Abstract
A 6-y-old neutered male ferret (Mustela putorius furo) was presented because of a 1-mo history of progressive weight loss, chronic cough, and hair loss. On clinical examination, the animal was coughing, slightly depressed, moderately hypothermic, and had bilateral epiphora. Thoracic radiography was suggestive of severe multinodular interstitial pneumonia. Abdominal ultrasound examination revealed hepatosplenomegaly and mesenteric and pancreaticoduodenal lymphadenopathy. Fine-needle aspiration of the pancreaticoduodenal lymph node, followed by routine Romanowsky and Ziehl–Neelsen stains, revealed numerous macrophages containing myriad acid-fast bacilli, leading to identification of mycobacteriosis. Autopsy and histologic examination confirmed the presence of disseminated, poorly defined, acid-fast, bacilli-rich granulomas in the pancreaticoduodenal and mesenteric lymph nodes, intestines, and lungs. Destaining of May-Grünwald/Giemsa–stained slides with alcohol, and then restaining with Ziehl–Neelsen, revealed acid-fast rods and avoided repeat tissue sampling without affecting the Ziehl–Neelsen stain quality and cytologic features. Tissue samples were submitted for a PCR assay targeting the heat shock protein gene (hsp65) and revealed 100% homology with Mycobacterium genavense. We emphasize the use of special stains and PCR for identification of this potential zoonotic agent.
Mycobacterium genavense is a non-tuberculous, slow-growing, acid-fast bacterium first reported in 1990. 7 This emerging opportunistic bacterium is responsible for disseminated diseases in immunocompromised human patients, 2 in many species of birds, 5 and in an increasing number of domestic animals including ferrets.9,11,19 Fever, diarrhea, weakness, weight loss, hepatosplenomegaly, and lymphadenopathy are the usual clinical findings in humans and animals. Involvement of the lung, skin, and kidneys is also reported.2,19 We report herein the postmortem, cytologic, and histopathologic description of disseminated M. genavense infection in a domestic ferret (Mustela putorius furo) in Europe.
A 6-y-old male neutered ferret was presented with a 1-mo history of progressive weight loss, chronic cough, and hair loss. The cough had been treated with unspecified doses of sulfamethoxazole and prednisolone for 1 wk. The ferret was slightly depressed, hypothermic (rectal temperature of 36.2°C), had a body condition score of 2 of 5, and had bilateral serous epiphora. A dry “barking” cough was easily induced by tracheal palpation. A blood sample and radiographs were obtained under general anesthesia. The complete blood count was within normal reference intervals (RIs); serum biochemistry analysis revealed mild hypoglycemia (0.52 g/L; RI: 0.54–1.44 g/L). 6 Other biochemical parameters were within normal RIs. Radiographs showed multifocal peribronchial interstitial opacification throughout the lung, consistent with severe multifocal interstitial pneumonia. Abdominal ultrasound examination revealed a mass in the pancreaticoduodenal area. We suspected a pancreatic mass or pancreaticoduodenal lymphadenomegaly.
Fine-needle aspiration of the pancreaticoduodenal mass was performed, and smears were stained with routine May-Grünwald/Giemsa (MGG). Cytologic examination revealed numerous epithelioid macrophages admixed with small lymphocytes and a few binucleate cells containing myriad intracytoplasmic, 2 × 0.5 µm, negative-staining rods. Subsequently, mycobacteriosis was highly suspected. One of the MGG-stained slides was dipped in alcohol until destained, and then restained with Ziehl–Neelsen (ZN) stain. Numerous acid-fast bacilli within macrophages supported a tentative diagnosis of mycobacterial lymphadenitis (Fig. 1). Given the poor prognosis and condition of the animal, the owners elected euthanasia.
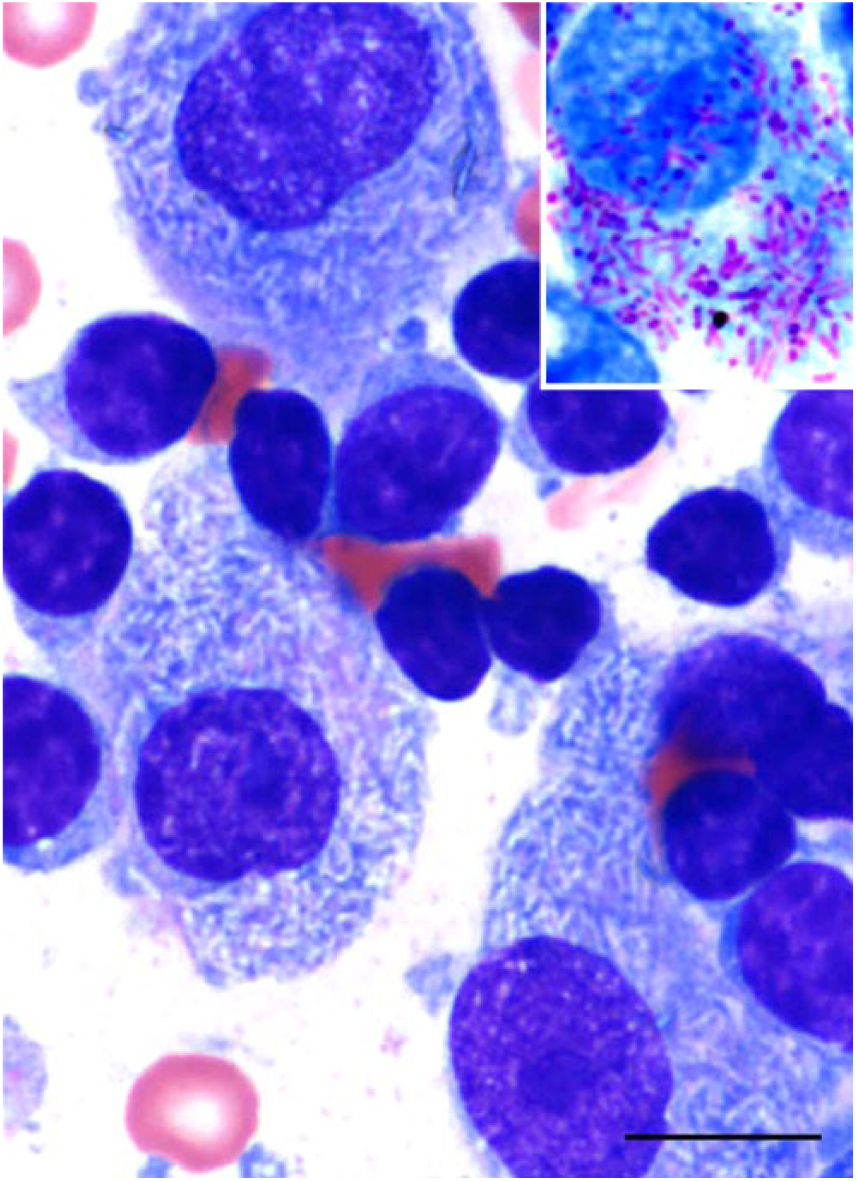
Cytologic smear of a pancreaticoduodenal mass in a ferret with Mycobacterium genavense infection, showing macrophages, multinucleate cells, lymphocytes, and myriad intracytoplasmic, negative-staining 2 × 0.5 µm rods. May-Grünwald/Giemsa stain. Bar = 20 µm. Inset: intracellular rods are acid-fast positive. Ziehl–Neelsen stain.
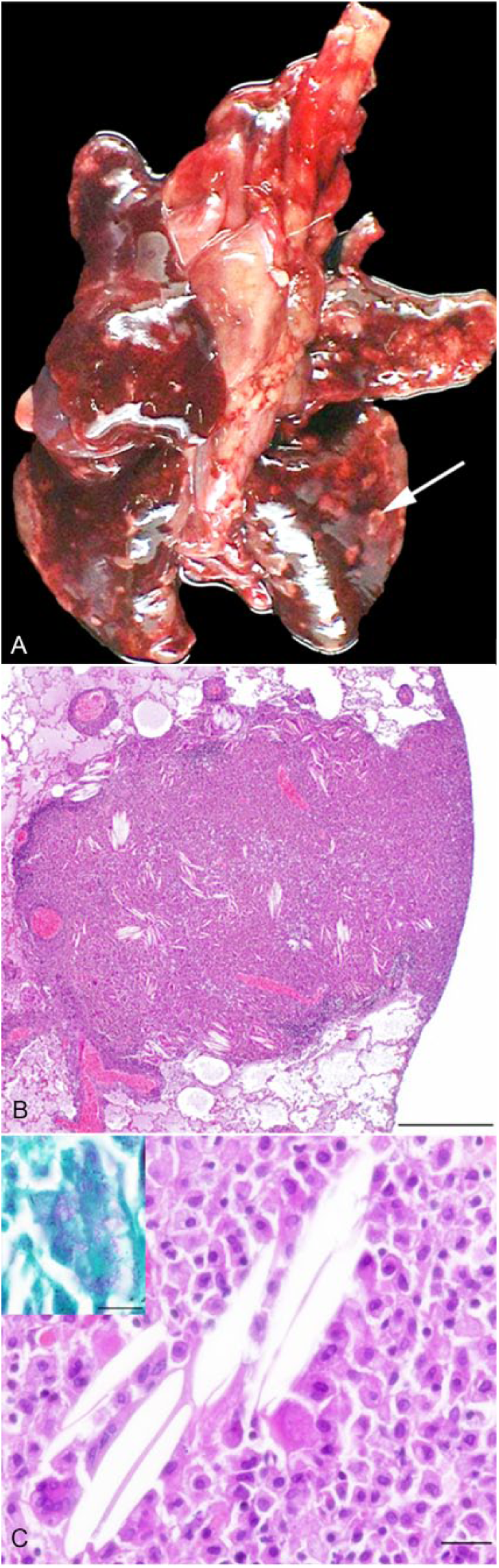
At postmortem examination, the pancreaticoduodenal and mesenteric lymph nodes were severely and moderately enlarged, respectively. The lungs had disseminated tan-white, firm 0.5–1 cm nodules (Fig. 2A). Diffuse splenomegaly and hepatomegaly were noted, and the small intestinal mucosa was moderately thickened. Representative samples from the pancreaticoduodenal and mesenteric lymph nodes, pancreas, liver, intestine, spleen, lung, and kidney were fixed in 10% neutral-buffered formalin and processed routinely for histologic examination. Microscopically, multifocal-to-coalescing poorly delineated granulomas containing numerous epithelioid and foamy macrophages were seen. Myriad intracytoplasmic, ZN-positive, rod-shaped bacteria were noted in all examined organs except the kidneys (Figs. 2B, 2C). In the lung, disseminated granulomas were observed and were often organized around blood vessels. Granulomas often contained acyclic (cholesterol) clefts surrounded by numerous multinucleate giant cells, a cuff of epithelioid macrophages, and small lymphocytes. Apart from rare macrophage aggregates in airway lumens, bronchi and bronchioles were essentially normal. In addition, the pulmonary parenchyma had moderate emphysema and edema. The liver had moderate portal lymphocytic infiltration, with rare macrophages, and diffuse moderate Kupffer cell hyperplasia. Rare ZN-positive bacilli were observed in macrophages. The spleen had marked extramedullary hematopoiesis.
Representative frozen samples of lung, spleen, liver, and tracheobronchial lymph node were submitted for PCR analysis to the French Agency for Food, Environmental and Occupational Health & Safety (ANSES; Maisons-Alfort, France), which is the national reference laboratory for identification of tuberculosis in animals. Briefly, tissues were processed as follows: after mechanical lysis of tissue, DNA was extracted on a column (QIAamp DNA mini kit, Qiagen, Hilden, Germany) according to the manufacturer’s instructions. 3 Then, 5 μL of the extracted DNA was submitted to PCR using primers targeting the 65-kDa heat shock protein gene (hsp65=groEL2 primers, Eurofins Genomics, Les Ulis, France; Tb11 [5′-ACCAACGATGGTGTGTCCAT-3′] and Tb12 [5′-CTTGTCGAACCGCATACCCT-3′]). 18 The sequenced PCR products were compared to the GenBank/EMBL/DDBJ databases using the Basic Local Alignment Search Tool (BLAST) and revealed 100% homology with M. genavense in all tissues tested.
In domestic mammals, infection caused by M. genavense has been reported in a young dog, a feline immunodeficiency virus (FIV)-positive cat, a dwarf rabbit, 2 ferrets, and a chinchilla.8–12 Ferrets are particularly susceptible to a wide range of mycobacteria, among which M. bovis and M. avium seem to be the most commonly involved. 4
Our case provides a cytologic, macroscopic, and microscopic description for this pathogen in ferrets. M. genavense affects the same organs affected by other non-tuberculous mycobacteria: lung, mesenteric lymph node, liver, spleen, and intestinal tract. 14 However, unlike the previous report of M. genavense in ferrets, our case did not have granulomatous conjunctival lesions. 11 Typical nonspecific histologic lesions caused by M. genavense include ill-defined coalescing granulomas composed of foamy and epithelioid macrophages and multinucleate giant cells filled with numerous intracytoplasmic acid-fast bacilli, and extensive peripheral interstitial fibrosis.5,9,11,13,19 The ferret in our case showed the same histologic pattern in the lung, intestines, mesenteric and pancreaticoduodenal lymph nodes, and spleen. However, the involvement of both the digestive tract and lung prevents us from determining the primary route of infection.
M. genavense is an emerging opportunistic pathogen seen with increasing frequency in immunocompromised humans and should be considered as a potential zoonotic agent, in view of epidemiologic reports of animal-to-human transmission of atypical mycobacteria.1,17 Owners should be warned of the potential zoonotic risks when M. genavense infection is suspected or identified in pets.
M. genavense is a fastidious, slow-growing bacterium that is likely under-detected in veterinary medicine given the intrinsic challenges of detecting disease caused by Mycobacterium spp. Under-detection is also likely a result of its similarity in clinical signs, imaging, and autopsy findings to 2 other diseases of the ferret: lymphoma, and systemic coronavirus-associated disease. In addition, the histologic appearance of multifocal pulmonary granulomas with foamy macrophages and cholesterol clefts may be mistaken for endogenous lipid pneumonia. Lipid pneumonia can be an incidental finding, but also has been reported in ferrets with severe respiratory disease. 16 However, endogenous lipid pneumonia is generally subpleural, in contrast to the perivascular distribution in our case.
Cytology was an important aid in identifying the etiology in our case. In multibacillary lesions, fine-needle aspiration of a lymph node or a granuloma, followed by ZN or Fite–Faraco staining, enables rapid, minimally invasive identification of acid-fast bacilli in macrophages. In our case, all cytologic smears were initially stained with MGG. After destaining with alcohol and restaining with ZN, acid-fast rods were identified, which avoided repeat sampling. In our experience, this technique yields results and staining intensity comparable to direct acid-fast staining. Moreover, destaining MGG-stained slides did not affect the ZN stain quality or cytologic features.
PCR using the hsp65 gene is reported as a rapid, sensitive, and specific method to identify slow-growing mycobacteria. 18 PCR using the hsp65 gene can distinguish closely related mycobacterial species. 15 Identification of M. genavense with molecular characterization excluded other less likely intracellular acid-fast mycobacteria and raised awareness of the potential zoonotic risk.
Most of the human cases of M. genavense infection have been associated with immune system impairment, primarily as a result of HIV infection, but have also been seen in transplant recipients, lymphoproliferative malignancies, and other disorders resulting from immunosuppressive therapy.2,17 Evidence of an impaired immune system is usually not found in animal infections with M. genavense, but rare cases have been reported such as a cat with FIV infection. 8 Although no virus has been reported to cause immunodeficiency in ferrets, some clinicians have speculated that the occurrence of cryptococcosis, lymphoma, and disseminated mycobacteriosis in ferrets suggests the existence of such a virus. 9
Footnotes
Acknowledgements
We thank the histology laboratory staff at the Ecole Nationale Vétérinaire d’Alfort (S Joubert, JL Servely, J Biet, and P Wattier) for histologic and cytologic processing of samples; Dr. C Pignon, D Laniesse, and Dr. F Stambouli for the clinical data; and Dr. T Donnelly for revising the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
