Abstract
Various pathogens gain access to the intestinal wall via specialized cells, the M cells, found among the follicle-associated epithelial cells overlying the domes of the Peyer's patches. The present study was undertaken to examine the uptake of live Mycobacterium avium subsp. paratuberculosis in the distal small intestine of goat kids. Following laparotomy, distal small intestinal segments of five goats were ligated and injected with bacterial suspension. After 1 hour, the intestinal segments were excised and fixed for light and electron microscopic studies. M. a. paratuberculosis organisms were observed by transmission electron microscopy at locations in the intestinal wall, suggesting transcellular transportation through the M cells. The organisms were present both in the cytoplasm of the M cells and in the cytoplasm of intraepithelial leukocytes found in M-cell pockets. Intercellular bacteria between M cells were occasionally seen. Bacteria were not observed in association with the absorptive epithelium. This study indicates that in goat kids, M. a. paratuberculosis enters the intestinal wall primarily through the M cells in the follicle-associated epithelium of the Peyer's patches.
Keywords
The mucosa of the gastrointestinal tract has a well-developed local immune system, the gut-associated lymphoid tissue (GALT). The major components of the GALT are the Peyer's patches, which are organized areas of lymphoid tissue. The Peyer's patches consist of follicles, domes with an overlying follicle-associated epithelium (FAE), and the interfollicular areas in the submucosa. 1 4 10 The FAE includes highly specialized cells, the membranous (M) cells. Apart from M cells, there are variable numbers of columnar epithelial cells, occasional goblet cells, and lymphocytes. 1 4 M cells have microvilli or microfolds on their apical surfaces, and they adhere to adjacent cells by tight junctions and desmosomes. The basolateral membrane is invaginated, forming a pocket (an extracellular space) containing leukocytes. 1 4 M cells are important in antigen sampling and transportation to the immunocompetent cells of the underlying lymphoid tissue. 1 4 9 There are species differences in morphology, function, and number of Peyer's patches and in the type of FAE in the small intestine. 5 7 9 Despite these differences, there are many similarities in functional properties of the FAE, including the ability to transport macromolecules and microorganisms into the intestinal mucosa. 5 10
In paratuberculosis, a chronic disease of ruminants caused by Mycobacterium avium subsp. paratuberculosis, infection occurs at an early age and, after a long incubation period, leads to granulomatous enteritis and lymphadenitis. 2 Infection is established following the ingestion of bacteria, their penetration of the intestinal mucosa, and subsequent phagocytosis by resident macrophages. The bacteria multiply within macrophages and with time cause severe granulomatous inflammation in the intestine and in the draining lymph nodes. 1 2 Early intestinal lesions in paratuberculosis are usually located in the submucosa, between or involving the capsule of the lymphoid nodules of the Peyer's patches. 13 This association with the lymphoid tissue of the Peyer's patches suggests that the FAE is involved in the uptake of the bacteria. 9
Although paratuberculosis is a well-known disease of goats, it has not been as extensively studied in goats as in sheep and cattle. The aim of the present study was to examine the uptake of M. a. paratuberculosis organisms in the distal small intestine of goats. Emphasis was placed on investigating the association of bacteria with the M cells, particularly in comparison to the adjacent absorptive epithelium.
Material and Methods
Animals and experiment design
Five male goat kids (Nos. 1–5) of the Norwegian breed were used in this study. The goat kids came from a herd with no history of paratuberculosis. However, animals within the herd were regularly vaccinated against the infection with live attenuated strains of M. a. paratuberculosis (National Veterinary Institute, Oslo, Norway). 12 The kids were allowed to suckle their dams until delivery to the National Veterinary Institute. Upon arrival, the goat kids were between 12 and 17 days old. At the Institute, the kids were fed pasteurized soured cow milk from suckling bottles, and hay was available ad libitum. The cow milk came from a source (and geographic region in Norway) where paratuberculosis in cattle has never been detected. When the experiment commenced, the kids were between 18 and 21 days old. The kids did not receive milk approximately 12 hours prior to surgery. The kids were anesthetized with 0.1 mg/kg xylazine (Rompun®, Bayer AG, Leverkusen, Germany) and 10 mg/kg thiopental (Pentothal-Natrium®, Abbott Scandinavia AG, Sweden). An incision was made into the right flank, and the distal small intestine was exposed. Four segments from the distal small intestine were ligated with a nonlacerating cotton string. Each segment was about 5 cm long and approximately 2, 17, 23, and 34 cm, respectively, from the ileocecal valve. After ligation, most of the intestinal content was aspirated with a hypodermic syringe, and 3 ml of a body-temperature bacterial suspension containing 2.365 mg of bacteria was injected into the lumen of each intestinal segment. Injections were performed close to the ligature at one end of each segment, away from the central area where tissue was later sampled. Care was taken to minimize the mechanical damage to the intestinal wall during this procedure and to maintain the mesenteric blood supply to each ligated intestinal segment; upsetting the integrity of the mucosal epithelium could be a source of erroneous results. After administration of the bacterial suspension, the intestine was repositioned into the abdominal cavity, and the incision was sutured. The animals were maintained under anaesthesia for 1 hour before the flank was reopened, and the segments were excised and immersed in fixative. The animals were then immediately euthanatized with an intravenous injection of 100 mg/kg pentobarbital (National Hospital Pharmacy, Oslo, Norway). From kid No. 5, two segments were removed 30 minutes after bacterial injection, and the remaining two segments were removed 60 minutes after injection.
Bacterial strains and culture methods
Bacteriologic isolation and identification were as previously described. 11 The strain of M. a. paratuberculosis used had recently been isolated from the intestine and several lymph nodes of a goat with clinical paratuberculosis. Bacterial colonies were scraped from the culture medium and ground finely in a mortar, and 47.3 mg (dry weight) was suspended in 10 ml of 0.1 M phosphate-buffered saline (PBS de Boer) solution, pH 7.2. The suspension was divided into five portions of 2 ml each and stored at 4 C until used for inoculation a few days later. When ready to use, each 2-ml suspension was diluted further in PBS to 12 ml. The five doses of 12 ml were then divided into 4 portions of 3 ml, each containing 2.365 mg bacteria.
Light and electron microscopic examination
Before resection of the intestine, the intestinal content with the excess bacterial suspension was aspirated from each ligated segment. The fixative, 3% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.2, was then injected into the intestinal lumen, and the segments were resected and immersed in the same fixative. After 5–48 hours in fixative, samples were taken from each segment, cut into small pieces (approximately 1 mm3), and placed in a new solution of 3% glutaraldehyde for transmission electron microscopy (TEM). The small pieces were postfixed in 2% osmium tetraoxide containing 0.1 M cacodylate buffer, pH 7.2, for 2 hours. The pieces were then dehydrated and embedded in LX 112 Resin (Ladd Research Industries). Semithin (1 µm) epoxy sections mounted on glass slides were stained with toluidine blue for correlation between light microscopy (LM) and TEM. Ultrathin sections (silver to gold interference colors) were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined with a Jeol transmission electron microscope. The remaining glutaraldehyde-fixed material was transferred to 10% buffered formalin for light microscopic studies. The material in formalin was fixed for up to 1 week, trimmed, and processed by routine paraffin embedment. Sections (2–3 µm) were cut, mounted, and stained with hematoxylin and eosin (HE) and with the Ziehl-Neelsen (ZN) method for acid-fast bacteria.
Two sections from each segment of intestine stained with both HE and the ZN method were examined by LM. Light microscopic examination revealed small hemorrhagic foci in 15/40 sections (37.5%), mainly in the lymphoid tissue of the Peyer's patches. There was mild to moderate edema in 14/40 sections (35%). Tissue sections for TEM examination were selected from intestinal segments showing minimal hemodynamic changes.
Results
LM
In HE-stained sections, the large lymphoid follicles of the Peyer's patches were present in all sections. Many mononuclear and polymorphonuclear leukocytes were present within the FAE. In ZN-stained sections, acid-fast bacteria were occasionally detected in the cytoplasm of M cells and in intraepithelial mononuclear leukocytes (Figs. 1, 2). Bacteria were observed within the domes and in the adjacent villi (Fig. 3) but were most numerous in the submucosal interfollicular areas of the Peyer's patches. At these sites, the acid-fast bacteria were both free and cell associated and were present in the interstitial tissue and in the lumina of lymphatic vessels. Bacteria were not observed in the absorptive epithelium of villi, between M cells, in intraepithelial polymorphonuclear leukocytes, or in the lymphoid follicles of the Peyer's patches.
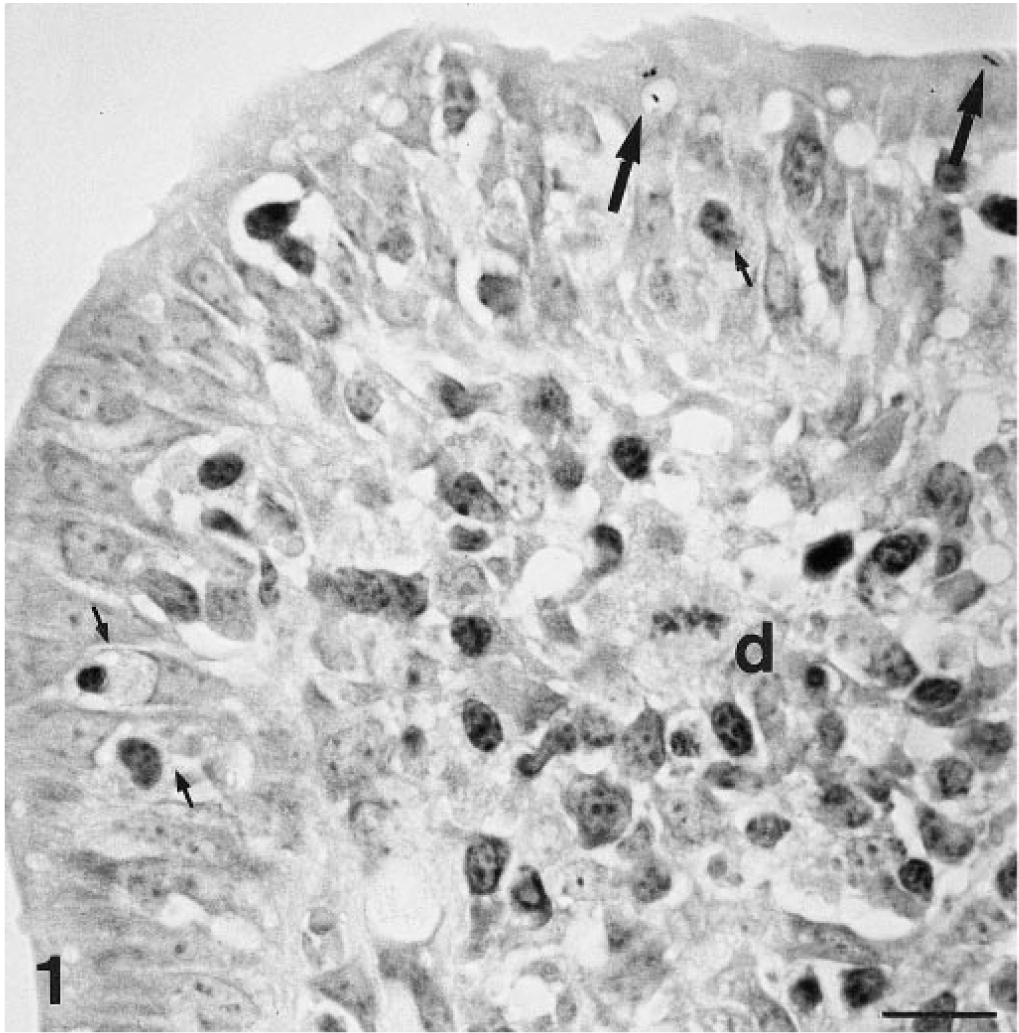
Intestine; goat. Acid-fast bacteria (long arrows) in the apical portion of M cells. Several mononuclear leukocytes (small arrows) are present within the follicle-associated epithelium. d = dome. ZN. Bar = 20 µm.
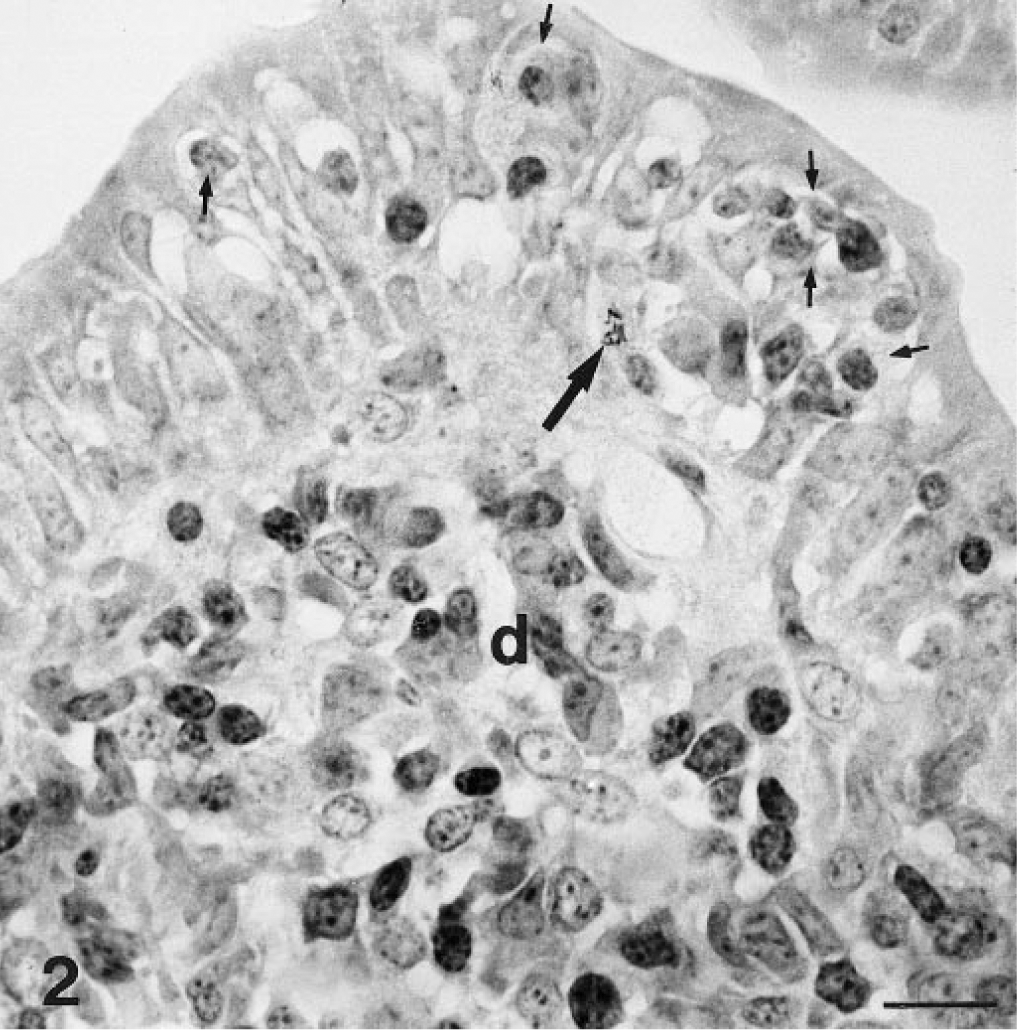
Intestine; goat. Acid-fast bacteria (long arrow) in a mononuclear leukocyte present within the follicle associated epithelium. Small arrows = intraepithelial mononuclear leukocytes; d = dome. ZN. Bar = 20 µm.
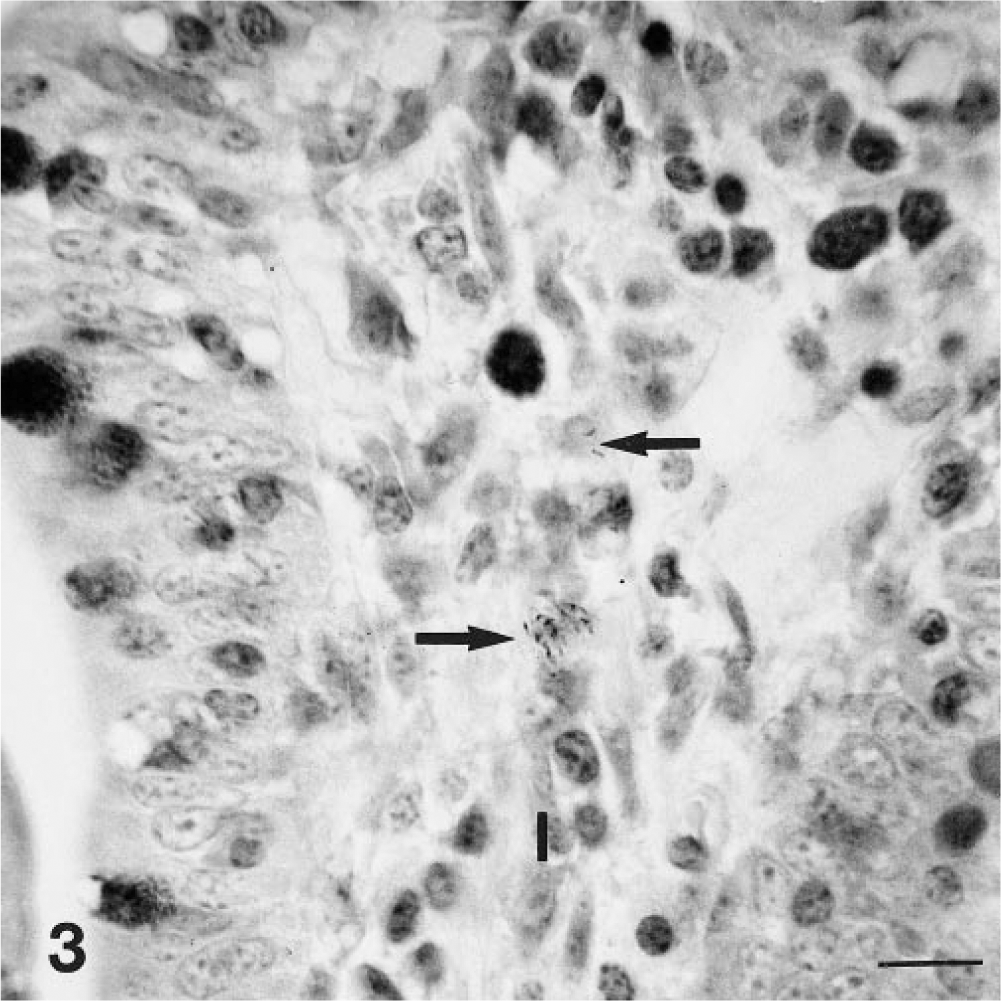
Intestine; goat. Intracellular acid-fast bacteria (long arrows) in the lamina propria (l) of a villus. ZN. Bar = 20 µm.
TEM
Electron microscopic examination revealed bacteria within M cells. A single bacterium was usually seen in these specialized cells (Fig. 4). Bacteria were also seen within the cytoplasm of leukocytes in the large pockets of M cells (Fig. 5). Only rarely were bacteria seen free in the M-cell pockets. Bacteria were also seen between M cells (Fig. 6). Transcellular transportation through M cells, whether free or in leukocytes, predominated over paracellular transportation. In the domes, free bacteria were more numerous than cell-associated bacteria, whereas phagocytes had engulfed most of the bacteria in the submucosal interfollicular areas. A few free and phagocytized bacteria were present in the villus lamina propria adjacent to domes, often in close proximity to lacteals.
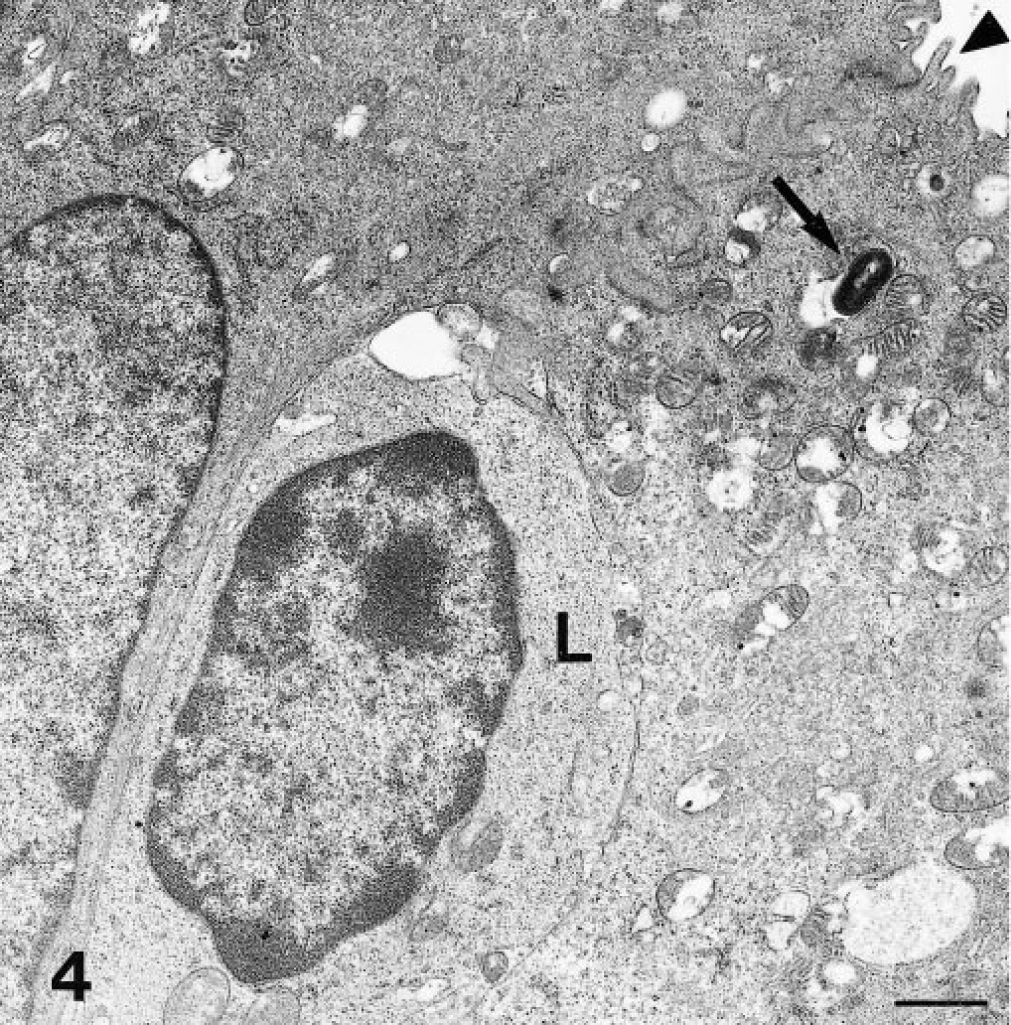
Transmission electron micrograph. Intestine; goat. A bacterium (arrow) is present in the apical portion of an M cell. Note microvilli (arrowhead) of M cells. L = lymphocyte in pocket of M cell. Uranyl acetate and lead citrate. Bar = 1 µm.
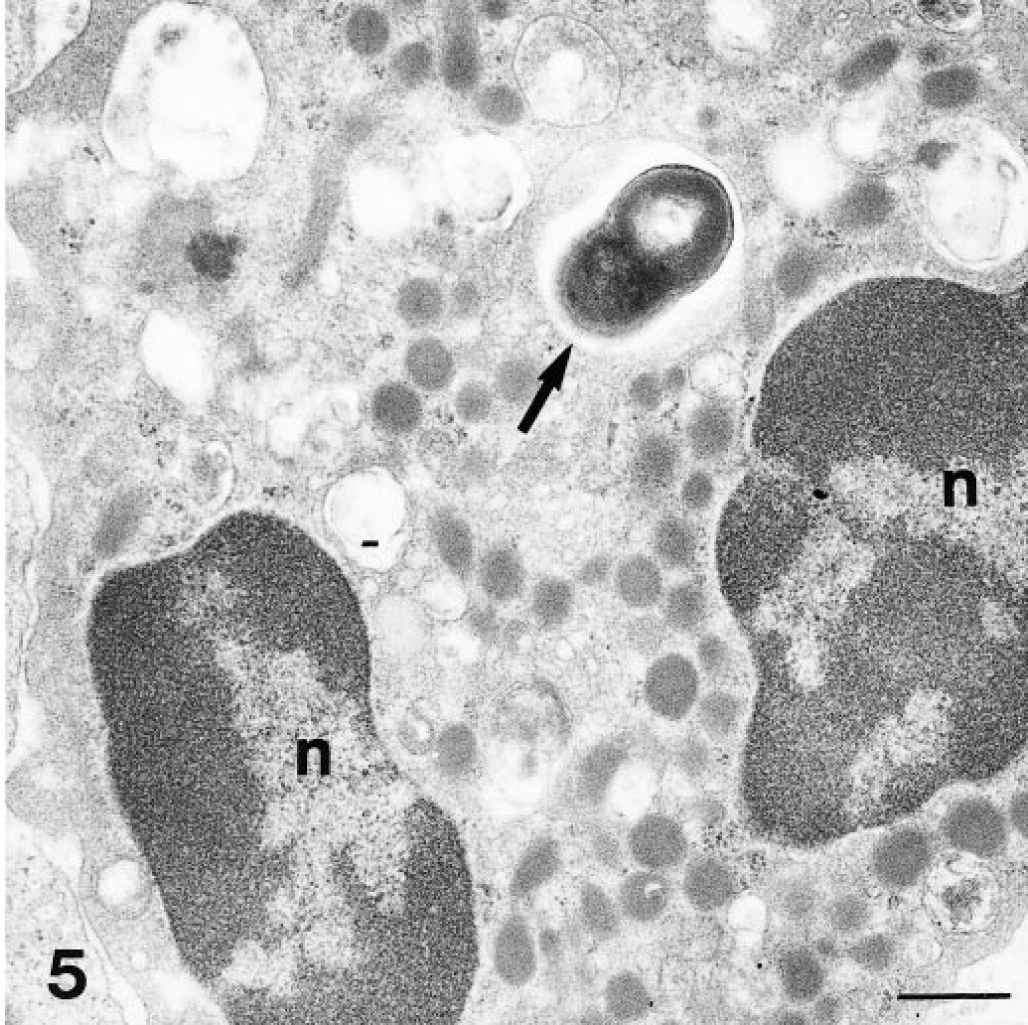
Transmission electron micrograph. Intestine; goat. A bacterium (arrow) is present in a neutrophilic leukocyte (NLS), located in the pocket of an M cell. n = nucleus of NLS. Uranyl acetate and lead citrate. Bar = 1 µm.
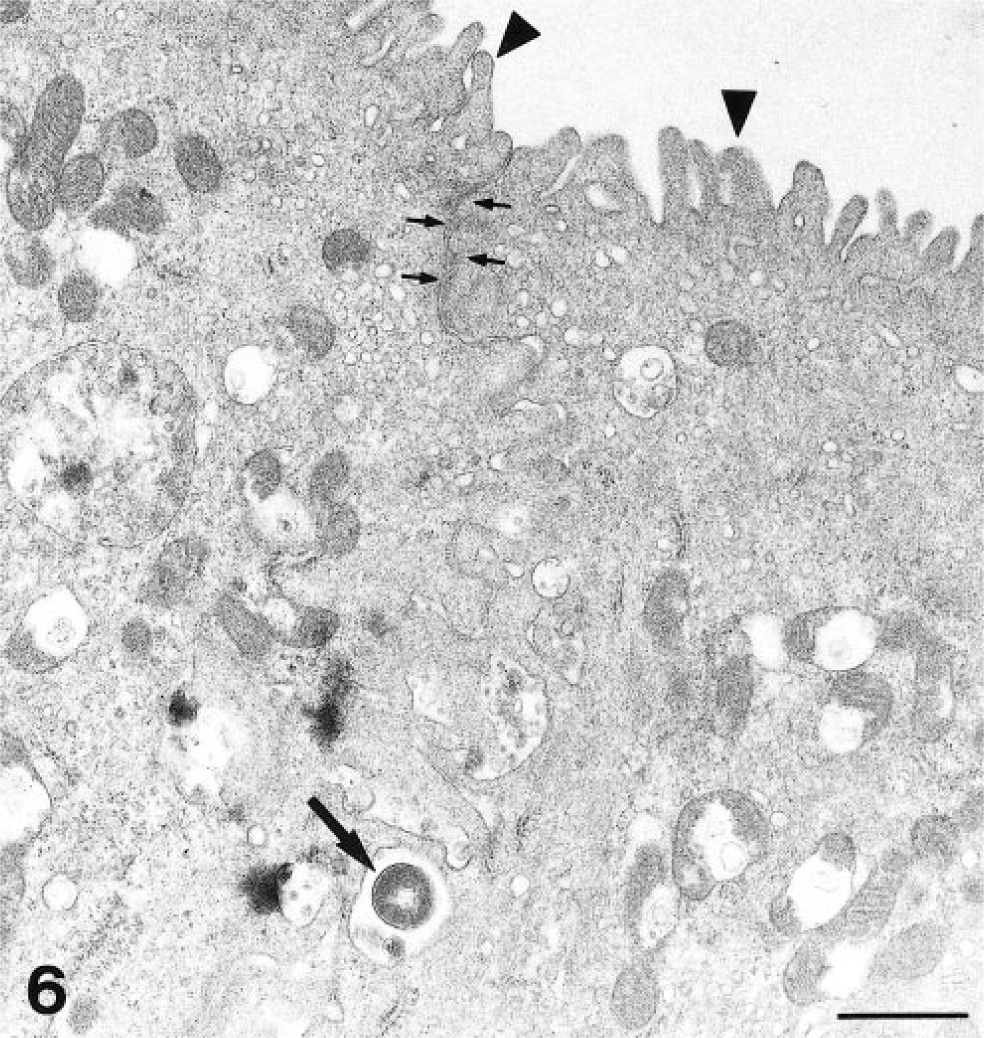
Transmission electron micrograph. Intestine; goat. A bacterium (large arrow) is located between two M cells. Arrowheads = microvilli on M cells; small arrows = desmosomes. Uranyl acetate and lead citrate. Bar = 1 µm.
There were numerous bacteria free in the intestinal lumen and some bacteria were present in the cytoplasm of intraluminal leukocytes. There was no obvious difference in the distribution and localization of bacteria between the intestinal samples collected after 30 minutes and after 60 minutes of incubation from goat No. 5.
Discussion
There is a rapid uptake of M. a. paratuberculosis in goat intestine, and the bacteria primarily enter the dome and interfollicular areas of the lymphoid tissue of Peyer's patches. Bacteria gain access to the subepithelial compartment of the intestinal wall by way of transcellular or paracellular transportation across M cells. The transcellular route was the more frequently observed method of bacterial uptake and could be demonstrated as early as 30 minutes after bacterial inoculation into the intestinal segments. This route of uptake is in accordance with previous studies of microorganisms, including M. a. paratuberculosis and BCG bacteria. 3 9 Although bacteria were most often demonstrated within the cytoplasm of the M cells, a few bacteria were also observed within the M cell pockets, either free or engulfed by leukocytes (Figs. 4, 5). This finding was interpreted as representing the sequential stages of transportation in the M cell. However, it is not certain whether all bacteria entering M cell pockets are engulfed by phagocytes before being expelled to the underlying interstitial tissue of the domes. In the domes, both cell-associated and free bacteria were seen.
Our findings are concordant with the results obtained in an experiment with rabbits, where BCG bacteria were injected into intestinal loops. 3 In these rabbits, there was a rapid uptake of the organisms, with organisms being present in M cells within 1 hour of injection. Paracellular uptake of bacteria was also evident. In addition, macrophages, both enfolded by the M cells and present below the FAE, contained bacteria at 1 hour postinjection. However, free bacteria were not described in domes or in the interfollicular areas of the rabbit intestines. 3 In a similar experiment in calves using live and killed M. a. paratuberculosis, the results were somewhat different. 9 Only a few bacteria were observed in M cells after 20 hours of incubation, and most of the bacteria showed signs of degeneration. Macrophages containing bacteria were however seen in the domes after 5 hours of incubation, and the cell-associated bacteria were especially numerous in the domes after 20 hours of incubation. Paracellular uptake of bacteria was not demonstrated in the calf experiment, and although both lymphocytes and neutrophils were present in M-cell pockets, these cells did not contain bacteria. The study performed with calves and the present study of goats differed in tissue collection time. In the present study, the bacterial uptake was rapid (< 1 hour); most of the bacteria injected into the calf intestines may have already passed the M cells by the time the material was collected. The duration of the experiment could also explain the frequent presence of free bacteria in the domes of the goats, whereas the bacteria were always cell associated in the calves. 9
In the goat kids, bacteria, both free and cell associated, were found in the villus lamina propria adjacent to domes (Fig. 3). In the previous calf study, 9 macrophages containing M. a. paratuberculosis bacteria were found in the same location 20 hours after inoculation of the bacteria into the intestinal lumen of calves; these macrophages were thought to have migrated from the dome area after phagocytosing bacteria. Inflammation, in the form of individual multinucleated giant cells, groups of macrophages, or small granulomas, is sometimes demonstrated in the lamina propria at the tip of villi in mild to moderate lesions of paratuberculosis and in early lesions of experimental infections. 2 13 The route followed by bacteria to reach the villus lamina propria in the present study is not known, but bacteria were not observed in the absorptive epithelium of the villi. Although the presence of free bacteria may suggest direct entry across the absorptive epithelium, the interconnecting network of branching lymphatics between the lamina propria and the dome areas of Peyer's patches may provide a route of passage for the free bacteria occasionally seen in the lumina of lacteals. 8 Further experiments are needed to address the origin of villi lesions and the role of absorptive epithelium of the villi in M. a. paratuberculosis infection.
M. a. paratuberculosis enters the intestinal wall via the specialized M cells of the FAE overlying the domes of Peyer's patches. The apparent preference for this route of entry may explain the association between paratuberculosis lesions and the lymphoid tissue of Peyer's patches. 6 9 13 Indications for other routes of infection were found, which may account for lesions in intestinal villi without obvious connection to submucosal lesions, suggesting direct invasion of villi or hematogenous infection. 1 13
Footnotes
Acknowledgements
We thank Professor T. Landsverk for helpful discussions and advice and S. Nilsen and E. Engeland for their skillful technical assistance. We also thank Professor G. Georgsson for cooperation in allowing part of this study to be performed at the Institute for Experimental Pathology, University of Iceland, Keldur, Iceland.
