Abstract
The immunophenotype of tumor cells and inflammatory infiltrate associated with cutaneous melanocytic lesions (29 melanocytomas, two malignant melanomas, and 23 residual lesions) from 54 adult Iberian and Iberian X Duroc pigs were examined using a panel of nine antibodies. All neoplastic cells were vimentin−, cytokeratin−, and alpha-1-antitrypsin− and the majority were S100+, whereas all pigmented macrophages were vimentin+, cytokeratin−, and S100− and most expressed alpha-1-antitrypsin. Regressing tumors were characterized by zones with low density of neoplastic cells accompanied by heavy infiltration of CD3+ T lymphocytes, whereas zones with high density of neoplastic cells showed very low numbers of CD3+ T lymphocytes. The infiltrate of CD79a+ B cells and IgG, IgM, and IgA plasma cells was low. The majority of lymphocytes of the peri- and intratumoral infiltrate were major histocompatibility complex class II+, but neoplastic cells did not express class II antigen. The 17 residual lesions examined were composed of macrophages containing abundant melanin pigment and low to moderate numbers of CD3+ T lymphocytes. The results of the present study suggest that the local cellular immune response plays a crucial role in the host response that induces regression of cutaneous melanomas and melanocytomas of the Iberian and crossbred Iberian X Duroc pigs.
Certain breeds of swine such as Duroc, Sinclair, and Hormel have a hereditary predisposition to develop cutaneous melanocytic tumors,11,12,15 whereas these tumors are rare in other swine breeds.22,28 Swine melanocytomas are more often reported than are malignant melanomas, which may metastasize to lymph nodes or internal organs. Both swine melanocytomas and malignant melanomas are often obvious at birth or develop early in life.15,28
Melanomas of Sinclair miniature swine often regress spontaneously; regression is often accompanied by intense intra- and peritumoral infiltration of macrophages and lymphocytes. Thus, melanoma in Sinclair swine is considered an excellent animal model for study of the immunologic mechanisms involved in tumor regression.10,15,18,21
Recently, an extremely high prevalence of melanocytic lesions was reported in 3,176 crossbred Iberian × Duroc pigs and purebred Iberian pigs.7 In this population, 95% of pigs had cutaneous lesions, 16% had hyperpigmentation in the oral cavity, 10% had lesions of the mediastinal lymph nodes, and 9% had lesions in the thymus. Only 0.15% of the cutaneous melanocytic lesions were malignant melanomas, whereas the remainder were either melanocytomas (often showing signs of regression and abundant lymphocytic infiltration) or residual lesions.7 This high prevalence of swine cutaneous melanocytic lesions has not been reported before, but familial predisposition was suspected because these lesions have been reported in Duroc pigs11,12 and all pigs were from the same geographic region (north of the province of Cordoba, Andalucía).7 The nature of the inflammatory infiltrate associated with regressing melanocytic tumors has been investigated in the Sinclair miniature swine, but no studies have been carried out in other swine breeds. The large number of melanocytic tumors at different stages of regression provided a good model for studying the nature of the local inflammatory infiltration and its relationship with regression of tumors in crossbred Iberian × Duroc and purebred Iberian pigs. These investigations were conducted to provide further information about the cellular components involved in the spontaneous regression of melanocytic tumors in swine.
Materials and Methods
Tissue samples
Skin samples from 54 crossbred Iberian × Duroc pigs or purebred Iberian pigs were taken at the slaughterhouse. Tissue samples were fixed in 10% buffered formalin and embedded in paraffin. For the histopathologic study, 4-μm-thick tissue sections were stained with the hematoxylin and eosin with and without blanching by incubation in 0.25% potassium permanganate for 30–60 minutes depending on the amount of pigment and then by incubation in 0.1% oxalic acid for 5–8 minutes.
Immunohistochemistry
The avidin–biotin–peroxidase complex (ABC) technique with blanching and the streptavidin–biotin–alkaline phosphatase (SBA) technique without blanching were used. Endogenous peroxidase activity was blocked by incubation with 0.3% hydrogen peroxide in methanol for 30 minutes. Sections for the detection of CD3 (Dako, Glostrup, Denmark), and IgG, IgM, and IgA (Serotec, Humbeek, Belgium) were incubated with 0.1% pronase (Sigma Chemical Co., St. Louis, MO) for 10 minutes, whereas those for the detection of CD79a (Dako) were incubated in 0.01 M citrate buffer in microwave oven at 100 C for 7 minutes. After three rinses in 0.01 M phosphate-buffered saline (PBS), pH 7.2, tissue sections were incubated with 5% normal goat serum for 30 minutes at room temperature (20–25 C). Details of primary antibodies used are given in Table 1. Cross-reactivity with porcine tissues was reported for vimentin (Eurodiagnostics, Appeldorn, the Netherlands) cytokeratins 5 and 8 (RCK 102, Eurodiagnostics),12 CD3, CD79a, major histocompatibility complex (MHC) class II antigen,26 and alpha-1-antitrypsin (A1AT) (Dako), and S100 protein (Master Diagnostica, Granada, Spain).25 After three-10 minute rinses in PBS, a biotinylated goat anti-rabbit IgG diluted 1:200 in PBS (Vector Laboratories, Burlingame, CA) was applied for 30 minutes as a secondary reagent for primary polyclonal antibodies, and a biotinylated goat anti-mouse IgG diluted 1:50 in PBS (Dako) was applied for 30 minutes as secondary reagent for primary monoclonal antibodies. For the ABC technique, an ABC solution (Vector Laboratories) diluted 1:50 in PBS was applied for 1 hour at room temperature. Tissue sections were then incubated in 3–3′-diaminobenzidine tetrahydrochloride (Sigma), diluted to 0.035% in Tris-buffered saline containing 0.01% hydrogen peroxide for 1 minute. Sections were rinsed in tap water, counterstained with Mayer's haematoxylin, dehydrated, and mounted. For the SBA technique, an SBA solution (Biogenex Laboratories, San Ramon, CA) diluted 1:50 was applied as a third reagent for 1 hour. Tissue sections were then rinsed in PBS and incubated in Tris-PBS (pH 7.4) containing 0.5% fast red (Biogenex Laboratories) for 5 minutes. After counterstaining with Mayer′s haematoxylin, sections were mounted with Immu-mount (Shandon, Pittsburgh, PA).
Details of primary antibodies and immunohistochemical techniques used to evaluate porcine cutaneous melanocytic lesions.
M = monoclonal; P = polyclonal. RCK 102 = cytokeratins 5 and 8; A1AT = alpha-1-antitrypsin; MHC = major histocompatibility complex.
ABC = avidin–biotin–peroxidase complex; SBA = streptavidin–biotin–alkaline phosphatase.
Tissue sections in which the specific primary antibodies were replaced by rabbit or mouse nonimmune isotypic serum were used as negative controls. Swine lymph nodes tissue sections were used as positive controls.
Cell counting
Tissue sections were examined by two pathologists. SBA immunolabeled tissue sections were evaluated for both pigmented cells (black color) and immunolabeled cells (red color). The density of both cell types was evaluated in 30 fields of 640× as follows: − = negative; ± = low (0–10 cells/field); + = moderate (10–25 cells/field); ++ = abundant (25–40 cells/field), +++ = very abundant (>40 cells/field).
Results
Cutaneous lesions were classified by their gross features according to published criteria.8 Type I lesions (12 pigs) showed black color, flat surface, and well-defined edges, type II lesions (31 pigs) showed black color, nodular surface, and well-defined edges, and type III lesions (11 pigs) had blue-brown color, flat or depressed surface, and poorly defined edges (Fig. 1).
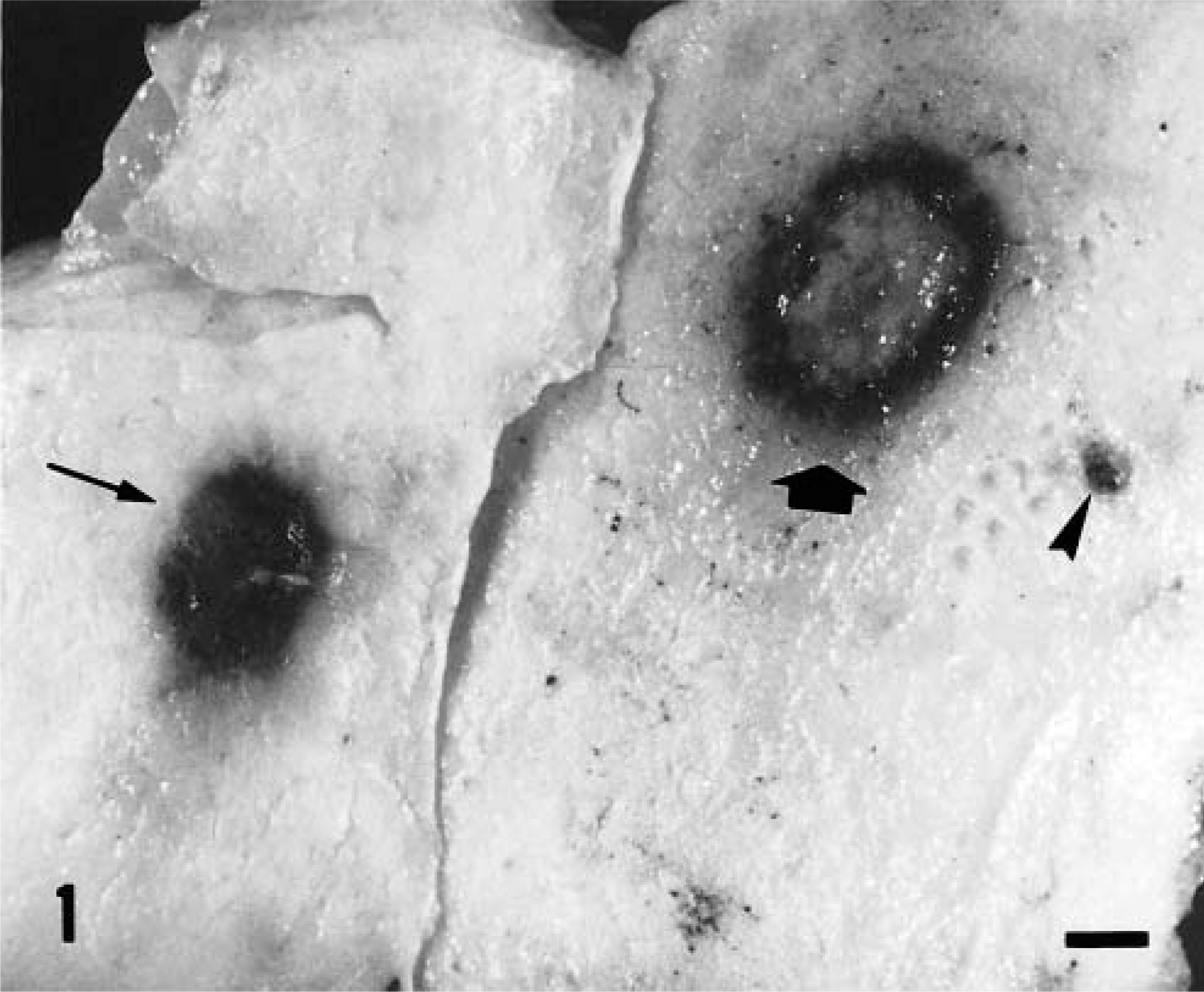
Shaved skin; pig. Note type I (arrowhead), type II (thin arrow), and type III (thick arrow) lesions. Note the poorly defined edges and depigmented center of the type III lesion.
The histologic study revealed that type I lesions were composed of large (15–35 μm in diameter) round or polygonal cells containing abundant, large granules of melanin pigment in their cytoplasm and were located in superficial dermis, sometimes with a perivascular or perifollicular pattern. On blanched sections, pigmented cells showed a round to oval nucleus without atypia or mitoses and large amounts of pale cytoplasm and were diffusely arranged with occasional lymphocytes among them. This cell type was identified as macrophages because of their morphologic features. Type III lesions were composed of small groups of pigmented cells located in deep dermis with low numbers of associated lymphocytes. On blanched tissue sections, these pigmented cells showed features of macrophages. Lesions of types I and III, which were easily differentiated grossly, showed similar histologic features, except that type III lesions were found in deep areas of the dermis. Both lesions were considered residual because tumor cells were not present.
Type II lesions were composed of well-defined, nonencapsulated dermal masses of round, polygonal, or spindle cells containing small melanin granules in the cytoplasm and of larger polygonal or round cells with large melanin granules. On blanched tissue sections, the former cells were identified as neoplastic melanocytes. In 29 lesions, nests of round or epithelioid cells (Figs. 2, 3) or bundles of flattened cells were observed. These cells had homogeneous nuclei without atypia or mitoses, and the lesions were identified as dermal melanocytomas. In the remaining two lesions of this type, neoplastic melanocytes showed considerable pleomorphism with occasional bizarre giant cells and were arranged in solid sheets. Atypia and mitotic figures were observed. These two lesions were identified as malignant melanomas. Tumor cells were vimentin+, cytokeratin−, and A1AT−, and the majority of them expressed S100 protein with both nuclear and cytoplasmic pattern (Figs. 2, 3). The second cell type showed features of macrophages; they were vimentin+, cytokeratin−, and S100−, and the majority of them expressed A1AT (Figs. 2, 3).
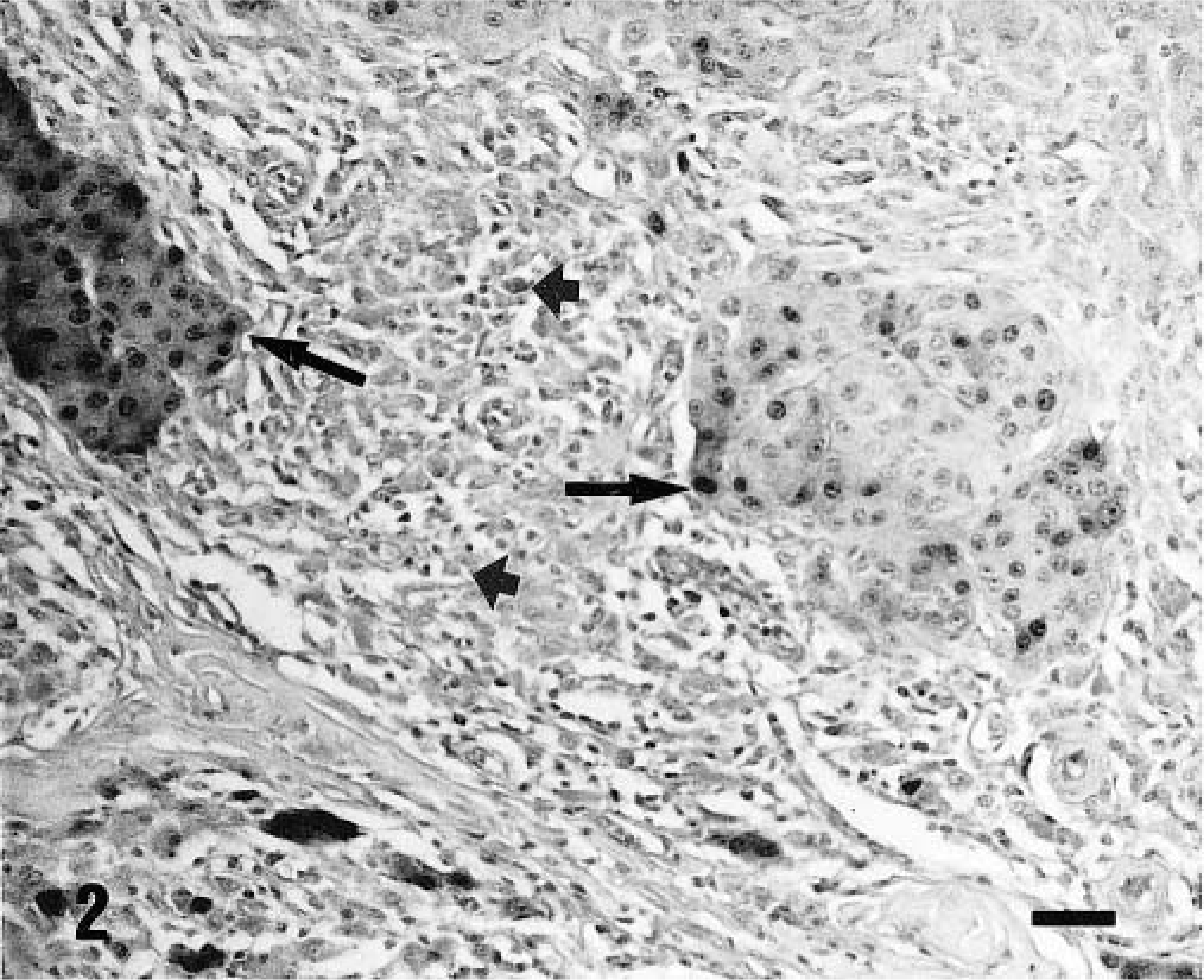
Skin; pig. Type II lesion. Nests of tumor cells show variable immunoreactivity for S100 (thin arrows), whereas the inflammatory infiltrate (thick arrows) is negative. ABC technique with bleaching. Mayer's hematoxylin counterstain. Bar = 42 μm.
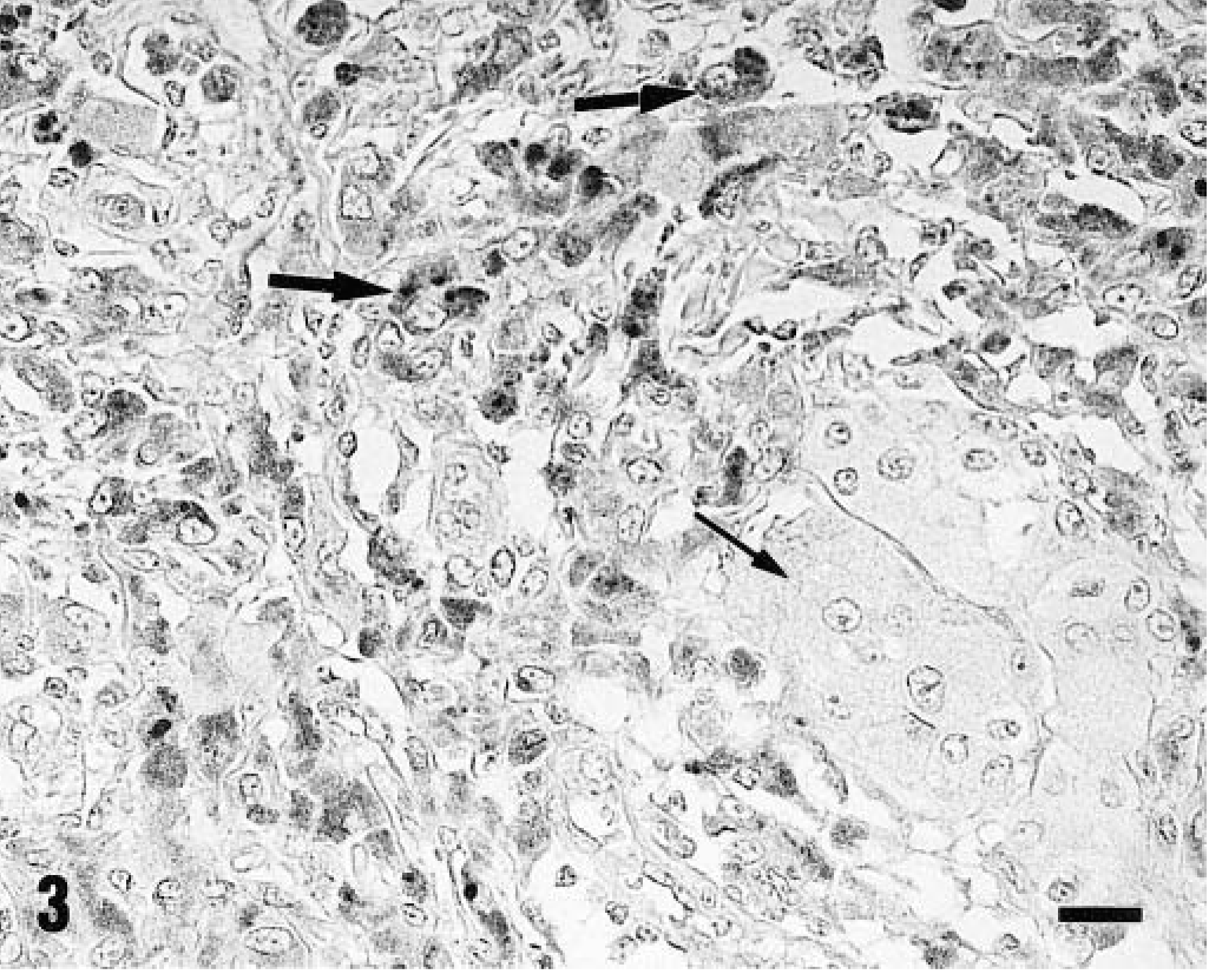
Skin; pig. Type II lesion. A nest of tumor cells is negative for A1AT (thin arrow), whereas some macrophages in the inflammatory infiltrate (thick arrows) are positive. ABC technique with bleaching. Mayer's hematoxylin counterstain. Bar = 30 μm.
The majority (80–90%) of intra- and peritumoral infiltrating lymphocytes were CD3+ T lymphocytes, and their distribution in the different types of lesions is summarized in Table 2. Type I and type III lesions showed low numbers of CD3+ lymphocytes, whereas type II lesions showed a variable and asymmetrically distributed infiltrate of CD3+ cells. Thus, the same tumor often had zones with heavy infiltration of CD3+ T lymphocytes and low numbers of tumor cells, whereas other areas had a high density of tumor cells and low numbers of CD3+ cells (Fig. 4).
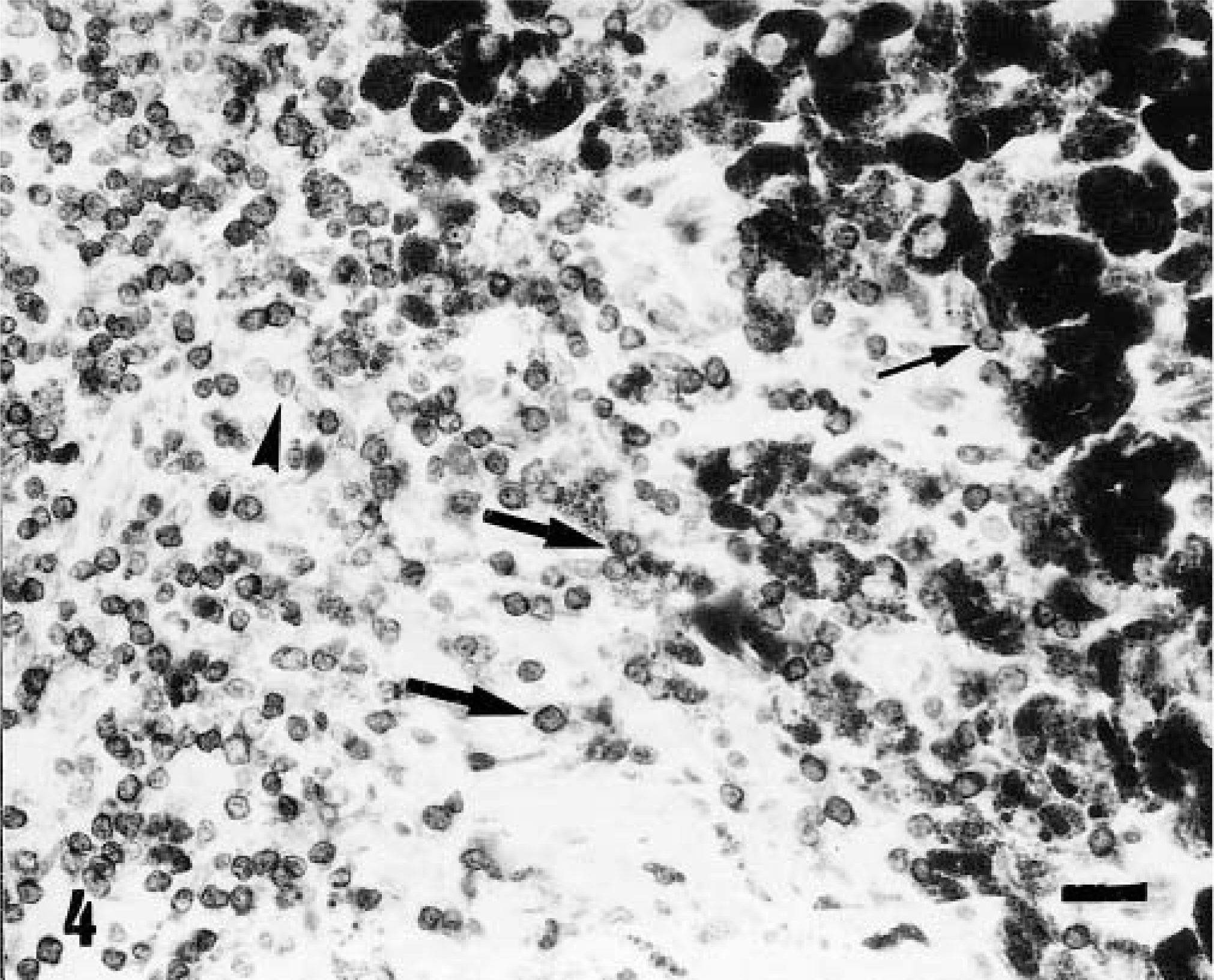
Skin; pig. Type II lesion. Peritumoral inflammatory infiltrate with numerous CD3+ T lymphocytes (thick arrows), some of them closely associated with pigmented cells (thin arrow). A few lymphocytes are CD3− (arrowhead). ABC technique with bleaching. Mayer's hematoxylin counterstain. Bar = 30 μm.
Density of CD3+ T lymphocytes in 54 porcine cutaneous melanocytic lesions.

Number of CD3+ cells per 640 X field: +/− = 0–10; + = 10–25; ++ = 25–40; +++ = >40.
The CD79a manoclonal antibody reacted with isolated lymphocytes and plasma cells located in the intra- and peritumoral inflammatory infiltrate. One type II lesion showed moderate infiltration of CD79a+ lymphocytes and plasma cells, the majority of which were IgG+ (Fig. 5). The remaining lesions presented occasional IgG+ plasma cells. The infiltration of IgM+ and IgA+ plasma cells was very low in all lesions examined.
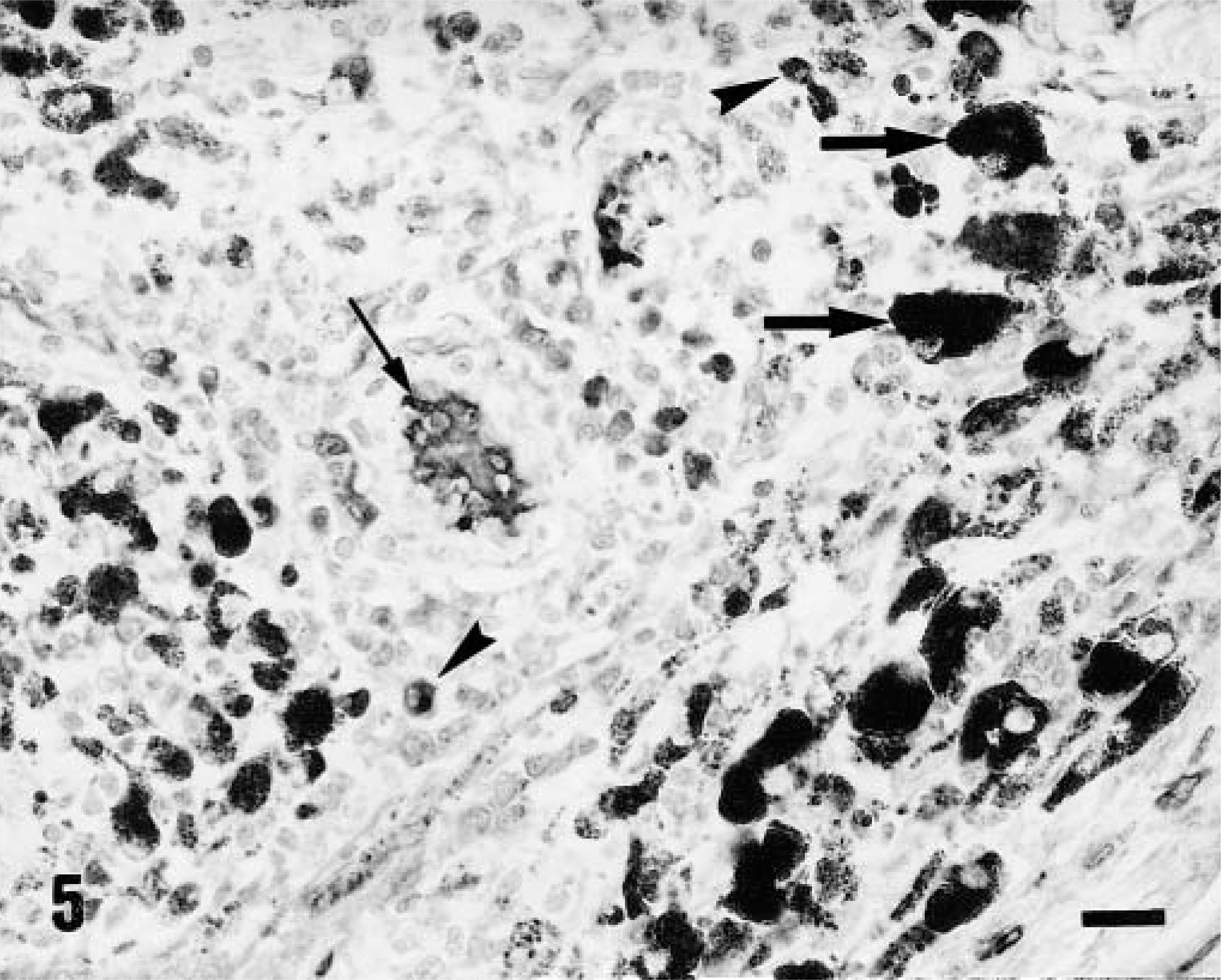
Skin; pig. Type II lesion. Intratumoral inflammatory infiltrate with some IgG+ plasma cells (arrowheads) among large pigmented cells (thick arrows). Diffuse immunostaining is visible within a blood vessel (thin arrow). ABC technique with bleaching, Mayer's hematoxylin counterstain. Bar = 30 μm.
The majority of intra- and peritumoral infiltrating lymphocytes strongly expressed MHC class II antigen (Fig. 6). The ABC technique carried out on blanched tissue sections allowed evaluation of MHC class II antigen expression by pigmented cells. Neoplastic cells did not express class II antigen (Fig. 6), whereas low to moderate numbers of pigmented macrophages expressed class II antigen.
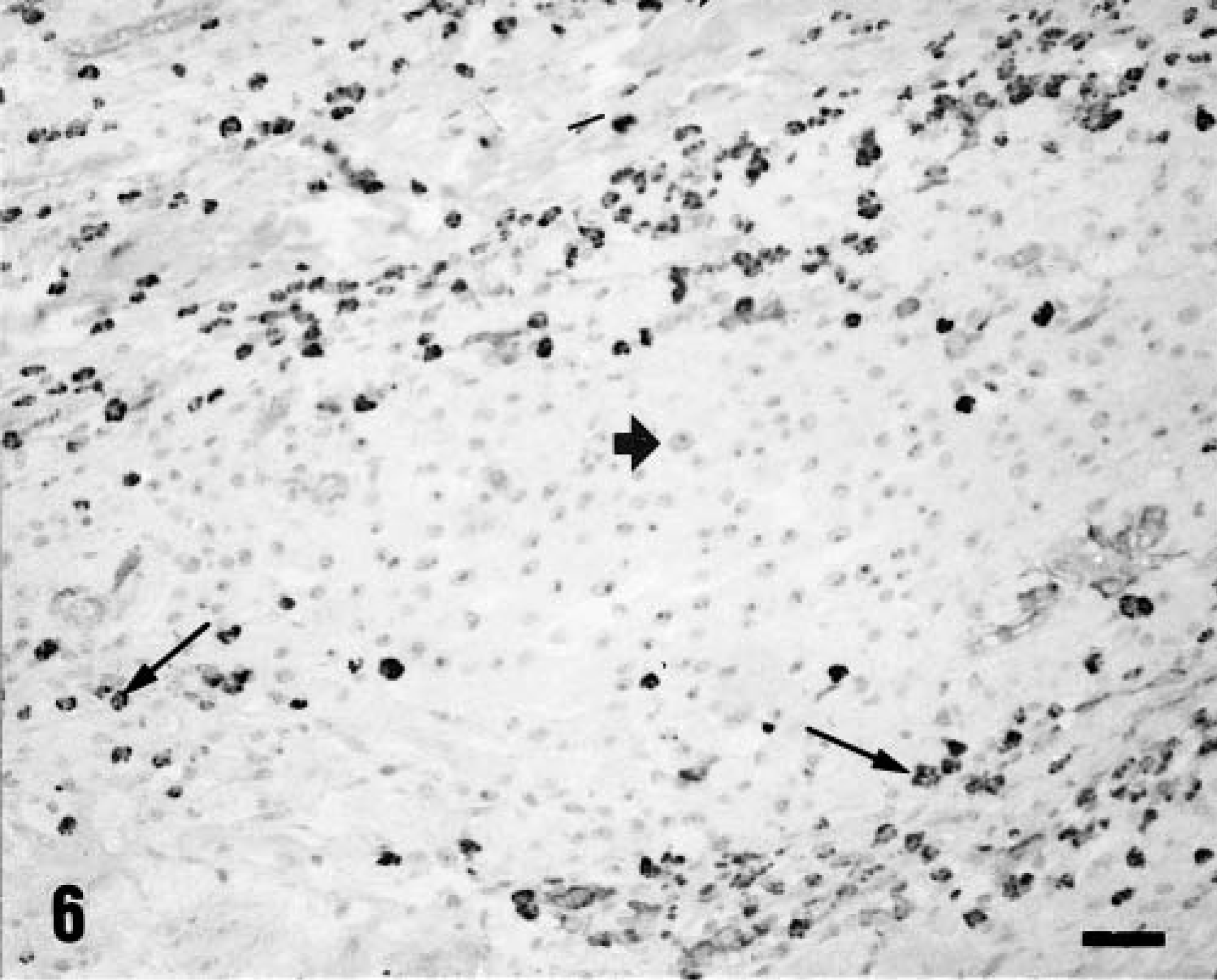
Skin; pig. Type II lesion. Lymphocytes and macrophages of the inflammatory infiltrate are immunoreactive for MHC class II antigen (thin arrows), whereas neoplastic cells, which show prominent nucleoli, are negative (thick arrow). ABC technique with bleaching, Mayer's hematoxylin counterstain. Bar = 45 μm.
Discussion
In the present study, normal and neoplastic porcine melanocytes were vimentin+, cytokeratin−, S100+, and A1AT−, an immunophenotype similar to that reported for human normal and neoplastic melanocytes.20,23 In a previous study, tumor cells of melanomas of Duroc pigs were vimentin+ and cytokeratin−.12 The majority of pigmented macrophages expressed vimentin and A1AT, and all of them were cytokeratin− and S100−, an immunophenotype reported for human macrophages.17 Therefore, A1AT and S100 protein were useful markers for an accurate identification of neoplastic melanocytes and pigmented macrophages on blanched tissue sections of swine cutaneous melanocytomas and melanomas. Pigment-laden macrophages in regressing melanocytic tumors of Sinclair swine have been described as terminally differentiated tumor cells.9 The results of the present study confirm that in regressing and residual lesions the majority of pigmented cells showed morphologic and immunophenotypic features of macrophages, but these finding do not support the assertion that they came from melanocytic tumor cells, unless neoplastic melanocytes can change immunophenotype to that of macrophages, which has not been demonstrated.
Malignant cutaneous melanoma of Sinclair miniature swine is considered a unique model to study the immunologic mechanisms involved in spontaneous tumor regression in vivo.2,10,16 Regression has been reported to occur in two phases, the first characterized by a rapid and massive infiltration of pigmented macrophages and a few lymphocytes and the second characterized by a heavy infiltration of lymphocytes with progressive loss of tumor cells.4,10 These two regressive phases could not be established in the pigs of the present study because the age of the lesions could not be determined. However, in 13 type II lesions low or moderate CD3+ lymphocyte infiltration was observed in zones with pigmented macrophages and a high density of tumor cells, coinciding with the features reported in the first regressing phase of melanomas in Sinclair swine. In the remaining 18 type II lesions, abundant or very abundant CD3+ lymphocyte infiltration was accompanied by loss of tumor cells, a feature reported in phase 2 of regressive melanomas in Sinclair swine.4,10 Type I and type III lesions were considered residual because tumor cells were not present, but they were composed of macrophages loaded with melanin and low numbers of lymphocytes, suggesting that once tumor cells are killed the lymphocyte infiltration decreases substantially.
The CD3 polyclonal antibody used in this study is considered a pan T cell marker that cross-reacts with porcine T lymphocytes.26 The majority of lymphocytes of the intra- and peritumoral infiltrates were CD3+. This result is similar to that reported in the inflammatory infiltrate associated with regressing melanoma of Sinclair swine, which showed a predominant immunophenotype of T lymphocytes labeled with a CD2 marker.18 In contrast, in familial melanoma of Duroc swine, low infiltration of CD3+ T lymphocytes was found.12 The presence of high numbers of CD3+ T lymphocytes in areas where tumor cells were sparse but low numbers of CD3+ T lymphocytes in areas where tumor cells were abundant coincides with the results reported in regressing melanoma of Sinclair swine10 and in regressing melanoma of human beings.19,27 These results suggest that T lymphocytes mediate, at least in part, spontaneous regression of this neoplasm. The study of apoptosis in tumor cells of these areas may elucidate the mechanisms by which tumor cells are killed.
The CD79a monoclonal antibody is considered a pan B-cell marker14 that cross-reacts with porcine B cells.26 The low number of CD79a+ B cells located in the peri- and intratumoral inflammatory infiltrates in the pigs of the present study was consistent with the low numbers of IgG+, IgM+ and IgA+ plasma cells in such infiltrates. These results suggest that the local antibody response does not play an important role in the spontaneous regression of swine melanomas and coincide with the low numbers of CD1+ B cells found in the inflammatory infiltrates associated with the regressing melanomas of Sinclair swine.18 However, antibodies to melanocytes and melanoma cells were found in 13 of 13 Sinclair swine with melanoma.5
MHC class II antigen was expressed by the majority of the lymphocytes infiltrating both tumor and residual lesions of the pigs examined in this study, suggesting cell activation.13,24 This result coincides with the high percentage of MHC class II+ cells in the inflammatory infiltrate of melanomas of human beings27 and Sinclair swine.18 Expression of MHC class II antigen by tumor cells of human melanomas mediated antigen presentation to CD4+ T lymphocytes;1,3 however, high expression of MHC class II antigen was found in tumor cells of progressing and metastasic malignant melanomas.3,6 In the present study, MHC class II antigen was not expressed by neoplastic cells of melanocytomas and melanomas of swine, suggesting that such expression is not necessary for an effective immune response and tumor regression.
Cutaneous melanomas and melanocytomas of crossbred Iberian × Duroc and purebred Iberian swine showed an immunophenotype similar to that of human melanomas. Spontaneous regression was associated with a heavy infiltration of CD3+ T lymphocytes, mainly located in zones with a low density of tumor cells, suggesting that the local cellular immune response plays a crucial role in the regression of this neoplasm. Future studies should be focused on the analysis of T-cell subsets and cytokines to further investigate the mechanisms of spontaneous regression of this tumor.
Footnotes
Acknowledgements
This work was supported by Junta de Andalucía (grant AGR137). We are grateful to R. De la Torre, who took most of the tissue samples used in this study.
