Abstract
A 4-year-old Quarter Horse gelding was presented with a history of weight loss of 6 months duration, along with extensive ventral subcutaneous edema. Clinicopathologic findings included a markedly low serum total protein (2.9 g/dl) and a low packed cell volume (24%). The mucosal surface of the distal jejunum and entire ileum were carpeted with numerous polypoid, papillary, and glandular masses comprised of pseudostratified tall columnar cells and large numbers of interspersed goblet cells. Neoplastic change was diffuse throughout the mucosa of each mass, but abrupt demarcation occurred between neoplastic masses and adjacent mucosa. Immunohistochemical staining for protein of the p53 tumor suppressor gene revealed only occasional cytoplasmic reactivity within polyps and normal mucosa. Nuclear staining for papillomavirus antigens was not observed. Electron microscopic examination revealed features of well-differentiated intestinal epithelial cells, including apical tight junctions and microvilli, desmosomes, and the presence of numerous goblet cells. Microorganisms were not detected. Small intestinal polyposis should be considered as a rare differential diagnosis for protein-losing enteropathy in the horse.
Intestinal adenomatous polyps (adenomas) are benign neoplastic lesions composed of tubular, villous, or tubulovillous intestinal epithelial proliferations.1–3 They are common tumors of humans, occurring most frequently in the colon.3 In the small intestine, the duodenum is the most common site, and the ileum is least commonly affected. Intestinal adenomatous polyps may occur spontaneously or as part of one of several inherited syndromes, with greater numbers of large intestinal polyps being more characteristic of the familial forms.2,3 Progression of colorectal polyps to carcinomas is a relatively well-defined event in humans.2 Intestinal adenomatous polyps are uncommon in domestic animals, occurring most frequently in the colon and rectum of dogs.1,5,11 Malignant transformation has been reported in only a small number of canine lesions.5,11 Adenomatous polyps have also been reported in the intestinal tract of cattle,1 sheep,1 and cats.1,7,9 In the Quarter Horse described in this report, the occurrence of numerous benign adenomatous polypoid tumors in the distal jejunal and ileal mucosa resulted in marked hypoproteinemia and mild anemia.
A 4-year-old Quarter Horse gelding failed to thrive and lost approximately 91 kg in body weight since being purchased 6 months previously. The horse presented to the University of Florida College of Veterinary Medicine with extensive ventral edema. Clinicopathologic abnormalities included a total protein of 2.9 g/dl (reference 5.5–7.5 g/dl), with albumin of 1.5 g/dl (reference 2.7–4.2 g/dl) and globulins of 1.4 g/dl (reference 2.1–3.8 g/dl). A normocytic and normochromic anemia also was present, with packed cell volume of 24% (reference 32–47%). Abdominocentesis revealed a white blood cell count of 33,700/μl, with the majority of cells (82%) being nondegenerate neutrophils. The animal was euthanatized due to a poor prognosis for recovery and submitted for necropsy.
Arising from the mucosa of the distal 6 m of the jejunum and of the entire ileum were numerous 0.5 to 5-cm-diameter, raised, multinodular purple to tan, moderately firm to friable, polypoid masses (Fig. 1). The masses were generally pedunculated, and were arranged both singly and in multilobulated botryoid clusters. The intervening mucosa was normal in appearance. Polypoid lesions were not noted in the stomach, duodenum, or large intestine. Other gross lesions included slight enlargement of the mesenteric lymph nodes, and marked expansion of the subcutis of the ventrum and prepuce by clear fluid. Tissues were immersed in 10% neutral buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin. Paraffin-embedded sections of a small intestinal polyp were processed using a standard avidin–biotin immunohistochemical protocol, using a polyclonal rabbit-origin primary antibody to human p53 tumor suppressor gene protein (Polyclonal antibody CM-1, Signet Laboratories, Dedham, MA).12 Additional serial sections were tested with a rabbit polyclonal antibody (Anti-BPV-1, DAKO, Santa Barbara, CA) directed against papillomavirus group-specific antigens.6 For electron microscopy, 1-mm cubes of fresh tumor were immersed for fixation in 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.2). The samples were postfixed in 2% osmium tetroxide, dehydrated through graded alcohols, then embedded in Spurrs/Epon (1:1) resin. Ultrathin sections were stained with uranyl acetate and Reynold's lead citrate, and examined on a Hitachi H-7000 transmission electron microscope.

Ileal adenomatous polyposis; horse. Multinodular polypoid masses are arranged singly and in multilobulated botryoid clusters in the mucosa. Only small areas of mucosa are free of tumor (arrow). Bar = 2 cm.
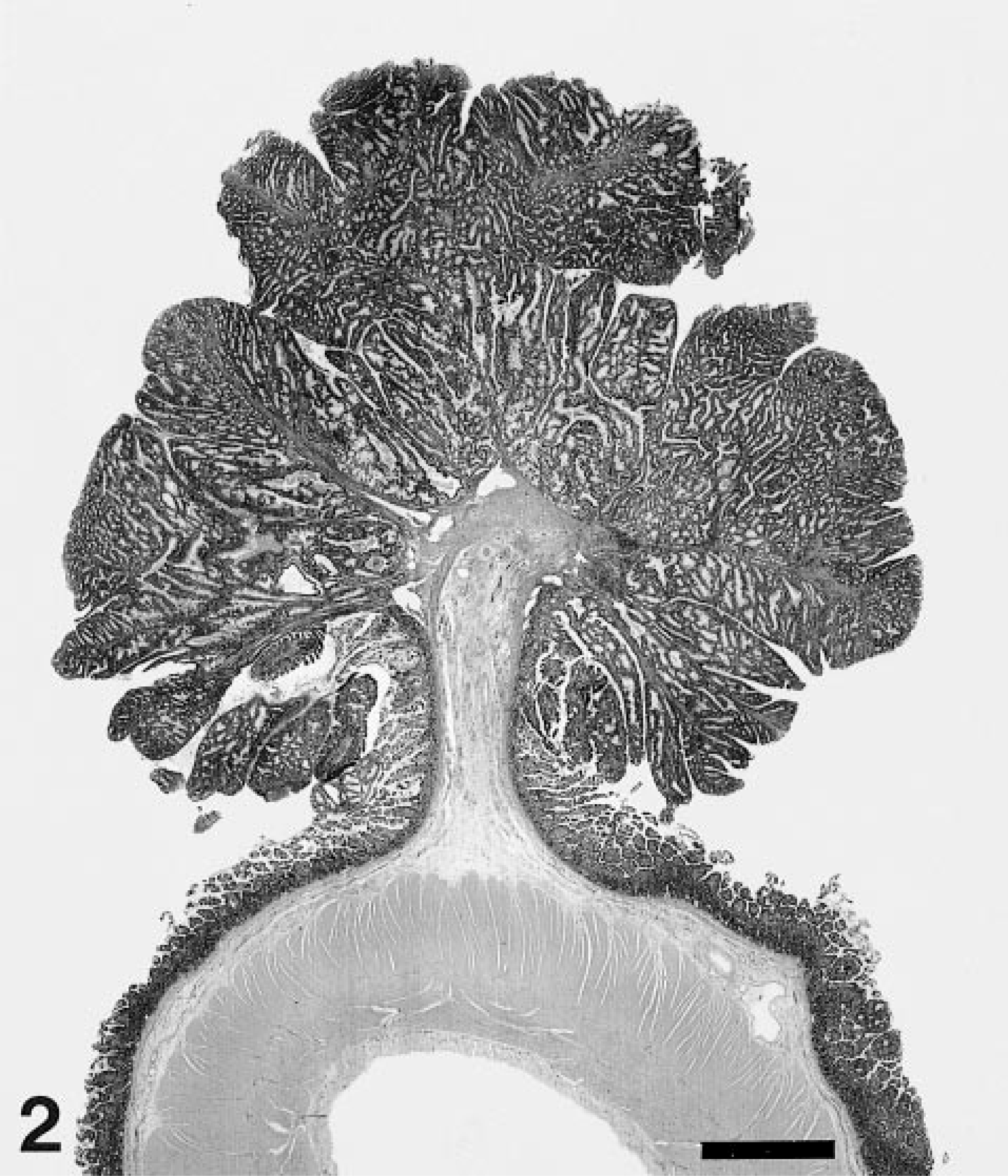
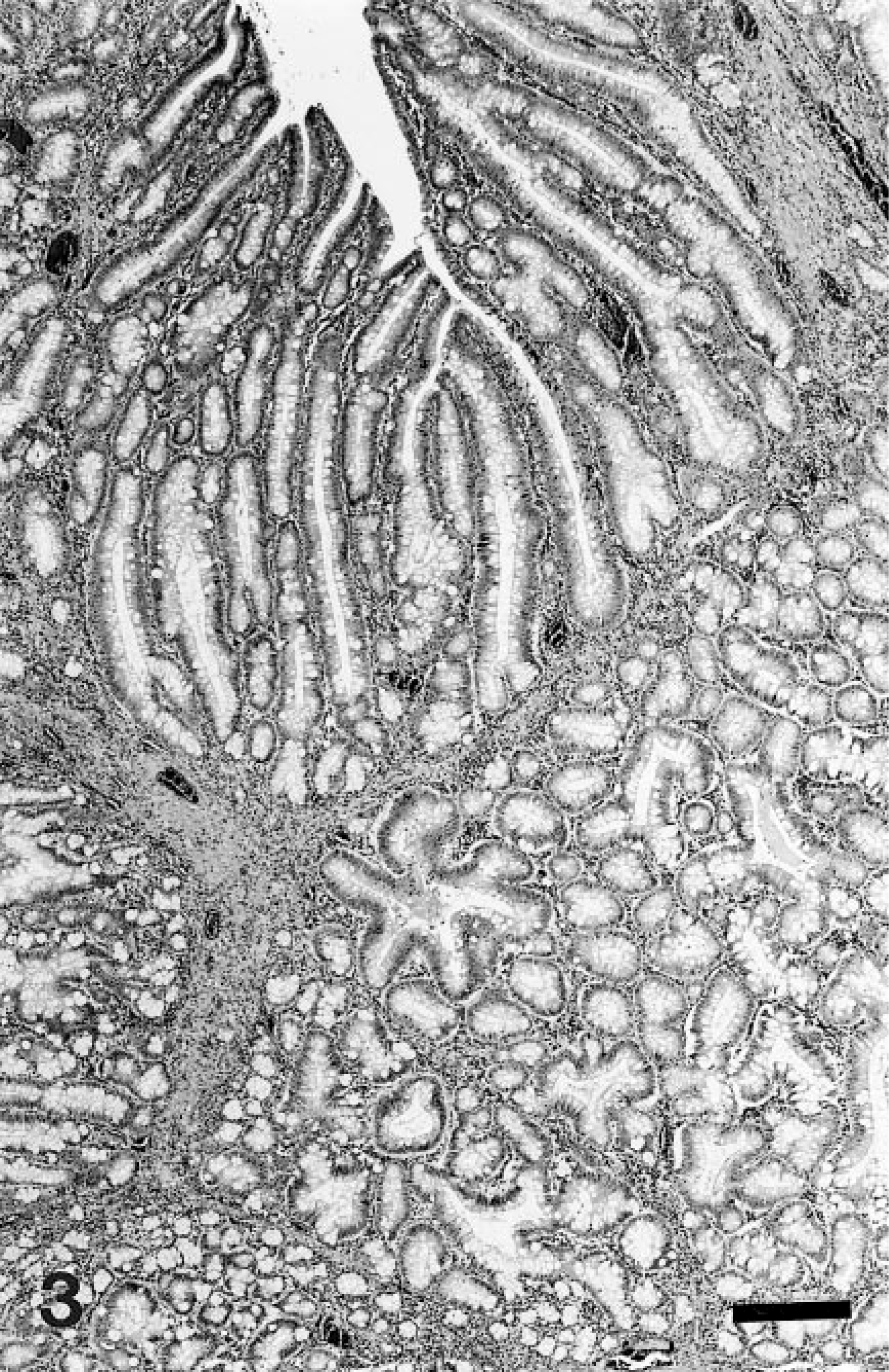
Microscopically, the small intestinal masses were irregularly shaped, papillary, and glandular, with abrupt transition from adjacent unaffected mucosa (Fig. 2). Branching papillary fronds with invaginated rows of epithelium and numerous variably dilated acinar structures were lined by closely packed tall columnar epithelial cells and large numbers of goblet cells (Fig. 3). Epithelial cell nuclei were elongate, vesicular, and often stratified; low numbers of mitotic figures were observed. Acinar structures were ovoid, irregularly shaped, branching, or tubular. Some dilated glands contained small to moderate numbers of degenerate neutrophils. Papillary stroma was composed of loose fibrous tissue subtended by moderately thick, branching stalks of dense fibrous tissue arising from the underlying muscularis mucosa and submucous. Small numbers of lymphocytes, plasma cells, neutrophils, and globular leukocytes were scattered through the stroma. A Warthin–Starry stain did not reveal bacterial organisms. Based on the gross and microscopic appearances, the lesions were diagnosed as tubular adenomatous polyps. Other histopathologic findings included moderate lymphoid hyperplasia of mesenteric lymph nodes, mild erythroid hyperplasia of the bone marrow, mild to moderate portal hepatitis, and mild interstitial nephritis without evidence of proteinuria.
Ileal adenomatous polyposis; horse. Mass is pedunculated, papillary, and glandular. The transition from adjacent to unaffected mucosa is abrupt. HE. Bar = 0.25 cm.
Ileal adenomatous polyposis; horse. In papillary component of mass are invaginated rows of pseudostratified columnar epithelium and goblet cells. Similar cells line acinar structures. A moderate stromal leukocytic infiltrate is present. HE. Bar = 350 μm.
Immunohistochemical staining of sections of a small intestinal polyp for p53 protein revealed scattered cytoplasmic staining of epithelial cells within the lesion, and within the adjacent intestinal mucosa. Positive nuclear staining was not observed. Nuclear staining for papillomavirus was not observed. Ultrastructurally (Fig. 4), tumor cells were closely apposed. Modest numbers of organelles were found within the cytoplasm, including clustered or interspersed mitochondria, dispersed endoplasmic reticulum, and scattered small vesicles. The cytoplasm of goblet cells was distended by 1- to 2-μm-diameter ovoid to round membrane-bound granules, sometimes with an eccentric, electron-dense, 0.5-μm round core, and contained abundant endoplasmic reticulum. Much of the lumenal surface was occupied by goblet cells. Apical borders of intervening epithelial cells were often lined by microvilli, and occasional tight junctions were seen. Elsewhere, scattered desmosomes were observed. Viral and bacterial organisms were not detected in the lesion.
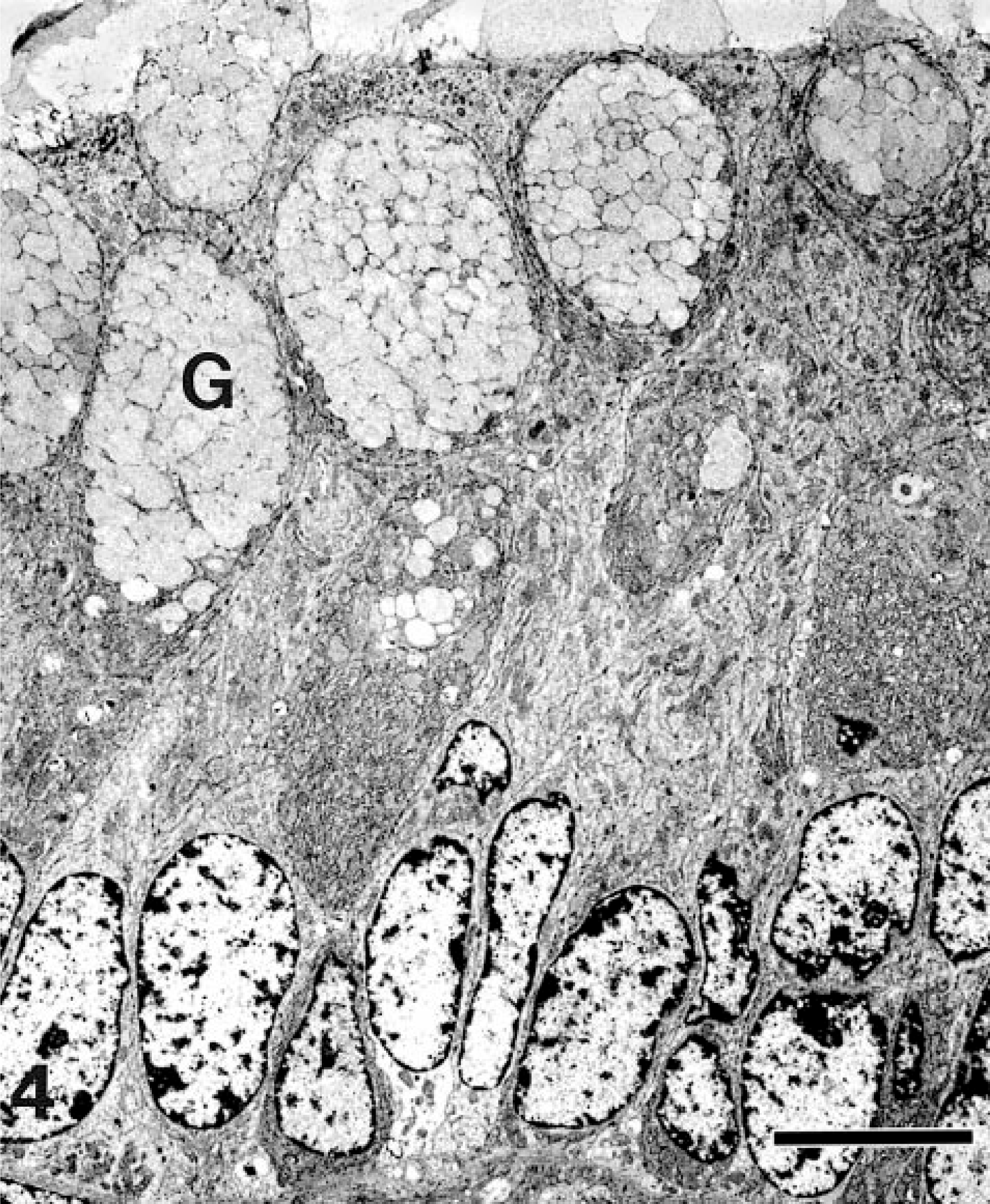
Transmission electron micrograph. Ileal adenomatous polyposis; horse. Cells are closely apposed in this well-differentiated tumor. Mucus-containing portions of goblet cells (G) line the lumenal surface. Goblet cells contain abundant endoplasmic reticulum. Other cells contain modest numbers of cytoplasmic organelles. Bar = 4 μm.
The numerous small intestinal tubular adenomas in this horse resembled tumors associated with adenomatous polyposis syndromes in humans, mice, and cats,2,3,9,10 except that tumors occur most frequently in the colorectum of humans.2 Ultrastructural features of tumor cells were consistent with well-differentiated intestinal epithelial cells.4 Adenomatous polyposis may occur as a familial or spontaneous disease in humans, with exposure to dietary carcinogens or carcinogenic by-products of bacterial metabolism being suggested as potential etiologic factors in the spontaneous form.2 A familial cause has also been suggested for some Asian cat breeds.9 The progression from adenoma to carcinoma in humans involves sequential mutations in several specific tumor-suppressor genes and in the K-ras oncogene, and forms the basis of the “multihit” concept of carcinogenesis.2 The initial step in the development of tumors is a mutation in the Apc gene, a tumor-suppressor gene. Individuals affected by familial syndromes may inherit one mutant allele of the Apc gene. The multiple intestinal neoplasia (Min) mouse, resulting from a nonsense mutation in the Apc gene, has been developed as an animal model to investigate the role of APC protein function in intestinal tumorigenesis.8,10 Subsequent mutations sequentially involve the remaining allele of the Apc gene, the K-ras oncogene, the deleted in colon cancer (DCC) tumor-suppressor gene, and the p53 tumor suppressor gene.2 The propensity for malignant transformation in small intestinal and colonic polyps is related to their size and number,2,3 implying a high likelihood of occurrence in individuals affected by polyposis. Although mutation of the p53 gene is a late event in the progression of human tumors, one study of canine benign and malignant colorectal tumors demonstrated greater overall positive immunohistochemical staining of benign tumors for p53 protein.12 The suggestion was made that mutation of the p53 gene may be an early occurrence in the progression of canine tumors. Malignant transformation was not detected in any tumors from the horse in this report, and immunohistochemical staining of tissue sections of a lesion from this horse demonstrated only occasional cytoplasmic staining for p53 protein. Thus, the progression of equine lesions may be similar to that of humans, with mutation of the p53 gene being a later event. Another possibility is that the antibody to human p53 protein binds poorly to the equine protein. Serial sections of the equine tumor were stained for papillomavirus antigens because papillomaviruses can induce benign epithelial neoplasms in animals.6 However, immunohistochemical staining and electron microscopic examination did not reveal a viral etiology.
Adenomatous polyposis in the small intestine of this horse was associated with extensive ventral edema and marked hypoproteinemia that could not be attributed to renal loss, endoparasitism, chronic hepatic disease, or poor diet. Involvement of the distal jejunal and ileal mucosa was extensive, and the hypoproteinemia was presumed to be result of hemorrhage and loss of serum from the more friable masses. Accelerated intestinal epithelial cell turnover, loss of mucoproteins, and altered permeability of tight junctions could have also contributed to the loss of protein.1 Small intestinal adenomatous polyposis should be considered as a rare differential diagnosis for protein-losing enteropathy in the horse.
Footnotes
Acknowledgements
Special thanks to S. Whittaker of the ICBR Electron Microscopy Laboratory (University of FLorida), B. Hall of the UFCVM Immunohistochemistry Laboratory, and Drs. R. Bronson, C. Buergelt, L. Fox, P. Ginn, and R. Smith for their insightful review of this manuscript. This paper is published as University of Florida, College of Veterinary Medicine, Journal Series Number 549.
