Abstract
Leproid granulomas from seven dogs in the United States were evaluated. Gross characteristics included nodular and ulcerated dermal and subcutaneous lesions primarily on the caudal aspects of the pinnae and to a lesser extent on the muzzle, face, and forelimbs. In all except one dog, there was complete regression of the lesions within 6 months, either with no therapy or after surgical resection. Cytology or histopathology revealed pyogranulomatous inflammation with few to many acid-fast mycobacterial bacilli within macrophages. The organisms could not be cultivated in vitro. DNA sequencing of part of the 16S ribosomal RNA gene region revealed 99–100% homology among fragments from five of these dogs and fragments from dogs in the South Pacific. This syndrome occurs in dogs in North America and the prognosis is excellent, in contrast to the prognosis for rapid-growing or tuberculous mycobacteriosis.
Nodular and ulcerative dermatitis in dogs can be associated with a number of conditions, including immune-mediated, infectious, parasitic, and other disease processes. We have observed a clinically distinct condition in North American dogs in which the skin of pinnae, lateral aspects of the muzzle, and occasionally forelegs develops discrete intradermal, sometimes ulcerating granulomas. Acid-fast staining of these lesions typically reveals acid-fast bacilli within macrophages, yet bacterial culture using appropriate protocols for a variety of mycobacteria is invariably unproductive. These lesions have some similarities with feline leprosy and atypical mycobacteriosis of dogs and cats yet are distinct based on culture results and clinical characteristics.
There have been published reports under the name leproid granuloma syndrome of a similar syndrome in dogs from Australia, New Zealand, Brazil, and South Africa.14,16,24 In those reports, single or multiple hard, painless nodules from 2 mm to 5 cm in diameter usually occurred on dorsal pinnae or the head. The nodules were granulomas in the subcutis or skin, with superficial alopecia or ulceration.2,16 The objective of the present study was to describe a series of leproid granulomas in dogs from the United States and to contrast findings from this series with those of Australian and New Zealand dogs.
Case Reports
Dog No. 1
A 5-year-old female Bulldog from the central California coast presented to a referring veterinarian with a raised, circumscribed mass on the tip of one pinna. The owner reported that there might have been a cat scratch to this region of the dog's ear. A needle aspirate was obtained for cytologic evaluation. The single lesion was surgically resected and did not recur following surgery.
Dog No. 2
An 8-year-old male mixed-breed German Shepherd Dog from the central California coast presented with raised, firm masses with serpiginous borders affecting the margins of the right and left pinnae. Several biopsy samples were acquired. The dog was treated with cephalexin (500 mg orally twice daily for 10 days) and then enrofloxacin (500 mg orally twice daily for 14 days). Lesions completely regressed within 3 months following biopsy, leaving focal areas of depigmented skin.
Dog No. 3
A 7-year-old male Boxer from Coral Springs, Florida, was evaluated because of extensive draining nodular lesions on the dorsal surfaces of the pinnae and sides of the face. Biopsies were submitted for histopathologic evaluation and bacterial and fungal culture. The dog was not treated. Nevertheless, all lesions resolved within 4 months.
Dog No. 4
An adult female Pit Bull Terrier from the central valley of California was evaluated because of extensive draining nodular lesions on the dorsal surfaces of the pinnae, sides of the face, and dorsal limbs (Figs. 1, 3, 5). A sample was submitted for histopathologic evaluation, but the dog was lost to follow-up.
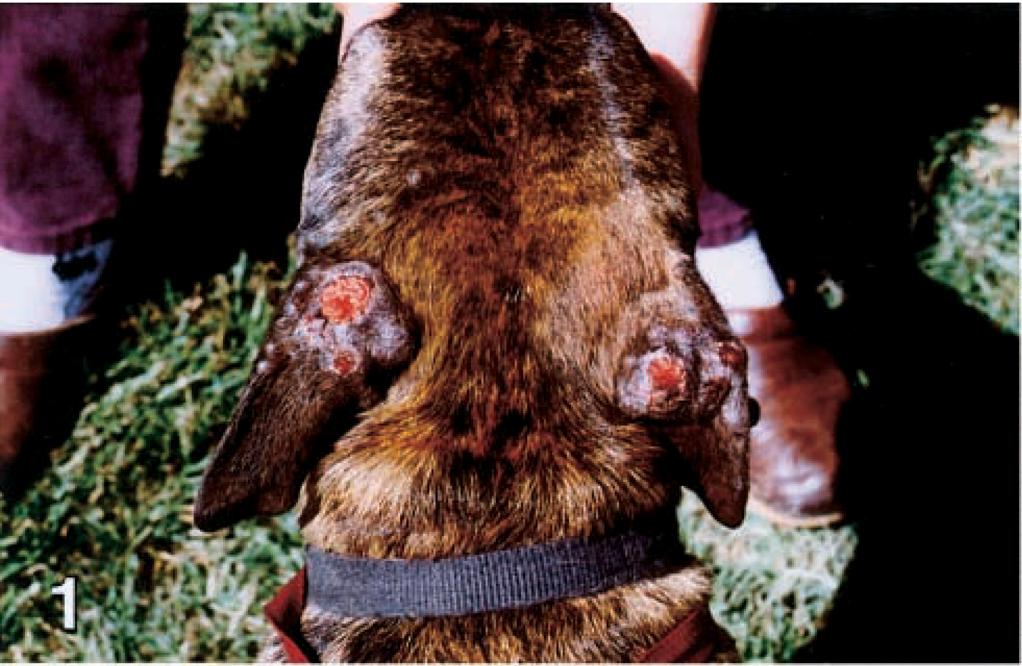
Dorsal head and pinnae; dog No. 4. The skin has extensive draining nodular lesions on the dorsal surfaces of the pinnae.
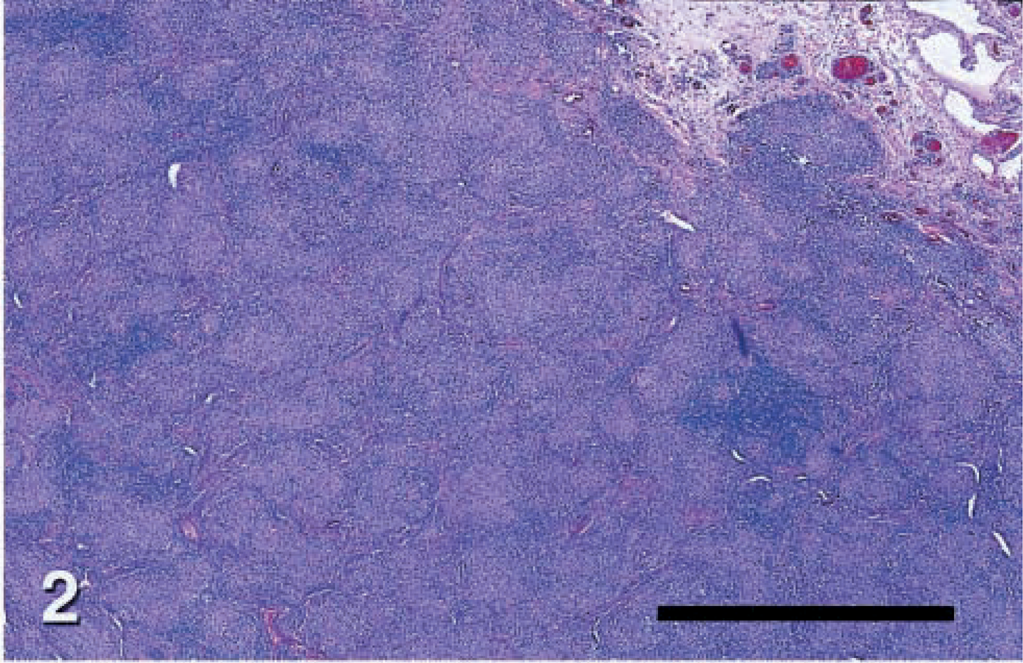
Skin; dog No. 5. Low magnification view of leproid granuloma lesion containing diffuse infiltrations of large macrophages, neutrophils, and fewer lymphocytes or plasma cells filling the dermis and panniculus. HE. Bar = 330 μm.
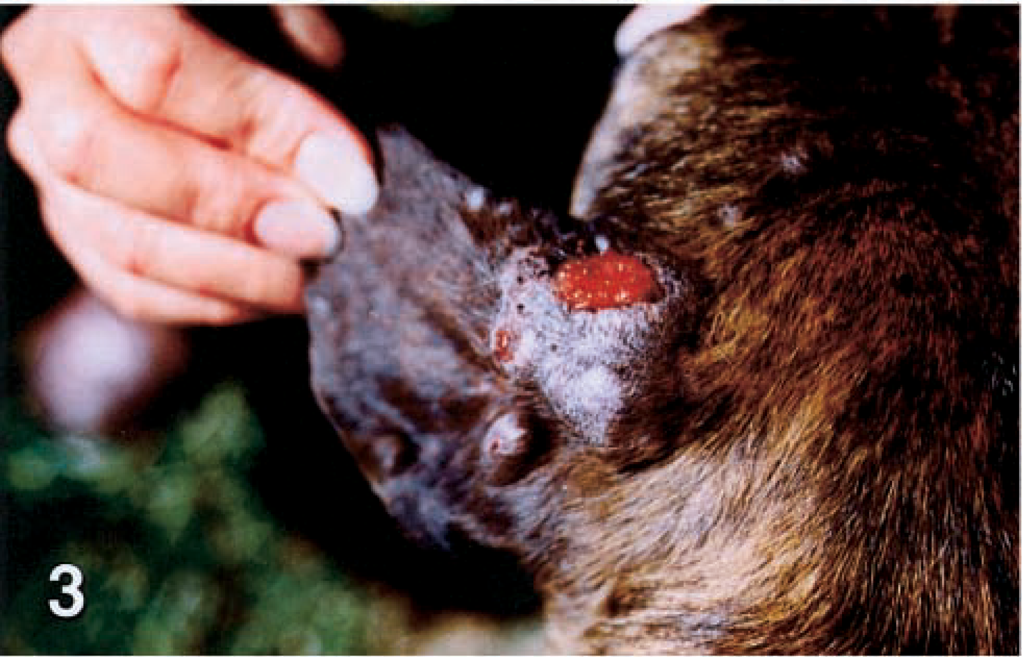
Left pinna; dog No. 4. Closer view reveals multiple nodules with variable ulceration.
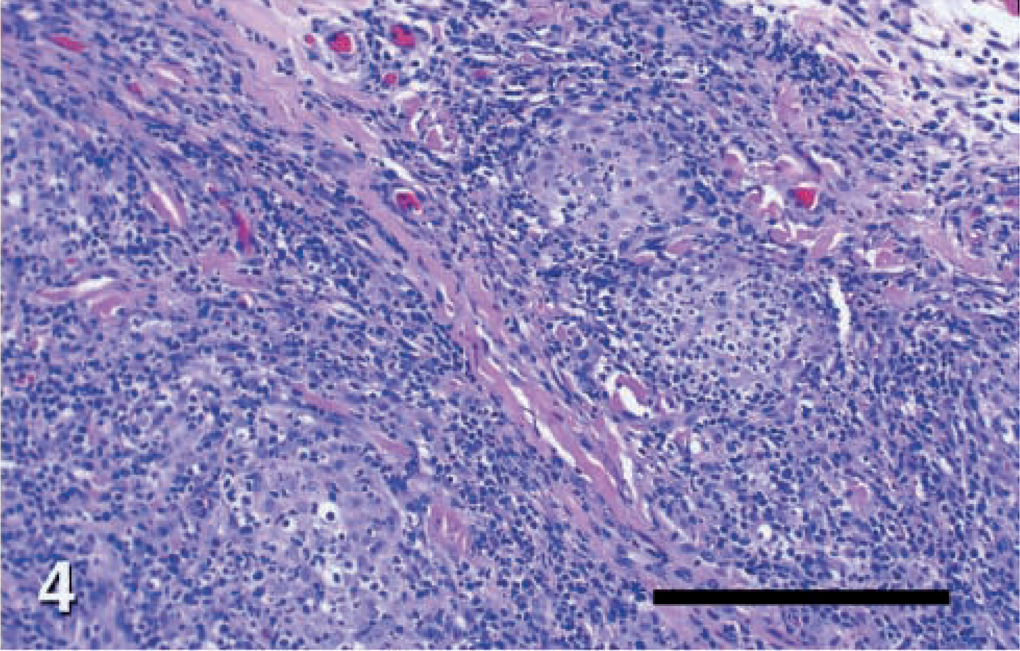
Skin; dog No. 7. Leproid granuloma lesion, showing loosely organized pyogranulomas with confluent and indistinct borders, with infiltrating macrophages, neutrophils, lymphocytes, and plasma cells interspersed with clusters of plasma cells and lymphocytes. HE. Bar = 82.5 μm.
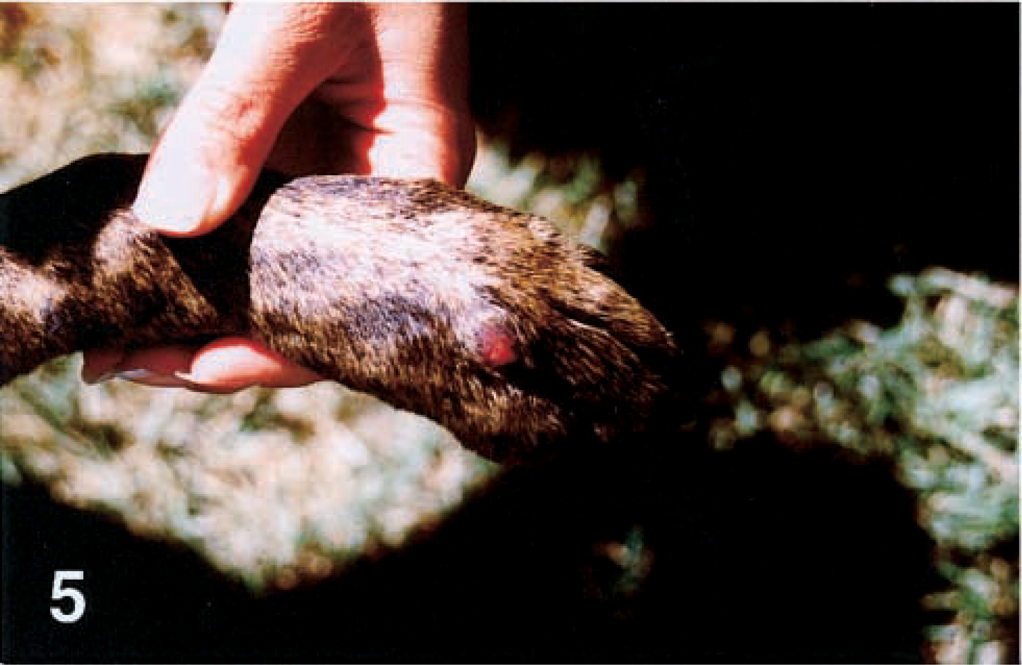
Dorsal left forepaw; dog No. 4. A single nodular ulcerated lesion in present at the base of the fourth digit.
Dog No. 5
A 12-year-old male Border Collie from Hawaii presented with raised, ulcerated intradermal masses on the dorsal right pinna and right cranial foreleg. The masses were surgically removed and submitted for histopathologic evaluation. The dog was treated with cephalexin (500 mg orally twice daily for 10 days). The masses did not recur.
Dog No. 6
A 10-year-old mixed-breed Chow-Chow/German Shepherd Dog from San Diego presented with a 3-cm area of raised inflammation and alopecia on the dorsum of the nose. Biopsies were submitted. The lesions were managed with topical application of amoxicillin powder, but there was no improvement of the clinical signs. After 3 months, treatment was switched to clarithromycin (250 mg orally twice daily for 30 days). At the time of publication, the lesions had not regressed.
Dog No. 7
A 7-year-old male German Shepherd Dog from the central valley of California presented with a complaint of multiple firm nodules on the dorsum of the right pinna. A needle aspirate was obtained for cytology. The lesions were surgically removed and did not recur.
Materials and Methods
Microbiology
Histopathologic and cytologic samples were stained using Ziehl-Neelsen or Fites's modification of acid-fast techniques. Culture of the bacteria was attempted using three different protocols. To evaluate for Mycobacterium avium paratuberculosis, samples from dog Nos. 1–6 were grossly decontaminated with a 30-minute soak in 0.1% hexadecyl pyridinium chloride, finely ground, and resuspended in 0.15% zephiran. The supernatant was removed after 20 minutes, and the sediment was inoculated onto Herrold's egg yolk agar with and without mycobactin. Samples were evaluated weekly and held at 37 C for 12 weeks before being considered negative. For assessment of M. avium, M. bovis, M. tuberculosis, and other slow-growing pathogens of humans, samples from dog Nos. 1 and 7 were sent to the National Jewish Medicine and Research Center, Mycobacteriology Laboratory, Denver, Colorado, for culture using their standard techniques. Minced samples from all dogs were cultured on blood agar at 37 C with 5% CO2 for 7 days for Runyoun group IV mycobacteria. Culture of M. leprae and M. lepraemurium was not attempted.
Polymerase chain reaction and DNA sequencing
Fresh or formalin-fixed, paraffin-embedded tissues from the lesions were evaluated for the presence of mycobacterial DNA by polymerase chain reaction (PCR) and DNA sequencing for five of the seven dogs (excluding cytology samples from dog Nos. 1 and 7). Fresh tissue was finely minced, suspended in buffer ATL from a kit (DNeasy tissue kit, Qiagen, Valencia, CA), ground with a mortar and pestle, boiled at 95 C for 20 minutes, and then mixed with 100 µl of 0.1 mm zirconia beads (zirconia/silica 0.1-mm beads, Biospec Products, Bartlesville, OK) and agitated forcefully for 5 minutes (Mini-Beadbeater, Biospec). If samples were submitted after embedment in paraffin, xylene removal of the paraffin and an ethanol wash were added. DNA extraction was performed according to the DNeasy kit instructions. Amplification of the 16S ribosomal RNA (rRNA) gene region was performed previously as reported10 using primers 246 (5′-AGAGTTTGATCCTGGCTCAG) and 247R (5′-TTTCACGAACAACGCGACAA) for the first round and M1 (5′-AGTGGCGAACGGGTGAGTAAC) and R7 (5′-TTACGCCCAGTAATTCCGGACAA) for the second round in a thermal cycler (MJ Research, Watertown, MA). Culture, DNA extraction, and PCR were all performed in separate rooms with separate equipment, and all PCR were run with a water negative control. The products were separated by electrophoresis through 1.5% agarose minigels and visualized with ethidium bromide. Amplicons were sequenced directly using primers M1 and R7 after purification with a kit (Microcon, Millipore, Bedford, MA). Big dye terminator cycle DNA sequencing (ABI Prism, Foster City, CA) was performed. Electropherograms were analyzed with Chromas (Technelysium, Helensvale, Queensland, Australia), and sequences were evaluated using the BLAST algorithm1 from the National Center for Biotechnology Information. The genetic relationships among 16S rRNA sequences from the USA and Australia (GenBank accession No. AF144747) were compared using the computer program GCG (Genetics Computer Group, Madison, WI). A representative fragment from one dog was submitted to GenBank (accession No. AF325531).
Results
Pathologic changes
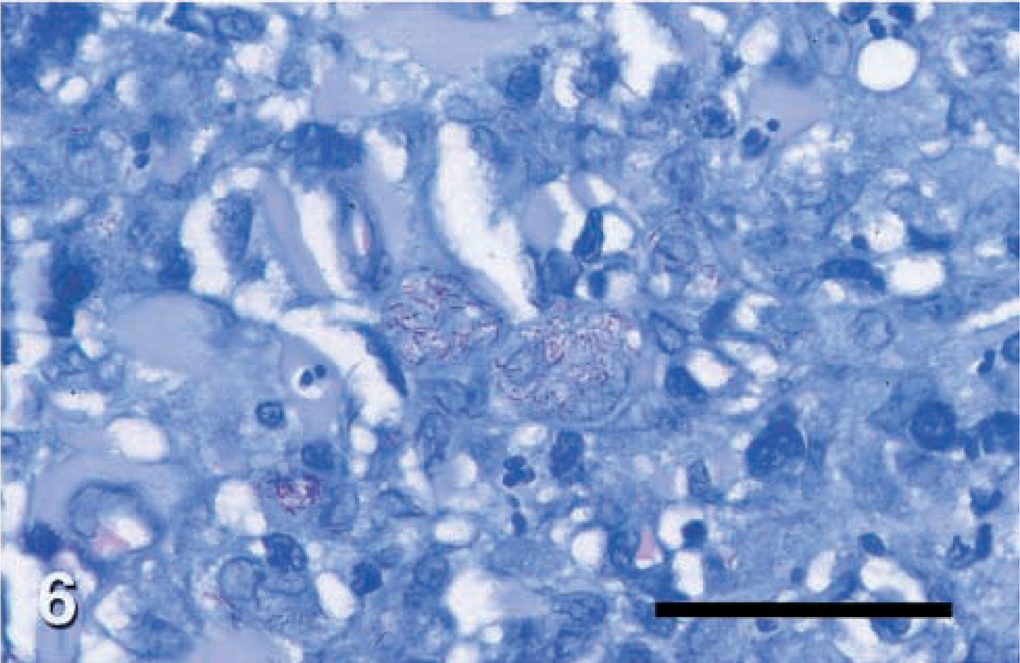
Histopathologic changes from dog Nos. 2–6 were evaluated from formalin-fixed sections stained with hematoxylin and eosin (HE) and acid-fast stain. Histopathologic findings in all cases were similar (Figs. 2, 4). There was variable ulceration and exudation of the surface of the nodules. Diffuse infiltrations of large macrophages, neutrophils, and fewer lymphocytes or plasma cells filled the dermis and panniculus. Inflammation was often loosely organized as granulomas or pyogranulomas with confluent and indistinct borders. These lesions were interspersed with clusters of plasma cells and lymphocytes. Filamentous acid fast bacteria were seen in small to moderate numbers within the cytoplasm of many macrophages throughout the infiltrate (Fig. 6). The distribution of organisms was often uneven. Most macrophages in some high power fields lacked visible organisms, whereas many macrophages in other fields contained organisms.
Skin; dog No. 2. Oil immersion view of acid-fast mycobacterial bacilli responsible for canine leproid granuloma syndrome within macrophages. Fites's Faraco stain. Bar = 33 μm.
Samples from dog Nos. 1 and 7 were obtained by aspiration cytology. The aspirate from dog No. 1 was highly cellular, with a mixed inflammatory population including plasma cells, lymphocytes, macrophages, and multinucleated giant cells. Abundant intracellular, negatively stained bacilli were noted within macrophages and giant cells. An acid-fast stain of the smear was positive. The sample from dog No. 7 was of moderate cellularity, with fibroblasts, occasional macrophages, small lymphocytes, and nondegenerate neutrophils. Macrophages contained large numbers of negatively stained bacilli, which were positive on an acid-fast preparation.
Microbiology
All samples contained acid-fast bacilli within macrophages. All samples were culture negative for rapidly growing mycobacteria. Dog Nos. 1–6 were negative for M. avium paratuberculosis, and dog Nos. 1 and 7 were negative for major slowly growing mycobacteria of humans.
PCR and DNA sequencing
A 519-base pair fragment of the 16S rRNA gene was amplified from DNA from the lesions of dog Nos. 2–6. DNA sequencing of the fragment was performed, and for each dog, the closest match in the GenBank database was a fragment named “Mycobacterium sp. Murphy 16S ribosomal RNA,” which was also identified in a dog in Australia with canine leproid granuloma.10 The homology between the North American fragments and the Australian fragment designated “Murphy” was 99–100% for all North American sequences.
Discussion
Canine leproid granuloma syndrome is a clinical manifestation of cutaneous infection with a novel, as yet unnamed, uncultivated species of mycobacterium. The disease has been reported from Africa24 and is a common problem in Australia and New Zealand.16 The signalment of affected dogs in previous reports reflects primarily Boxer and Boxer-cross dogs and other short-coated breeds such as Staffordshire Terriers and Doberman Pinschers.16 German Shepherd Dogs may be overrepresented in the present series. This breed also is more susceptible to infection with several other intracellular or fungal organisms, such as Ehrlichia canis and Aspergillus spp.3,17
In canine leproid granuloma, firm or ulcerated, nodular pyogranulomatous lesions are present in dermal and subcutaneous tissue of affected dogs, predominantly on the ears and to a lesser degree on the head or thoracic limbs. Lesions usually regress spontaneously within weeks or months, with or without surgical removal, although chronic disease occurrence has been reported.16 As previously reported for other dogs, most dogs in the present study experienced lesion regression within a few weeks to months, with or without surgery and without anti-mycobacterial treatment. There was no evidence for involvement of the facial nerves or draining lymph nodes, as occurs with human and occasionally feline leprosy.7
Histopathologically and cytologically, leproid granulomas are composed of diffuse granulomatous or pyogranulomatous infiltrates; macrophages are unevenly invested with acid-fast bacilli in small to moderate numbers. In contrast to infection with rapidly growing (Runyoun group IV) mycobacteria, discrete pyogranulomas with clear central spaces are absent in leproid granulomas.6 Although there are often large numbers of Ziehl-Neelsen acid-fast bacilli within macrophages, the organisms have not been cultivated, despite the use of protocols for Runyoun group IV mycobacteria, M. avium paratuberculosis, M. avium complex (MAC), M. genavense, M. bovis, M. tuberculosis, and others.
Recently, PCR assay and DNA sequencing of canine leproid granulomas from 12 Australian dogs were reported.10 In that study, there was no genetic variability in the short product obtained by PCR from the 12 dogs, and the closest phylogenetic relatives to this novel mycobacterium were M. tilburgii, M. simiae, and M. genavense. The fragments reported in the present study had 99–100% homology with those from Australia and New Zealand. The 16S rRNA target used in that study has several advantages. There are conserved primer sequences at the 5′ and 3′ ends of the gene, facilitating a universal PCR approach. The 16S rRNA target was the first used to identify an uncultivated bacterium (Bartonella henselae) via universal PCR22 and has since been used extensively enough that a relatively large database is available for comparison of any new bacterial strain. The variable regions 2 and 3 of the 16S rRNA gene have been reported for numerous mycobacteria and are useful for discrimination at the species level.12,23 The characterization of the primary agent of feline leprosy as M. lepraemurium was accomplished by DNA sequencing of the 16S V2 and V3.9 In contrast, the 65-kda heat shock protein gene is also an important target for discriminating mycobacteria.4,27 This gene contains greater microheterogeneity than does the 16S gene and is potentially useful in molecular epidemiologic studies.21
Other small animal dermal lesions with acid-fast bacilli are caused by rapidly growing mycobacteria, tuberculous mycobacteria, and other slowly growing nontuberculous species. Atypical mycobacteriosis with Runyoun group IV mycobacteria (M. fortuitum, M. smegmatis, M. chelonae, M. thermoresistable, and others) typically presents as nodular dermatitis and panniculitis with draining tracts, especially in cats, and is characterized by pyogranulomatous inflammation with small numbers of visible organisms, often in clear spaces.5,13,26,29,30 There is rapid growth on culture. M. fortuitum from at least two dogs with canine leproid lesions has been cultured on Lowestein–Jensen medium, but this positive culture may have been due to environmental contamination, given that these organisms are common saprophytes.24 Case reports have documented cutaneous MAC organisms and other slowly growing nontuberculous mycobacteria in cats and dogs.15,28 Tuberculosis skin lesions in dogs and cats, although rare in the present era, are characterized by a variable number of bacteria that propagate readily although slowly in culture.11,18,20,25 The agent of feline leprosy, M. lepraemurium, is abundantly present in classical leprosy lesions but may be rare in the tubercular form of leprosy.8 This organism is difficult to culture, requiring special techniques.19 In several cases of feline leprosy, an M. malmoense-like organism was detected by DNA sequencing,9 possibly representing a novel species.
The initially described geographic distribution of canine leproid granuloma superficially resembles that of feline leprosy, i.e., predominant in cool, moist areas of the world, commonly near Pacific port cities. However, in the present study, several affected dogs were from the central valley of California (which is usually dry and very warm in summer). This novel organism probably is an environmental saprophyte and is introduced by percutaneous inoculation via wounds or arthropod vectors.16 There may have been inoculation via cat scratch in one of the dogs in the present study. The anatomic distribution of lesions, i.e., restricted primarily to ears, is not explained by percutaneous inoculation alone, unless growth of the mycobacterium is optimal on cooler areas of the skin. Given that the clinical syndrome in the present dogs was so similar to that previously reported and that the DNA sequences were identical, there may have been an introduction of this organism into the Western Hemisphere, resulting in emergence of this condition in the USA. Alternatively, there may be increased awareness of a condition that has been present for many years.
This report documents several cases of canine leproid in the USA with clinical, culture, and molecular similarities to the syndrome in Australia and New Zealand. Clinicians and pathologists should be aware of the features of this syndrome and should recognize that, at least in cases where the underlying organism is this novel uncultivated mycobacterium, the prognosis is excellent without treatment.
Footnotes
Acknowledgements
We acknowledge the contributions and support of S. Klintworth, M. Armington, S. Sawyer, C. Zatkin, M. Espinoza, R. Malik, S. Jang, L. Tell, C. Krach, C. Vieira, and J. Milan. This study was supported by the University of California School of Veterinary Medicine, Center for Companion Animal Health.
