Abstract
Mycobacterium genavense is a common cause of mycobacteriosis in passerine birds. In a research colony of zebra finches (Taeniopygia guttata), 8 birds were diagnosed with mycobacteriosis. The finches had granulomatous inflammation of the heart and heart-base, most with medial expansion of the great vessels containing foamy macrophages and acid-fast bacilli. Non-cardiac inflammatory lesions associated with acid-fast bacteria were found in 2 birds, and extracardiac bacteria were often in lower quantities. Pan-mycobacterial in situ hybridization detected periaortic bacteria in one bird with similar cardiac lesions that was negative for bacteria via acid-fast staining. Mycobacterium genus PCR and sequencing of pooled fecal samples confirmed the presence of M. genavense within the colony. Heart and great vessel lesions have not been previously recognized as a site of localized infection in passerines. To facilitate diagnosis of mycobacteriosis in zebra finches, routine necropsies should include microscopic examination of the heart base great vessels.
Mycobacteriosis affects a variety of avian species but is not often identified in research bird colonies. Rarely reported in zebra finches (Taeniopygia guttata), mycobacteriosis has been described as a multi-organ granulomatous disease.2,10,20 One of the most common agents of avian mycobacteriosis in passerines and psittacines is Mycobacterium genavense, a ubiquitous fastidious slow growing nontuberculous mycobacterium first isolated in a human patient with acquired immunodeficiency syndrome in 1990.3,9 Transmission of M. genavense is mainly fecal-oral from contaminated birds in the environment.2,3 Aerosol transmission has also been proposed due to involvement of the respiratory system in some avian species. 20 Infections can be subclinical and are often only diagnosed based on the presence of acid-fast positive bacteria in infected tissues on histology or detection of genetic material in fecal samples by PCR. 3 Due to the non-specific nature of the clinical presentation in infected animals and the zoonotic potential, thorough investigation during necropsy and accurate diagnosis are important to prevent animal and human infections. 18 While cardiac involvement has been documented for mycobacteriosis in psittacines, this association has not been established in passerines. 13 This report documents heart-base lesions associated with M. genavense infection in a research colony of zebra finches used to study song learning.
In a 2-year period (2022–2024), within a closed colony of approximately 150 birds, 20 animals that were either found dead or euthanized were submitted for diagnostic necropsy. Of the 20 animals, 9 were identified to have great vessel and heart-base inflammation, and 7 were diagnosed with mycobacteriosis based on the presence of intralesional acid-fast (AF) positive bacteria. Examined birds ranged from 2 to 5 years of age, representing both sexes (Table 1). Clinical signs prior to postmortem evaluation were often non-specific, including respiratory distress, lethargy, feather loss, and ataxia resulting in the inability to fly or ambulate. Three animals were clinically normal prior to being found dead within their enclosure. At necropsy, internal gross lesions were only noted in 2 animals and consisted of cardiomegaly and multifocal to coalescing peritoneal hemorrhage. The heart (including great vessels), lungs, liver, kidney, spleen, head (with brain), skeletal muscle, pancreas, gastrointestinal tract, reproductive tract, and skin were collected for histology.
Summary of histopathologic findings in zebra finches with cardiac lesions associated with mycobacteriosis.
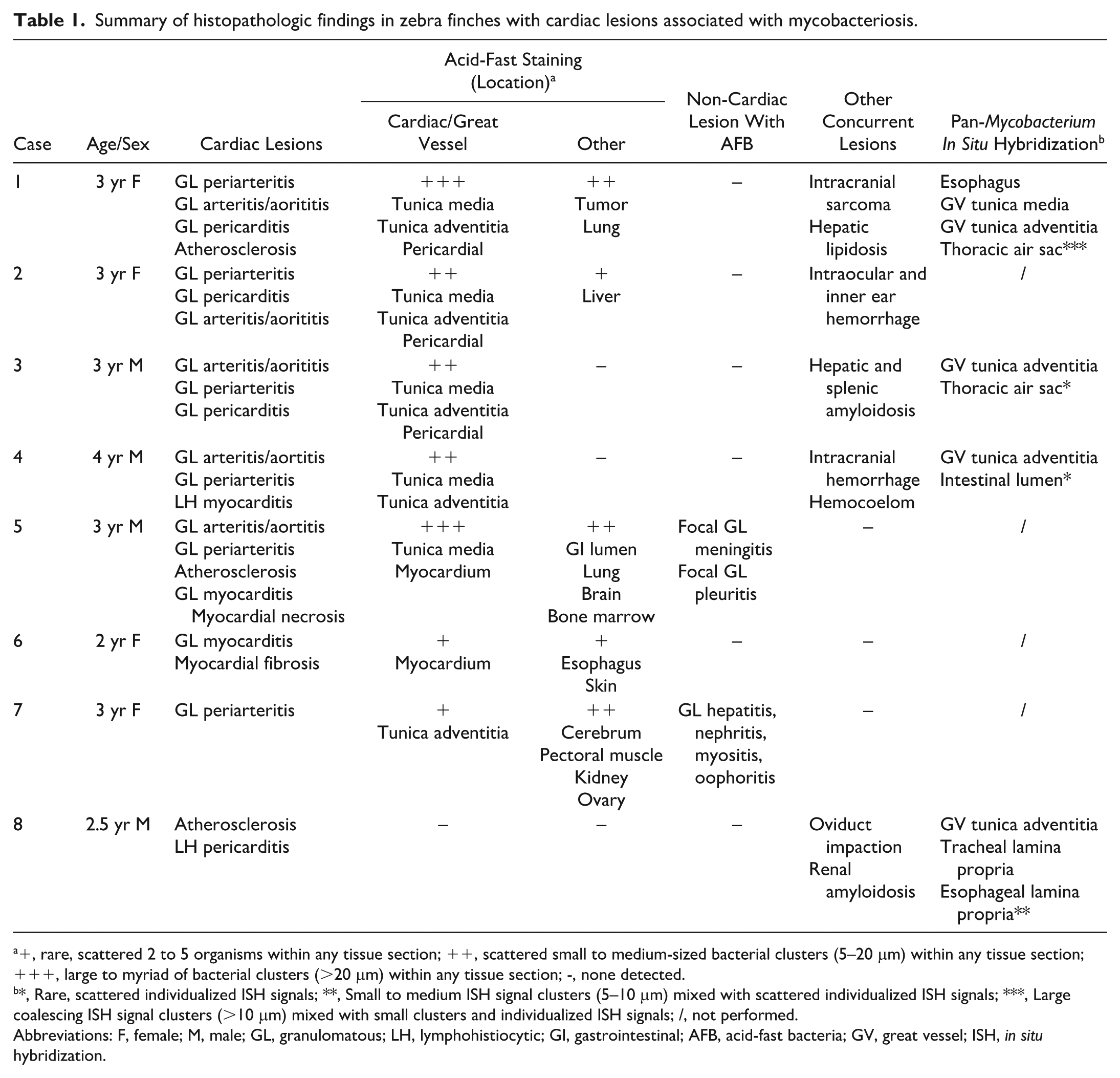
+, rare, scattered 2 to 5 organisms within any tissue section; ++, scattered small to medium-sized bacterial clusters (5–20 μm) within any tissue section; +++, large to myriad of bacterial clusters (>20 μm) within any tissue section; -, none detected.
*, Rare, scattered individualized ISH signals; **, Small to medium ISH signal clusters (5–10 μm) mixed with scattered individualized ISH signals; ***, Large coalescing ISH signal clusters (>10 μm) mixed with small clusters and individualized ISH signals; /, not performed.
Abbreviations: F, female; M, male; GL, granulomatous; LH, lymphohistiocytic; GI, gastrointestinal; AFB, acid-fast bacteria; GV, great vessel; ISH, in situ hybridization.
Microscopically, all 8 birds had granulomatous inflammation at the base of the heart within the large arteries and the pericardium with associated perivascular lymphohistiocytic foci (Fig. 1a, b). In the great vessels, the mid to outer tunica media was predominately affected, with expansion by clusters of foamy macrophages (Fig. 1c). In most birds (7/8), approximately 1 to 2 μm AF bacilli were observed within the histiocytes or as free bacteria within the inflammatory foci (Fig. 1d). Intrahistiocytic or free AF bacilli were observed within inflammatory foci surrounding great vessels of 4 birds. Three birds had concurrent myocarditis within the ventricular walls, accompanied by myocardial degeneration, necrosis, or fibrosis. Myocardial inflammation was either lymphohistiocytic with no detectable AF bacteria or granulomatous when intralesional AF bacteria were present. With the exception of one case, all birds had AF bacteria sparsely present within the parenchyma or small blood vessels in other organs in lesser quantities that were associated with little to no inflammation (Table 1). Other concurrent histopathologic findings included hepatic lipidosis, interstitial nephritis, hepatic and splenic amyloidosis, and an intracranial sarcoma (Table 1).
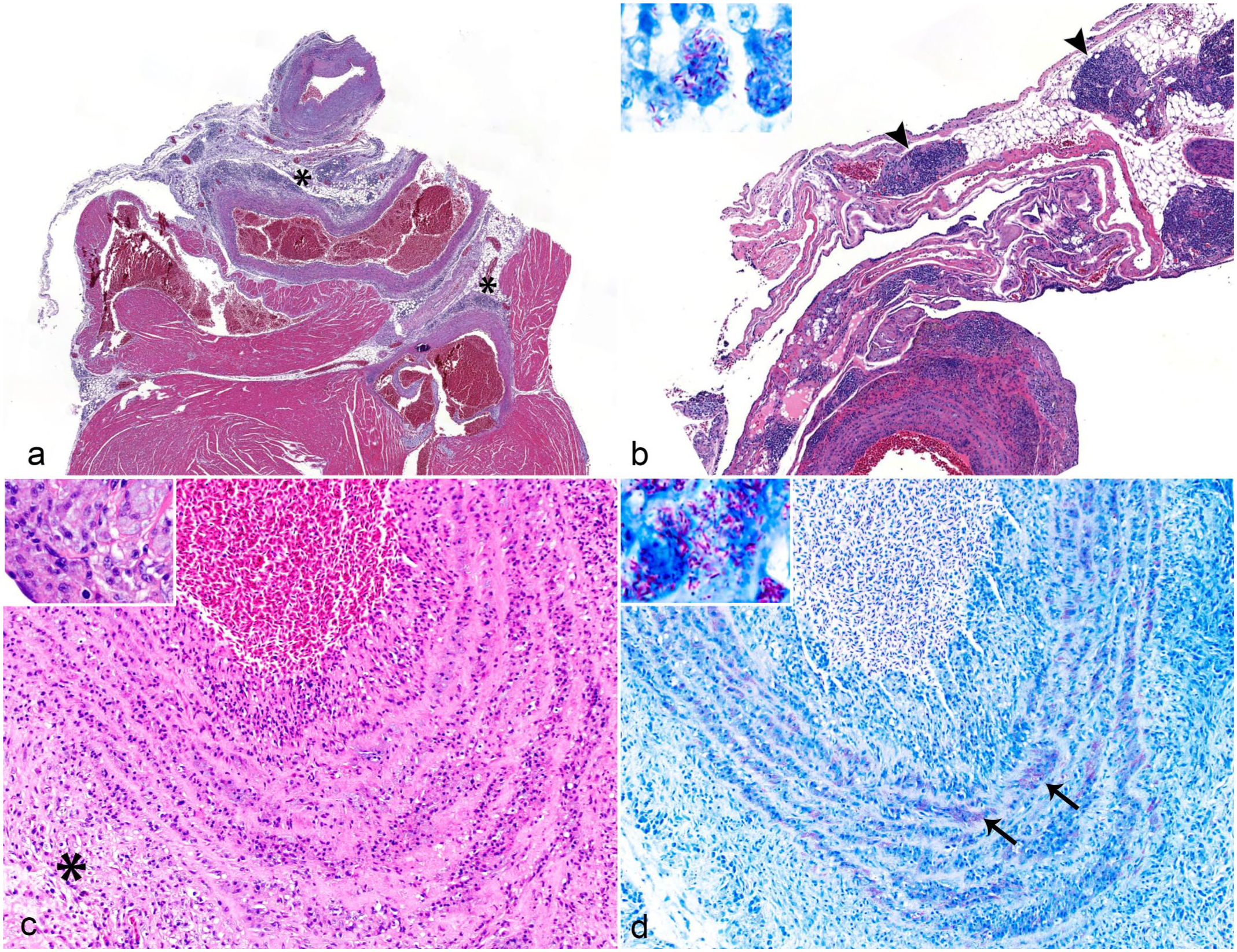
Mycobacteriosis, heart and great vessels, zebra finches. Granulomatous arteritis, periarteritis, and pericarditis in the heart-base of zebra finches. (a) Multifocal lymphohistiocytic foci (asterisks) at the base of the heart that surround or infiltrate the aorta and other great vessels. Hematoxylin and eosin (HE). (b) Multifocal inflammatory foci at the heart-base and periarterial tissues are frequently laden with acid-fast positive bacteria (arrowheads). HE. Inset: higher magnification. Ziehl-Neelsen (ZN) acid-fast. (c) The tunica media of the aorta is expanded by foamy macrophages and lymphocytes, mixed with flocculent eosinophilic material (inset). The tunica adventitia also has mixed mononuclear inflammatory cells (asterisk). HE. (d) Numerous intracellular, slender, rod-shaped, acid-fast-positive bacteria within the aortic tunica media (arrows, inset). ZN.
Five birds were selected to further probe for the presence of Mycobacterium spp. using a pan-mycobacterial RNAscope in situ hybridization (ISH) assay (Table 1 and Supplemental Materials). 17 One bird (case 1) was used as positive control (Fig. 2a, b). Two birds (cases 3 and 4) with inflammation and AF bacilli solely detected in the heart base had small numbers of strong hybridization signals adjacent to the great vessels (Fig. 2c), thoracic air sac, and intestinal lumen. One bird (case 8) that had granulomatous pericarditis without AF bacilli showed scattered hybridization signals adjacent to the aorta, esophagus (Fig. 2d), and trachea. One bird with granulomatous aortitis had no AF bacilli or hybridization signals detected in any tissue sections.
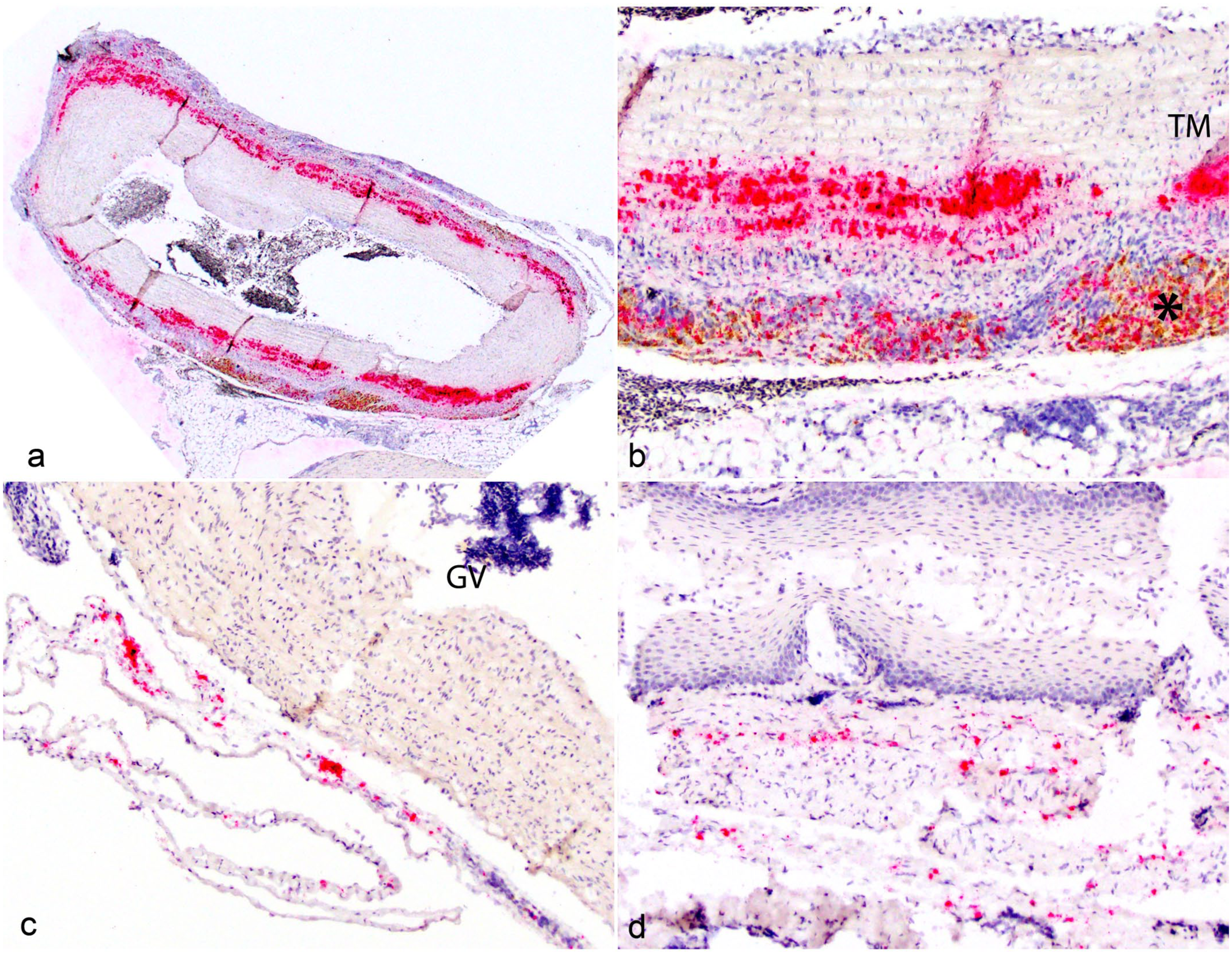
Pan-mycobacterial RNAscope in situ hybridization in great vessels and non-cardiac tissue in zebra finches. (a) Case 1, positive control. Strong, coalescing hybridization signals distributed circumferentially throughout the aortic tunica media (red). (b) Higher magnification of (a) demonstrating strong clustering hybridization signals within the aortic tunica media (TM), intermixed with pigment-laden macrophages in the outer aortic wall (asterisk). (c) Case 4. Few strong hybridization signals adjacent to a great vessel (GV). (d) Case 8. Scattered moderate hybridization signals within the lamina propria of the esophagus.
To further characterize the bacterial species, a pooled fecal sample collected from the colony was submitted for Mycobacterium genus PCR panels, with primers targeting 16S rRNA and heat shock protein.22,23 The amplicons were further sequenced and identified as Mycobacterium genavense (GenBank reference: MT107781.1, 100% identity) (Athens Veterinary Diagnostic Laboratory, GA). Paraffin scrolls from blocks containing AF bacteria were also submitted for the same test, but the results were negative results due to poor DNA quality.
Mycobacterium avium subsp. avium remains the most common cause for avian mycobacteriosis, but several other Mycobacterium species have also been known to be pathogenic to birds, including M. genavense. Mycobacterium genavense infections are primarily reported in pet birds such as passerines and psittacines and may have gross and histopathological lesions analogous to that of the Mycobacterium avium complex.11,21 Outbreaks or spontaneous M. genavense infections have been reported in other avian groups as well, such as Columbiformes, Ciconiiformes, Coliiformes, Coraciiformes, Galliformes, and Sphenisciformes. Mycobacterial lesions in these species primarily consisted of the typical patten of Mycobacterium avium complex infections with multifocal to widely disseminated granulomas containing AF bacteria.8,12,18
In passerines and psittacines, the true prevalence of mycobacterial infections remains unknown, as it depends on multiple factors such as age, housing conditions, and origin of animals (captive bred vs wild caught). 21 Commonly reported gross lesions are often non-specific but have included hepatomegaly, splenomegaly, peritonitis, hypertrophy of the small intestine with rare nodules present in the mucosa, pneumonia, and congestion of the lung and brain.7,9,11,13,16 Histologically, M. genavense infection in psittacines and passerines appears to mostly resemble lepromatous granulomatous inflammation rather than distinct granulomas. These lesions are comprised of poorly organized epithelioid or foamy macrophages with or without multinucleated giant cells laden with AF-positive bacilli, and lack distinct aggregation or structured formation of a granuloma. 20 AF bacteria are most abundantly found in the intestinal lumen, likely due to the predominant fecal-oral route of transmission. 15 Lesions can be present in the hepatic parenchyma, lamina propria of the small intestine, white pulp of the spleen, lungs, thymus, and meninges. Localized M. genavense infection without systemic dissemination can also occur in skin, conjunctiva, and central nervous system.6,18,20 Cardiac lesions associated with mycobacteriosis in passerines and psittacines, especially involving the great vessels, have been sporadically described, documenting some similarities between the 2 groups. In an extensive retrospective study regarding mycobacteriosis in psittacines, the cardiac lesions included infiltration of macrophages and rare multinucleated cells in the epicardium and pericardium that infrequently involve the myocardium. A cockatiel was also reported with granulomatous aortitis and infiltration of AF bacteria. Among the few case reports of passerines, one European goldfinch (Carduelis carduelis) and one fairy bluebird (Irena puella) diagnosed with mycobacteriosis had cardiac lesions that included granulomatous aortitis and peri-aoritis with intrahistiocytic AF bacteria. 13 The goldfinch also had concurrent myocardial degeneration, fibrosis, and necrosis. 11
There is limited documentation of gross and histopathologic lesions associated with M. genavense infection in zebra finches, and great vessels lesions in these reports were rarely mentioned or identified.2,10,20 Two zebra finches from separate institutions have reported similar aorta and heart-base lesions, but intralesional AF bacteria were not always present.10,19 In our study, the heart-base and great vessels were a key index location in all animals diagnosed with mycobacteriosis, as inflammatory lesions were typically lacking in tissues other than the heart. In one animal (case 3), inflammation and bacteria were exclusively localized to the heart-base. In some animals, besides the heart-base inflammation, aortitis resembled atherosclerosis on hematoxylin and eosin-stained sections, with the tunica media expanded by moderate to large numbers of foamy macrophages containing AF bacteria. While we were unable to identify the species of Mycobacterium in individual animals, broad-range Mycobacterium genus PCR with sequencing of pooled fecal samples detected the presence of M. genavense within the colony.
One additional animal from this colony had similar cardiac and great vessel lesions, but neither acid-fast staining nor ISH identified mycobacterial organisms. This may be due to the low sensitivity of histochemical staining in detecting etiologic agents or bacterial RNA degradation from prolonged tissue fixation. In 3 animals, ISH was able to detect organisms in tissues that were negative for AF staining. This discrepancy may reflect the increased sensitivity of ISH compared to AF, or that the bacteria were AF-negative due to their latent state and decreased AF staining affinity secondary to an altered cell wall composition. 24 In most animals, bacteria were present within tissue parenchyma without an associated inflammatory response, suggesting potential immune evasion during early infection or bacterial dormancy. 5 Our findings demonstrate that contemporary diagnostic techniques, such as ISH, can effectively complement traditional staining methods for detecting mycobacterial infections in paucibacillary cases or those with atypical histopathologic patterns.
The zoonotic risk for M. genavense presents a potential public health concern. Mycobacterium genavense infection has been reported across several other mammalian species, including dogs, ferrets, cats, and humans, most with a history of an immunosuppressive health status. 7 In humans, especially in human immunodeficiency virus (HIV) immunocompromised individuals, fecal-oral and airborne transmission may occur. 4 While there is limited published data documenting direct spread from birds to humans, transmission has occurred from close contact with infected animals and contaminated water.1,4 Infection has also been identified in non-HIV patients after immunosuppressive therapy or hematopoietic stem cell/organ transplantation. 1 This underscores the importance of accurately diagnosing mycobacterial infections in research or pet bird colonies, as well as utilizing appropriate personal protective equipment (respirator, gloves, gown, and face shield) in a research setting, primarily for personnel that are immunocompromised and working directly with a Mycobacterium-positive colony.
In conclusion, our study describes heart-base and great vessel localization of mycobacterial infection in zebra finches in a research setting. The inability to diagnose this disease antemortem due to non-specific clinical signs suggests mycobacteriosis in passerines may be widely underdiagnosed. Furthermore, Mycobacterium could remain latent in the great vessels resulting in asymptomatic chronic infections or nonspecific clinical disease. 14 Therefore, all bird colonies should be routinely screened for the presence of Mycobacterium spp. via PCR, and any potential clinical manifestation of the disease should be investigated. Diagnostic necropsies including histologic examination with AF staining of the heart and great vessels is recommended, especially when Mycobacterium spp. has been detected in zebra finch colonies or other passerines.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251372573 – Supplemental material for Granulomatous arteritis/aortitis associated with Mycobacterium genavense in a colony of zebra finches (Taeniopygia guttata)
Supplemental material, sj-pdf-1-vet-10.1177_03009858251372573 for Granulomatous arteritis/aortitis associated with Mycobacterium genavense in a colony of zebra finches (Taeniopygia guttata) by Rachel R. Howie, Nicholas M. Tataryn, Katherine A. Shuster, Katherine N. Gibson-Corley, Agustín Rebollada-Merino, Elena A. Demeter and Tzushan S. Yang in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the Translational Pathology Shared Resource Center which is supported by NCI/NIH Cancer Center Support Grant P30CA068485 for their diagnostic support and services. In addition, we acknowledge Dr Nicole Creanza of Vanderbilt University for her collaboration and technical assistance.
Author Contributions
RRH and TSY contributed to writing the original draft, investigation, and editing. NMT, KAS, and KNG-C provided case materials, resources, and contributed to editing the manuscript. AR-M and EAD conducted and assisted in interpreting the ISH assay and contributed to editing the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
