Abstract
The role of tumor suppressor genes in the pathogenesis of canine melanoma is incompletely understood. The genes encoding the tumor suppressors p53, Rb, p21 (
Melanoma is a common tumor in dogs that arises from melanocytes or melanoblasts. It is the most common malignancy found in the oral cavity and on the digits in the dog.21,65 Dermal melanomas are usually benign, but uveal, digital, and oral melanomas generally are malignant and respond poorly to standard therapies.46 There are, however, exceptions to these generalities; a few dogs with oral melanoma are cured of the disease, whereas others with dermal melanoma die from metastasis. Thus, shared genetic characteristics may be as or more important than location.
The natural history and pathogenesis of canine melanoma are incompletely understood, but recent work has begun to elucidate the role of growth regulation genes in the development of this disease.46 Cellular growth and division is a complex pathway governed by competing growth inhibitors and promoters. Proto-oncogenes encode proteins that promote cell cycle progression, proliferation, and survival in normal cells; tumor suppressor genes do the opposite, encoding proteins that inhibit cellular proliferation and promote cell death.68 For cells that have undergone malignant transformation, the balance between proliferation and inhibition is upset.
For this study, we used reverse transcription (RT) polymerase chain reaction (PCR) assays, immunoblotting, and immunocytochemistry to examine gene expression and protein accumulation of the p53, p21 (
Reduced or absent expression of p16 and PTEN and accumulation of p53 in an inappropriate subcellular location were the most frequent abnormalities identified in this study. Loss of these tumor suppressor pathways seems to be a common occurrence that may contribute to the pathogenesis of canine melanoma. Further studies are necessary to confirm the prognostic significance of these biomarkers in canine melanoma.
Materials and Methods
Cell lines and normal tissues
The melanoma cell lines utilized for this study were derived from primary tumors of dogs with oral melanoma (CML-2, CML-13, JENNY, SCOOTER, TLM-1), a lymph node metastasis of cutaneous melanoma (CML-6MC2), and a lung metastasis of oral melanoma (SHADOW) and have been described previously.12,33,56,73 The Cf2Th line, originating from normal canine thymic epithelial cells, was obtained from the American Type Culture Collection (Rockville, MD). Normal tissue samples obtained from healthy dogs at the time of euthanasia were generously provided by Dr. John Bauer. The procedures for handling and euthanasia of the dogs and for the transfer of samples to this study were approved by the Institutional Animal Care and Use Committee of Texas A&M University.
Clinical cases
Clinical cases were retrieved from the medical records database of the Texas Veterinary Medical Center. Every case that had a histologic diagnosis of melanoma between 1994 and 1997 was reviewed for eligibility; 31 tumor samples with formalin-fixed archival material that were suitable for immunostaining were utilized. Samples were excluded when 1) blocks did not contain sufficient material for serial sectioning, 2) tumor material was not evident on routine hematoxylin and eosin–stained recuts, 3) blocks had evidence of improper or incomplete fixation, such as loss of cellular detail along plasma membrane and nuclear membrane or decreased staining intensity, 4) blocks had evidence of improper or incomplete infiltration of paraffin (based on their gross appearance), and 5) tissues were negative for vimentin control stains, where normal stromal tissues in the block were evaluated for vimentin expression by immunostaining. The features of some of these cases were reported previously.33 Some tissue blocks did not yield sufficient tissue to analyze all of the markers included in this study. The eligible cases included 11 primary oral tumors (one tongue, three lip, seven gingiva), 18 dermal melanomas (three digit, three limb, two eyelid, three trunk, one ear, one scrotum, five unspecified), and two lymph node metastasis (one from an oral tumor and one from a digital tumor). Twenty-one tumors were pigmented, and ten were amelanotic. The tumors were categorized as malignant or benign based on pleomorphism, degree of proliferation, local invasion, and documentation of metastasis. Pleomorphism and proliferation were not by themselves considered adequate indicators of malignancy in the absence of known recurrence, invasiveness, or documentation of metastasis. Using these criteria, 14 tumors were benign, 11 were identified as malignant, and the behavior of six was not evident from the histology or history. The samples represented 29 dogs and 17 different breeds: four Gordon Setters, four Doberman Pinschers, three Labrador Retrievers, two Miniature Schnauzers, two Boxers, one each of Giant Schnauzer, Brittany Spaniel, Bichon Frise, Cairn Terrier, Bull Terrier, Weimaraner, Bull Mastiff, Dachshund, Shetland Sheepdog, Golden Retriever, English Pointer, and Greyhound, and two mixed-breed dogs. The dogs ranged in age from 2 to 14 years, with an average age of 11 years and a median age of 12 years. The age of one dog was unknown. Thirteen of the dogs were neutered males, three were intact males, 12 were spayed females, and one was an intact female.
Cell culture
Tissue culture materials were obtained from Nalge Nunc (Naperville, IL). Cells were cultured in Dulbecco's modified Eagle medium (DMEM, Gibco BRL, Grand Island, NY) containing 10% heat-inactivated fetal calf serum (FCS, Hyclone, Logan, UT) in a humidified atmosphere containing 5% CO2 at 37 C. All the cell lines grew as monolayer cultures and were maintained by passage when they reached >90% confluence.
Identification of a partial cDNA for canine ink-4a
The sequence for canine
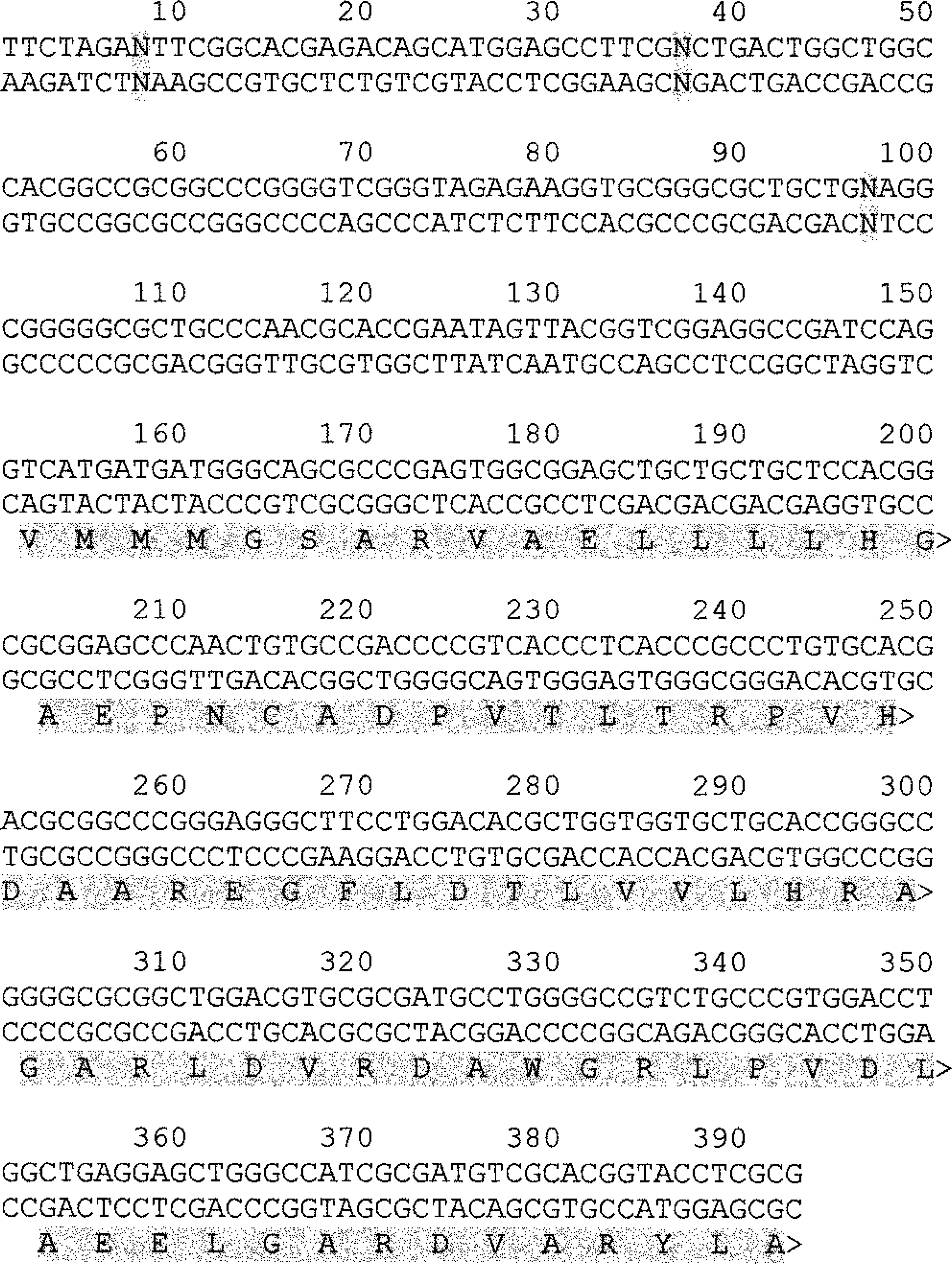
Double-stranded nucleotide sequence of a partial canine

Aligned canine and human amino acid sequences for p16 exon 2. Asterisks represent sequence identity. Conserved substitutions are represented by a dash under the sequence. A blank space under the sequence represents nonconserved substitutions. The regions of the human protein for which the matching canine sequence is unknown are highlighted in gray.
Gene expression
Gene expression was analyzed by RT-PCR as previously described.55 RNA was isolated using the RNAWiz kit (Ambion, Austin, TX) as recommended by the manufacturer. Complementary DNA synthesis was accomplished using a kit (Roche Diagnostics, Chicago, IL) followed by PCR amplification with dog-specific primers. The sequence of the complete coding domains for the
Immunocytochemistry and immunohistochemistry
For cell lines, at the end of the culture period plastic chambers were removed, and slides were rinsed in phosphate buffered saline (pH 7.4), fixed in acetone at 4 C for 5 minutes, and air dried. For archival tissues, 5-μm serial sections from paraffin-embedded blocks were mounted onto positively charged slides (Probe-on, Fisher Scientific, Pittsburgh, PA). Immunostaining was performed using a modified streptavidin–biotin complex method previously described.55,56 For paraffin-embedded sections, antigen retrieval procedures included microwave heating for 6 minutes in a buffer of 0.1 M sodium citrate (pH 6) for p21, p16, Rb, and PTEN staining and microwave heating for 6 minutes in Retrieve All (Signet Pathology Systems, Dedham, MA) for p53. Antigen retrieval was not used for staining of the cell lines, except in the case of p21, where microwave heating for 6 minutes in sodium citrate buffer was used to unmask nucleolar staining. Slides were incubated for 1 hour at room temperature with primary antibodies against p53 (antibody CM-1, diluted 20-fold, Signet Pathology Systems), p21 (antibody C-20, 5 µg/ml, Santa Cruz Biotechnology, Santa Cruz, CA), Rb (mouse monoclonal antibody G3–245, 5 µg/ml, Pharmingen, San Diego, CA), p16 (antibody H-156, 0.5 µg/ml, Santa Cruz Biotechnology), or PTEN (antibody A2B1, diluted 100-fold, Santa Cruz Biotechnology). Secondary antibodies consisted of goat anti-rabbit IgG or goat anti-mouse IgG conjugated to biotin (Kirkegaard & Perry Laboratories, Gaithersburg, MD). The presence of the relevant antigens was detected using streptavidin conjugated to alkaline phosphatase. Following a rinse in Tris buffer, the color reaction was accomplished using the Histomark red kit (Kirkegaard & Perry Laboratories). Negative controls were prepared by using irrelevant isotype matched antibodies in place of the primary antibodies. Sections obtained from canine tissues (liver, kidney, brain, spleen, gut, skin) served as positive controls for the immunostains. To our knowledge, there are no previous reports of immunohistochemical staining for p16 and PTEN in fixed and embedded canine tissues. The conditions and specificity for staining were optimized in one of our laboratories (J. Wojcieszyn) based on those reported by Levine.37 Expression of p16 in the normal tissues examined was most intense in hepatocytes, with localization to the cytoplasm and prominent accumulation in Golgi areas. Moderately intense staining also was seen in cell bodies of the gray matter in the brain. Mononuclear leukocytes showed faint cytoplasmic staining. Expression of PTEN in the normal tissues examined was prominent in vascular structures, with the most intense staining in renal glomeruli.
Cell cultures and cell lines were graded for intensity and percentage of positive cells. Samples were considered negative when no staining was seen above that seen in negative controls. Staining that was fine, diffuse, and not always visible at low magnification (40–100×) was considered weak. Staining that was punctate or regional but prominent enough to be seen at low magnification was considered moderate. Staining that was diffuse, prominent, and easily visualized at low magnification was considered strong. In negative to weak (− to −/+) samples, weak staining was visible in <10% of the cells in the tumor. Samples were only considered positive when expression of these proteins was clearly detectable in melanocytic tumor cells (versus supporting stroma or inflammatory cells) based on the expression of S100a protein, Melan A, or neuron-specific enolase in serial sections from the tumors.
Immunoblotting
Whole cell lysates were made by disrupting cells in a high-salt buffer (300 mM sodium chloride, 50 mM Tris, pH 7.6, 0.5% Triton X-100, 1 mM
Statistics
Statistical analyses were performed at the Biostatistics Core facility of the AMC Cancer Research Center and the University of Colorado Cancer Center. Data were analyzed using the SAS statistical package. Correlation among the different variables was determined using Spearman rank correlation analysis.
Results
Expression of p53
Alterations in p53 appear to be a late event in the progression of melanoma. Such alterations can include deletion of the gene or mutations that silence the gene, disrupt the DNA binding domains, or alter the stability or subcellular localization of the protein.26,36 However, many other proteins that interact with p53 affect its stability and subcellular localization,26,36 thus, overexpression of p53 protein in tumor cells does not necessarily imply mutation or loss of the
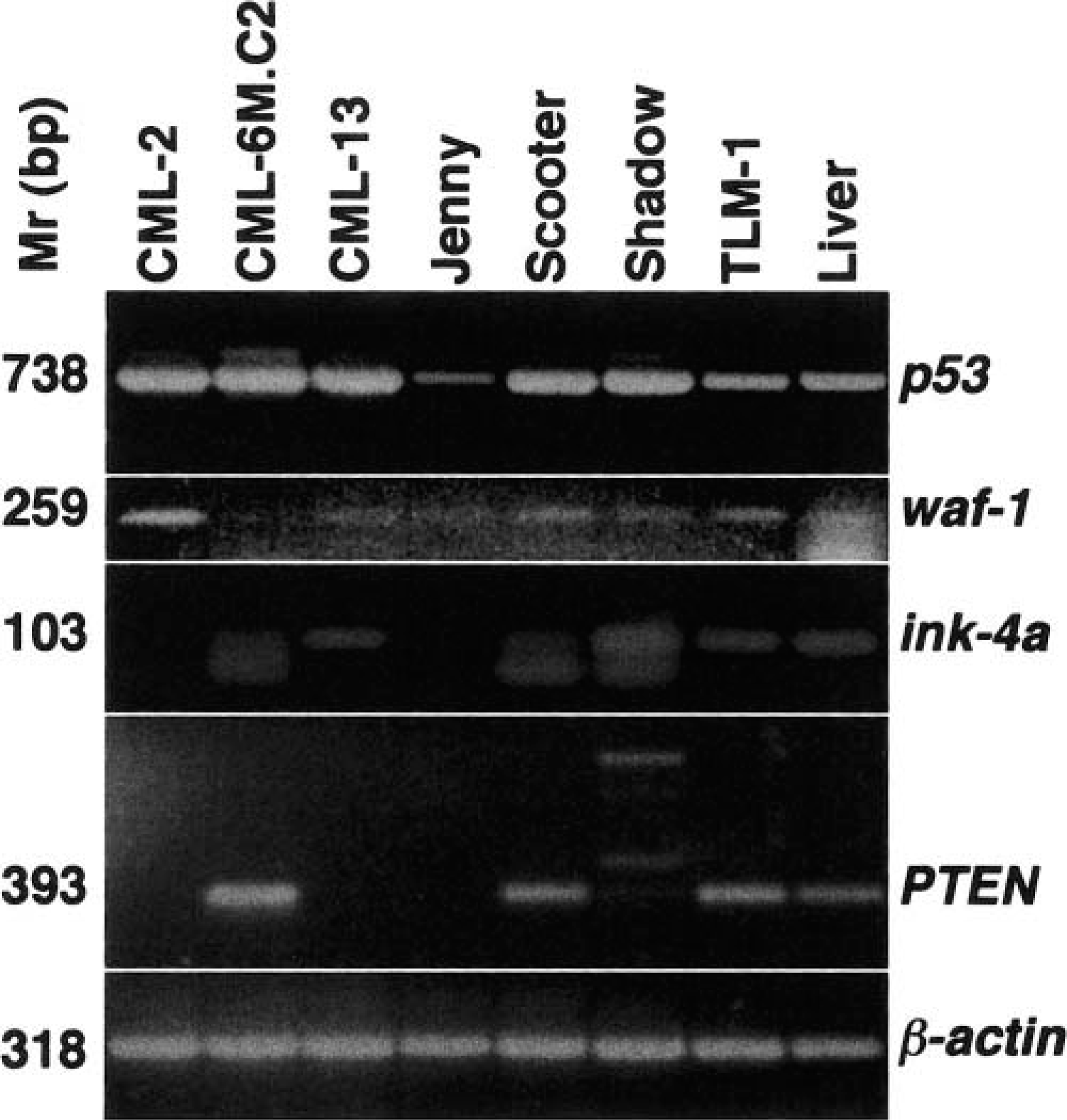
Expression of mRNA for each tumor suppressor gene (
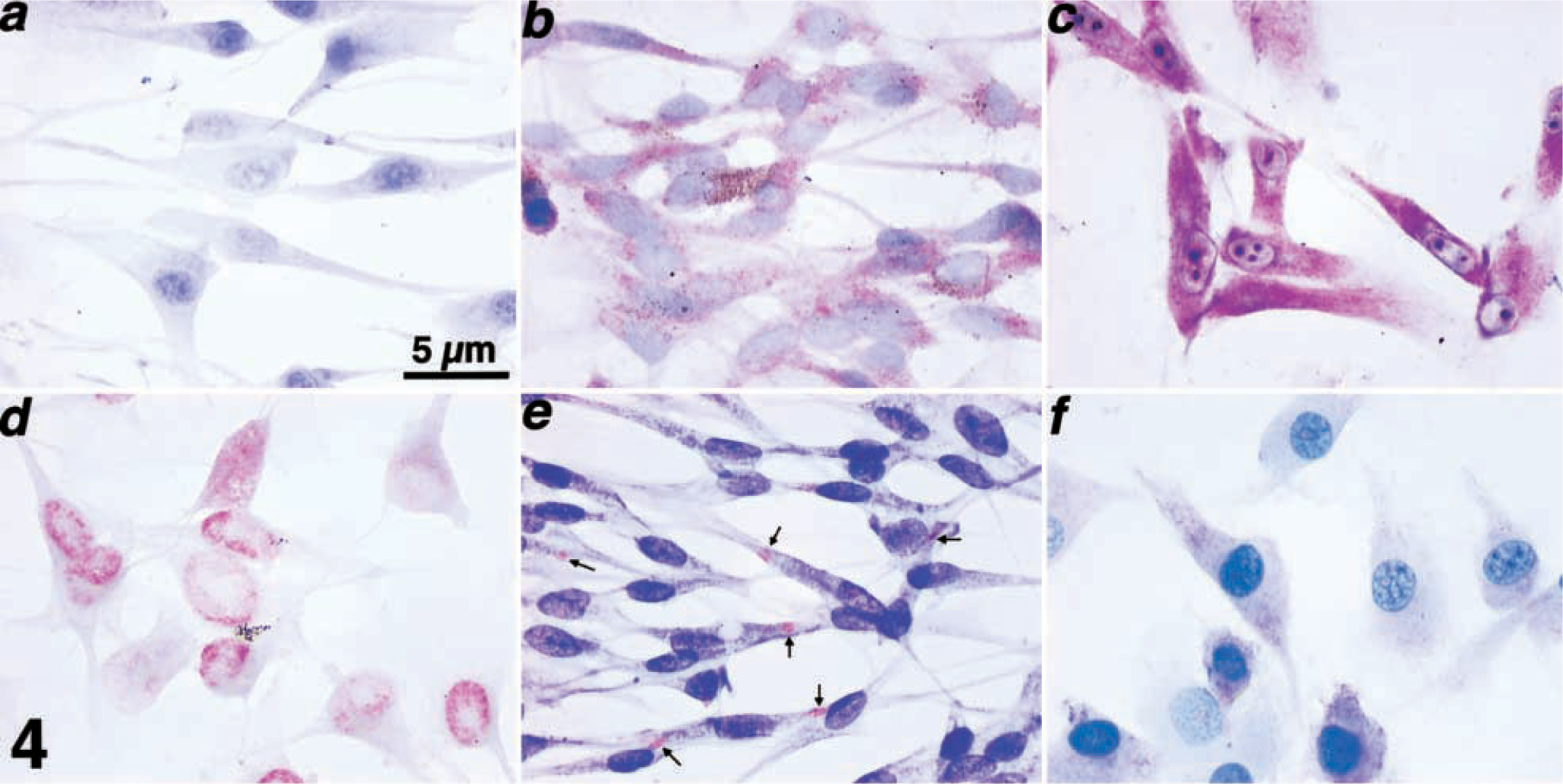
Immunocytochemical assessment of the expression and accumulation patterns for p53, p21, Rb, p16, and PTEN in canine melanoma cell lines.
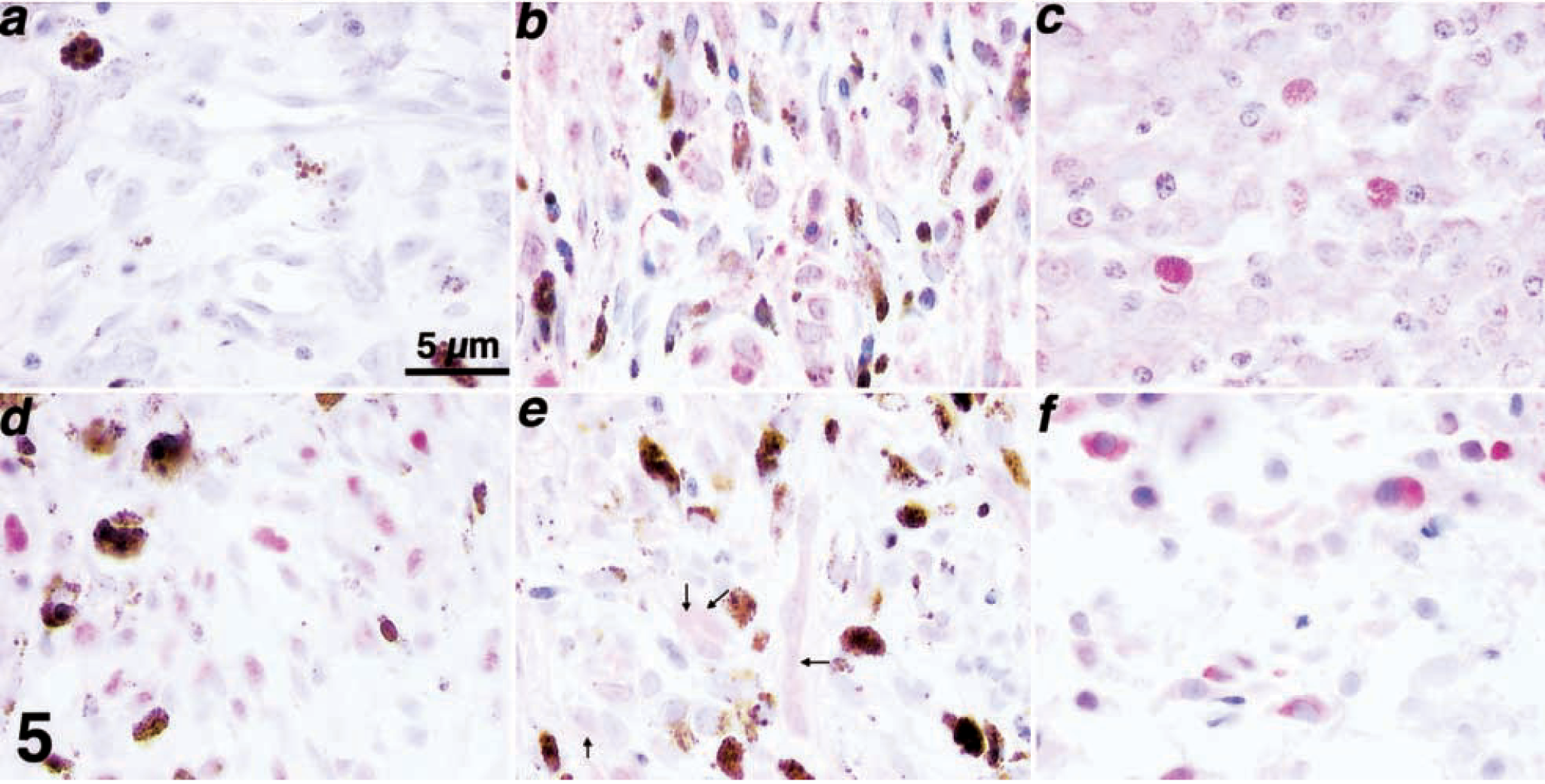
Immunohistochemical assessment of the expression and accumulation patterns for p53, p21, Rb, p16, and PTEN in samples from spontaneous cases of canine melanoma.
Immunostaining results for the seven canine melanoma cell lines.
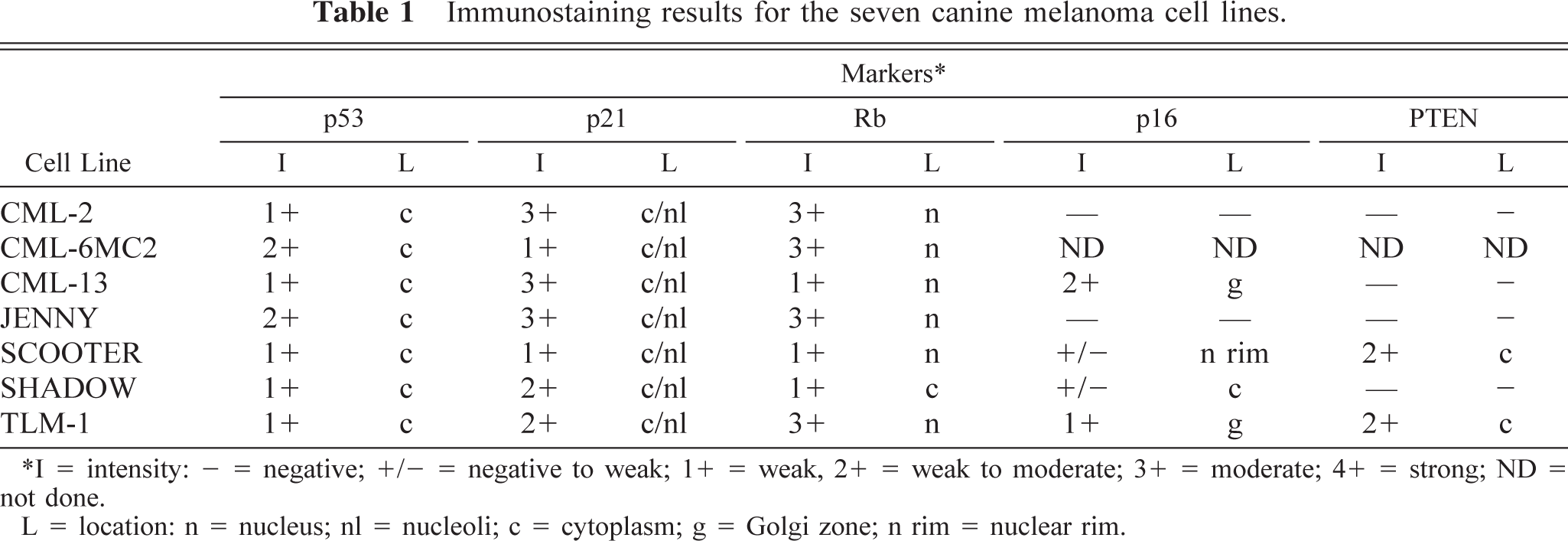
I = intensity: - = negative; +/− = negative to weak; 1+ = weak, 2+ = weak to moderate; 3+ = moderate; 4+ = strong; ND = not done.
L = Location: n = nucleus; nl = nucleoli; c = cytoplasm; g = Golgi zone; n rim = nuclear rim.
Loss or dramatic reduction of p53 expression (graded as negative or negative to weak) was seen in nine of 30 samples (five benign, three malignant, one of indeterminate behavior, Table 2). Three of these samples (Nos. 8, 10, 11) were from dogs with multiple, cutaneous, benign melanocytic tumors, with loss or reduction of p53 in every independent tumor examined. Exclusion of p53 from the nuclear compartment occurred in 18 of 25 samples with detectable p53 staining (eight benign, seven malignant, three of indeterminate behavior; Table 2). Figure 5b shows the p53 staining for sample No. 2, where the protein was shown to localize to the cytoplasmic and nuclear compartment. The two samples (Nos. 4, 8) from dogs with multiple benign melanocytic tumors with detectable p53 expression that were part of this latter group similarly showed localization of p53 exclusively in the cytoplasm in every independent tumor examined.
Immunostaining results for the 31 archival canine tumors.
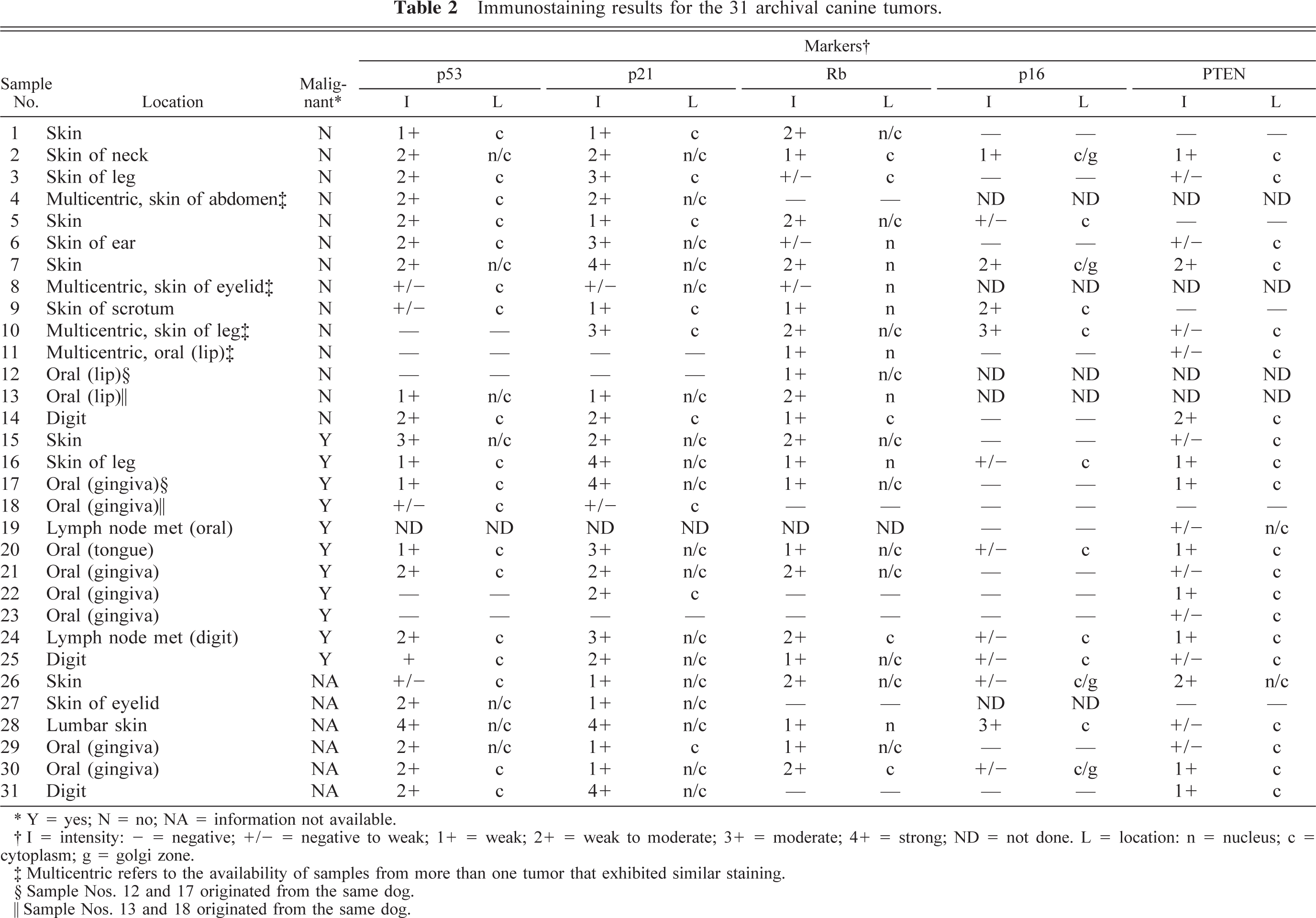
Y = yes; N = no; NA = information not available.
I = intensity: + = negative; +/− = negative to weak; 1+ = weak; 2+ = weak to moderate; 3+ = moderate; 4+ = strong; ND = not done. L = location: n = nucleus; c = cytoplasm; g = golgi zone.
Multicentric refers to the availability of samples from more than one tumor that exhibited similar staining.
Sample Nos. 12 and 17 originated from the same dog.
Sample Nos. 13 and 18 originated from the same dog.
Expression of p21
The
Loss or dramatic reduction of p21 expression (graded as negative or negative to weak) was seen in six of 30 samples (three benign, two malignant, one of indeterminate behavior; Table 2). Two of these samples (Nos. 8, 11) were from dogs with multiple, cutaneous, benign melanocytic tumors, with loss of p21 in every independent tumor examined. Exclusion of p21 from the nuclear compartment occurred in nine of the 27 samples with detectable p21 staining (six benign, two malignant, one indeterminate, Table 2). One of the three samples from dogs with benign multiple melanocytic tumors with detectable p21 expression (sample No. 10) that was part of this latter group similarly showed localization of p21 exclusively in the cytoplasm in every independent tumor examined. Figure 5c shows the p21 staining for sample No. 21, where the protein was localized in the cytoplasmic and nuclear compartments.
Expression of Rb
Mutations of Rb do not appear to be prevalent in human melanoma, possibly because mutations of other genes that participate in the same growth control pathway occur so often as to render Rb expression inconsequential.8,51,54,59,60,75 The expression of Rb in canine melanoma has not been evaluated extensively. We used immunocytochemistry to evaluate the expression and subcellular localization of Rb in the canine melanoma cell lines. We previously verified the specificity of the G3–245 antibody against canine Rb by immunoblotting.47,55 The results show nuclear accumulation of Rb in six of the seven cell lines (Table 1, Fig. 4d). Not all the cells from the CML-13 and SCOOTER lines showed Rb staining, although this inconsistency could be attributable to the fact that these cells were growing asynchronously. Alternatively, loss or reduction of
Loss or dramatic reduction of Rb expression (graded as negative or negative to weak) was seen in nine of 30 samples (four benign, three malignant, two of indeterminate behavior; Table 2). Two of these samples (Nos. 4, 8) were from dogs with multiple, cutaneous, benign melanocytic tumors, with loss of Rb in every independent tumor examined. Exclusion of Rb from the nuclear compartment occurred in four of the 24 samples with any detectable Rb staining (three benign, one of indeterminate behavior; Table 2) but was not seen in any of the samples from dogs with multiple benign melanocytic tumors. Figure 5d shows the localization of Rb staining restricted to the nuclear compartment in sample No. 13.
Expression of ink-4a
Mutation of
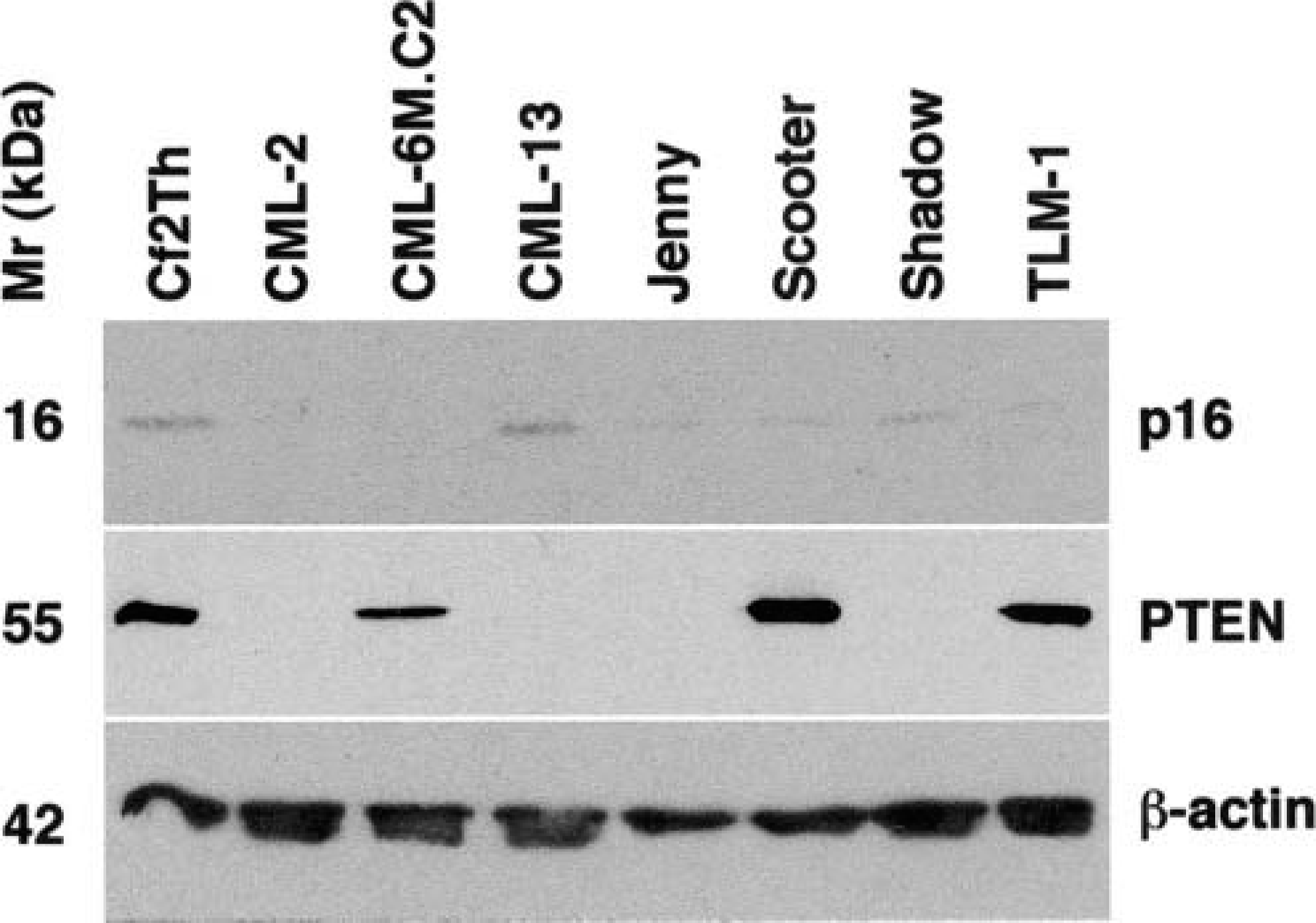
Accumulation of p16 and PTEN tumor suppressor proteins in canine melanoma cell lines CML-2, CML-6MC2, CML-13, JENNY, SCOOTER, SHADOW, and TLM-1 was examined by immunoblotting. Analysis of β-actin was used as a loading control. Normal canine Cf2Th cells were used to evaluate the expression of these proteins in a nonneoplastic tissue sample. The sizes for the proteins are 16 kd for p16, 55 kd for PTEN, and 42 kd for β-actin.
Loss or dramatic reduction of p16 expression (graded as negative or negative to weak) was seen in 21 of 26 samples (six benign, 11 malignant, four of indeterminate behavior; Table 2). When present, staining of p16 was generally confined to the cytoplasm, although intense staining in the Golgi area was seen in four cases (two benign and two indeterminate). The pattern of p16 accumulation is illustrated by the immunostaining for sample No. 7 (Fig. 5e).
Expression of PTEN
The importance of the
Discussion
Melanomas are malignant tumors of melanocytes or melanoblasts. Cultured cells usually resemble the morphology of the parent tumor from which they were derived;15,33,49,56 however, cultured melanoma cells are not the same as tumors in vivo. Theoretically, cell lines can mutate and may not possess all the same characteristics of the parent tumor. Variation in environmental conditions may also influence growth and differentiation. Nevertheless, cultured melanomas are a useful preliminary model to study the origin and progression of melanoma. Combined with analysis of primary archival sections, this approach provides a robust model to define the frequency with which the expression of particular genes or proteins may be lost (or amplified) in these tumors and to investigate possible biological consequences of these abnormalities.
In this study, we used RT-PCR and immunocytochemistry to examine tumor suppressor gene and protein expression in seven cultured melanoma cell lines and 31 archival samples of melanoma from 29 affected dogs. The gene products we chose to examine, p53, p21, Rb, p16, and PTEN, have each been implicated to some extent in the origin or progression of melanoma in humans or other animal models.1,11,22,28,46,55,56,58,59,70,71 We specifically sought to investigate whether the pathways of growth control, survival, and DNA integrity in which these proteins participate might contribute to the genesis of canine melanoma. Our working hypotheses were 1) mutations in these pathways are common in canine melanoma and 2) mutations in more than one pathway predict a worse outcome. The results presented here are consistent with the first hypothesis. Our results also show that these genetic lesions are common in both the benign and malignant forms of canine melanoma, but additional work is necessary to document their prognostic significance.
We previously proposed a predisposition of individual dogs to develop multiple melanocytic tumors as a result of a mutation in a melanocyte precursor, based on a shared genetic characteristic in the tumors.56 This study includes data from the same dog (sample No. 11) and three additional dogs with multiple, cutaneous, benign melanocytic tumors. Two additional dogs were included that developed two distinct tumors (sample Nos. 12, 17; sample Nos. 13, 18), but in both of these dogs, one of the tumors was benign and one was malignant. Three of these dogs were Gordon Setters, and three of the four Gordon Setters in the study developed more than one melanoma in their lifetime. The results from this study confirm the presence of a shared phenotype in each of the tumors from dogs with multiple, cutaneous, benign melanocytic tumors, supporting the premise that these tumors may have arisen from a common precursor. However, such shared phenotype was not seen in the dogs that developed more than one tumor with different biological behavior (i.e., one benign and one malignant), suggesting that different factors may underlie the pathogenesis of these two apparently related tumors.
The observation that abnormalities indicative of loss of p16/Rb function (including reduced or absent expression of either gene or exclusion of Rb from the nuclear compartment) occurred most frequently in both the cell lines (six of seven) and the clinical samples (22 of 26) suggests that inactivation of this pathway is a critical step in the pathogenesis of benign and malignant melanoma in dogs, as it is in humans and other animals.48,59,61,72 Mutation of
Mutations of Rb do not appear to be prevalent in human melanoma,39 possibly because mutations of
Exclusion of p53 from the nuclear compartment occurred almost as frequently as loss of p16/Rb. The cytoplasmic accumulation of p53 in the cell lines and tumor samples was not a feature of the antibody or the conditions used or an artifact of tissue culture; p53 was localized almost exclusively in the nuclear compartment of Cf2Th cells,55 and seven tumor samples contained abundant nuclear p53. In human melanomas, mutations of the
Loss or reduction of PTEN expression also was seen frequently in the cell lines but less so in the tumor samples. The cell line that did not exhibit abnormalities of p16 or Rb (CML-13) showed loss of PTEN expression, suggesting that enhanced survival or growth mediated through growth factor pathways that use PI3K may help to overcome the effects of normal p16/Rb pathways in canine melanoma. Three cell lines (CML-2, JENNY, and SHADOW) showed concurrent abnormalities of p16 or Rb and PTEN, suggesting that concurrent loss (or reduction) of both the p16/Rb and PTEN pathways may facilitate progression of the disease. At least two mechanisms may account for loss of PTEN expression. Of the four cell lines that showed mutations of the
An important role for p21 in melanocyte growth and differentiation has been documented.30,32,44,47,55 The p21 protein is a CDKI that has nonoverlapping functions with p16, but it also participates in contact-induced growth arrest and controls the function of stress-activated protein kinases, polymerases involved in DNA synthesis, proteins that regulate survival and apoptosis, and possibly cell division (cytokinesis).2,6,23,24,64,67,74 There was no clear correlation between the presence of p21 protein and the morphology or growth properties of the cell lines. One mechanism through which p21 expression can be upregulated is through transcriptional activation mediated by p53,13 and p21 appears to be important in p53-dependent growth arrest in cells that must undergo DNA repair.67 However, p21 expression can also be induced through pathways independent of p53.42,76,77 We saw no apparent correlation between p21 and p53 in the seven cell lines. Loss or reduction of p21 was also an infrequent occurrence in the clinical samples, but exclusion of p21 from the nuclear compartment was disproportionately seen in cutaneous melanomas, suggesting that loss of function of this protein may play a role in this form of the disease.
In this group of samples, anatomic location remained the best predictor for tumor behavior. Multiple abnormalities in these proteins were not significantly more common in malignant lesions than in benign lesions. This lack of significance may be due to the limited statistical power afforded by the relatively small sample size. Alternatively, its could signify that the function of these genes is important to prevent malignant transformation of melanocytes, but once transformation takes place their loss of function does not significantly impact disease progression. Nevertheless, the results showed distinct patterns with shared abnormalities. For example, the absence of functional p53 was significantly correlated with absence of nuclear p21. Similarly, loss or reduction of p16 was correlated with abnormalities of Rb. These results were predictable because p53 is an important regulator of p21 expression and because abnormalities in a single member of the p16/Rb pathway render other members of the pathway inconsequential.
The results from this study show that abnormalities of tumor suppressor genes and proteins are common in canine melanomas. These abnormalities likely reflect mutations that may contribute to the origin of the tumors in vivo. The possibility that malignant tumors accumulate mutations in multiple tumor suppressor gene pathways at an accelerated rate that may influence prognosis merits further investigation in spontaneous canine tumors.
Footnotes
Acknowledgements
We thank Dr. Steve Dow, Dr. Lauren Wolfe, Dr. Elaine Ostrander, and Dr. David Beach for providing cell lines and reagents, Josy Mayor and Dr. Vijayanagaram Venkatraj for assistance cloning canine
