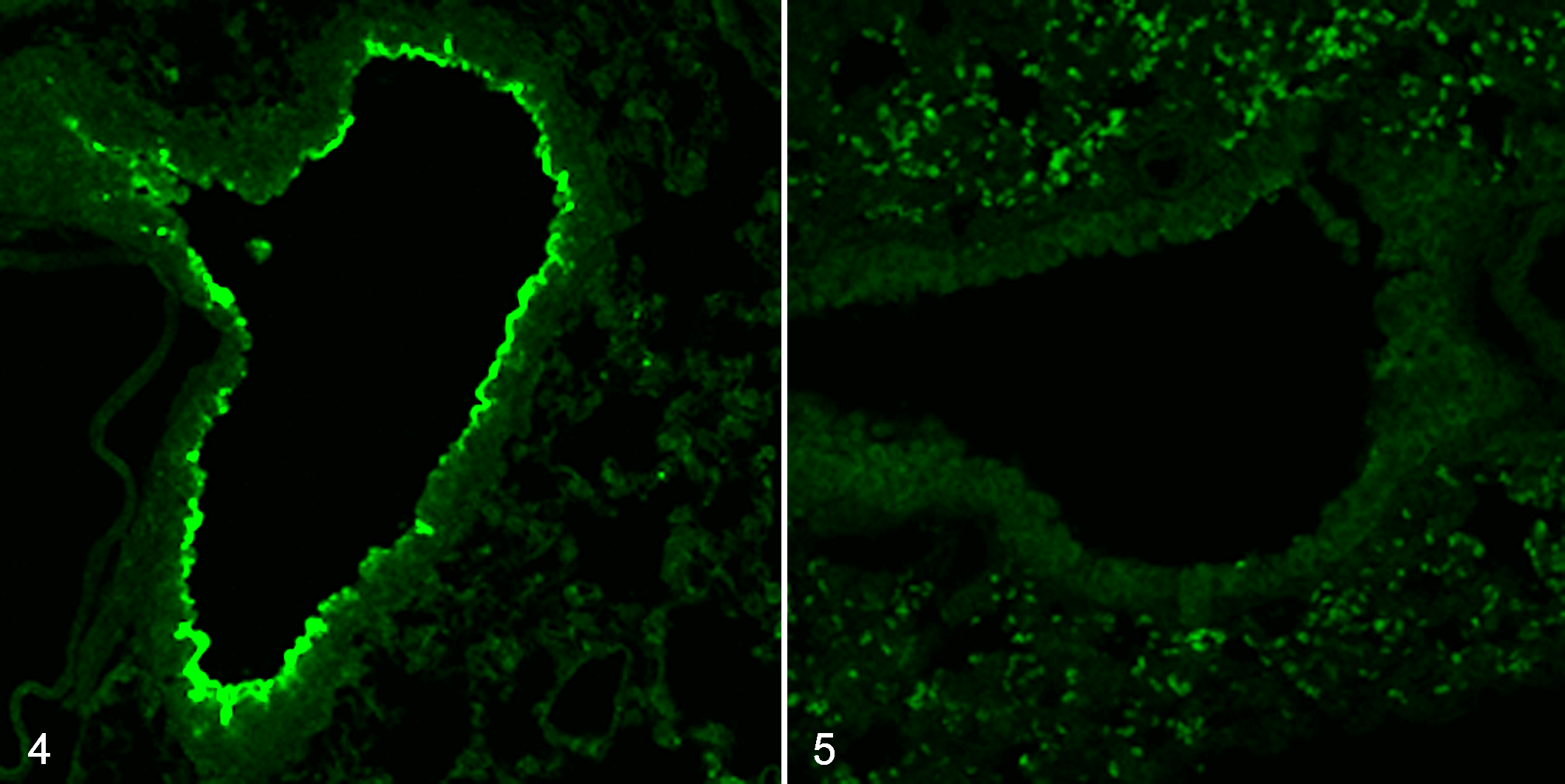
Exogenous expression of equine major histocompatibility complex (MHC) class I, lung, mice. Immunofluorescent staining using an anti-V5 antibody (green).
Experimental Infection
Starting at 1 day p.i., all EHV-1–inoculated mice, Tg and WT, began to show tachypnea and lethargy with ruffled fur. At 3 days p.i., all mice inoculated with EHV-1 had postmortem lesions of bronchointerstitial pneumonia, with peribronchiolar and perivascular infiltration of lymphocytes, macrophages, and neutrophils. Necrosis of the bronchiolar epithelial cells was more evident in Tg mice than in WT mice (Figs. 6a, 8a, 10; P < .05). Using immunohistochemistry (IHC), we detected EHV-1 in the nucleus and cytoplasm of bronchiolar epithelial cells and macrophages in both groups of mice (Figs. 6b, 8b; Suppl. Figs. S1, S2). More EHV-1–positive cells were found in the lungs of Tg mice than those of WT mice (Fig. 11; P < .05). Furthermore, perivascular edema was much more prominent in the Tg mice; however, virus antigen was not detected in the vascular endothelium (Figs. 7a, 7b, 9a, 9b). Inflammatory cells infiltrating the lung were primarily lymphocytes. The number of CD3-positive cells in the lung did not differ between the 2 groups of mice (Fig. 12; P > .05). Notably, neither histological lesions nor viral antigen were detected in the liver, spleen, kidneys, heart, and brain of Tg or WT mice inoculated with EHV-1.
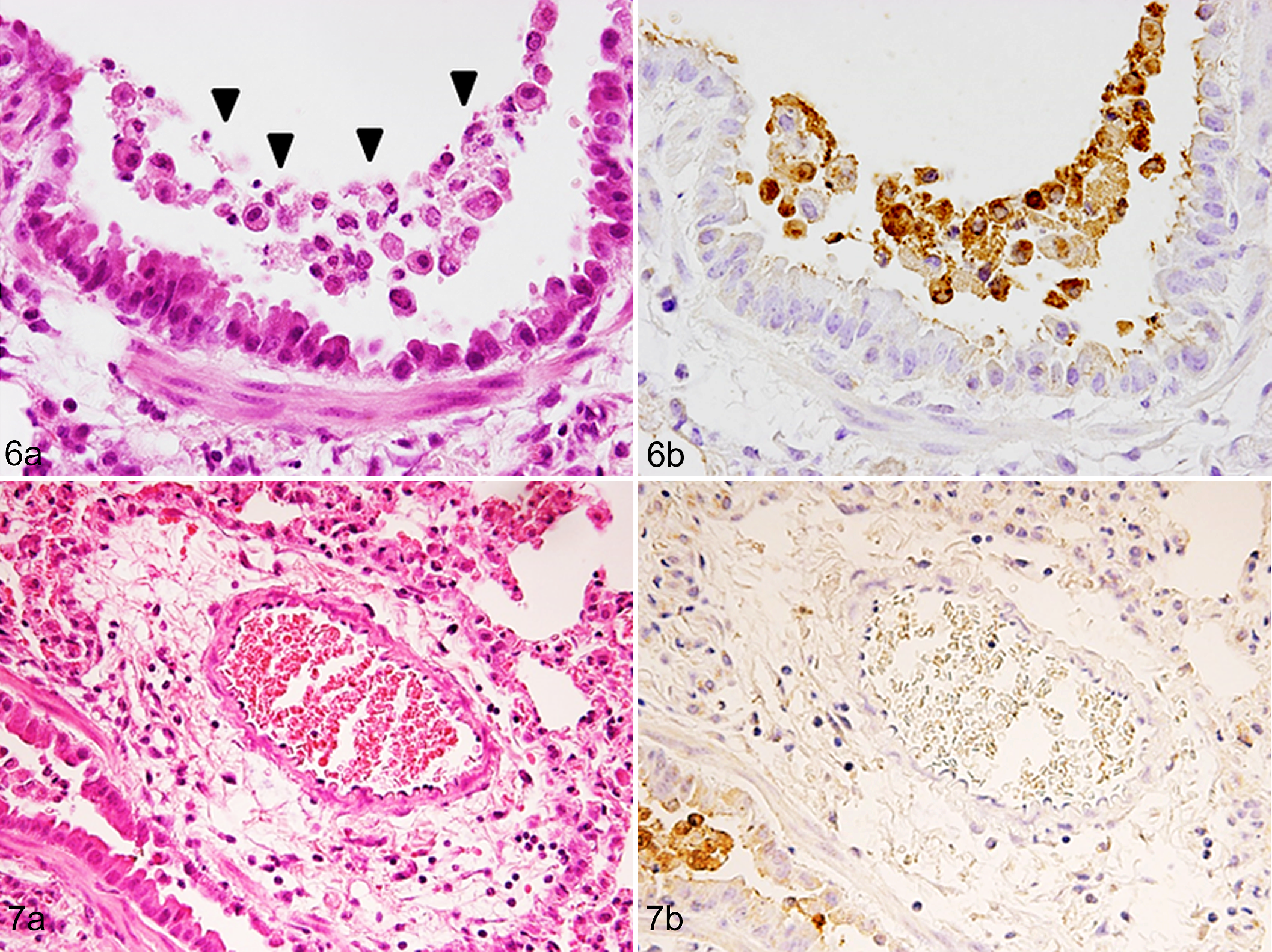
Equine herpesvirus 1 (EHV-1) infection, lung, mice transgenic for equine major histocompatibility complex (MHC) class I.
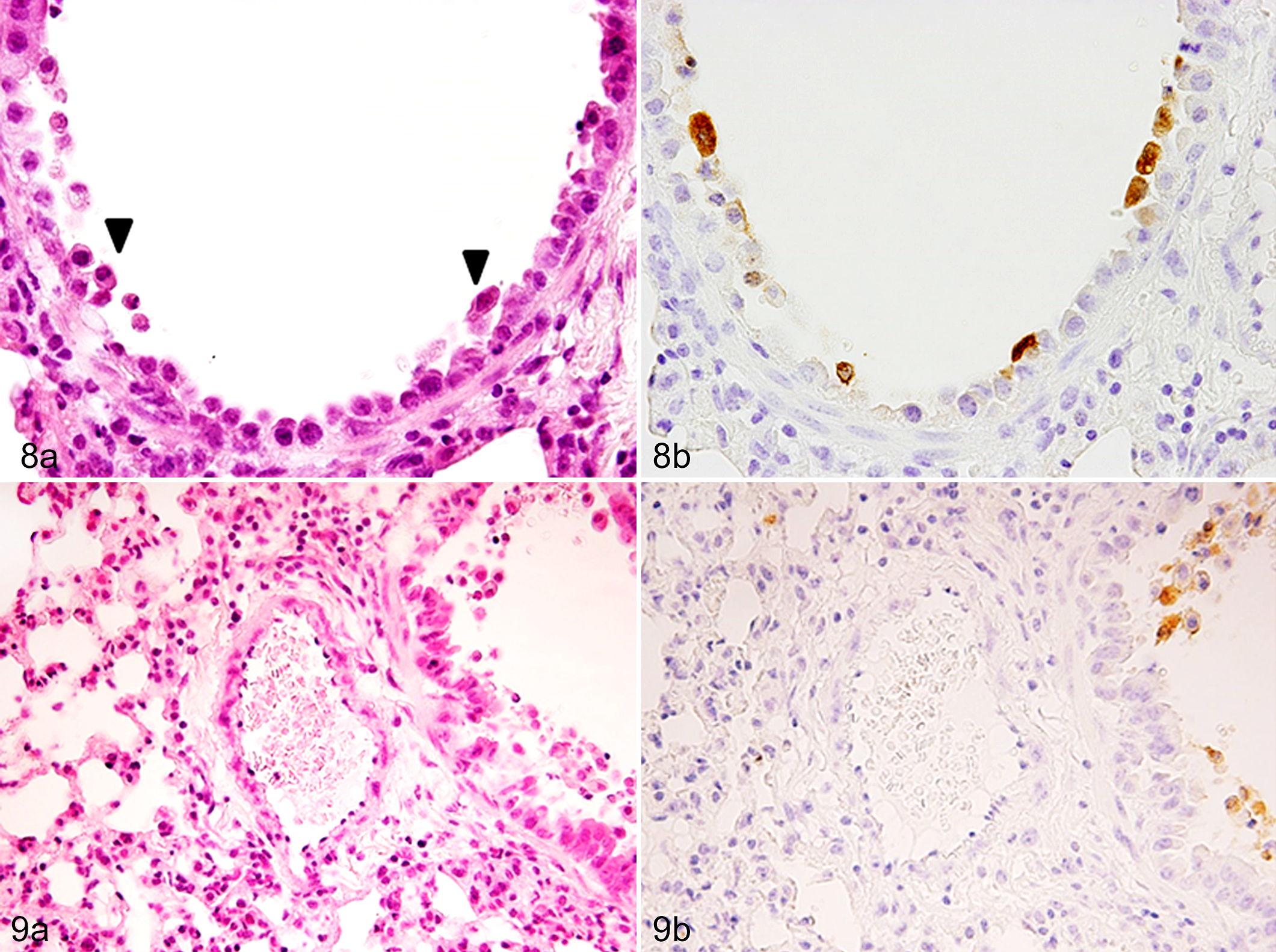
Equine herpesvirus 1 (EHV-1) infection, lung, wild-type littermate mice.
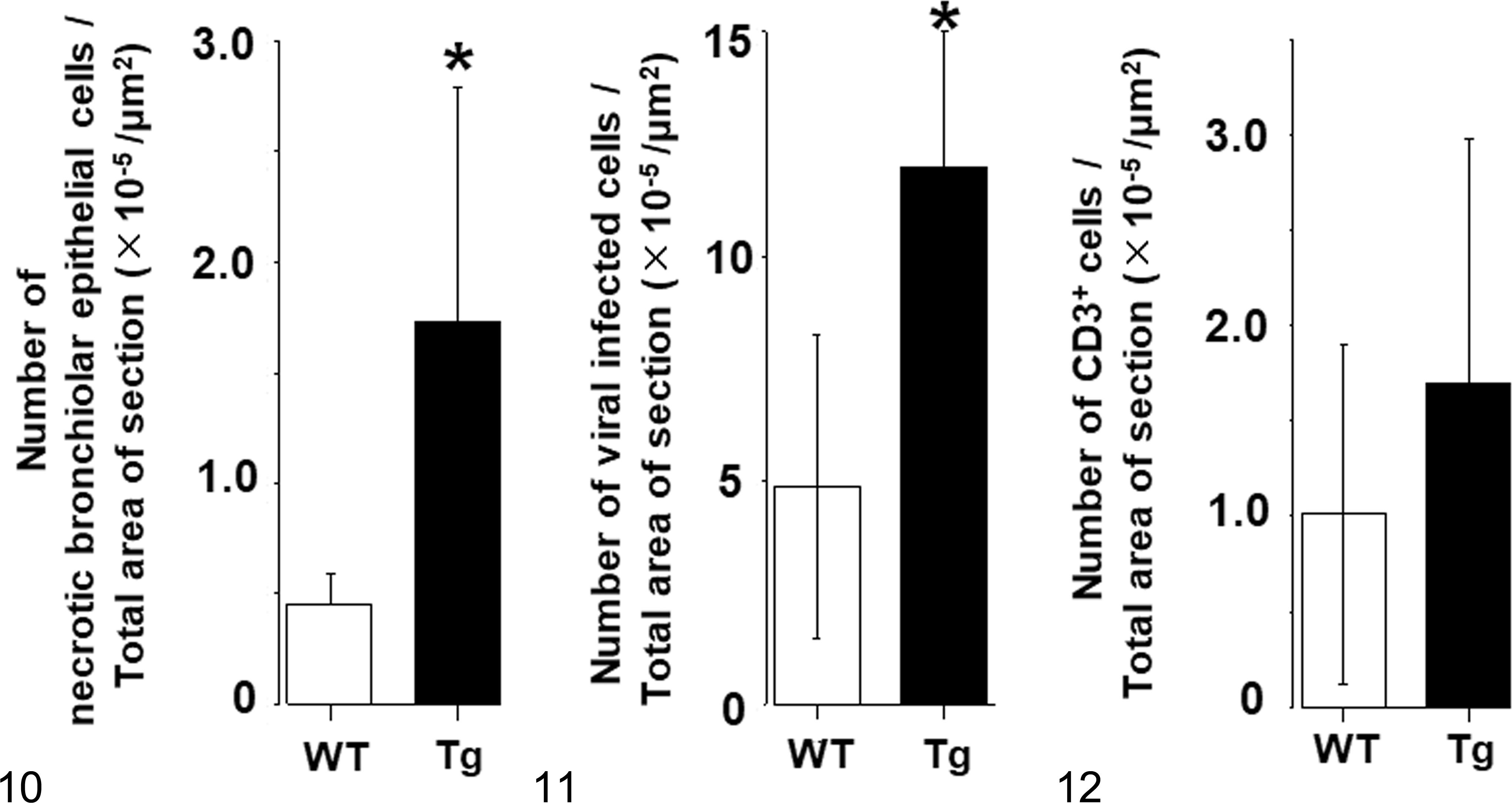
Quantitative histopathologic and immunohistochemistry data from wild-type (WT) mice and mice transgenic for equine MHC class I. The data show mean +/- standard deviation.
Discussion
We generated Tg mice expressing equine MHC class I molecules, which are known to act as entry receptors for EHV-1 in vitro. Western blot analysis showed the expression of equine MHC class I proteins in various organs, including the lung. After experimental nasal infection with EHV-1, the number of EHV-1–infected cells observed in the lung of Tg mice was higher than in that of WT mice. These results suggest that exogenous expression of equine MHC class I increased the susceptibility of mice to EHV-1 infection.
Intranasal infection with EHV-1 in adult BALB/c mice causes bronchiolar epithelial infection as well as pulmonary lesions with intranuclear inclusions and peribronchiolar and perivascular mononuclear cell infiltrates. 2 Our mice developed similar lesions upon EHV-1 intranasal infection. However, infection and necrosis of the bronchiolar epithelium were more severe in Tg mice expressing equine MHC class I compared to the WT mice. This finding was consistent with the result of immunofluorescent staining that revealed selective expression of equine MHC class I expression in the bronchiolar epithelium of Tg mice.
Perivascular edema surrounding pulmonary arteries was a characteristic lesion in both Tg and WT mice but was more severe in Tg mice than in WT mice. Because neither vascular endothelial cells nor vascular smooth muscle cells were positive for viral antigen, the edema may have reflected the severity of adjacent bronchiolar damage.
The role of leukocytes in lung lesion pathogenesis remains unclear. The numbers of CD3-positive cells in the lung were similar in WT and Tg mice, although necrosis and desquamation of bronchiolar epithelial cells were more evident in the Tg mice. Foals infected with EHV-1 show necrotizing bronchiolitis and infiltration of mononuclear cells 6,26 similar to the lesions of the Tg mice in this study. Smith et al 23 previously reported that mice infected with EHV-1 RacL11 strain developed more severe lung lesions than those infected with attenuated KyA strain. Interestingly, the level of proinflammatory β-chemokines produced in the bronchiolar lavage fluid by RacL11 was higher than that by KyA, despite identical T-cell responses and viral loads in the lungs of both strains. 23 Therefore, pathogenesis of the relatively severe lung lesions in EHV-1 in Tg mice may involve a contribution of the immune response in addition to a direct cytolytic effect of the viral infection.
Immunofluorescence staining of the V5 epitope tag did not show positivity in the liver, spleen, and brain of Tg mouse, although reverse transcription–PCR (RT-PCR) and Western blot analyses demonstrated the gene and protein expression of equine MHC class I in these organs. It may be that expression levels of equine MHC class I in cell types other than bronchiolar epithelial cells were below the detection limit of the indirect immunofluorescent technique. The CAG promoter used in this study is known to induce high-level gene expression in mammalian cells; however, at times, it fails to attain sufficient protein expression levels, depending on the nature of the cargo gene and/or the type of host cell. 16 An unknown mechanism was potentially involved in determining the final stationary expression levels of the equine MHC class I in the mice.
Neither histological lesions nor viral antigens were detected in the liver, spleen, kidneys, heart, and brain of the Tg mouse at 3 days after intranasal inoculation with EHV-1. This was consistent with the paucity of equine MHC class I antigen-specific signal obtained in these tissues by immunofluorescent staining. Notably, Western blot analysis showed the expression of exogenous protein in all examined organs, albeit at levels below the detection limit of immunofluorescent staining. Therefore, it will be interesting to investigate the susceptibility of Tg mice to EHV-1 by using alternative routes of virus inoculation.
This study suggests that transgenic expression of equine MHC class I is a useful method for increasing the in vivo susceptibility of mouse cells to EHV-1 infection. New Tg mice with more widespread overexpression of equine MHC class I, including additional EHV-1 target cells (eg, endothelial cells and leukocytes) in addition to bronchiolar epithelial cells, may provide a suitable model for the study of EHV-1 pathogenesis.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819834616 - Exogenous Expression of Equine MHC Class I Molecules in Mice Increases Susceptibility to Equine Herpesvirus 1 Pulmonary Infection
Supplemental Material, DS1_VET_10.1177_0300985819834616 for Exogenous Expression of Equine MHC Class I Molecules in Mice Increases Susceptibility to Equine Herpesvirus 1 Pulmonary Infection by Erina Minato, Keisuke Aoshima, Atsushi Kobayashi, Naomi Ohnishi, Nobuya Sasaki and Takashi Kimura in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Scientific Research (B) from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
