Abstract
Histopathologic and immunohistochemical examinations were performed to determine the origin of host cells parasitized by Eimeria in the small intestines collected from five foals. Eimeria organisms at various stages (mainly microgametes and macrogametes) were frequently found in the cytoplasm of hypertrophied host cells in the lamina propria at the tips of villi of the jejunum and ileum. The cytoplasm of the host cell was immunohistochemically positive for cytokeratin AE1/AE3 and cytokeratin 13 and was negative for vimentin, desmin, α-smooth muscle actin, chromogranin A, neuron-specific enolase, and factor VIII. The host cells parasitized by Eimeria species had the immunostaining characteristics of epithelial cells but not of mesenchymal cells, endothelial cells of lacteals or capillaries, smooth muscle cells or neuroendocrine cells. These results suggest that the host cell of Eimeria species is possibly derived from intestinal epithelial cells and then displaced into the lamina propria of the small intestine.
Infection with Eimeria species (E. leuckarti, E. solipedum, and E. uniungulsti) infection is a worldwide protozoal disease of horses.1,3 E. leuckarti is the only coccidium in horses noted in Japan.8 Routine histopathologic examinations of foals and older horses, with or without clinical symptoms of lower alimentary tract disorders, frequently reveal infection with the protozoan in the small intestine. The gametocytes develop in the cytoplasm of hypertrophic host cells in the lamina propria of the small intestine.
The life cycle and histopathologic characteristics of Eimeria species have been well described by early investigators;2 however, the origin of hypertrophic host cells is still unclear. Some early investigators proposed that host cells may be derived from mesenchymal, epithelial, or endothelial cells of lacteals or smooth muscle cells in the lamina propria.1,2,8 In the present study, histopathologic and immunohistochemical examinations were performed to investigate the charactaristics of the host cells parasitized by Eimeria species.
Five Thoroughbred foals with various clinical conditions, such as intractable mycotic inflammation of the guttural pouch, gastric ulcer, and obscure diarrhea, were examined. At necropsy, tissue samples from the small intestine were collected for histologic and immunohistochemical examinations. Samples were fixed in 10% neutral buffered formalin and dehydrated in a series of alcohols. Sections for histopathologic examination were cut at 4 μm from paraffin-embedded blocks and stained with hematoxylin and eosin (HE).
Serial sections were immunohistochemically stained using the avidin–biotin–peroxidase complex (ABC) procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA). Details of the primary antibodies used in this study are summarized in Table 1. Deparaffinized sections were blocked for endogenous peroxidase in 3% H2O2 for 10 minutes. All sections were incubated with primary antibody at 4 C for 16 hours, with biotinylated secondary antibody for 30 minutes at room temperature, and with avidin–peroxidase conjugate for 30 minutes. Sections were developed in 0.05% 3,3′-diaminobenzidine solution and counterstained with Mayer's hematoxylin. Control incubation using excess normal mouse and rabbit sera as the primary antibody and with the primary antibody omitted resulted in the absence of specific staining.
Immunohistochemical staining results for host cells in the small intestine of horses parasitized by Eimeria species.
MEC = mucoepithelial cells; SMC = smooth muscle cells; NEC = neuroendocrine cells; FC = fibrocytes; EC = endothelial cells; BM = basement membrane. + = immunopositive; - = negative.
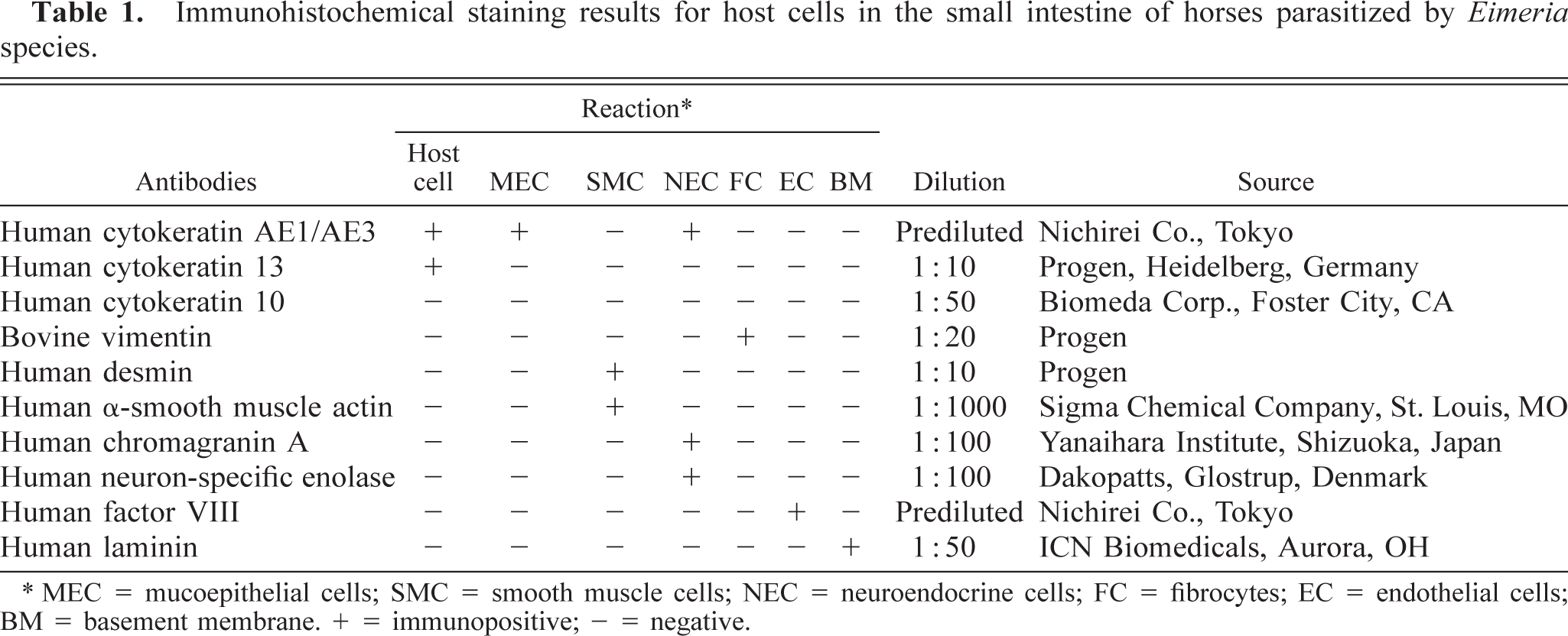
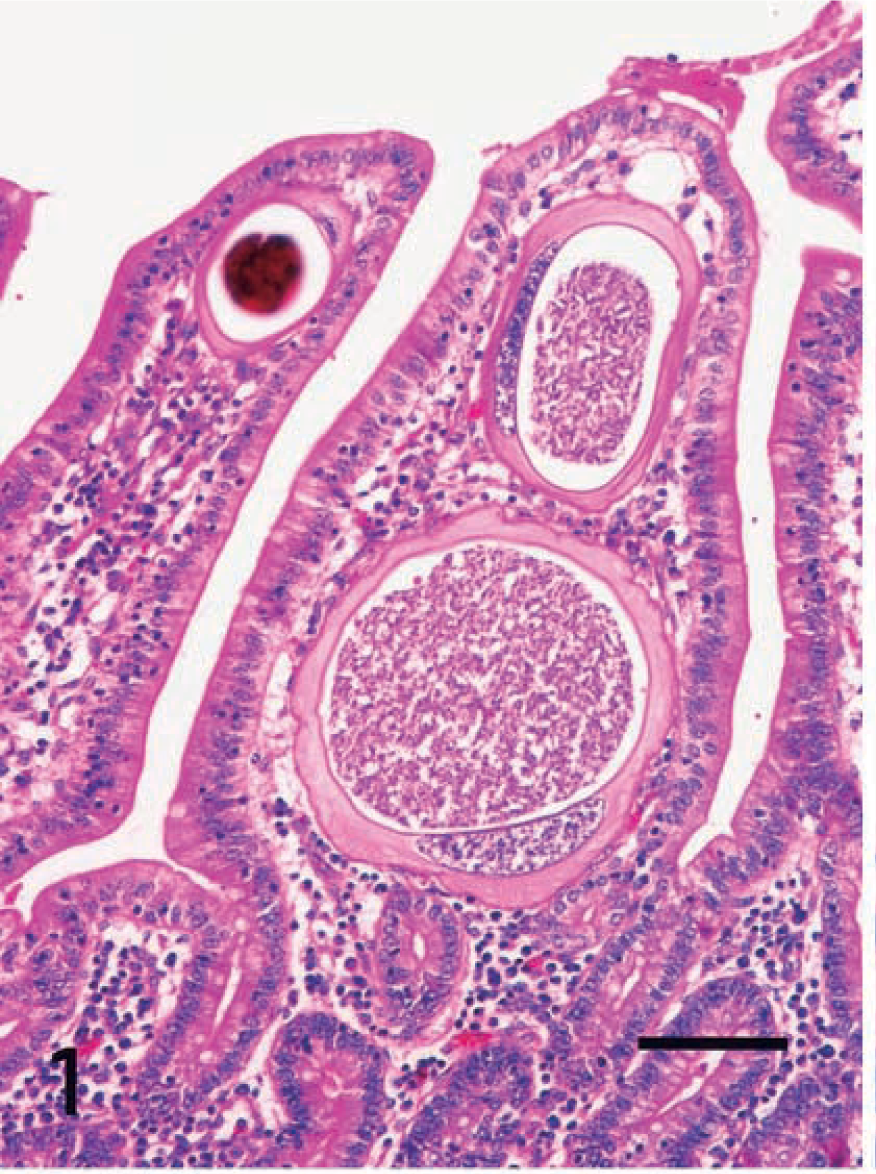
Eimeria organisms were frequently found in the cytoplasm of hypertrophied host cells in the lamina propria at the tips of villi of the jejunum and ileum. Atrophy of the villi usually occurred, although no erosions or ulcers were present. There was no accumulation of macrophages or lymphocytes around the host cells parasitized by Eimeria. Smaller numbers of eosinophils were present mostly in the tela submucosa. The host cells ranged from approximately 10 to 200 μm in diameter. The markedly hypertrophied cells were >250 μm in diameter. Smaller host cells were ovoid or spindle shaped and had round single nuclei and a small single vacuole. Markedly hypertrophied host cells had very large flattened single nuclei and large single vacuoles containing various stages of Eimeria organisms (early gametocytes, microgametes [Fig. 1], macrogametes [Fig. 2], and oocysts) in the cytoplasm. Large cells were flattened, with a signet ring appearance (Fig. 1). Occasionally, the host cells disappeared, in which cases the free extracellular parasites were frequently surrounded by macrophages and lymphocytes in the lamina propria. Giant cells occasionally contained the fragments of parasites in their cytoplasm.
Intestine; horse. Two hypertrophied host cells in the lamina propria of the jejunum contain microgametes of Eimeria species. HE. Bar = 40 μm.
Intestine; horse. A hypertrophied host cell contains macrogametes of Eimeria in its cytoplasm (lower left). A single vacuole is visible in the cytoplasm of a mildly hypertrophied cell (upper right). HE. Bar = 10 μm.
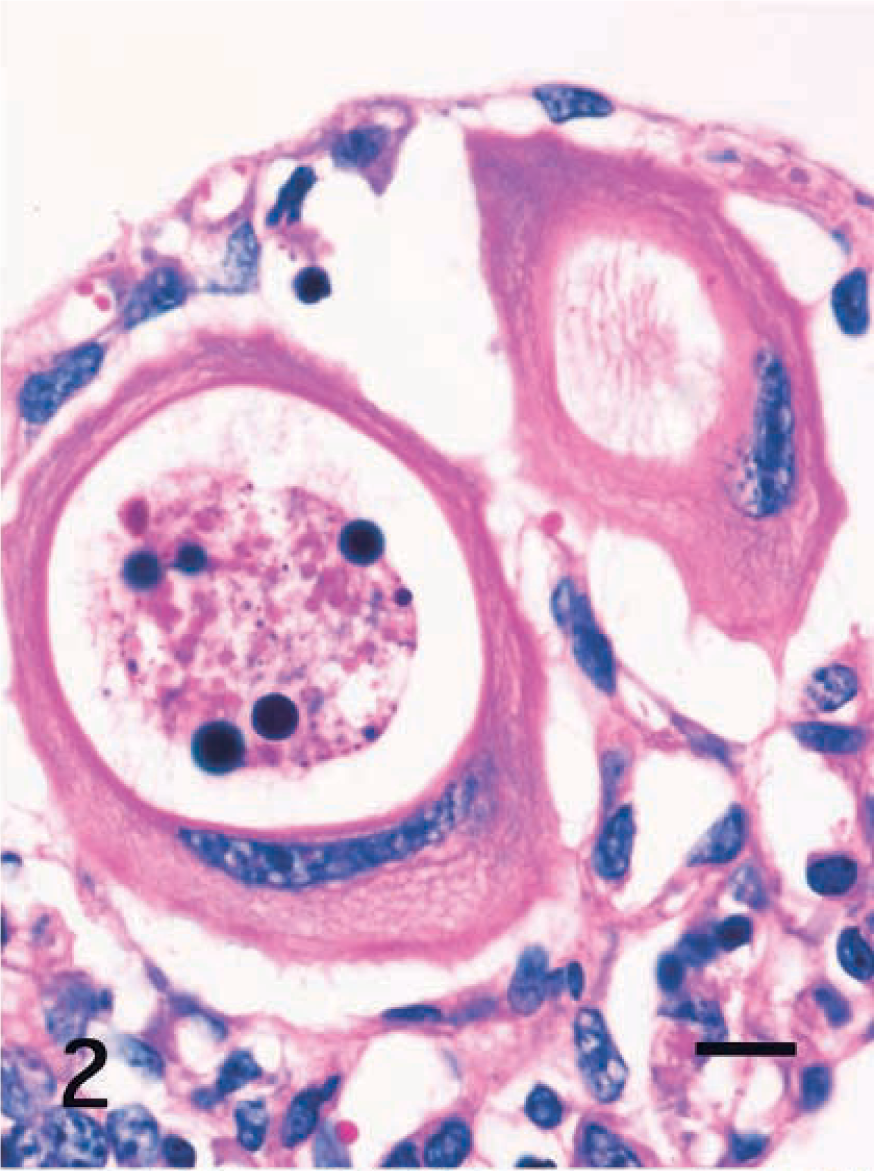
Immunohistochemical properties of the host cells are summarized in Table 1. In all cases, the cytoplasm of small and large host cells was strongly positive for cytokeratin (CK) AE1/AE3 (Fig. 3) and CK13 (Fig. 4) but did not react to CK10, vimentin (Fig. 5), desmin, α-smooth muscle actin (α-SMA), chromogranin A, neuron-specific enolase (NSE), or factor VIII. Smooth muscle cells in the lamina propria reacted to desmin and α-SMA. Mononuclear cells including macrophages and lymphocytes around host cells reacted only to vimentin (Fig. 5). Endothelial cells of lacteals or capillaries reacted to factor VIII and vimentin. The thin fibrillar layer around the hypertrophied host cells reacted positively to laminin (Fig. 6). The intact epithelial cells of the small intestine were positive for CKAE1/AE3 (Fig. 3) and negative for CK10 and CK13 (Fig. 4). Chromogranin A–positive cells were occasionally observed in the epithelial layer.
Intestine; horse. A markedly hypertrophied Eimeria host cell has a large, flattened single nuclei, and the cytoplasm reacts to cytokeratin AE1/AE3, giving a signet-ring appearance. ABC, Mayer's hematoxylin counterstain. Bar = 40 μm. Inset: A small host cell with a single vacuole has cytoplasm that reacts to cytokeratin AE1/AE3. ABC, Mayer's hematoxylin counterstain. Bar = 10 μm.
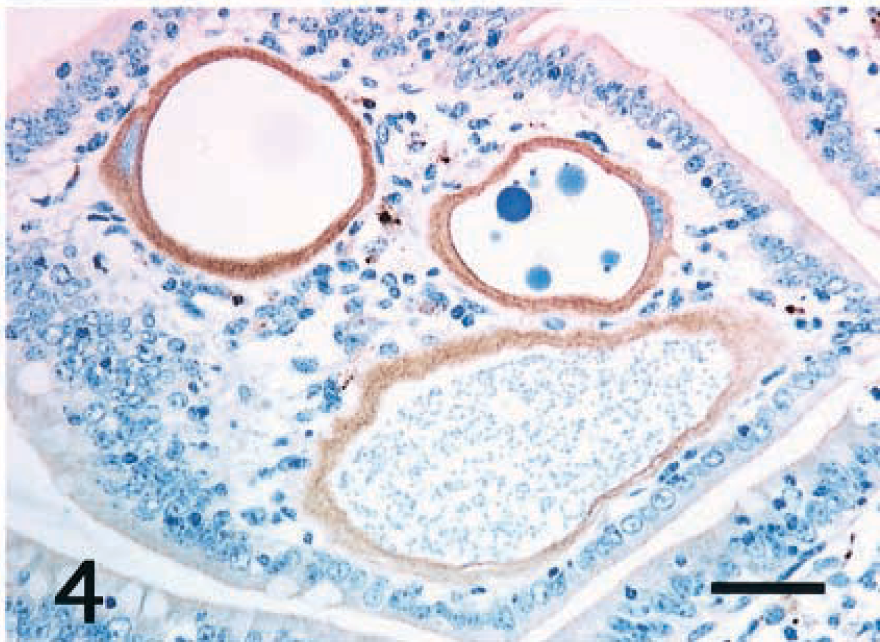
Intestine; horse. The cytoplasm of three hypertrophied Eimeria host cells reacts positively to cytokeratin 13. Epithelial cells of the jejunum do not react to cytokeratin 13. ABC, Mayer's hematoxylin counterstain. Bar = 40 μm.
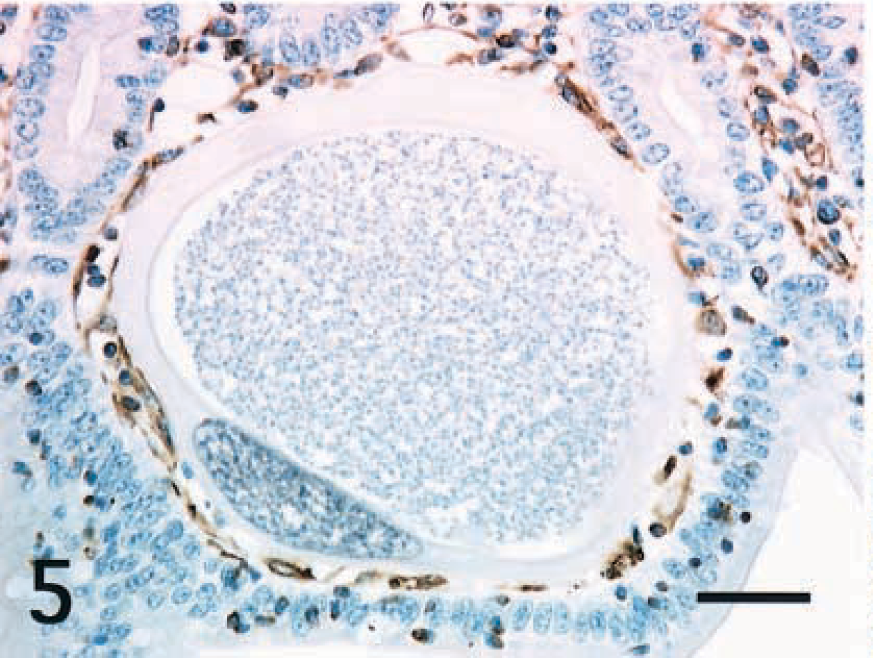
Intestine; horse. The cytoplasm of a hypertrophied Eimeria host cell does not react to vimentin. Interstitial cells around the host cell react to vimentin. ABC, Mayer's hematoxylin counterstain. Bar = 40 μm.
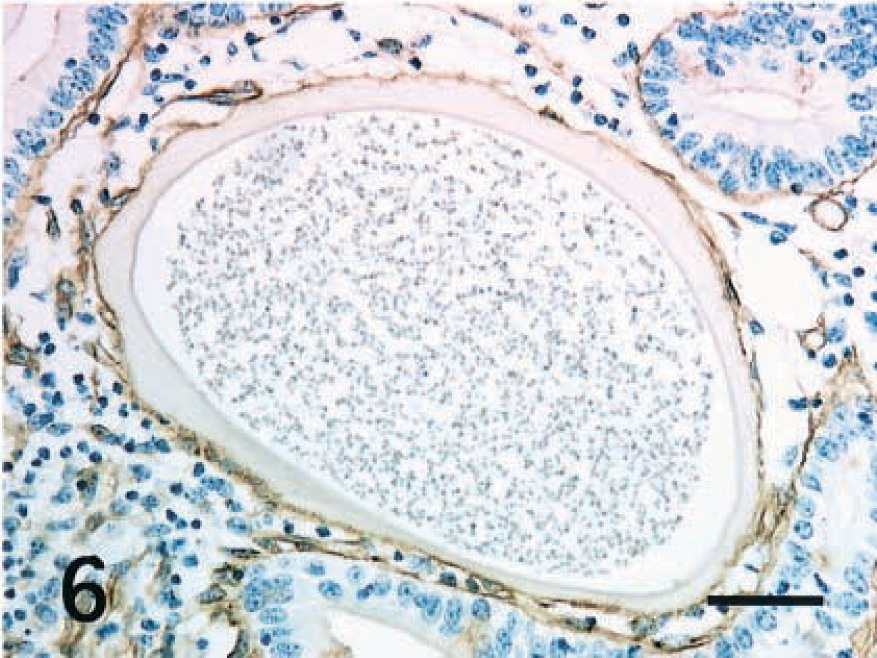
Intestine; horse. Thin fibrillar layer around the hypertrophied host cell reacts positively to laminin. ABC, Mayer's hematoxylin counterstain. Bar = 40 μm.
In the present study, histopathologic and immunohistochemical examinations revealed that the host cells parasitized by Eimeria species had characteristics of epithelial cells but not mesenchymal cells, endothelial cells of lacteals or capillaries, neuroendocrine cells, or smooth muscle cells. The cytoplasm of the host cells was strongly immunopositive for cytokeratin intermediate-size filaments of epithelial cells and immunonegative for vimentin filaments of mesenchymal cells. These results suggest that the host cells of Eimeria organisms are probably derived from the epithelial cells of the small intestine but not from any mesenchymal cells, including fibrocytes and fat cells in the lamina propria. Early reports concerning the origin of host cells mentioned that they might be smooth muscle cells or endothelial cells because their spindle-shaped appearance is similar to that of smooth muscle cells of the lamina muscularis mucosae or endothelial cells of the lacteals.1 Other workers assumed the host cells to be mesenchymal cells in the lamina propria because of the presence of early stage gametocytes.2,8
Anti-CKAE1/AE3 antibody is very useful for distinguishing between epithelial calls and mesenchymal cells because of its reaction against a wide range of cytokeratin intermediate-size filaments in the epithelial cells. CK13 is a characteristic major component of noncornified stratified squamous epithelia such as tongue mucosa, anal canal epithelium and tracheal epithelium.7 Increasing expression of CK13 is accompanied by squamous differentiation in the tracheobronchial epithelial cells under physiologic conditions.5 The expression of CK13 polypeptide in squamous metaplasia of the human uterine cervix was investigated by immunocytochemical techniques. The development of metaplastic foci was accompanied by the expression of CK13 polypeptide.4 Thus, cytokeratin synthesis in the metaplastic epithelial cells may possibly be altered with commitment to terminal differentiation of the cells under pathologic conditions. In these foals, the expression of CK13 suggests that the host cells took on the nature of noncornified stratified squamous epithelial cells as a result of infection by Eimeria species. Chromogranin A and NSE are useful markers for neuroendocrine cells in the small intestine. Many neuroendocrine cells that reacted positively to chromogranin A and NSE antibodies were distributed in the epithelial layer of the small intestine. However, the host cells parasitized by Eimeria did not react to either antibody.
The life cycle of E. leukariti has been well studied experimentally.1 Early gametocytes were found in the host cells in the lamina propria of villi in the small intestine at 14 days postinoculation (PI). By 23 days PI, macrogametes and microgametocytes could be microscopically distinguished in the host cells. At 28 days PI, macrogametes had begun formation of an oocyst wall in the cytoplasm of host cells.1 These findings suggest that the lifespan of host cells parasitized by E. leukariti is probably at least 28 days, even though the lifespan of normal intestinal epithelial cells may be 2–3 days. Thus, host cells parasitized by E. leukariti have a much longer lifespan than normal epithelial cells. Noncornified stratified squamous epithelia with expression of CK13, such as tongue mucosa and anal canal epithelium, have a much longer lifespan than do epithelial cells of the small intestine. Therefore, CK13 expression by the host cells may mean that the lifespan of host cells was eventually extended because of intracellular development of Eimeria organisms. How the host cells were dislocated to the lamina propria and achieved this long life span remains unclear.
Laminin is a major component of the basement membrane in many tissue types, contributes to the extracellular matrix architecture, and influences cell adhesion, spreading, and migration. Moreover, laminin regulates cell proliferation and has the capacity to specify cell and tissue development, differentiation, and function.6 The basal lamina is necessary for the polarity of normal intestinal epithelial cells. In these foals, all of the small and large host cells had a surrounding thin basal lamina that reacted positively to laminin. These findings suggest that the epithelial host cells might lose their polarity and then migrate into the lamina propria or might lose their polarity as a result of the migration. Thus, laminin may regulate the displacement of the host cells parasitized by Eimeria into the lamina propria from the epithelial layer.
The results of the present study suggest that Eimeria organisms develop in the cytoplasm of epithelial cells displaced into the lamina propria of the small intestine.
