Abstract
Rainbow trout (Oncorhynchus mykiss) were bath challenged with viral hemorrhagic septicemia (VHS) virus or infectious hematopoietic necrosis (IHN) virus or with both viruses simultaneously. The viral distribution and development of histologic lesions were examined using immunohistochemistry, while virus titer in kidney was determined by viral titration in cell culture. Single infections with VHS virus and IHN virus showed similar distributions of virus in internal organs. The early identification of virus in gill epithelium, 1 and 2 days postinfection (PI) for VHS virus and IHN virus, respectively, indicates that this organ is the point of entry for both viruses. The detection of VHS virus at 1 day PI and 3 days PI for IHN virus is indicative of kidney and spleen being the target organs for these viruses. A simultaneous infection of VHS virus and IHN virus resulted in both viruses establishing an infection. Further double infection did not result in a statistically significant lower titer of both viruses in kidney but a more restricted distribution of IHN virus in internal organs compared with the single infected group. The most striking finding is that, for IHN virus, virus was not detected in the brain in situ in the double-infected group. This study provides support for the conclusion that simultaneous infection with two piscine rhabdoviruses in a susceptible host results in some degree of interaction at the cell level, leading to a reduced systemic distribution of IHN virus.
Keywords
Viral hemorrhagic septicemia (VHS) virus and infectious hematopoietic necrosis (IHN) virus are both important disease agents in farmed rainbow trout, and disease outbreaks cause major losses in fish farming worldwide.
There are many similarities between these two viruses and the diseases they cause. Both VHS virus and IHN virus are viruses of the Novirhabdovirus genus (Rhabdoviridae family). As the name indicates, VHS is a hemorrhagic disease. Further, the pathologic changes observed during IHN in salmonids are similar to VHS. The organs affected during an infection with both viruses are usually kidney and spleen, though most organs and tissues are affected in the later stages of the disease. Hematopoietic tissue in kidney and white pulp of the spleen are the most frequently affected tissues, but there are cases in which virus was isolated from the brain only.15,20,33 Leukocytes and endothelial cells are anticipated to be important sites for virus replication.29,32
It has been demonstrated that VHS virus effectively infects by waterborne challenge. Both gills and intestine are suggested as a primary site of infection19,29 and Yamamoto and coworkers31 showed that epithelial cells from skin and gills are capable of supporting early VHS virus and IHN virus replication. In contrast, others have not been able to demonstrate primary infection in gill epithelial cells using immunohistochemistry for in situ identification.11
Interactions between viruses during double or simultaneous infection have been studied thoroughly in mammals. Vesicular stomatitis virus (VSV) has an inhibitory effect on influenza virus transcription during coinfection,14 while vaccinia virus stimulates the growth of VSV 10–20-fold at the protein synthesis level in mouse L cells. The enhanced growth of VSV is correlated with the ability of vaccinia virus to inhibit the VSV-mediated damage to the host translational machinery.27 In cell culture, the synthesis of both VSV and mengovirus was inhibited during a simultaneous infection.21 This shows that viruses can interact during coinfections, resulting in stimulatory or inhibitory effects on viral replication. In fish, very few coinfection studies have been undertaken addressing this topic. In vitro studies have shown that catfish reovirus inhibits replication of channel catfish herpesvirus, the mechanisms implicated being both interference and induction of interferon-like factors.7 In vivo studies have been carried out on stocks of Atlantic salmon persistently infected with infectious pancreatic necrosis (IPN) virus. The study showed that, when these fish were infected with infectious salmon anemia virus, the IPN virus carrier stage was not activated.17 Further to this, a field case of coinfection of IPN virus and IHN virus has been reported in farmed rainbow trout in Spain.26 However, the possible interaction between the two viruses was not assessed. A more recent study showed that IPN virus infection in BF-2 cells in vitro interfered with the replication of IHN virus and resulted in a log 3 lower titer of IHN virus in double-infected cells.3
The aim of this study was to characterize the interactions between VHS virus and IHN virus during an experimental coinfection of rainbow trout in terms of viral distribution, as assessed by immunohistochemistry and concomitant histopathologic changes, and the extent of viral replication in kidney.
Material and Methods
Challenge of rainbow trout by bath
Full siblings of rainbow trout (Oncorhynchus mykiss) with a maximum weight of 10 g were infected with a VHS virus isolate, DK-3592B (with four to five passages in BF2-cells), serologically related to the reference strain F1 and with proven high pathogenicity in rainbow trout. For the IHN experiment, a European IHN virus isolate, 32.87 (sixth passage in EPC-cells), was used. The fish were split into four groups. For group I, the VHS virus-infected fish, all individuals were infected by bathing at concentrations of 105 TCID50 virus per milliliter of water for 3 hours at 10 C. Group II was infected with 106 TCID50 IHN virus per milliliter of water for 3 hours. Group III was infected with 105 TCID50 VHS virus and 106 TCID50 IHN virus per milliliter of water for 3 hours. Control fish (group IV) were exposed to an equal volume of virus-free cell culture medium (CCM) and handled as the infected fish. Groups I, II, and III consisted of 600 fish that were divided in three 200-liter tanks containing 200 fish each. In group IV, 50 fish were kept in a 50-liter tank.
Virus replication in kidney
Samples from posterior kidney from three fish from each group at each time point (see below) were subjected to virologic examination, including titration. Virus collected from cell culture supernatant was identified according to standard procedures by ELISA using monoclonal antibodies IP5B11 or 136-3 against VHS virus N protein (reference strain F1) or IHN Virus N-protein (strain 32.87), respectively. Titration of virus was performed according to standard procedures for quantification of virus.
Tissue sampling
Organ samples from three fish from each of the four different groups mentioned above were collected at 0 (before onset of the experiment), 24, and 48 hours and 3, 5, 7, 8, 10, 12, 14, 21, and 28 days postinfection (PI). Tissue specimens from skin, gills, brain, liver, spleen, kidney, pyloric caecae including pancreas, and muscle were sampled and submerged in 10% phosphate-buffered formalin (pH 7.4). The samples were embedded in paraffin wax according to standard procedures.
Immunohistochemical identification of virus in tissue
Single immunohistochemistry
The immunohistochemical procedure was performed as described previously12 with a few modifications. After deparaffinization, the sections were incubated with 5% (w/v) bovine serum albumin (BSA) in Tris-buffered saline (TBS, pH 7.4) for 20 minutes. The solution was removed from the slides and a monoclonal antibody specific for nucleoprotein from VHS virus (IP5B11) or IHN virus (136-3) was added to the slides at a dilution of 1:700 for 1 hour at 37 C. After washing for 5 minutes in TBS, the secondary antibody, biotinylated anti-mouse immunoglobulin diluted 1:300, was added. The solution was incubated for 30 minutes at room temperature. The sections were washed 5 minutes in TBS before incubation with streptavidin-biotin alkaline phosphatase diluted 1:500 for 1 hour at 37 C. After washing, Fast red (1 g/liter; Sigma, St. Louis, MO, USA) and naphthol AS-MX-phosphate (0.2 g/liter; Sigma) with 1 mM levamisole (Sigma) in 0.1 M TBS was added to develop for 20 minutes. Sections were washed in tap water before being counterstained with Mayer's hematoxylin and mounted in aqueous mounting medium (Aquamount, BDH Laboratory, Dorset, UK).
Performance controls included application of heterologous monoclonal antibodies (of the same isotype) at almost identical protein concentrations as the specific antibody. Tissue sections from noninfected rainbow trout were also incubated with specific or heterologous monoclonal antibodies. Estimation of end-point dilution value (the highest dilution giving a positive reaction that was discernible from background) was performed as previously described.12
Paired immunohistochemistry
The immunohistochemical incubations were performed as described previously10 with some modifications. Sections were incubated with 5% BSA for 20 minutes before solution was removed. The double staining included two separate sequences. The first sequence included a monoclonal antibody specific for nucleoprotein from IHN virus (136-3) that was added at a dilution of 1:700 for 1 hour at 37 C. After washing for 5 minutes in TBS, an alkaline phosphatase-labeled secondary anti-mouse antibody was added at a dilution of 1:500 for 1 hour at 37 C. After washing for 5 minutes in TBS, Fast blue was added and allowed to develop for 20 minutes. Sections were washed in TBS. Prior to the second incubation sequence, a normal mouse serum was added to block any free binding sites of the secondary antibody of the first sequence. This was followed by washing in TBS and subsequent incubation with a biotinylated monoclonal antibody specific for nucleoprotein from VHS virus (IP5B11) at a dilution of 1:700. Incubation was for 1 hour at 37 C. The sections were washed for 5 minutes in TBS before incubation with avidin–biotin peroxidase diluted 1:500 for 1 hour at 37 C. After washing, aminoethylcarbazole (AEC) was added to develop for 20 minutes. Sections were washed in tap water before being mounted in aqueous mounting medium (Aquamount, BDH Laboratory). No counterstaining was used. Control sections positive for VHS virus or IHN virus and a virus-free section were used to verify the specificity of the method.
Statistical analyses
The statistical analyses were performed in the SAS-PC System® Version 8 for Windows (SAS Institute Inc., Cary, NC, USA, 1999). The descriptive analyses were conducted using PROC FREQ. The odds ratios (OR) calculated from the estimated coefficients in the final models were used to express the chance of getting a positive sample. The two experiments with repeated measurements over time conducted in the study were analyzed separately with two regression models for both. The repeated effect in all models was the variable day of sampling. The independent variables, type of infection (1 = single, 2 = double) and the organs that were investigated in each fish, were included as fixed effects. The best model fit was found by forcing the variable type of infection into the model and including the organs investigated.
The outcome variables in the first experiment were the levels of VHS and IHN virus (measured in plaque forming units/gram kidney). The distributions of the outcomes were very skewed to the left, and therefore the logarithmic transformation was applied to the model. Twenty-four of the fish sampled had a virus titer equal to zero, and therefore we added the value 10 to all the titers. Two repeated measures analyses were conducted using PROC MIXED, and the kidney sample was the statistical unit. The Type III F-test was used to test the significance (P < 0.05) of the variables.
The outcome variables in the second experiment were the results from the immunohistochemical identification of VHS and IHN virus in the specimens (1 = positive, 0 = negative). Two repeated measures regression models using PROC GENMOD were assessed2,23 and the tissue specimen was the statistical unit in both. The type III chi-square value was used to test the significance (P < 0.05) of the variables. The interaction terms between the two independent variables were also tested for significance.
One important assumption for traditional statistical methods is that the observations are independent of each other, which is assumed not to be the case in the present study. In a repeated-measures analysis, it is assumed that the observations within each cluster (fish in this case) over time (sampling day) are correlated. In repeated measurements analysis, it is also possible to specify the structure of the correlations among the observations within each cluster.2 In this study, we assumed that the best fitting approach was a first-order autoregressive model (AR1) in which one measurement is correlated with the previous one.
Results
Virus replication in kidney
There was no statistical difference in virus titer in kidney samples for VHS virus at early times after challenge, days 2, 3, 5, 7, and 8 PI, when double-infected fish were compared with single-infected fish (Fig. 1). The same observation was made for single- and double-infected groups of IHN virus (Fig. 2).
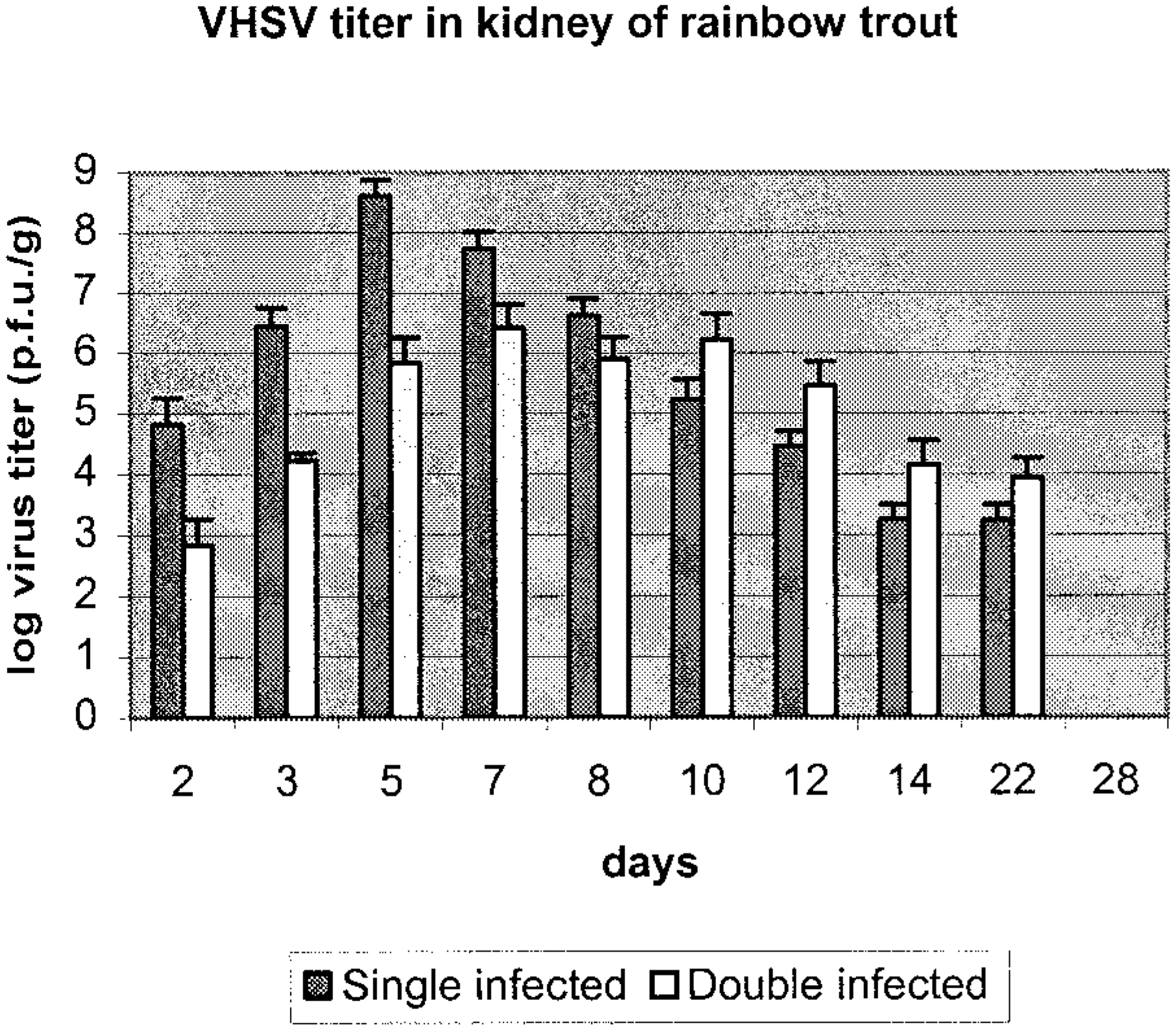
Titer of VHS virus in kidney samples of rainbow trout in single- (solid bar) and double-infected groups (open bar). Data are given as an average of three fish with standard deviation of the mean.
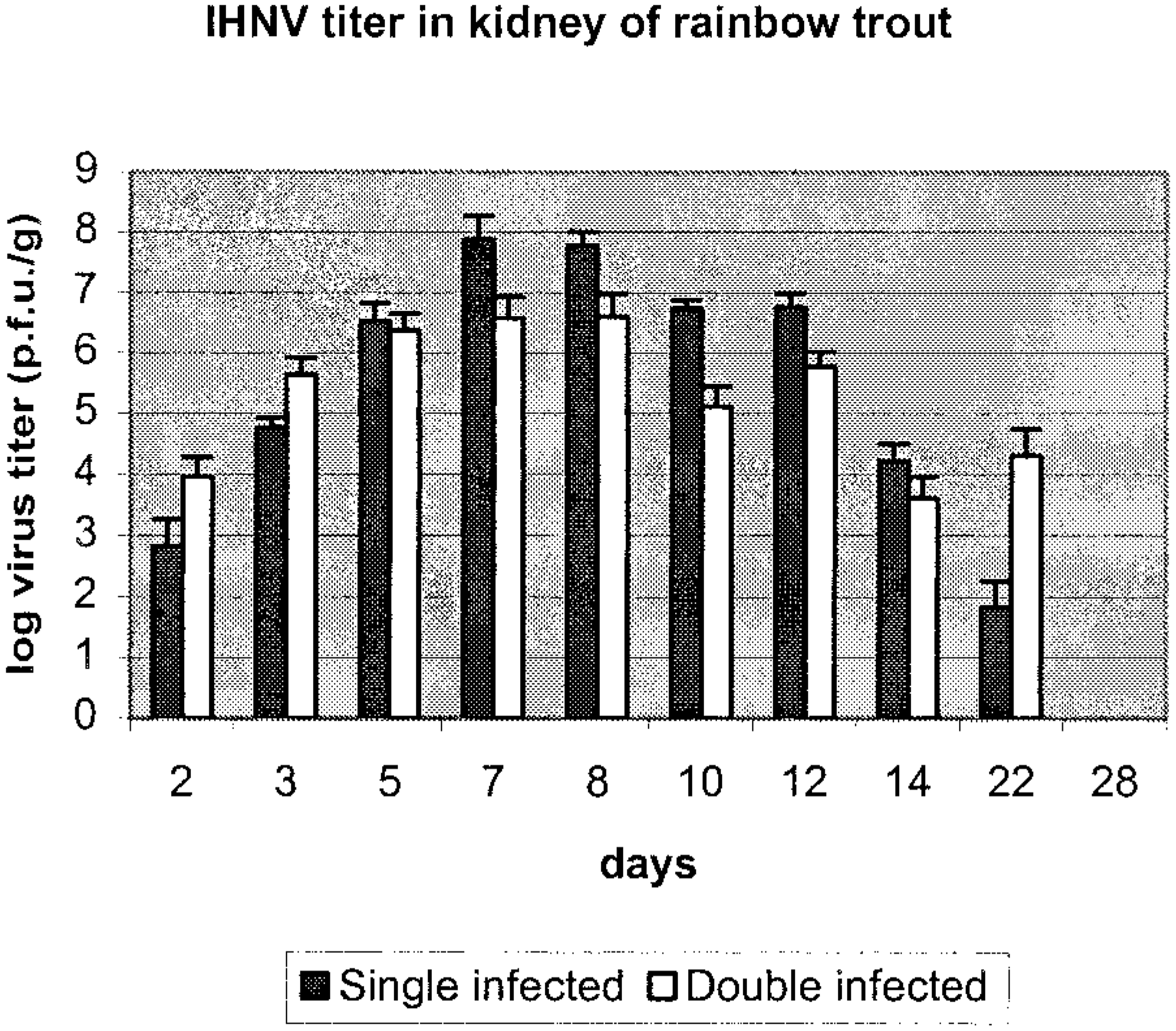
Virus titer of IHN virus in kidney samples of rainbow trout in single- (solid bar) and double-infected groups (open bar). Data are given as an average of three fish with standard deviation of the mean.
Viral distribution and pathologic changes
Group 1—VHS virus infected
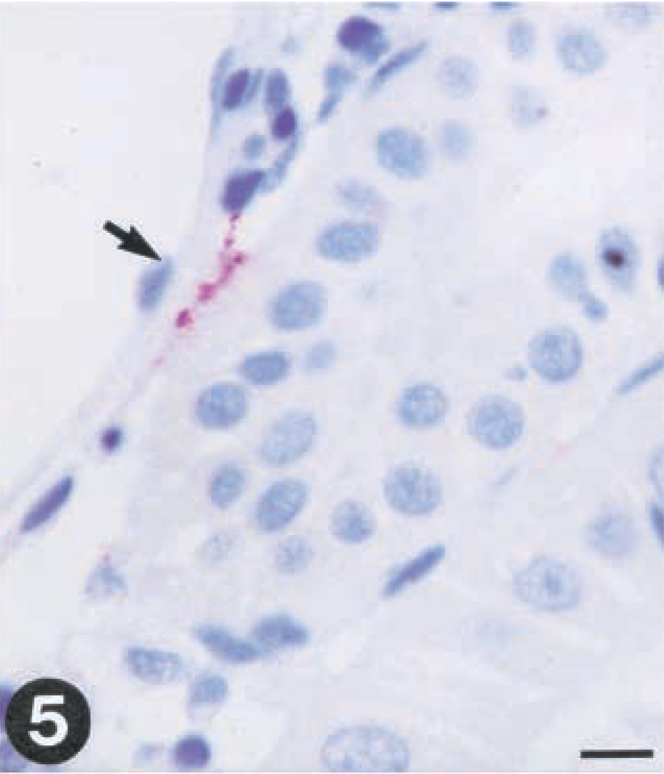
The virus was detected by immunohistochemistry in gill (one out of three fishes) and kidney (two out of three fishes) as early as day 1 postinfection (PI). Only a few epithelial cells of the gills were found positive for virus (Fig. 3). A few endothelial cells of the posterior kidney also exhibited positive reaction for virus at this time (Fig. 5).
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
At day 3 p.i., virus was detected in the endothelial cells lining the vessels of the primary gill arch (Fig. 4). In kidney and spleen, the reaction was multifocal in appearance and mainly located in the cytoplasm of endothelial cells, without any concomitant pathologic changes.
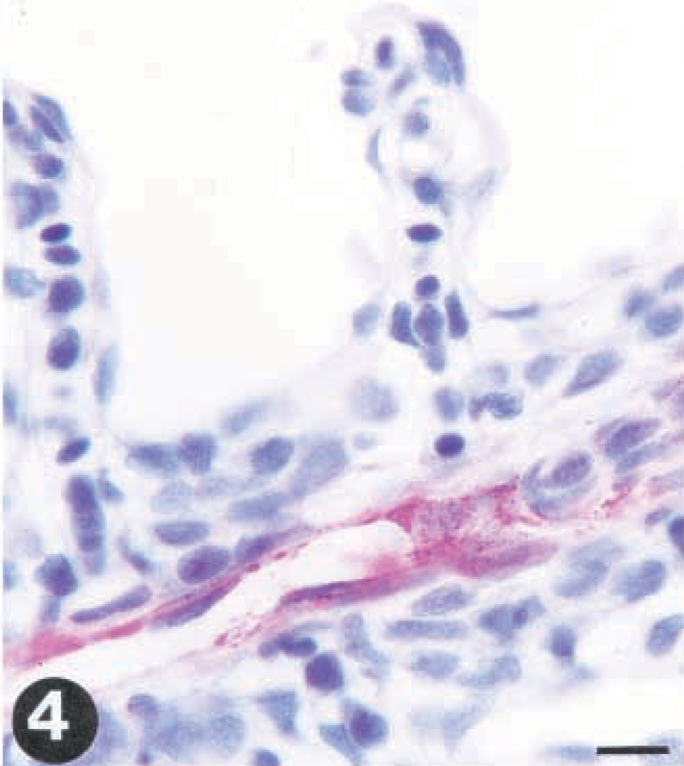
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
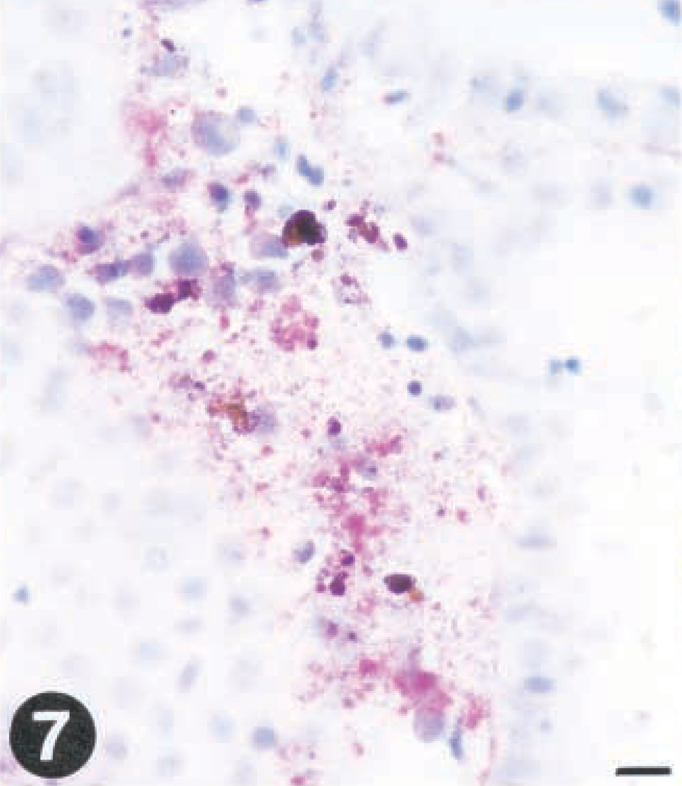
At day 5 PI, all fish sampled were found virus positive, although with some variation between fish regarding which organ was positive (Table 1). Macrophage-like, melanomacrophages, and hematopoietic cells in the interstitial stroma of kidney and spleen were positive and showed early histomorphologic signs of degeneration and necrosis, typically observed as indistinct cell borders and initial fragmentation of the nucleus. At the later stages, the spleen and kidney exhibited focal areas of necrosis with a faint immunohistochemical staining (Fig. 7). VHS virus was not detected in renal tubules of posterior kidney. Severe congestion of the spleen sinusoids was observed at late stages (not shown).
Immunohistochemical identification of VHS virus in tissue of single-infected rainbow trout. Numbers indicate positive fish (out of three examined).
Total of two fish sampled.
The pancreas had focal areas of virus confined to the exocrine tissue at 5 days PI. At later stages, virus was identified in situ and pathologic changes were observed throughout the pancreas, including the endocrine tissue. The liver exhibited a multifocal distribution of virus in hepatocytes from day 5 PI and onward, and virus was detected in association with focal necroses. Similar observations were made for heart samples, and virus was detected as early as 5 days PI. Virus was present in myocardial and endothelial cells of both atrium and ventricle, also associated with single-cell necrosis at day 8 PI. At 12 and 14 days PI, necrotic foci were observed. VHS virus was detected in brain specimens as early as day 5 PI, and virus was present in the neurons and neuroglia cells, usually without concomitant cell degeneration. All sections of the brain were infected without any pattern of cell specificity. In skin specimens, VHS virus was detected in epithelial cells at day 5 PI, without prominent pathologic changes being observed, usually with a few leukocytes of the skin being observed virus positive.
At days 21 and 28, most sampled fish were negative by immunohistochemical staining for VHS virus, and pathologic changes were absent.
Group II—IHN virus infected
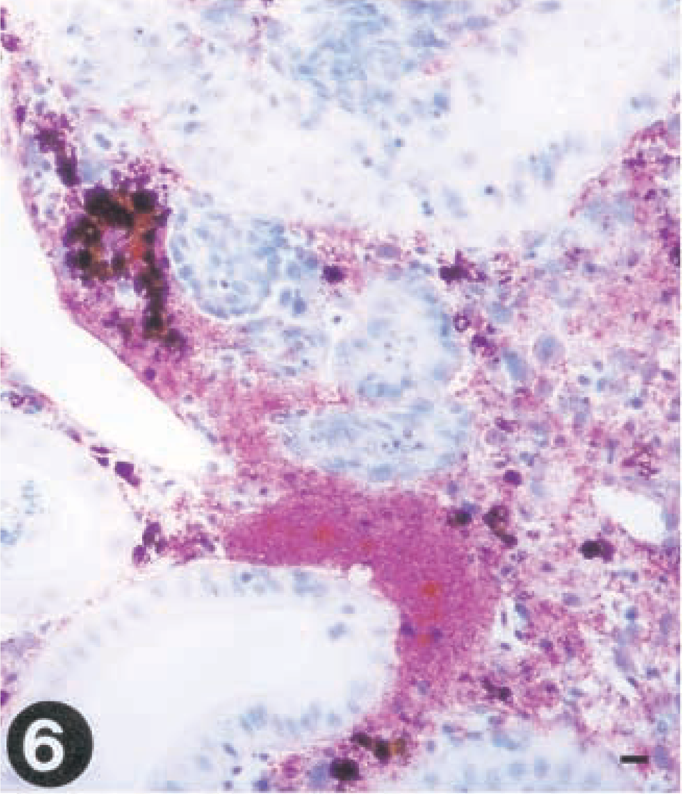
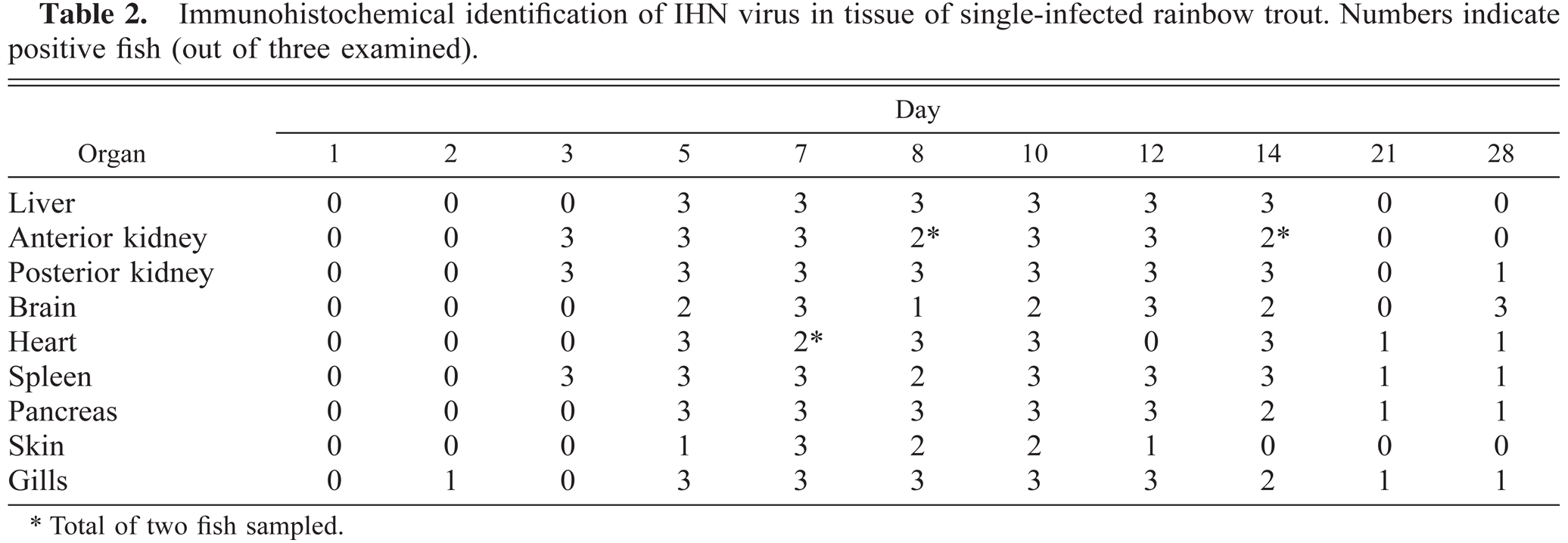
The detection of virus in the different organs and the distribution of virus as revealed by immunohistochemistry in infected fish did, to a large extent, resemble what was found for group I. IHN virus was detected in gills at day 2 and in spleen and kidney within 3 days PI and in liver specimens by day 5 (Table 2). In kidney, early identification of virus was associated with the endothelial cells lining the sinusoids, and the viral localization in the reticulo-endothelial stroma soon spread to the interstitial leukocytes (Fig. 6). Melanomacrophages were also virus positive at early stages of infection (5 days PI). In the spleen, the virus was localized in ellipsoidal macrophages. The viral distribution in skin, pancreas, heart, and brain closely resembled the distribution in single VHS virus-infected fish (Tables 1, 2). Brain samples were found positive from day 5 throughout the remainder of the experiment (Table 2, Fig. 8).
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
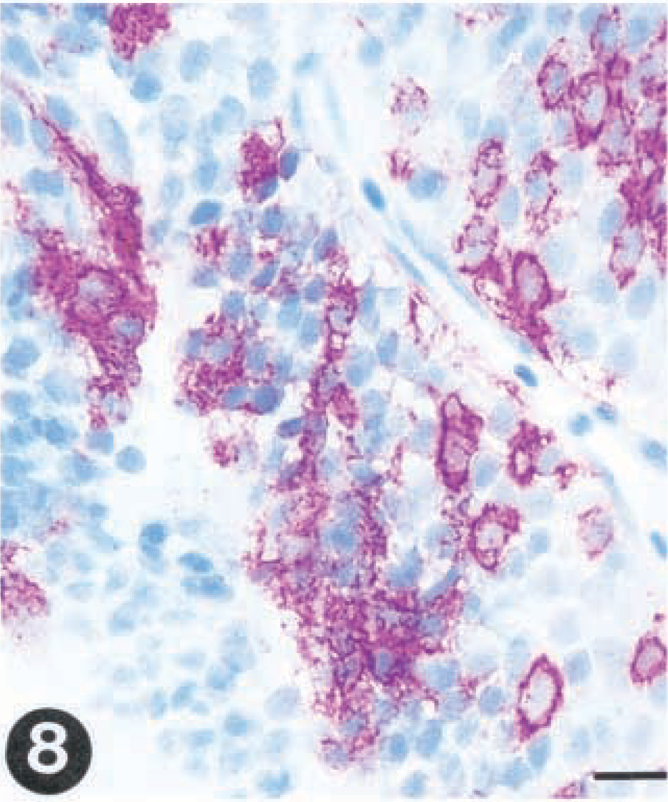
Tissue sections from rainbow trout (Oncorhynchus mykiss) infected with VHS virus or IHN virus. Presence of virus is demonstrated by use of a streptavidin-alkaline phosphatase method using Fast red as a chromagen.
Immunohistochemical identification of IHN virus in tissue of single-infected rainbow trout. Numbers indicate positive fish (out of three examined).
Total of two fish sampled.
Group III—double-infected fish
VHS virus was not detected at early time points in gills in double-infected fish, in contrast with what was found for group I (Table 3). Besides these findings, the distribution of VHS virus in internal organs was similar to what was found in group I.
Immunohistochemical identification of VHS virus in tissue of double-infected rainbow trout. Numbers indicate positive fish (out of three examined).


Total of two fish sampled.
For IHN virus, fewer fish were found positive in double-infected fish compared with group II, reflecting that the distribution of virus was more restricted (Table 2 versus Table 4). The most striking observation was that IHN virus was not detected in brain samples at any time during the experiment (Table 4).
Immunohistochemical identification of IHN virus in tissue of double-infected rainbow trout. Numbers indicate positive fish (out of three examined).
Total of two fish sampled.
By double immunohistochemistry, VHS virus and IHN virus were detected in the same cells in heart (Fig. 9) and liver (Fig. 10) specimens, showing that a single cell can support the simultaneous replication of both viruses.
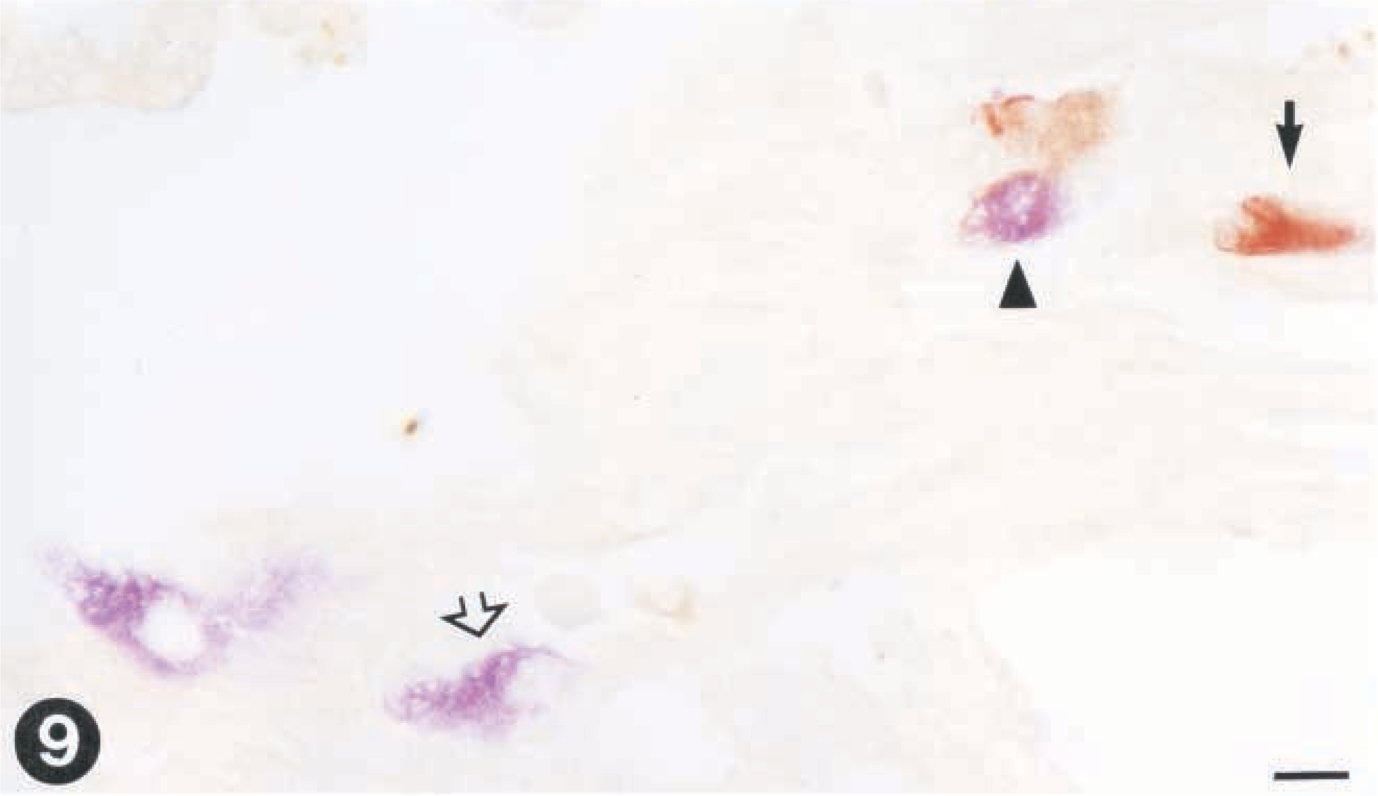
Tissue sections from rainbow trout infected with VHS virus and IHN virus. Virus is demonstrated by use of paired immunohistochemistry.
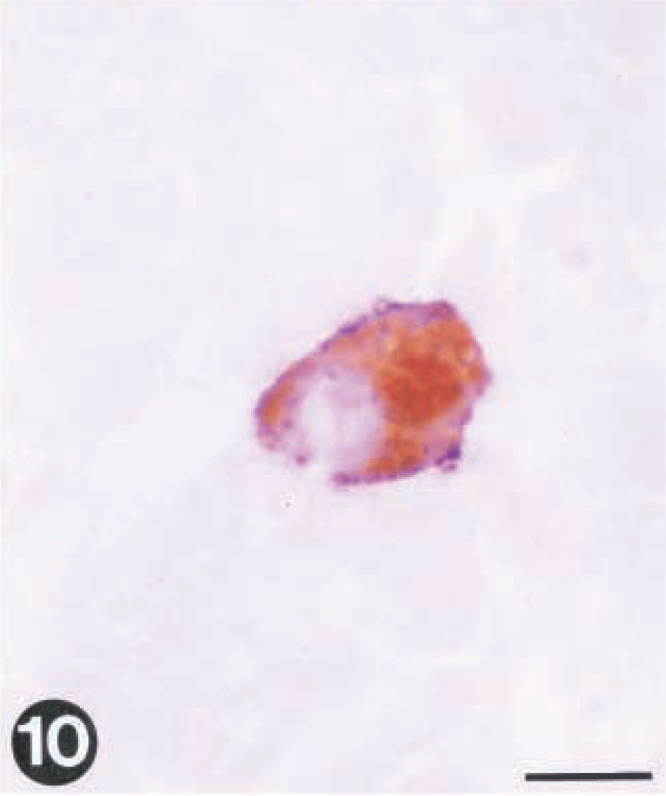
Tissue sections from rainbow trout infected with VHS virus and IHN virus. Virus is demonstrated by use of paired immunohistochemistry.
Statistical analyses
Virus replication in kidney
No statistically significant difference was identified between the two groups (single and double) for each day of sampling.
Immunohistochemistry
The analyzed VHS virus immunohistochemical data showed no significant difference in the OR between double- and single-infected fish.
The analyzed IHN virus immunohistochemical data showed significant differences in the odds ratios when comparing samples from double- and single-infected fish in liver, anterior kidney, posterior kidney, heart, pancreas, skin, and gill. The data demonstrated small differences between single- and double-infected samples in target organs for infection such as kidney (OR = 2.0 and 1.9), spleen (OR = 1.5), and liver (OR = 2.5), while heart (OR = 5.6) and pancreas (OR = 7.6) have a more reduced distribution in double-infected fish. When analyzing immunohistochemical samples from skin and gills, the chance of getting a virus-positive specimen was 12 times higher for single- compared with double-infected fish. Since no IHN virus-positive specimen was found in brain of double-infected fish, no OR value could be estimated.
Discussion
The main conclusion to be drawn from this study is that a simultaneous infection of VHS virus and IHN virus results in both viruses establishing an infection in a susceptible host. Further, double infection results in a more restricted distribution of IHN virus in internal organs; however, a significantly lower virus titer was not detected in kidney. The most striking finding is that, for IHN virus, no virus was detected in the brain in situ in the double-infected group.
Double infection resulted in a more restricted distribution of IHN virus during the acute stage of the infection (days 3–14). The statistical analysis of immunohistochemical findings showed significantly higher OR values for IHN-single compared with IHN-double infected for all organs analyzed except spleen. The difference in OR was less in target organs such as spleen and kidney compared with heart, pancreas, skin, and gills. No significant difference was detected between single and double VHS virus-infected fish (Table 5).
Results from the statistical analysis of immunohistochemical data in a study of single and double infection with VHS and IHN virus.∗
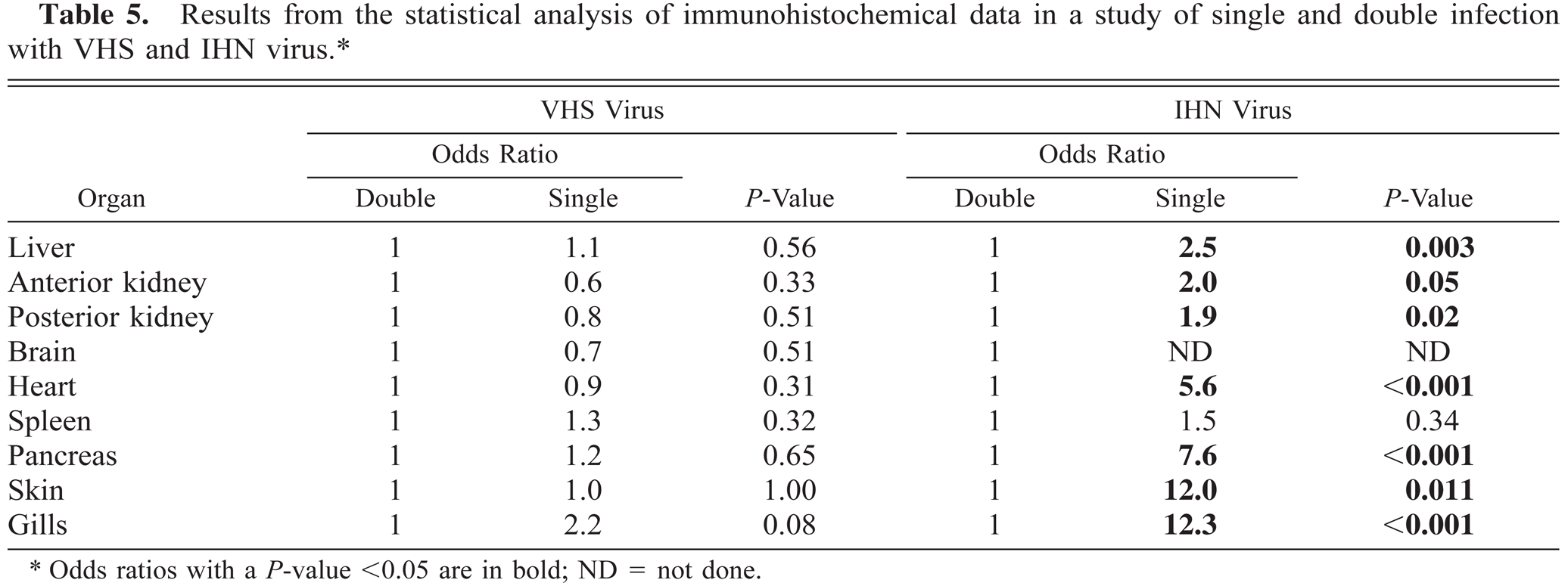
Odds ratios with a P-value <0.05 are in bold; ND = not done.
The virus titer in kidney showed no significant difference between groups, but the peak virus titer at days 5 and 7 PI for VHS virus and day 7 and 8 PI for IHN virus was 1–2 log10 lower for the double-infected group. Further, no IHN virus was detected in situ in brain samples in contrast with what was found for VHS virus in the double-infected group. The reason for this is not known, but one tempting speculation is that the two viruses compete for the same cell receptor or closely related cell receptors on the target cells in the brain tissue, resulting in only VHS virus gaining entrance. These observations are interesting in light of recent studies where it was shown that entry of VHS virus and IHN virus into the salmonid-derived RTG-2 cell line in vitro is associated with fibronectin or a fibronectin complex associated with the cell membrane.4 Moreover, it was also demonstrated that blocking of this receptor with monoclonal antibodies resulted in the cell line becoming resistant not only to VHS virus but also to IHN virus, pointing to the two viruses using the same receptor or receptor complex to gain entry into the cells.4 Specific receptors on neuronal cells of higher vertebrates have been identified for another rhabdovirus, rabies virus.24,25 The mechanisms of host–virus interaction are poorly understood for rhabdovirus infection in fish, and further studies should be pursued to elucidate the mechanism of cell entry, replication of virus, and spread of virus during infection.
It is fairly well documented that the prime port of entry for VHS virus is the gills. This conclusion is corroborated in reports for other susceptible fish species such as kokanee,30 sockeye,5,18 and rainbow trout.20,28 Previous studies of IHN virus infection in rainbow trout also support this finding.32 In the present study, VHS virus (day 1 PI) and IHN virus (day 2 PI) were detected in epithelial cells of the gills, findings that are in concert with previous reports. Others have obtained comparable results employing similar methods,8 describing a short-lived transient IHN virus infection of epithelial cells. At later stages of the infection (days 3–14 PI), VHS virus and IHN virus are primarily associated with endothelial cells and cells surrounding the endothelia in the gills.
The importance of the skin as a port of entry has not been well documented, but there are studies showing that VHS virus and IHN virus are capable of infecting skin tissue in vitro,31 but in vivo documentation is lacking. The results reported herein provide no indication of virus infecting the epithelial layer in the early stage of infection. Virus in skin was primarily detected in fish with a systemic infection.
The target organs for piscine rhabdoviruses have been identified as the hematopoietic tissues of kidney and spleen, described for spring viremia of carp,1 VHS virus,15,16 and IHN virus.33 Our findings are in support of these observations, but more interestingly, it seems that the endothelial cells of the kidney (posterior and anterior part) are the first cells of internal organs to become infected, as early as 1–3 days postexposure. As the infection progressed, a spread of the virus from the reticulo-endothelial stroma to the interstitium was observed, i.e., leukocytes and macrophages/melanomacrophages became infected. The importance of macrophages in early replication of VHS and IHN virus has previously been discussed,6,9 but those studies were mainly based on in vitro data. Results presented in this study and in previous studies using immunohistochemical methods9 show that VHS and IHN virus are detected in macrophages and melanomacrophages. The data suggest that macrophages also support virus replication. However, these findings are in contrast with the observation that it is difficult to infect isolated macrophages under culture conditions (Lorenzen, personal communication; own observations). A previous study of chronically infected rainbow trout without clinical signs of disease also showed macrophages loaded with VHS virus in spleen.12 These observations were made without there being any signs of cell degeneration or necrosis. Thus, the role of macrophages in rhabdovirus infection in rainbow trout needs further scrutiny.
In contrast with what was observed for brain specimens, both VHS and IHN virus were detected in liver, even in the same cell, as revealed by paired immunohistochemistry. Typical lesions characterized as multifocal necroses were observed in liver specimens from day 5 PI in both single- and double-infected groups, and immunohistochemistry showed strong staining in necrotic areas. It seems that the liver is not a prime target organ during rhabdovirus infections, and virus is typically detected later during the infection, first identified at day 5 for both IHN and VHS virus. Another interesting observation is the complete absence of virus infection in endothelial cells lining the sinusoid, concordant with previous studies.11 This is in contrast with what has been found for infectious salmon anemia (ISA) virus.13 One explanation might be that the hepatic endothelial cells lack the receptor for the VHS and IHN viruses. In contrast with VHS and IHN virus, ISA virus is not found in hepatocytes during experimental studies or clinical outbreak of disease.13,22
In conclusion, this study provides data supporting the notion that simultaneous infection with two piscine rhabdoviruses results in some degree of interaction at the cell level, leading to reduced replication of virus in the prime target organ, the kidney. The result is a more restricted distribution of IHN virus during the course of infection. Further studies should be pursued to study the interaction between virus and the host cell at the cellular and subcellular levels to better understand the pathogenesis of the diseases.
Footnotes
Acknowledgements
The authors thank Anita Lauve for help with conducting the infection trial, Ms. Randi Terland with valuable help in immunohistochemistry, and Torbj⊘rn Brudeseth for linguistic advice.
