Abstract
Despite recent advances in surgical technology, resection is not an option for many brainstem tumors. Experimental models have played essential roles in examining new approaches to therapy. The objective of the present study was to generate models by determining coordinates for safe inoculation into the brainstem of mice and rats, and to establish whether the implantation of heterotopic cells would create reproducible survival curves. Morbidity and survival studies were used to map stereotactic coordinates allowing successful inoculation of tumor cells. Survival studies were used to investigate the time course of tumor growth. Tumor location was examined by light microscopy and magnetic resonance imaging. Mice survived injections of 2 μL of saline at interaural, lateral, and depth coordinates of −2.5, 1.0, and 3.5 mm and −1.5, 1.0, and 3.5 mm. Rats survived injections at interaural, lateral, and depth coordinates of −2.0, 2.0, and 7.0 mm and −3.0, 0, and 7.0 mm. Median survival of mice challenged with 5 × 105 EMT6 and 104 B16 tumor cells was 11 and 10 days, respectively. Median survival for rats challenged with 104 9L and F98 cells was 14 and 13 days, respectively. The present study demonstrates a feasible approach to preparing models of brainstem tumors. Limitations of these models are discussed.
Brainstem tumors, a heterogeneous set of neoplasms, account for 10–20% of lesions arising within the central nervous system (CNS), and 25% of tumors arising in the posterior fossa of children.10,13,18 Each year, 200–300 new cases are diagnosed in the USA. Low-grade lesions can respond to surgery, chemotherapy, and radiation. These lesions, which account for 15–25% of cases, have a somewhat optimistic prognosis, with 5-year survival rates of more than 50%.24,26 However, high-grade, diffuse tumors, which account for the majority of cases, have 2-year survival rates of 10–15%.9,33
Surgery for diffuse lesions has been limited regardless of the species because even small iatrogenic lesions may yield significant deficits in tissues with little functional redundancy.4,15,30 Radiation has not prolonged survival.12,28 Chemotherapy has not been successful, and has been accompanied by organ failures and lethal infections.14,17,21
Brainstem tumor models are needed to test new therapies. However, several challenges face development of such models. Few tumor cell lines have invasive characteristics of the predominant brainstem cancers. One might assume that stereotactic implantation techniques that have been widely used in the brain would directly translate to techniques for the brainstem. The absence of brainstem models suggests otherwise. We are unaware of reports describing coordinates for implanting tumor cells in the brainstem. Cell implantation methods and stereotactic ablation have been used in mice and rat brains for decades, but the neurologic sensitivity of the brainstem has limited work in this area. Schabert and coworkers injected human medulloblastoma cells into the cisterna magna of nude rats. They found leptomeningeal growth in the cisterna magna invading the medulla oblongata, and tumor growth in the fourth ventricle invading the pons.27 Their results demonstrate that human tumor cells can colonize the nude rat brainstem, and suggest that other heterotopic cells, which may colonize the cortex, would grow in the pons and medulla. Klopp and coworkers recently demonstrated an elegant surgical approach to the caudal brainstem in dogs.19 Their report provides a necessary step in tumor research, that is, the availability of canine or nonhuman primate models to bridge the gap between rodent studies and safety studies in humans.
We hypothesized that tumor cells could be injected directly into permissive areas of the brainstem parenchyma because stereotactic biopsies can be used to sample mass lesions in the brainstem.30 Intratumoral drug delivery vehicles could offer therapy to lesions in surgically inoperable tissues.34 Based on these observations, the Johns Hopkins Animal Care and Use Committee approved a study to document implantation coordinates in mice and rats. The present report describes the preparation of these models.
Materials and Methods
Experimental design
Sites for placement of cell implants were evaluated by postoperative monitoring of neurologic symptoms after the injection of saline at target coordinates. Coordinates are reported based on standard stereotaxic atlases for the rat,22 and the mouse brain.11 Responses to tumor cell implants were assessed from survival data and histology.
Animals
Female Fischer 344 rats (180–220) g, female Nu/Nu rats (150–180 g), female Balb/C mice (18–25 g), and female C57BL/6 mice (18–25 g) were obtained from Harlan (Indianapolis, IN) and housed according to Johns Hopkins Animal Care and Use policies and federal guidelines. The numbers of animals used in each experiment are given in Tables 1–3.
Stereotactic coordinates for brainstem injections in mice and rats.
Brainstem injection coordinates associated with significant morbidity in rodents.

Survival after brainstem inoculation with tumor cells.∗

Data were collected from one to six control groups of eight animals each in efficacy studies.
Cells
The EMT6 breast carcinoma line and B16 melanoma line were obtained from the DCT Tumor Repository (NCI-Frederick Cancer Research and Development Center, Frederick, MD). The F98 glioma cells were from R. Goodman (Ohio State University, Columbus, OH). The 9L gliosarcoma line was obtained from the Brain Tumor Research Center (University of California, San Francisco, CA). Cells were maintained in Dulbecco modified Eagle medium (DMEM) and 10% fetal calf serum supplemented with penicillin–streptomycin and tested by the Gen-Probe Rapid Detection System (Fisher Scientific, Pittsburgh, PA) to rule out mycoplasma contamination. Cells were harvested with 0.25% trypsin, counted, and resuspended in DMEM solution before intracranial implantation.
Chemicals
Tissue Marking Dye, a gift of Triangle Biomedical Sciences (Durham, NC), was used to mark injection sites. Cell culture supplies including calf serum, DMEM, and trypsin were from Life Technologies (Rockville, MD).
Surgery
Animals were anesthetized with a stock solution containing 25 mg/ml ketamine hydrochloride, 2.5 mg/ml xylazine, and 14.25 % ethyl alcohol in saline; 0.65 ml was used for rats and 0.20 ml of a dilution of the stock solution was used for mice. The mouse dilution consisted of two parts saline and one part stock anesthetic solution. Surgical surfaces were shaved, washed with 70% ethyl alcohol, and painted with Betadine. With the aid of a Zeiss operating microscope, a 2-mm burr hole was made at locations described in Table 1.
Animals were moved to a stereotactic head frame after creation of the burr hole. After placement in the frame, a 10-µl 26-gauge Hamilton Gastight 1701 syringe needle was inserted to a specified depth (as described in Table 1). Over a period of 4 minutes, 2 µl of the dye, saline, or tumor cell suspension was injected into the brainstem. The needle was removed. The site was irrigated with 5–10 ml of sterile saline solution. The animal was then removed from the frame and the wound was closed with 4.0 vicryl or surgical staples.
Animals were monitored immediately after surgery and daily for the development of signs and symptoms of discomfort. Animals demonstrating neurologic signs were euthanatized with sodium pentobarbital by intraperitoneal injection of 0.5 ml of a 50 mg/ml solution, a dose that is equivalent to 100 mg of drug/kg of animal weight.
Histology
Tissues for histology were prepared from formalin-fixed sections. Brains with their brainstems and cervical spinal cords were isolated as a whole and immersed in 4% buffered formalin. Sections were prepared from paraffin-embedded specimens. Samples were stained with hematoxylin and eosin (HE) solutions. Tumor volume was estimated by the method of Cavalieri.6
Magnetic resonance imaging (MRI)
Imaging was performed on an Omega 4.7 Telsa General Electric spectrophotometer (Milwaukee, WI), equipped with Acustar actively shielded gradients obtained from General Electric (Fremont, CA). Before imaging, animals were injected intraperitoneally with 1.0 ml of gadolinium contrast solution (Magnevist, Berlex Laboratories, Wayne, NJ).
Statistical analyses
Version 3 of JMP software was used for statistical analyses (SAS Institute Cary, NC). Values of P < 0.05 were considered to be statistically significant.
Results
Injection coordinates in mice and rats
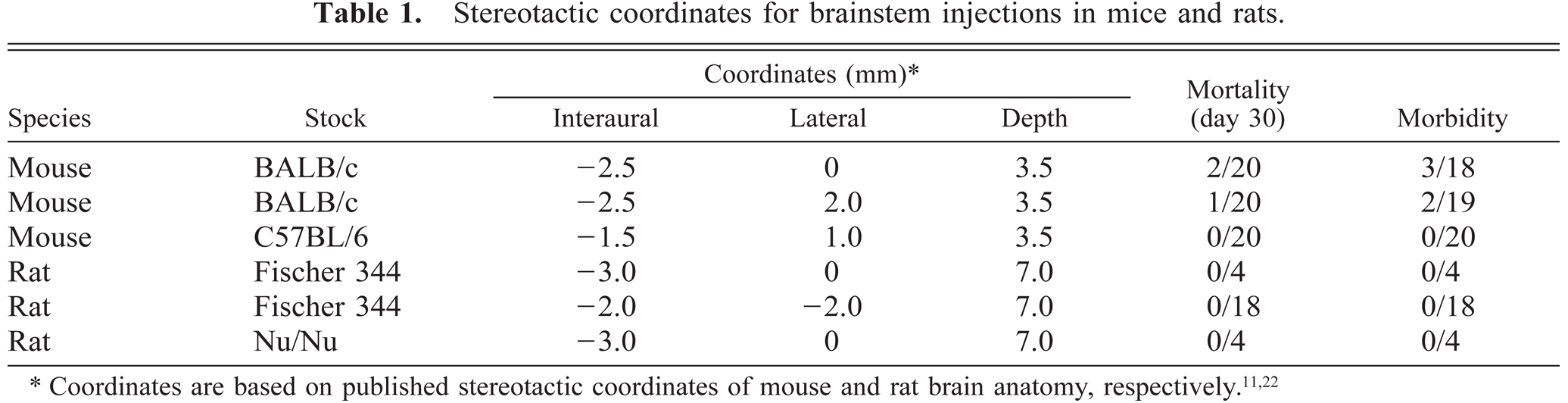
The coordinates for injection of 2 µl of saline through the cerebellum and fourth ventricle into the mouse and rat brainstem are given in Table 1. Figure 1A and B show injection sites of a tissue marking dye in a BALB/c mouse and Fischer 344 rat, respectively. Figure 1C and D show the tumor location in mice injected with EMT6 and B16 tumor cells. The BALB/c mouse described in Fig. 1C was euthanatized 10 days after inoculation with 104 EMT6 tumor cells. Median survival of mice inoculated with 5 × 105 EMT6 cells is 11 days (see below); thus, the tumor volume probably had not reached a lethal mass. The C57BL/6 mouse shown in Figure 1D was euthanatized 8 days after inoculation with 104 B16 tumor cells. The median survival for mice challenged with this dose of cells is 10 days.
Target marking (HE stain) with tissue dye and tumor cell inoculations.
Midline injections and injections 1 and 2 mm to either side of the midline were tolerated in mice and rats. The animals in Table 1 were observed for at least 30 days; no indication was found of neurologic symptoms or mortality in mice or rats injected with saline.
Examination of the data in Table 2 indicates the sensitivity of the brainstem to injections outside the coordinates listed in Table 1. Significant morbidity was apparent in the immediate postoperative period. The primary symptom in mice was failure to revive from the anesthesia. Shallow or irregular respiration was followed by death within 8 hours. Rats usually revived from anesthesia, but were lethargic and failed to drink or eat. Alternatively, rats exhibited a hemiplegia and moved in a circle with their nose down in their bedding. These animals were euthanatized.
Tumor cell challenges
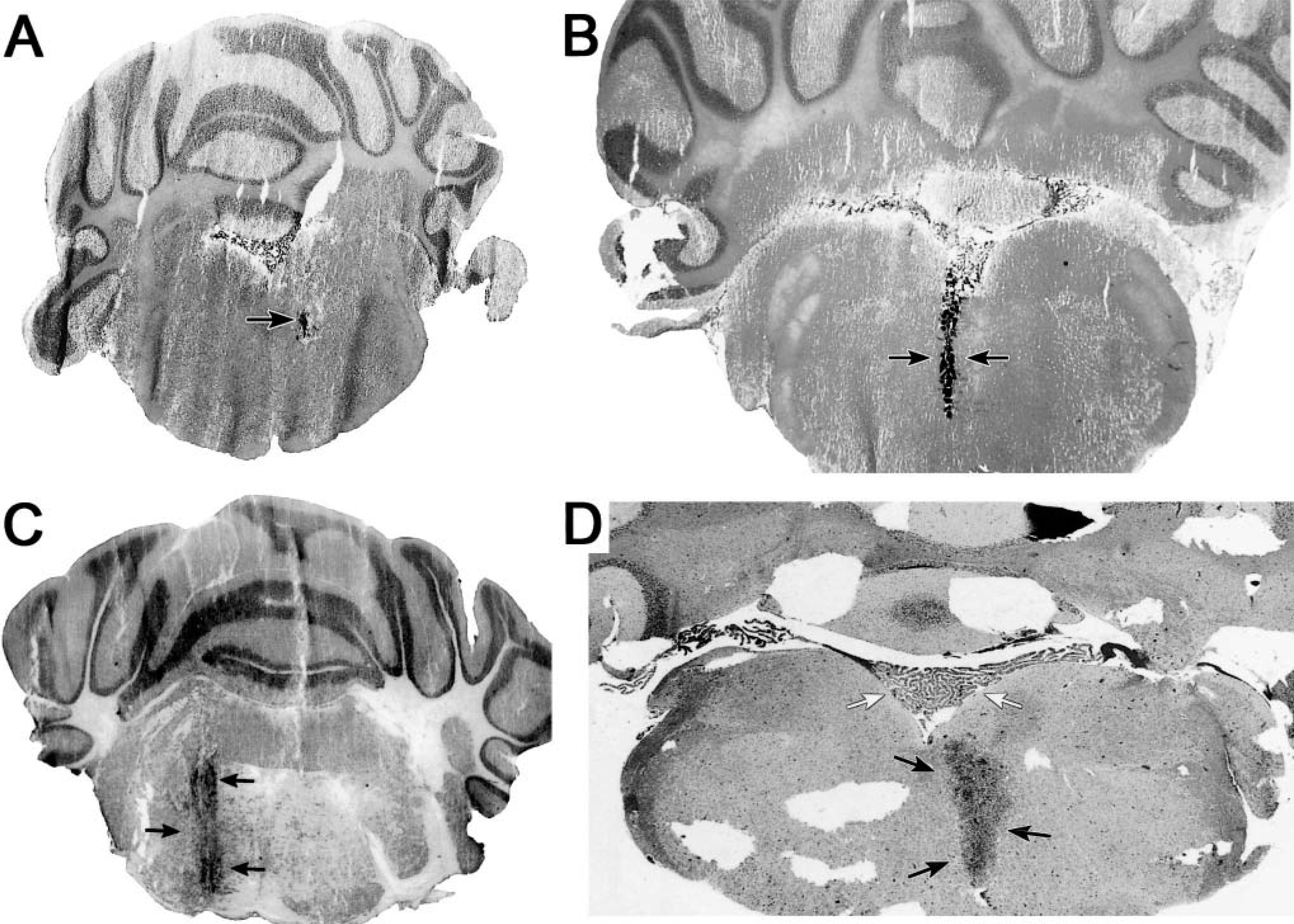
Examination of the survival data given in Table 3 indicates the capacity of heterotopic cells to colonize and proliferate in the brainstem. Inoculations with the four tumor cell lines proved uniformly lethal. The median survival time of BALB/c mice injected with 5 × 105 of the EMT6 metastatic breast cell line is 11 days. The median survival time of C57BL/6 mice inoculated with 104 B16 cells is 10 days. The median survival time of Fischer rats inoculated with 104 9L or F98 cells is 14 and 13 days, respectively. These times are notably shorter than those obtained when the same numbers of cells are injected into the parietal cortex. For example, in our laboratory, median survival after cortical injections of 5 × 105 EMT6 cells in mice and 104 9L cells in rats is about 16 and 20 days, respectively. Survival is dose dependent. In an experiment with three Fischer 344 rats per group, survival decreased in proportion to increasing numbers of F98 cells injected into the brainstem. Survival was 24 ± 1 days for the group injected with 103 cells, 14 ± 1 days for animals injected with 104 cells, and 11 ± 1 days for animals injected with 105 cells (Fig. 2).
Survival depends on the dose of a tumor cell inoculate. Three Fischer 344 rats in each group were inoculated with the quantity of F98 cells indicated. Data points indicate the median (± SD) days of survival. Differences for survival times for each group are significant, P < 0.05.
The development of neurologic symptoms in tumor-challenged animals is consistent with mass effects of the tumor on centers of respiration and consciousness. Animals sometime developed mild to moderate limb paralysis in the 24 hours before expiration. They generally became lethargic, lapsed into a coma, and died within 12–24 hours.
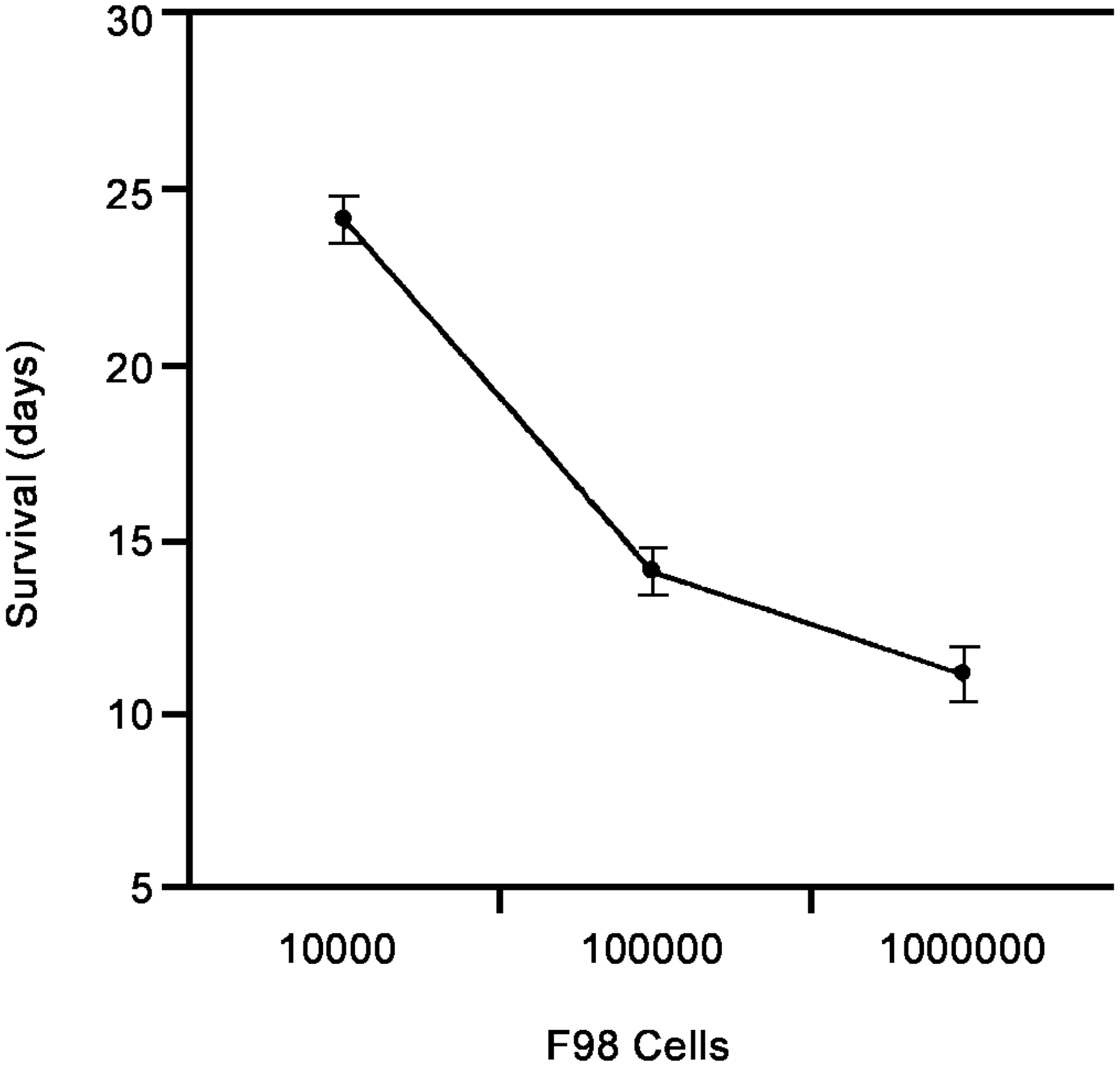
Although making histologic comparisons in immersion-fixed tissues is difficult, the appearance of the brainstem tumors is similar to the histology seen in cortical inoculations of the same cells. An exception is that cortical tumors can show evidence of central necrosis at an autopsy. Figure 3 shows a 9L glioma in the brainstem of a Fischer 344 rat. The left panel shows HE-stained tissue at day 10. The right panel, a gadolinium-enhanced MRI image at day 8 after inoculation, shows the bulk of the tumor at the site of the injection, and slight enhancement along the path of the needle. Little evidence was found of necrosis. The relatively small size of the tumor in the days before death supports the concept that the mass effects of the tumors are pronounced in tissues having limited functional redundancy; however, further research is needed to determine the cause of death.
Tumor location.
Tumor location and volume
Microscopic examination of the tumor cell–challenged brains demonstrated that the tumors grew primarily at the site of the inoculation and spread along the path of the injection. To verify this impression, we examined tumor location macroscopically and by histology in a group of eight rats inoculated with 104 F98 cells. Tumor volume at autopsy was estimated to be 1.9 ± 0.8 mm3 (range 0.8–3.2 mm3) by the method of Cavalieri.6 Gross examination revealed the bulk of the tumor at the site of the injection in the pons below the floor of the fourth ventricle. Histology revealed additional substantial tumor growth into the fourth ventricle in five animals. Tumors spread to cerebellar space in two rats, and to subarachnoid space in one rat. The extrabrainstem growth in all cases appeared to follow the path of the injection.
Discussion
Spontaneous tumors of the CNS are rare in most mouse and rat strains. Tumors can be induced by chemical carcinogens, although the efficiency of tumor induction varies greatly.16 Experimental tumors can be efficiently induced by avian sarcoma virus in Fischer rats. The mean induction time and frequency of tumors varied with the age of inoculation.16 Tumors occur commonly in dogs, but the incidence and natural history have not been established.19 The present work demonstrates that four rodent cell lines that efficiently colonize supratentorial regions of the brain efficiently grow in the brainstem. The inoculations produce reproducible survival curves, which is an essential prerequisite to efficacy studies. One may reasonably expect that other neuronal and glial tumor cell lines that colonized the rodent brain will perform in a similar manner.
Cell lines used in this study were selected from general experience with animal models and efficacy studies. For example, rat F98 and 9L glioma cell lines have been used to study tumor cell metabolism in animal brains,20,31 chemotherapy for brain tumors,3,23 neurotoxicity of heavy metals,29 radiation toxicity,32 radiation therapy,1,25 and gene therapy.2,5 The EMT6 breast tumor line has been used because breast cancer is the second most common cause of CNS metastases after lung cancer.8 By the same token, the B16 melanoma cell line is used because melanoma is the third most common source of intracranial metastases.7
The surgical approach was selected because of our laboratory's experience in using stereotactic frames in brain tumor models. After a burr hole is created in an appropriately prepared mouse or rat, the animal can be rapidly placed in a stereotactic frame for inoculation by a dorsal approach to the pons via the cerebellum and fourth ventricle. This is a convenience that facilitates the numbers of animals that must be tested to obtain statistical validation in toxicity and efficacy tests. Lateral and ventral approaches, which would be optimal for surgical resections, were considered but not explored after a significant effort to map coordinates that allowed safe inoculations through a dorsal approach. In work to be reported, we have used a similar dorsal approach to implant continuous infusion cannulas into the brainstem of cynomolgus monkeys.
The pattern of tumor growth appears to be similar to the models of tumor cells in the brain. Tumor cells expand at the site of injection, leading to mass effects on adjacent structures. Growth along the injection track is seen, but in many cases, tumor growth remains centered about the point of injection. Not surprisingly, survival is significantly decreased in brainstem-challenged animals compared to similar tumor cell challenges in the parietal cortex. One may assume that the difference is related to the low tolerance of the brainstem to tumor mass and edema. However, the limited sensitivity of immersion-fixed histology prevented further assessments with our preparations. Histology with perfused specimens and specific stains is needed.
The present work demonstrates that inoculations of 1–2 µl of saline can be accomplished without apparent morbidity in normal mice and rats and in immunodeficient (Nu/Nu) rats. In studies to be reported, we have looked for signs of tissue damage resulting from the inoculation process. Although it is difficult to find the needle track in tissue sections, microscopic hemorrhages and gliosis have been observed near injection wounds. These results imply that injections of local therapies, which have shown promise in the brain, may be applicable to brainstem lesions.
The EMT6 metastatic line and B16 melanoma cells offer tumor models for metastases to the brainstem. Although such metastases are regarded as quickly lethal, they are rare. None of the cells used in the present study faithfully mimic invasive characteristics of diffuse gliomas. Availability of the present rodent models may encourage efforts to develop highly invasive cell lines characteristic of the more prevalent tumors. Also note that the typical age range of diffuse pontine gliomas is 2–9 years, a period when the human brain is still undergoing developmental changes. To increase utility of the model, additional work is needed to characterize the capacity of tumor cell lines to colonize immature rodent brainstems. Other surgical approaches could be considered. The dorsal approach works well for inoculations, but ventral approaches would be superior for surgery in rodents and dogs.19
Conclusions
Tumor cells can be implanted in rat and mouse brainstems to create laboratory models to investigate new therapies for the treatment of brainstem lesions.
Footnotes
Acknowledgements
This work was generously supported by funds from The Children's Cancer Foundation.
