Abstract
Hepatobiliary disease was diagnosed in eight of 34 genetically unrelated cohabitating pet ferrets (Mustela putorios furo) during a 7-year period. The eight ferrets ranged in age from 5 to 8 years and exhibited chronic cholangiohepatitis coupled with cellular proliferation ranging from hyperplasia to frank neoplasia. Spiral- shaped argyrophilic bacteria were demonstrated in livers of three ferrets, including two with carcinoma. Sequence analysis of a 400-base pair polymerase chain reaction product amplified from DNA derived from fecal bacteria from one ferret demonstrated 98% and 97% similarity to Helicobacter cholecystus and Helicobacter sp. strain 266-11, respectively. The clustering of severe hepatic disease in these cohabitating ferrets suggests a possible infectious etiology. The role of Helicobacter species and other bacteria in hepatitis and/or neoplasia in ferrets requires further study.
Keywords
Hepatic disease in ferrets has been linked with physiological status, diet, toxins, and infectious agents.2,13 Aleutian disease virus (ADV) infection in ferrets causes hepatitis characterized by bile duct hyperplasia and periportal fibrosis.13 Hepatocellular and biliary neoplasms are not common in this species.13,31 Previously, hepatic hemangiosarcomas were diagnosed in a colony of ferrets, suggesting a common environmental etiology; however, no causative agent was identified in that study.7 Here, we describe a spectrum of hepatobiliary disease, including cancer, affecting eight cohabitating pet ferrets (mustela putorius furo). Spiral argyrophilic bacteria were identified in some animals. The continuum of lesions and shared environment suggest the possibility of an infectious etiology.
Materials and Methods
Animals
Eight clinically affected ferrets ranging in age from 5 to 8 years were examined during a 7-year period. These ferrets were privately owned and were part of a colony of 34 animals. Affected animals were genetically unrelated and included two neutered males and six spayed females. All the animals were fed a commercial ferret diet and were kept in a common enclosure with no known exposure to toxins. Water was provided ad libitum. The most common clinical sign was weight loss. Affected, ferrets also exhibited anorexia, lethargy, and diarrhea. Blood was collected during clinical evaluations and immediately prior to euthanasia by intravenous injection of pentobarbital sodium.
Viral serology and polymerase chain reaction
Serum from each ferret was tested at two or more time points for ADV during the study. Counterimmunoelectrophoresis (CIEP; United Vaccines, Harlan Sprague Dawley, Madison, WI) and polymerase chain reaction (PCR) were performed as described previously.11
Microaerobic culture
Fecal and liver samples from ferrets were homogenized with phosphate-buffered saline. Media used for culture included trypticase soy agar with 5% sheep blood (BAP), medium impregnated with trimethoprim, vancomycin, and polymyxin (TVP; Remel Laboratories, Lenexa, KS), and medium impregnated with cefoperazone, vancomycin, and amphotericin B (CVA; Remel). In addition, selective antibiotic medium (ABM) contained the following components: blood agar base No. 2 (Becton Dickinson Microbiology Systems, Sparks, MD), 5% horse blood (Remel), 50 µg/ml amphotericin B, 100 µg/ml vancomycin, 3.3 µg/ml polymyxin B, 200 µg/ml bacitracin, and 10.7 µg/ml nalidixic acid (Sigma, St. Louis, MO).
Approximately 100 µl of the homogenized fecal supernatant or the homogenized liver was applied directly to TVP medium, CVA medium, and ABM. About 0.5 ml of the homogenized fecal supernatant or homogenized liver was filtered through a 0.45-μm (pore size) filter and plated onto BAP. The plates were incubated at 37 C under microaerobic conditions for 2–4 weeks in vented jars containing N2, H2, and CO2 (80:10:10). Bacterial isolates from pure cultures were used for DNA analysis.
DNA extraction, PCR analysis, and sequencing
DNA was extracted from cultured bacterial colonies with InstaGene Matrix (Bio-Rad Laboratories, Hercules, CA). Bacteria were harvested from CVA plates and resuspended in 1 ml double-distilled H2O in microfuge tubes. After centrifugation and removal of the supernatant, 200 µl of InstaGene Matrix was added to the pellet and incubated at 56 C for 30 minutes. The samples were boiled for 10 minutes and centrifuged for 5 minutes at 12,000 rpm. Ten microliters of the supernatant was used for the PCR. PCR and 16S ribosamal RNA (rRNA) sequencing were performed as described previously.15,16
Helicobacter genus-specific primers C97 (GCTATGACGGGTATCC) and C98 (GATTTTACCCCTACACCA) were used to amplify the 400-base pair (bp) fragment for Helicobacter sp. In addition, Campylobacter genus-specific primers C98 and C99 (GCGTGGAGGATGACACCT) were used to amplify genomic DNA of Campylobacter species. Ten microliters of the DNA preparation was added to a 100-µl reaction mixture containing 1X Taq polymerase buffer (as supplied by the manufacturer but supplemented with 1 mol/L MgCl2 to a final concentration of 2.25 mmol/L), 0.5 μm of each of the two primers, 200 μm of each deoxynucleotide, and 200 µg of bovine serum albumin per milliliter. Samples were heated at 94 C for 4 minutes, briefly centrifuged, and cooled to 58 C. Two microliters of Taq polymerase (Roche Molecular Biochemicals, Indianapolis, IN) and 1 µl of polymerase enhancer (Perfect Match; Stratagene, La Jolla, CA) were then added and covered with a layer of 100 µl mineral oil. The following conditions were used for amplification: denaturation at 94 C for 1 minute, annealing at 58 C for 2 minutes, and elongation at 72 C for 2 minutes. A 15-µl aliquot of the sample was electrophoresed on a 1% agarose gel, stained with ethidium bromide, and visualized under ultraviolet light.
A pGEM-T vector (Promega, Madison, WI) was used for cloning the PCR products. The PCR products were purified with a QIAquick PCR purification kit (Qiagen, Valencia, CA) from a low-melting-point agarose gel. Fifty nanograms of purified PCR product was ligated with 50 ng pGEM-T vector at 4 C overnight and used to transfer into JM109-competent cells. Ampicillin plates with X-gal were used to select positive clones. Plasmid DNA was isolated from Escherichia coli using a Qiaprep mini spin kit (Qiagen).
Purified DNA from PCR products was sequenced with an ABI prism cycle-sequencing kit (BigDye Terminator Cycle Sequencing kit with Ampli-Taq DNA polymerase FS; Perkin Elmer Applied Biosystems Division, Foster City, CA). The primers used for sequencing have been previously described.9 Quarter-dye chemistry was used with 80 μm primers and 1.5 µl of PCR product in a final volume of 20 µl. Cycle sequencing was performed with an ABI 9700 DNA sequencer (Perkin Elmer) with 25 cycles of denaturation at 96 C for 10 seconds and annealing and extension at 60 C for 4 minutes. Sequencing reactions were run on an ABI 377 DNA sequencer (Perkin Elmer). The 16S rRNA sequence data were analyzed as previously described using customized RNA software.9 Our database contains over 1,000 sequences obtained in our laboratory and over 500 from GenBank.
Histopathology
Necropsies were performed on all eight ferrets. Selected tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5–6 μm, and stained with hematoxilyn and eosin (HE). Liver sections were also stained with Masson's trichrome and Steiner's and Warthin–Starry silver stains.
Hepatic and portal inflammation were scored from normal (0) to most severe (5) at 40X using the following criteria: 1=minimal to mild inflammation with <15 scattered leukocytes; 2=mild to moderate inflammation with approximately 15–30 scattered leukocytes; 3=moderate inflammation with approximately 30–50 leukocytes, occasionally having formed small aggregates; 4=moderate to severe inflammation with approximately 50–75 leukocytes often in aggregates; 5=severe inflammation with >75 leukocytes in aggregates of nodules.
Results
ADV profile
All 34 ferrets living in this colony were negative for ADV by CIEP and PCR.
Bacterial culture
Bacteria were isolated from fecal samples only and included Helicobacter and Campylobacter species from ferret Nos. 6 and 7, respectively. The identity of the organisms was confirmed by PCR and sequence analysis.
PCR and sequence analyses
PCR analysis of fecal bacterial isolates from ferret No. 6 using gene-specific primers of the 16S RNA gene for the genus Helicobacter yielded a 400-bp fragment. The sequence generated by this PCR was 98% similar to Helicobacter cholecystus and 97% similar to Helicobacter sp. strain 266-11, which have been previously isolated from hamsters and ferrets, respectively.19,20 Limited bacterial growth precluded full sequence determination of the 16S rRNA gene. In addition, gene-specific primers for Campylobacter amplified a 297-bp fragment from a fecal isolate from ferret No. 7. The complete 16S rRNA gene of the Campylobacter isolate was sequenced and identified as Campylobacter mucosalis.
Histopathology
The eight ferrets in this study had variable degrees of hepatobiliary disease (Table 1). All the animals exhibited hepatitis, bile duct hyperplasia, and other proliferative and/or degenerative lesions. Livers from affected ferrets had chronic portal and parenchymal inflammation associated with proliferation of biliary epithelium and bipotential oval cells. Inflammatory lesions centered on and sometimes radiated from portal triads. These lesions consisted of aggregates of various sizes and nodules of lymphocytes, plasma cells, and macrophages with fewer neutrophils and occasional eosinophils (Fig. 1). Oval cell hyperplasia was present adjacent to many areas of significant inflammation (Fig. 2). In many inflamed portal areas, marked duplication and cystic dilation of bile ductules and biliary epithelial hyperplasia and dysplasia were present (Fig. 3). Inflammation often extended beyond the hepatic limiting plate and was in some cases associated with bridging portal fibrosis, as demonstrated by trichrome stain. Parenchymal inflammatory changes were in general less severe and consisted of random small aggregates of lymphocytes, macrophages, and scattered neutrophils throughout lobules. In some areas, mononuclear cells surrounded and replaced individual necrotic (apoptotic) hepatocytes. Additional changes seen in liver sections were variable degrees of sinusoidal dilation, including marked telangiectasia, hepatocellular cytoplasmic hydropic and/or fatty vacuolation, and accumulations of macrophages containing dark brown cytoplasmic pigment (hemosiderin).
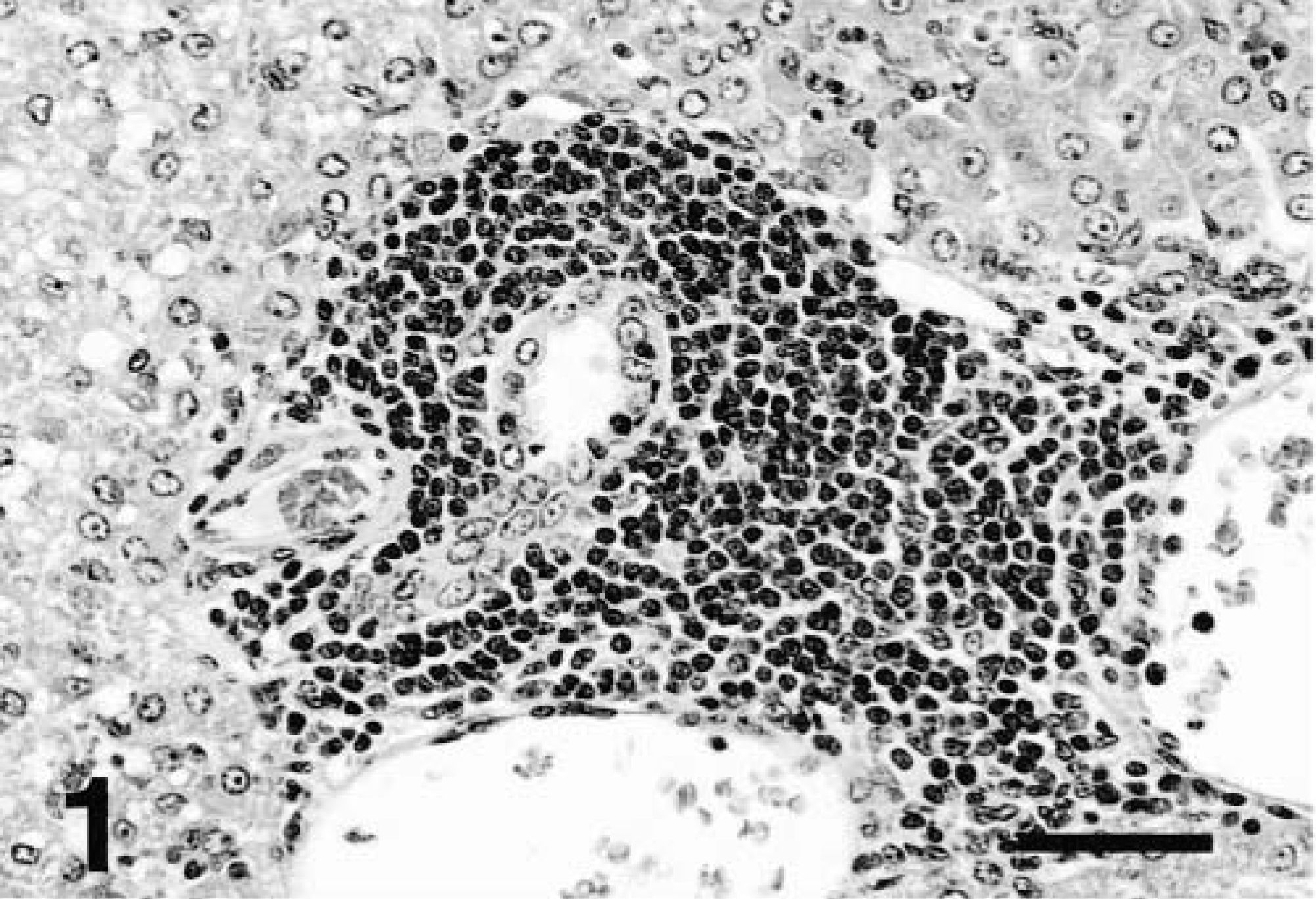
Liver; ferret No. 6. Periportal mononuclear cell infiltrate extending through hepatic plate into adjacent parenchyma. HE. Bar = 125 μm.
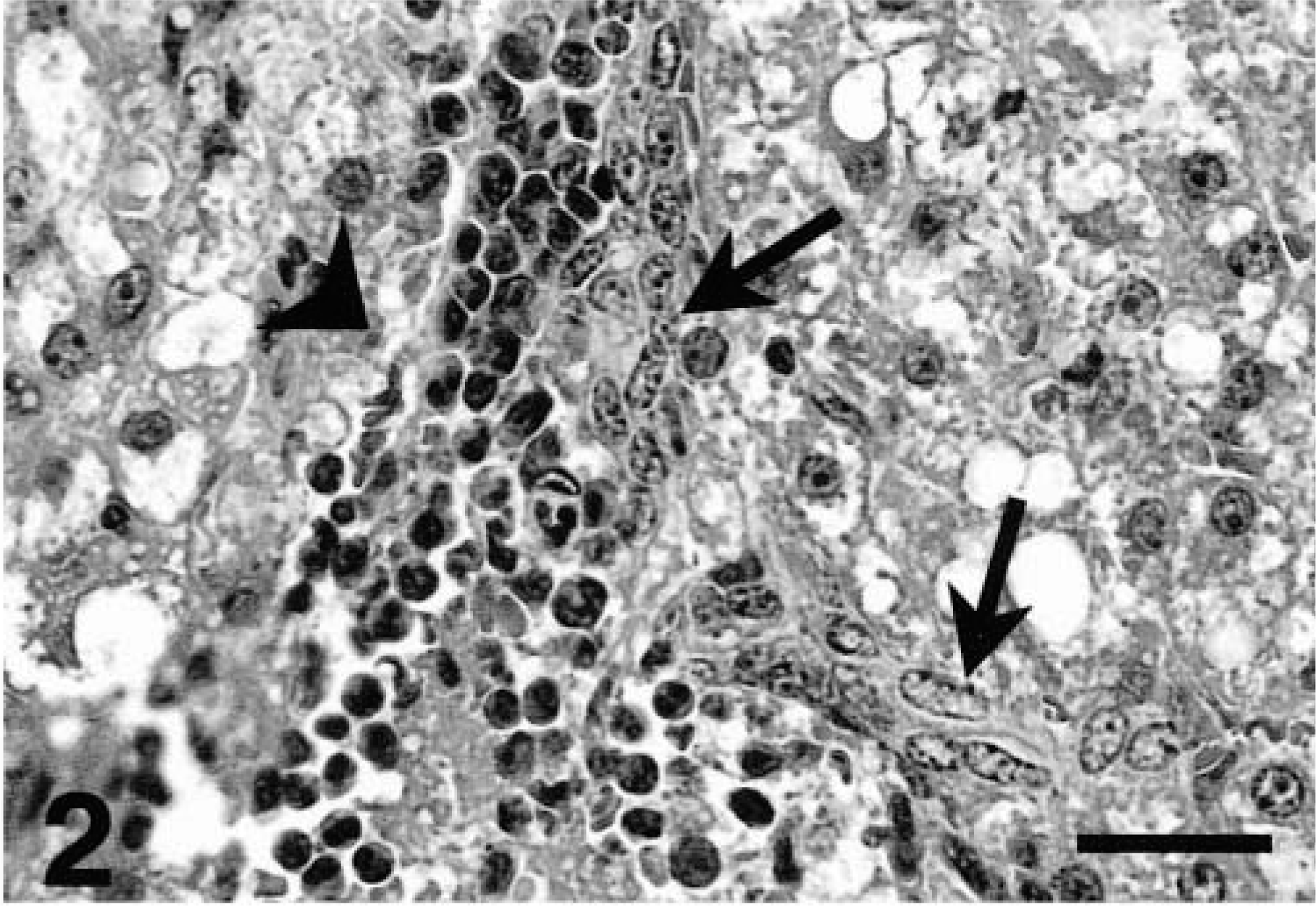
Liver; ferret No. 2. Hyperplastic oval cells (arrows) abutting a focus of mixed inflammatory cells (arrowhead). HE. Bar = 50 μm.
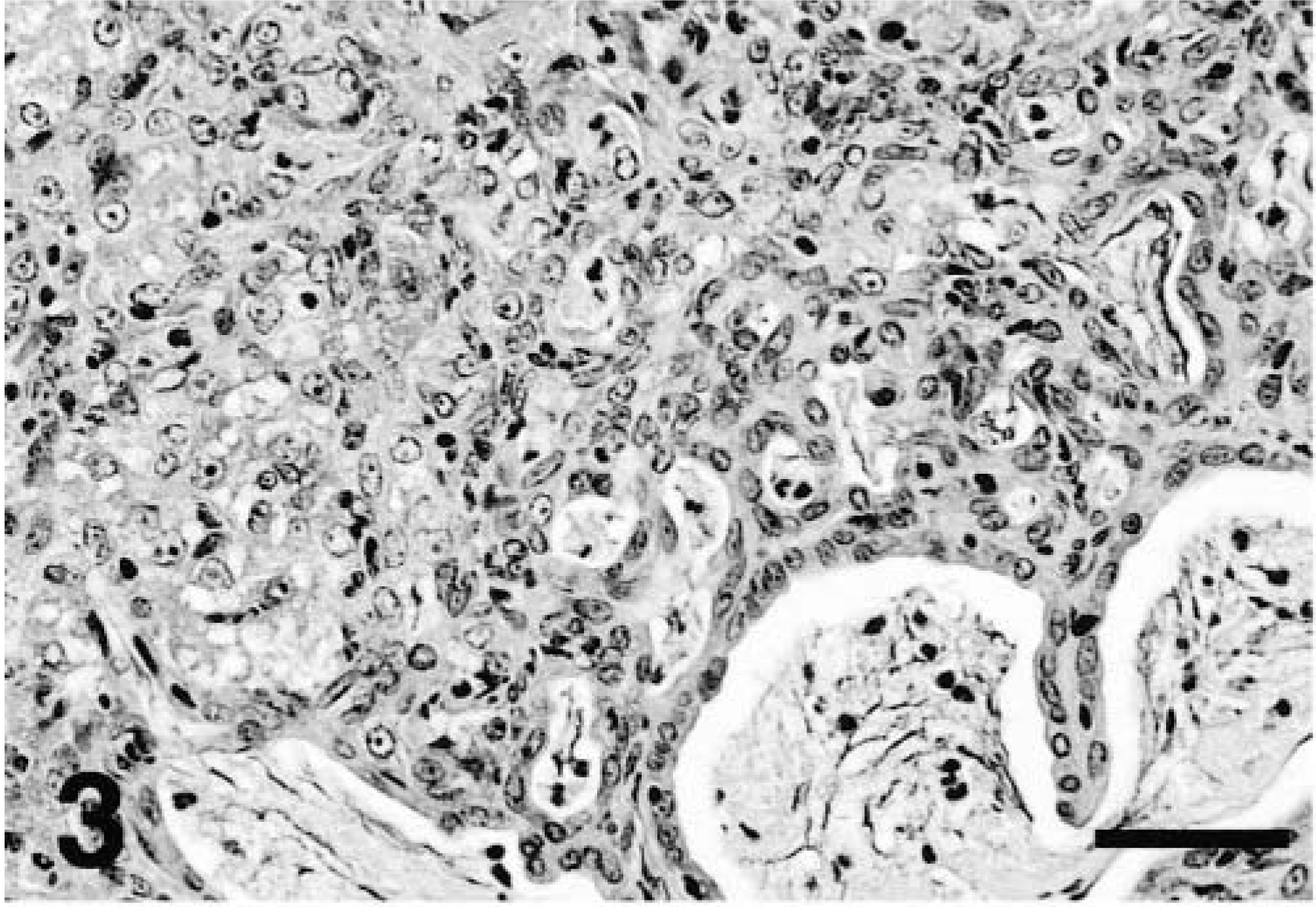
Liver; ferret No. 1. Biliary hyperplasia and dysplasia with cystic dilatation, inspissated bile, and intraluminal degenerate cells. HE. Bar = 125 μm.
Case summaries for eight cohabitating ferrets with primary hepatobiliary pathology.

M/C = castrated male; F/S = spayed female.
Biliary fibrosis, cystic dilatation, telangiectasia, hepatocellular apoptosis and/or nodular hyperplasia.
Isolated by culture (C) or detected with silver stain (S).
Two ferrets had a cholangiocellular cystadenoma consisting of a nonencapsulated, expansile mass of closely spaced ectatic and irregular ducts lined by low cuboidal or flattened simple epithelium (Fig. 4).17 Accumulation of amorphous eosinophilic luminal material was occasionally evident in some multilocular ductular proliferations. Two animals had cholangiocellular carcinoma characterized by formations of indistinct aggregates of polygonal epithelial cells and irregular ductlike structures that had extended into adjacent hepatic cords (Fig. 5). One ferret also had multicentric lymphoblastic lymphoma with hepatic involvement. All ferret livers exhibited chronic cholangiohepatitis and biliary hyperplasia of variable severity with or without additional degenerative and/or neoplastic changes.
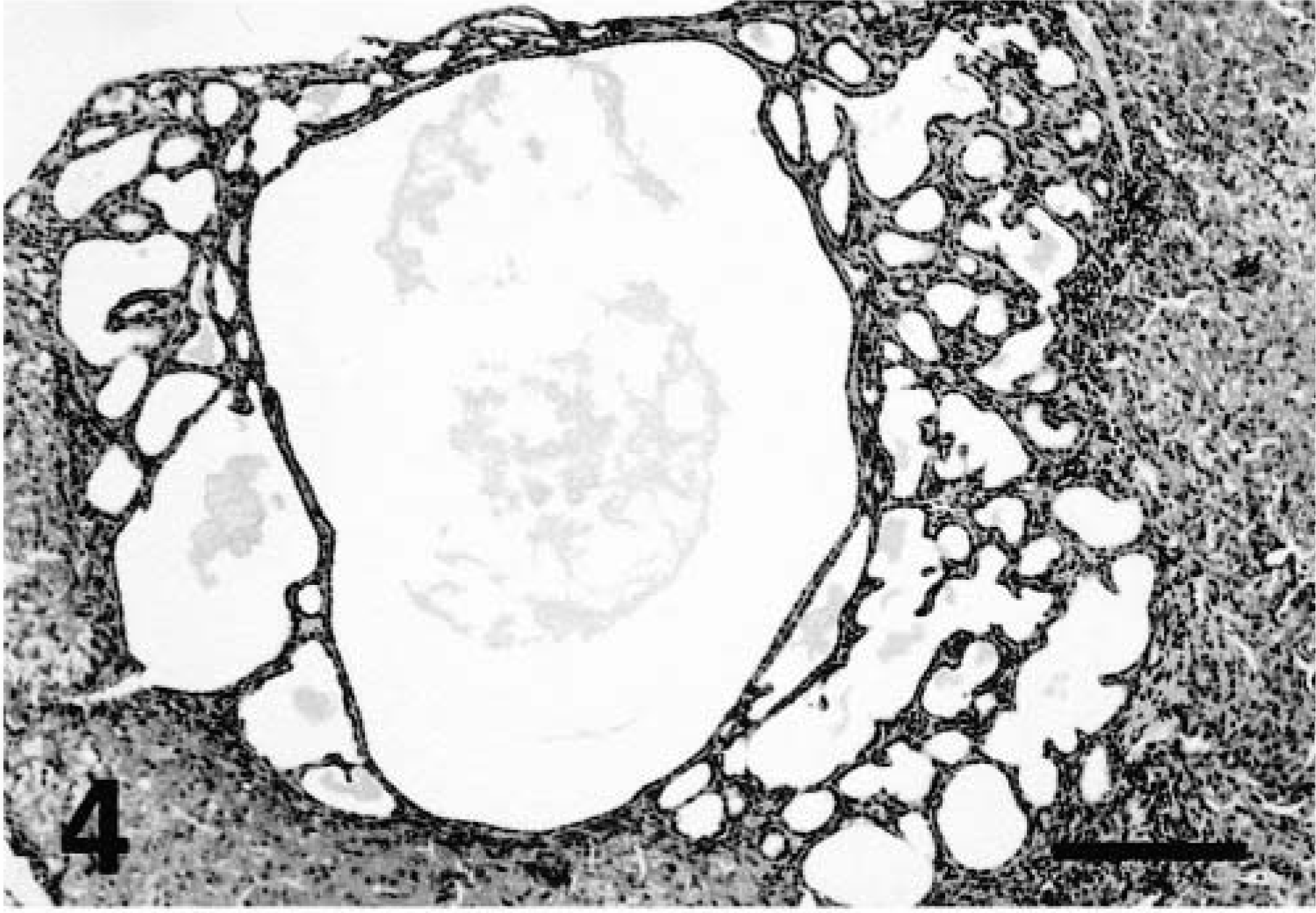
Liver; ferret No. 7. Biliary cystadenoma. Note inflammatory cells lining the well demarcated tumor border. HE. Bar = 500 μm.
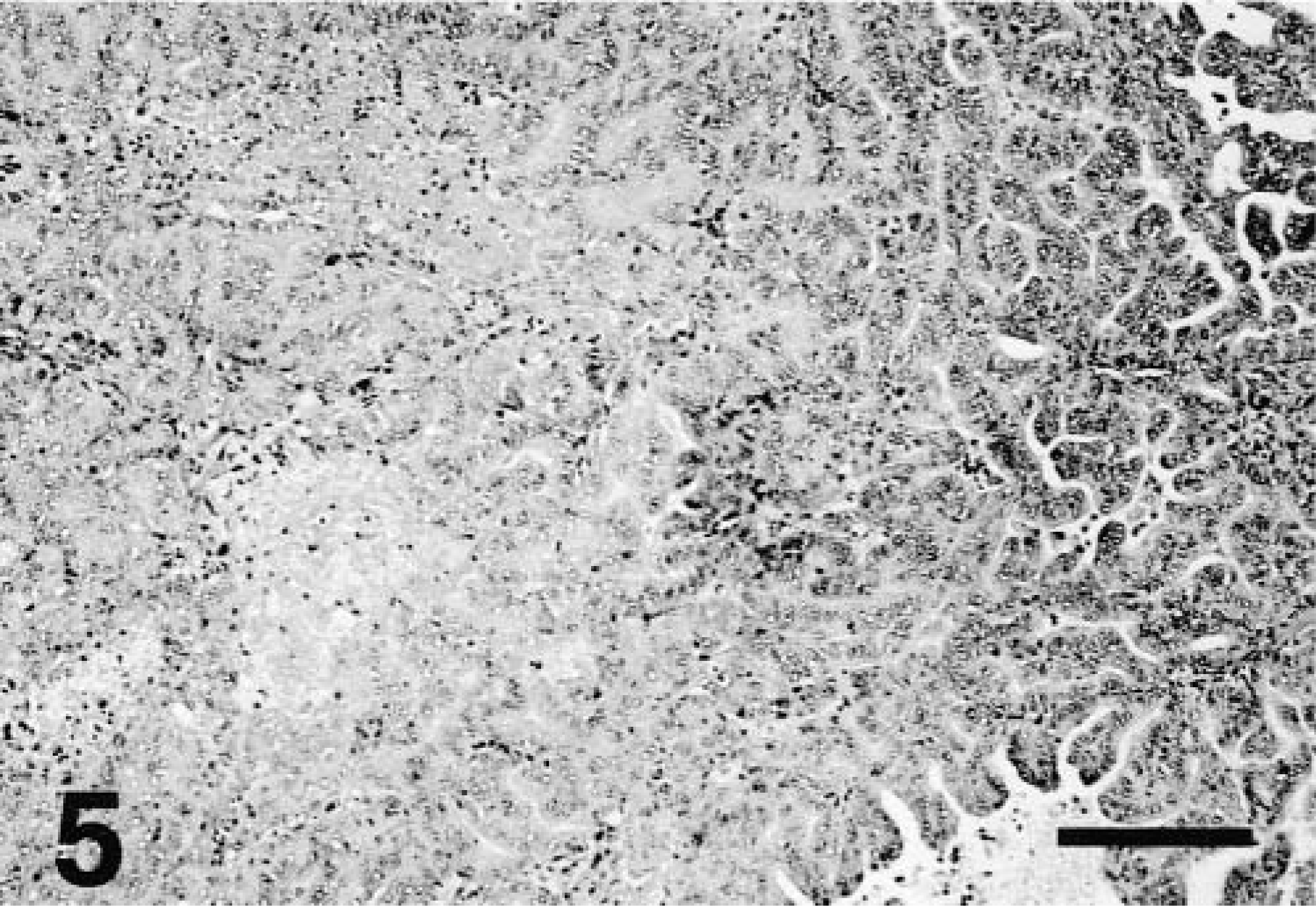
Liver; ferret No. 5. Cholangiocellular carcinoma with solid (left) and papillary (right) organization. HE. Bar = 250 μm.
Curved bacilli and/or spiral-shaped organisms were observed with silver stains in liver sections of three animals (Nos. 1, 3, 5), including those with carcinoma (Fig. 6). The bacteria were pleomorphic but generally were ∼1 × 4–5 μm and were located within hepatocytes or Kupffer cells or extracellularly in bile canaliculi. Bacteria were usually in clusters ranging of 2–5 per 100X field.

Liver; ferret No. 1. Curved and spiral argyrophilic bacteria (arrow) in a bile canaliculus. Warthin-Starry silver stain. Bar = 25 μm.
Discussion
In the present study, we investigated a spectrum of hepatobiliary disease in a colony of genetically unrelated ferrets that shared the same environment. The most common lesions were hepatitis, bile duct hyperplasia, and oval cell hyperplasia. This clustering of severe hepatic disease suggests an infectious etiology. Although ADV may cause hepatic lesions in ferrets, infection with this virus was ruled out by CIEP and PCR.8,13
Chronic cholangiohepatitis and biliary hyperplasia of variable intensity were present in all animals. Oval cell proliferation frequently accompanied severe lesions. Oval cells are bipotential precursors that have the ability to differentiate into either hepatocytes or bile duct epithelial cells.6,34,35 The uniform finding of biliary hyperplasia suggests that oval cells were preferentially differentiating into bile duct epithelial cells, as documented elsewhere.37 Oval cell proliferation in these ferrets reflected the degree of inflammation, as previously observed in other studies in animal models and humans.24,27,32,34 This type of hyperplasia may represent a precancerous stage of hepatobiliary cancer.30,34
There is precedent in other species for neoplastic transformation of chronic infectious and/or inflammatory hepatic disease processes. Cholangiocellular carcinoma is a potential sequel to primary sclerosing cholangitis in humans.3–5,g38 Cholangiocellular carcinoma has also been documented in dogs, cats, horses, and ruminants.1,10,25,28,33 The lesions observed in this group of ferrets shared certain features with human primary sclerosing cholangitis.6,18,22,24,34 Helicobater pylori infection is associated with primary gastric lymphoma and adenocarcinoma in humans.21 Viruses such as those causing hepatitis B and hepatitis C in humans and woodchuck hepatitis virus in woodchucks are known infectious causes of hepatic neoplasia.40,42 The presence of a multicentric lymphoma in one ferret also suggests that these ferrets may have been infected with a putative retrovirus.11,12 Thus, infection with a variety of agents may play a role in dysregulation of cell population homeostasis, ultimately resulting in tumors.
In the present study, the hepatic changes in affected ferrets were similar to the lesions observed in A/JCr, B6C3F1, and AXB recombinant inbred mice infected with Helicobacter hepaticus.14,24 Cholangiohepatitis and inflammatory bowel disease may also be induced by a novel Helicobacter species in A/J and Tac:ICR:HascidfRF mice.39 Recently, in a study assessing the impact of H. hepaticus infection in B6C3F1 mice used in National Toxicology Program carcinogenesis assays, hepatocellular neoplasms and hemangiosarcomas of the liver were consistently increased in control male mice.22 Previously, a high incidence of hemangiomas and hemangiosarcomas was reported in a closed colony of ferrets, suggesting the possibility of an infectious etiology, although a causative agent was not identified.7
The observation of spiral argyrophilic bacteria in liver tissue of three of the affected animals in combination with the molecular identification of a Helicobacter sp. suggests a bacterial etiopathogenesis for the liver lesions comparable to that described in mice infected with H. hepaticus.14,16,19,41
In the present study, C. mucosalis was identified in the feces of one of the affected ferrets. Although this species was initially isolated from pigs with proliferative enteritis, it is now known that that disease in pigs is caused by Lawsonia intracellularis.29,36 Campylobacter species have also been identified by PCR from DNA extracted from bile and biliary epithelium in humans with hepatolithiasis.23 In addition, mice experimentally infected with Campylobacter jejuni develop severe hepatitis.26
The spectrum of lesions described in this study, including variable degrees of inflammation, biliary hyperplasia, and oval cell hyperplasia, may represent a continuum of precancerous stages of hepatobiliary disease with the potential for malignant transformation. More studies are needed to fully characterize ferret hepatobiliary disease and the putative role of spiral bacteria in inflammatory and neoplastic induction.
Footnotes
Acknowledgements
We thank Janice K. Miller for her assistance in this study. This work was supported by National Institutes of Health grants RO1-CA67529 and T32-RR07036.
