Abstract
Fourteen 4- to 18-month-old vaccinated Greyhounds (10 males, 4 females) from three kennels in southern Ireland presented over a 2-year period with acute or insidious onset neurological signs. Head tilting, ataxia, recumbency, circling, and blindness were commonly observed, and animals were dull, dehydrated, and had lost weight. Hematologic and biochemical parameters reflected dehydration but were otherwise unremarkable. Microscopic examination revealed severe diffuse and focal gliosis and gemistocytosis accompanied by mononuclear cell perivascular cuffing in caudate nucleus and cortical gray matter of the cerebrum and in the periventricular gray matter of the anterior brainstem. Milder lesions were noted in the caudal brainstem, cranial spinal cord, and in the molecular layer of the cerebellum. This was accompanied by a lymphocyte and plasma cell infiltration of the cerebral and cerebellar meninges. Demyelination, neuropil necrosis, neuronophagia, and vasculitis were not observed. No inclusion bodies, fungi, or protozoal cysts were seen. Additional serologic and molecular pathology tests also failed to determine a cause, suggesting that these cases may represent a previously undiagnosed condition in the dog.
Inflammatory conditions of the canine central nervous system (CNS) are caused by many agents including bacteria, viruses, parasites, fungi, and saprophytic algae. 26 Additional inflammatory CNS lesions in which agents have not been implicated include those of immune-mediated origin, granulomatous meningoencephalitis (GME), hydrocephalus with periventricular encephalitis, and breed-associated encephalitis. 31 Specific breed-associated inflammatory conditions have been recognized since the early 1980s and include meningoencephalomyelitis in Pointer dogs, Pug dog encephalitis, and necrotizing encephalitis in Yorkshire Terriers. 4,5,30 The hallmarks of brain inflammation are perivascular cuffing and gliosis, and these reactions may be accompanied by neuronophagia, demyelination, and parenchymal necrosis. 15,25 The nature of the perivascular cuffing reaction often provides clues to the possible etiologic agent, with nonsuppurative reactions rich in lymphocytes and plasma cells being more typical of viral infection in the first instance but also suggestive of protozoal, rickettsial, or spirochaete agent involvement. 25
A novel neurologic syndrome in young Greyhounds in Ireland that is characterized by a severe nonsuppurative meningoencephalitis centering on the cerebrum and periventricular gray matter of the anterior brainstem is reported in this paper. Clinical, pathologic, and serologic findings have failed to implicate many of the conventional agents and would suggest that this may be a new syndrome.
Materials and Methods
Animals
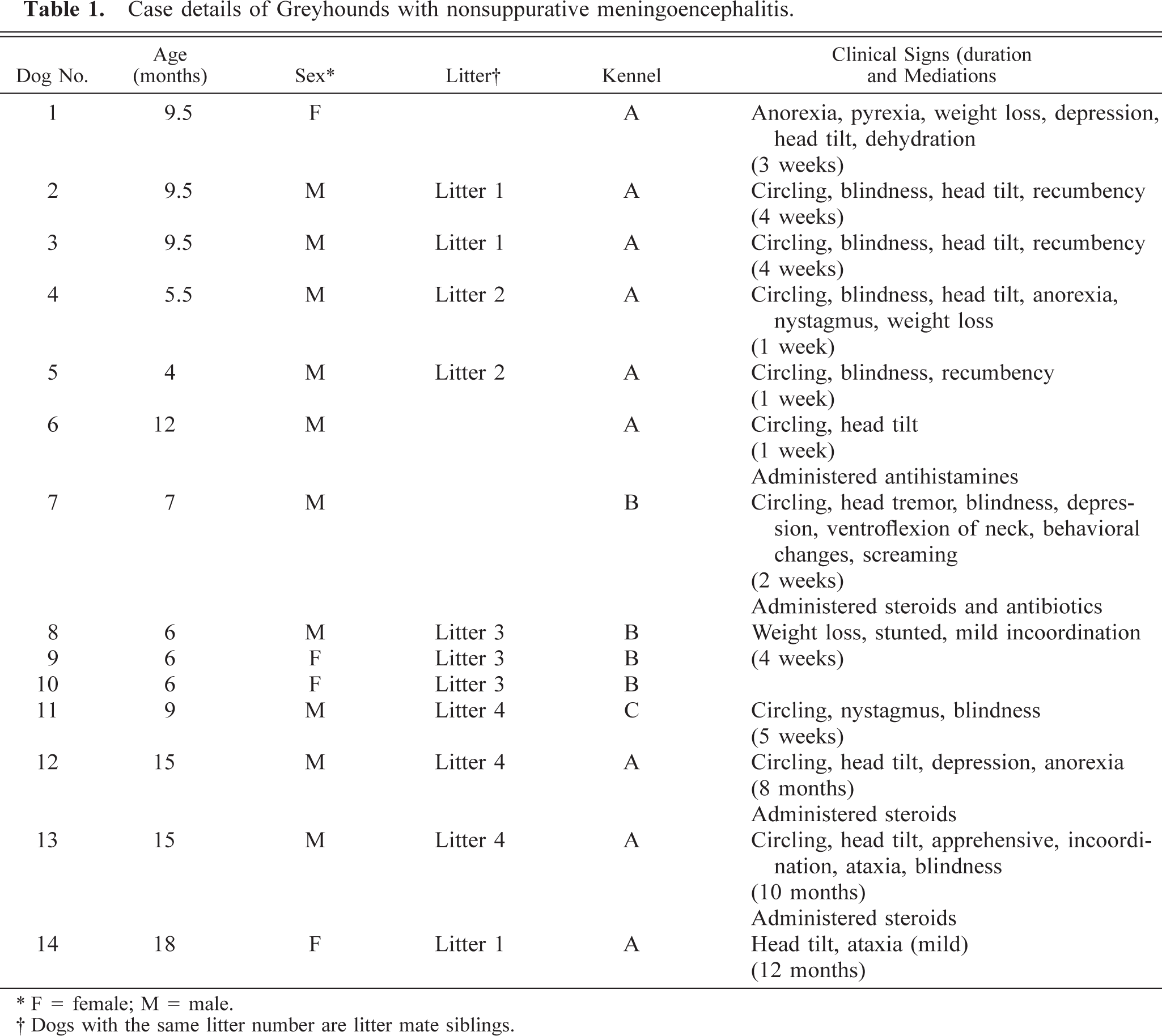
Fourteen Greyhounds were examined (Table 1). Eight Greyhounds (dog Nos. 1–4, 7, and 12–14) were submitted to the Small Animal Teaching Hospital at The Faculty of Veterinary Medicine, University College Dublin for clinical and postmortem examinations. Formalin-fixed brain tissue was submitted from five Greyhounds (dog Nos. 6 and 8–11). Dog No. 5 was submitted to the Department of Agriculture and Food Research Laboratory for postmortem examination. Nine Greyhounds (dog Nos. 1–6 and 12–14) were from one kennel (kennel A) in central Ireland that on average contained 300 dogs. Four animals (dog Nos. 7–10) were submitted from a second kennel (kennel B) that held 150 dogs and was located approximately 15 miles southeast of kennel A. One Greyhound (dog No. 11) was from kennel C, which contained 40 dogs and was located on the southeastern coast of Ireland approximately 45 miles south of kennel A and 35 miles south of kennel B. All kennels were racing and breeding establishments and interaction of personnel, vehicles, and animals would have occurred at race meetings. In addition, prominent contact and transfer of animals occurred between kennels A and B and racing establishments in the state of Texas (USA). Detailed histories and clinical examinations, including neurologic assessments, were taken.
Case details of Greyhounds with nonsuppurative meningoencephalitis.
∗ F = female; M = male.
† Dogs with the same litter number are litter mate siblings.
Postmortem examinations and histopathology
Brains were collected for histologic examination from all animals. Spinal cord was sampled from eight animals receiving full postmortem examinations. Representative samples of cardiac and skeletal muscle, eye, kidneys, bladder, liver, pancreas, stomach, intestines, lungs, lymph nodes, spleen, tonsils, adrenal glands, and thyroid tissues were also obtained. Specimens were fixed in 10% neutral buffered formalin and processed for paraffin embedding. Sections of approximately 4 µm were cut and stained with hematoxylin and eosin. Standard sections were processed from brain and cord to allow examinations of cerebrum, cerebellum, brainstem, medulla, pons, choroid plexus, midcervical cord, caudal thoracic cord, cranial lumbar cord, dorsal root ganglia, meningeal tissue, and optic nerves within the optic chiasm. Portions of kidney and liver were sampled for lead estimations in one animal (dog No. 5).
Serology
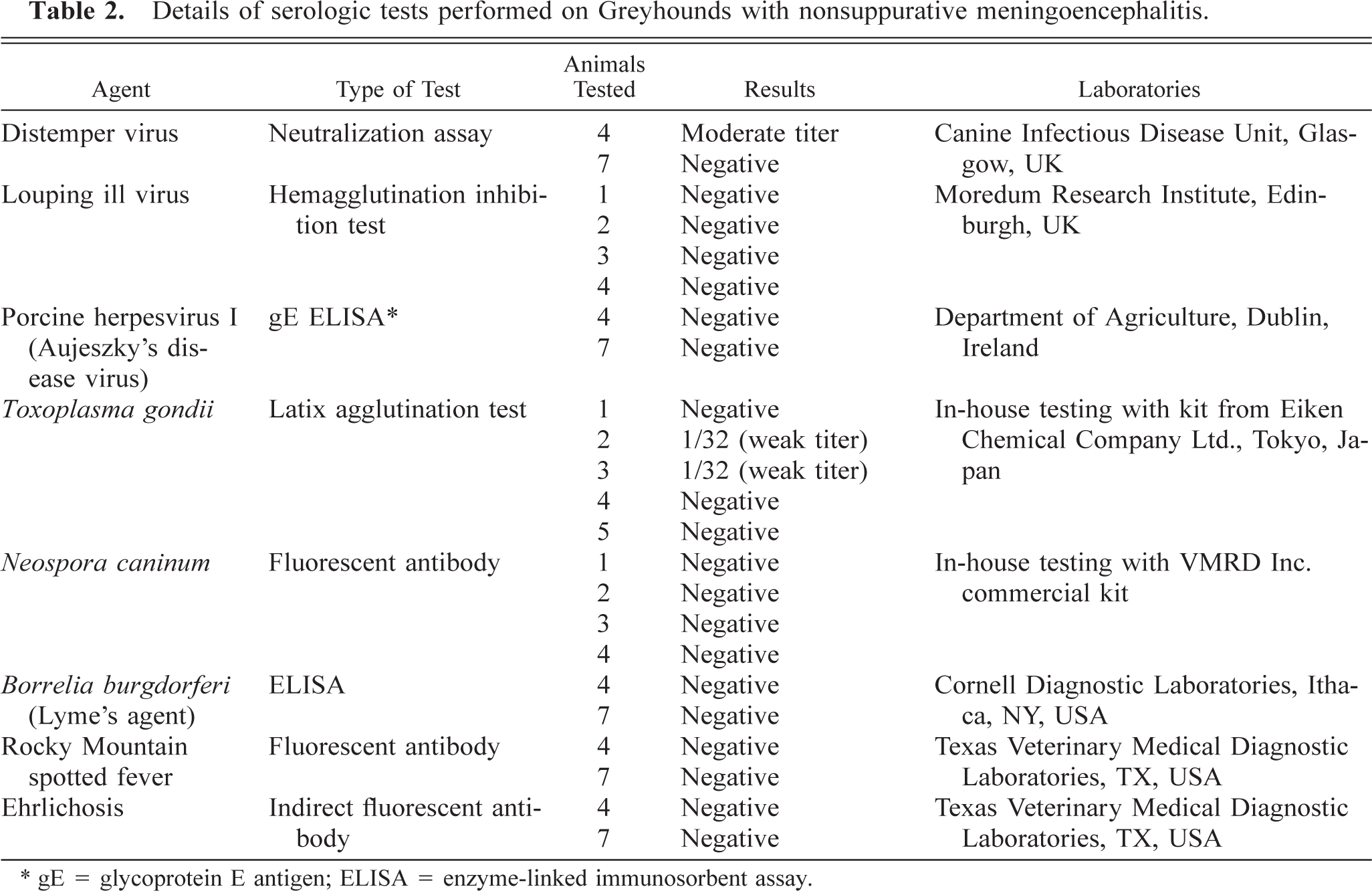
Serology was performed on six dogs for antibodies to canine distemper virus (CDV), louping ill virus, porcine herpesvirus I,
Details of serologic tests performed on Greyhounds with nonsuppurative meningoencephalitis.
∗ gE = glycoprotein E antigen; ELISA = enzyme-linked immunosorbent assay.
Immunohistochemistry
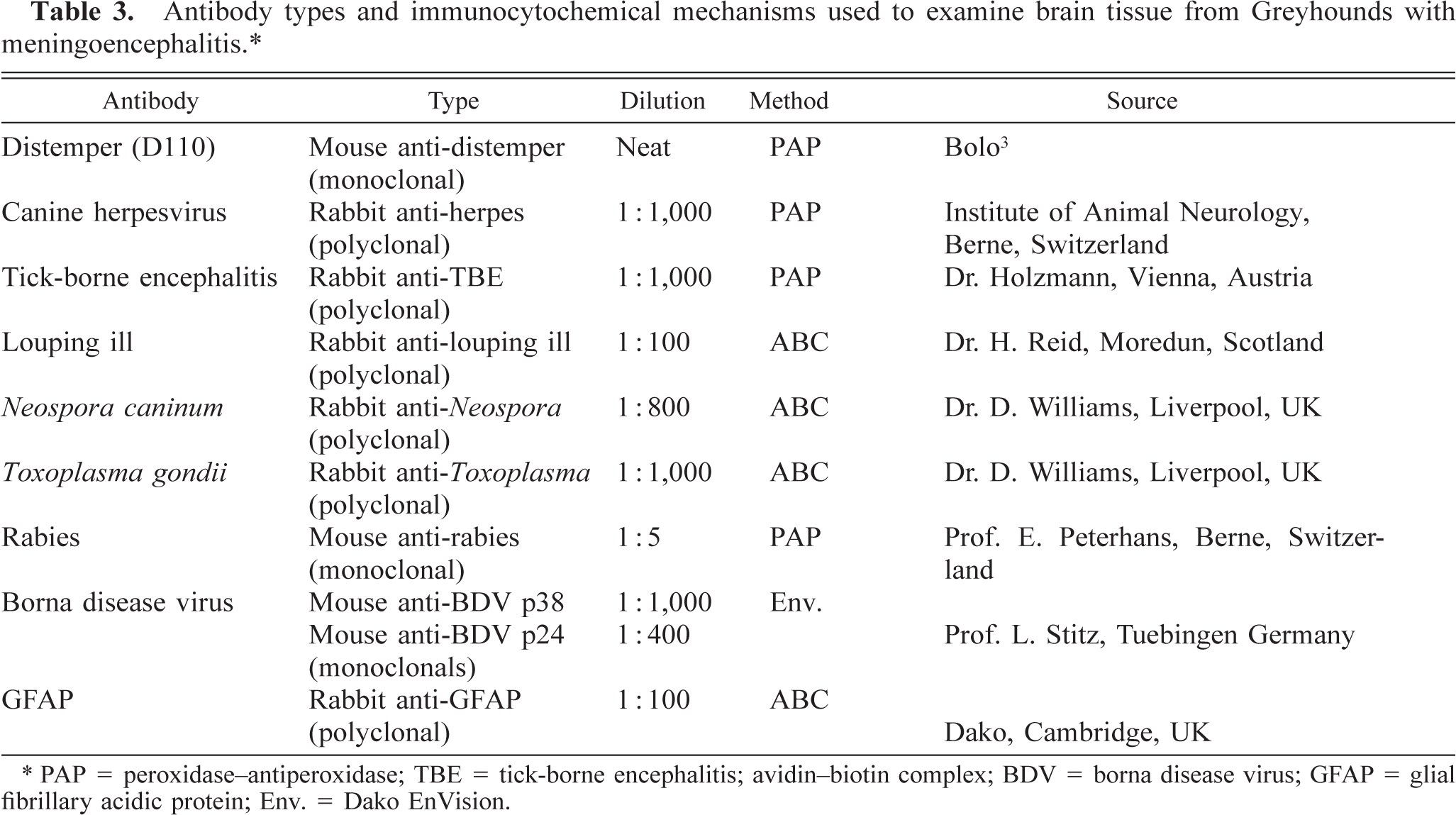
Immunohistochemistry was performed on formalin-fixed paraffin-embedded sections of cerebrum from dog Nos. 2, 3, and 4. Avidin–biotin complex (ABC) or DAKO EnVision methods were employed with commercial kits (Vectastain ABC Kit, Vector Laboratories, Peterborough, UK; DAKO, Cambridge, UK). Peroxidase–antiperoxidase procedures were performed in-house with peroxidase–antiperoxidase rabbit or mouse complexes (DAKO, Zug, Switzerland). The antibodies used, their specificity, and sources are listed in Table 3. Briefly, 5-µm-thick sections were deparaffinized in xylene and rehydrated through graded alcohols. Endogenous peroxidase activity was blocked by incubation with 0.3% hydrogen peroxidase in methanol at room temperature for 30 minutes. Sections were washed in Tris-buffered saline (TBS, 0.1 M Tris-HCl, 0.9% sodium chloride, pH 7.8). To demonstrate
Antibody types and immunocytochemical mechanisms used to examine brain tissue from Greyhounds with meningoencephalitis.∗
∗ PAP = peroxidase-antiperoxidase; TBE = tick-borne encephalitis; avidin-biotin complex; BDV = borna disease virus; GFAP = glial fibrillary acidic protein; Env. = Dako EnVision.
Immunohistochemical procedures with distemper antibody D110 were as previously described without modifications. 3 These techniques were also used for the detection of canine herpesvirus (CHV), European tick-borne encephalitis, and rabies antigens. Borna disease virus antigens were detected using DAKO EnVision detection systems (DAKO, Cambridge) after pronase (Sigma, Poole, UK) treatment for 10 minutes at 37 C. Examinations were performed with positive and negative controls in all situations.
In situ hybridization
In situ hybridization studies to detect CDV mRNA were performed on formalin-fixed, paraffin-embedded sections of cerebrum from dog Nos. 2, 3, and 4, as previously described.
20
Briefly, deparaffinized sections were rehydrated, washed with phosphate-buffered saline (PBS), and treated for 10 minutes with 0.2 M HCl. The samples were incubated with proteinase K (1 µg/ml) in 20 mM Tris-HCl (pH 7.5) and 2 mM CaCl2 for 20 minutes at 37 C. After pretreatment, samples were fixed with 4% paraformaldehyde in PBS, followed by the addition of 0.2% glycine PBS, and washed again in PBS. Slides were then prehybridized at 52 C for 1 hour in 50% formamide 5 × standard saline citrate (SSC), 10 × Denhardt's reagent, and 500 µg/ml salmon sperm DNA. Pretreated sections were hybridized at 52 C overnight in 50% formamide, 10 × dextran sulfate, 5 × hybridization salts (20 × hybridization salts: 0.1 M ethylenediaminetetraacetic acid, 0.1 M piperazine-
Results
Case histories
Animal details and presenting clinical signs are summarized in Table 1. Dog Nos. 1–11 presented with acute onset clinical signs that developed up to 6 weeks before euthanasia. In contrast, the clinical signs in dog Nos. 12–14 were insidious and chronic in nature and were monitored for a period of 8–12 months before euthanasia. Circling, a head tilt, and blindness were frequently recognized clinical signs. On ophthalmologic examination, papilloedema was noted and central blindness was diagnosed. These signs were accompanied by depression, anorexia, dehydration, and weight loss. However, the only clinical signs noted in siblings 8, 9, and 10 were subtle incoordinations and stunting. In situations with acute onset clinical signs, neck ventroflexion and ataxia with a progression to recumbency was noted in the most severe cases. In the chronic cases a head tilt and circling were the common clinical signs.
Routine blood biochemistry and hematology were performed in dog Nos. 1–4, 7, 12, and 13 and revealed elevated hematocrit and elevated concentrations of hemoglobin, total proteins, urea, and creatinine, which reflect dehydration.
Animals were all born in Ireland and came from eight litters from three kennels. In seven litters, multiple, but not all, siblings were affected. This was confirmed by histopathology in four litters. An association with the state of Texas (USA) was noted with three litters. The dam of dog No. 1 was born in Texas and imported to Ireland. She gave birth to two healthy litters before the affected litter. The dam of dog Nos. 2, 3, and 14 was born in Ireland but had raced in Texas. The litter containing dog No. 7 was bred in Texas but the pups were born in quarantine in Ireland.
The animals in this study were the product of many different dam and sire matings. No repeat mating using similar dam and sire combinations was used. Some sires and dams produced multiple affected litters with different Greyhounds: the dam of dog No. 1 produced a subsequent affected litter (containing dog Nos. 4 and 5), and the dam of dog No. 7 produced a subsequent affected litter containing dog Nos. 8, 9, and 10. One sire was common to three litters that produced dog Nos. 4, 5, 8–10, 12, and 13.
All three kennels were considered to be well managed. No changes had occurred in feeding or vaccination protocols or in husbandry practices, particularly in relation to the exposure to potential toxic agents used in the cleaning of premises or in the maintenance of buildings. Few problems required veterinary assistance. Each kennel used different commercially available vaccines administered in accordance with standard practices. Feeding consisted of fresh meat (usually bovine) supplemented with commercial feeds. Ticks and fleas were occasionally observed on dogs.
Postmortem examination and histopathology
At necropsy, all animals were in reasonable body condition. A diphtheritic plaque covered the surface of the left tonsil in dog No. 2. No additional gross abnormalities were detected on examination of all organ systems in animals that received full postmortem examinations (dog Nos. 1–5, 7, and 12–14). In both acute and chronic cases, histopathologic findings were confined to the central nervous system.
Acute cases
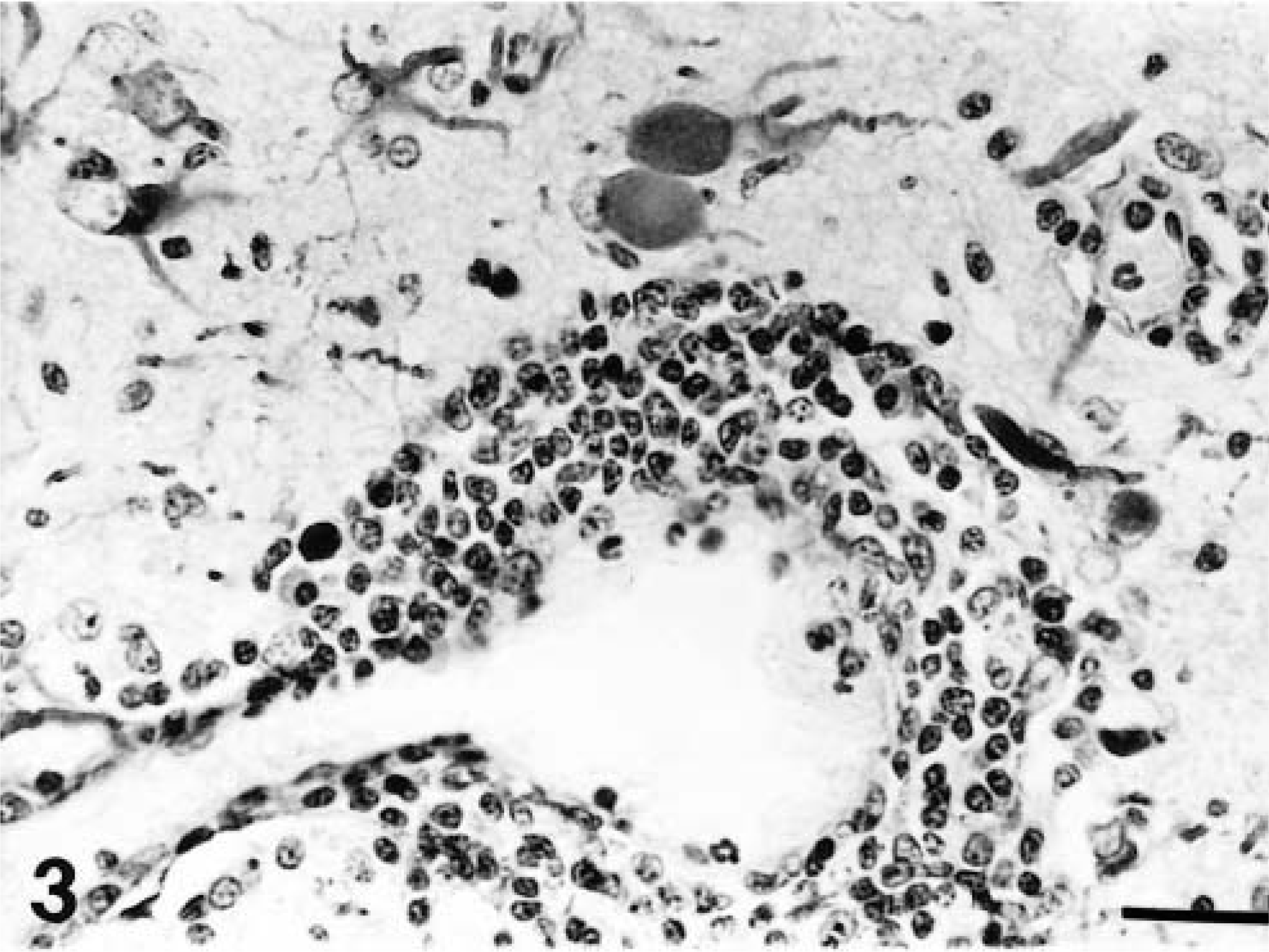
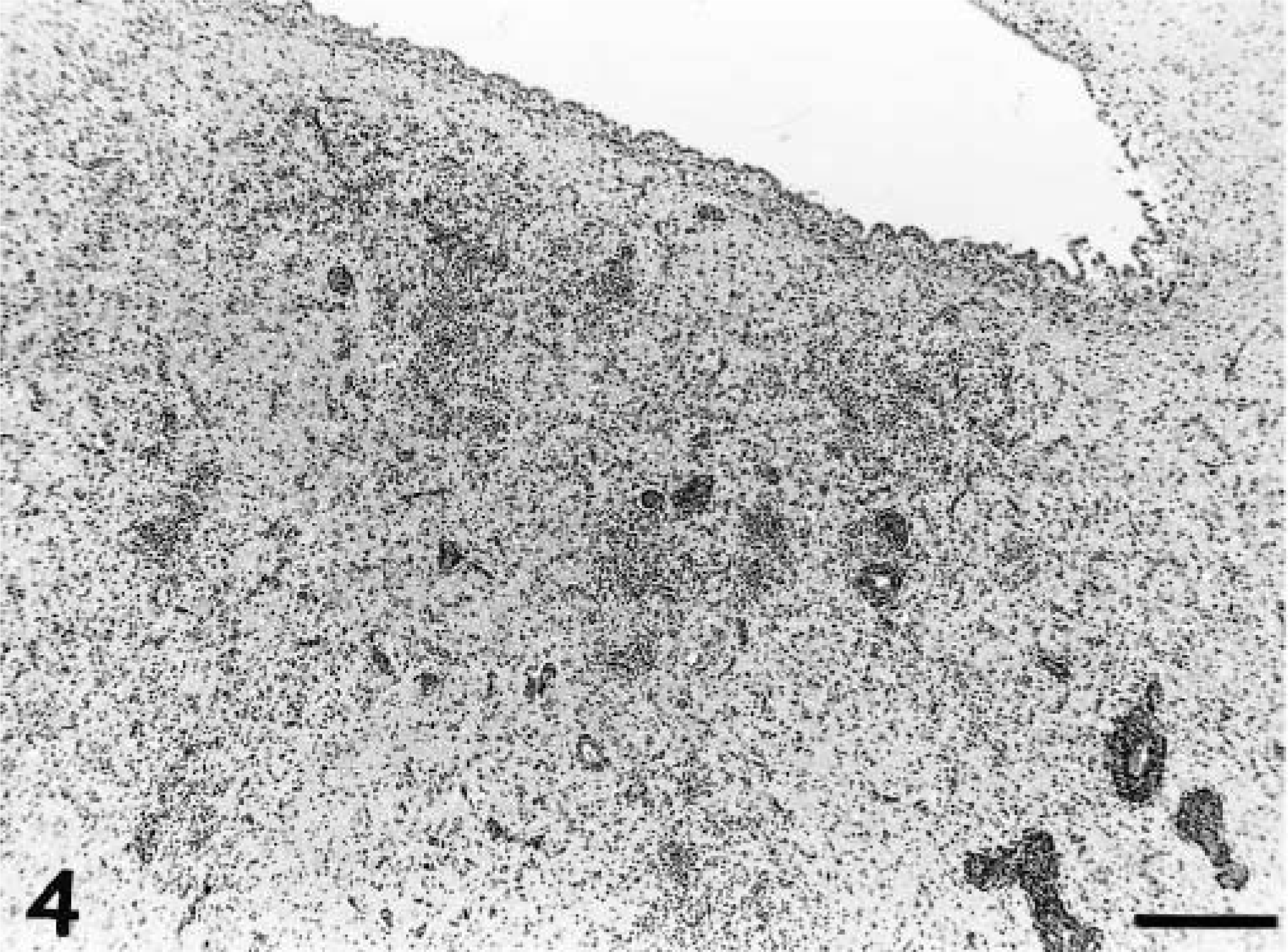
A distinct pattern of inflammation was observed in all the acutely affected dogs. The predominant lesion was diffuse and nodular gliosis of cerebral gray matter due to prominent microglial and astrocytic cells (Figs. 1, 2). This was accompanied by perivascular cuffing (lymphocytes and plasma cells), prominent endothelial swelling, and marked gemistocytosis (Fig. 3). In one animal (dog No. 11) rarefaction of parenchyma was also observed. This cerebral reaction was generally confined to cortical gray matter where the inflammation had a multifocal distribution. The region of the caudate nucleus was the second cerebral location that was involved (Fig. 4). In two animals (dog Nos. 1 and 4) single perivascular infiltrates also contained aggregations of histiocytes (Fig. 5).
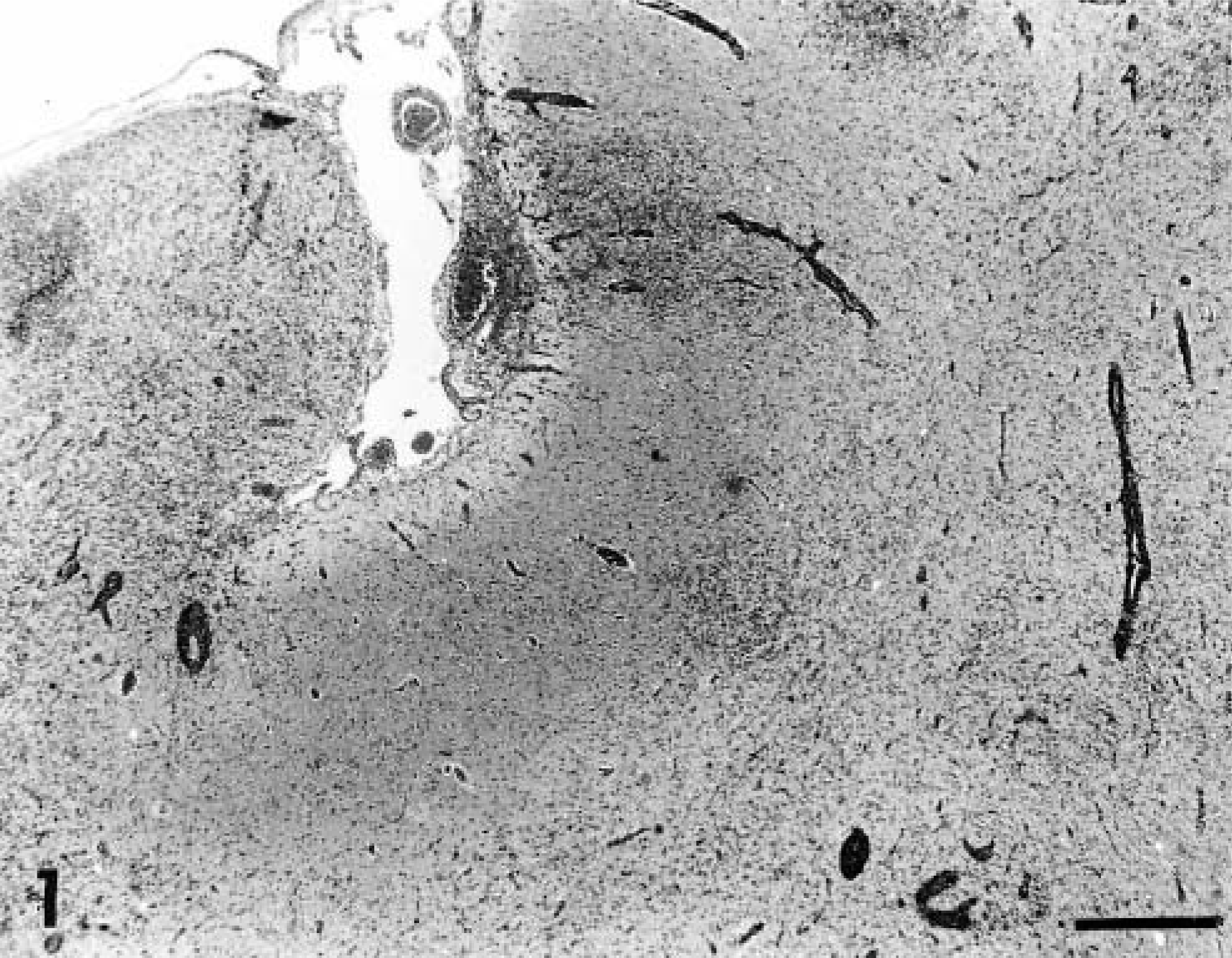
Cerebral cortex; dog No. 1. Nodular and diffuse gliosis with perivascular cuffing. HE. Bar = 570 µm.
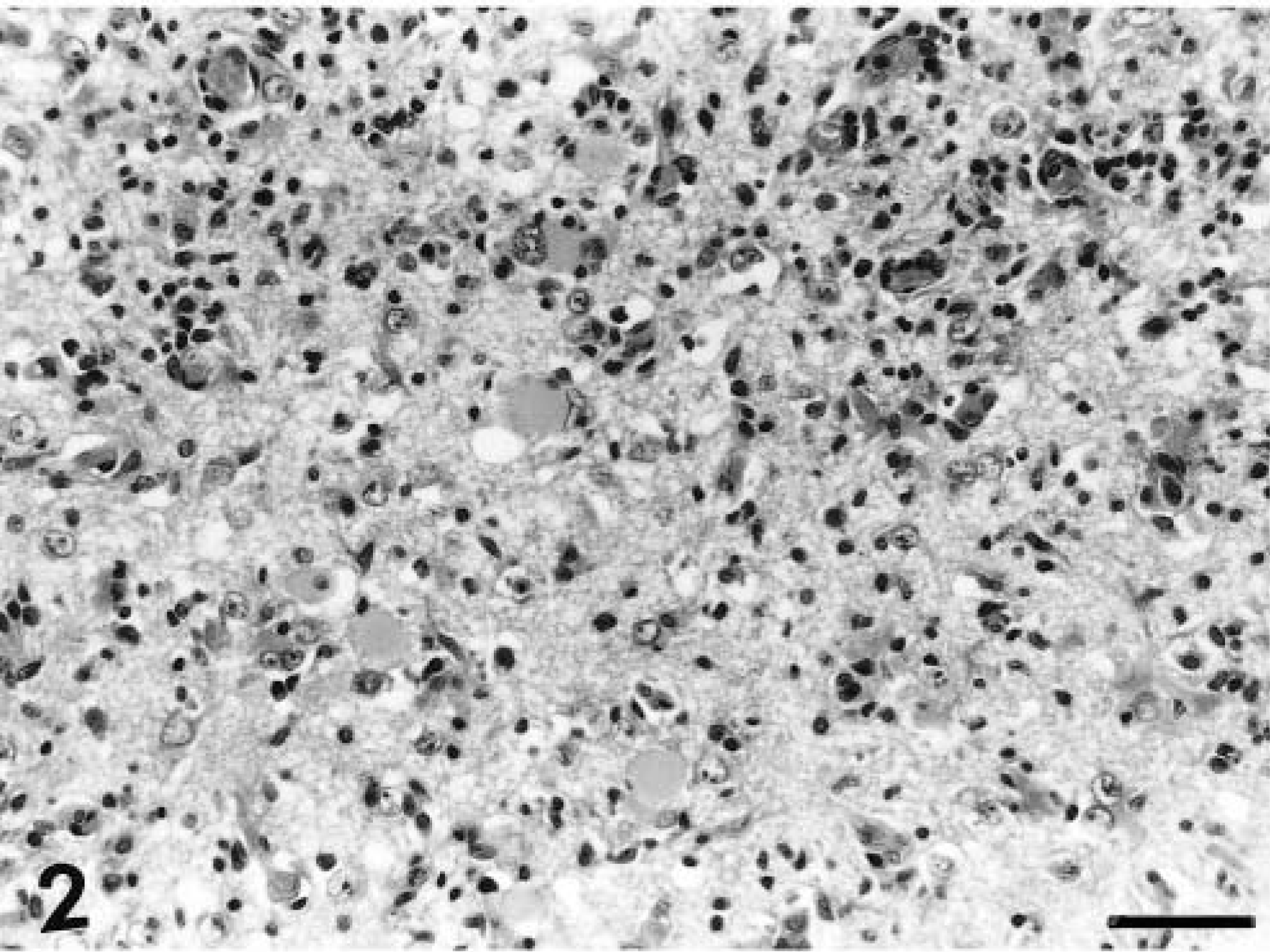
Cerebral cortex; dog No. 1. Extensive gliosis with gemistocytosis. HE. Bar = 57 µm.
Cerebral cortex; dog No. 8. Mononuclear cell perivascular cuffing, gliosis, and prominent GFAP-stained astrocytes. ABC method, hematoxylin counterstain. Bar = 28.5 µm.
Cerebrum; dog No. 8. Severe gliosis and perivascular cuffing in the caudate nucleus. HE. Bar = 570 µm.
Cerebral cortex; dog No. 4. Perivascular cuff containing an aggregation of histiocytic cells (arrows). HE. Bar = 114 µm.
Gliosis with prominent gemistocytic astrocytosis and perivascular cuffing also was documented in the gray matter of the midbrain, pons, and medulla. In the latter two regions, the reaction was milder than in the cerebrum and midbrain. The choroid plexus contained a minimal lymphocyte infiltrate in the fourth ventricle in dog Nos. 3, 5, and 7 (Fig. 6). Histopathologic changes within the cerebellum were confined to the molecular layer, in which solitary or multifocal (usually three or fewer) regions of gliosis were noted in 5 of the 11 acutely affected animals (dog Nos. 2, 3, 5, 6, and 8; Fig. 7). The spinal cord (examined in six dogs) was characterized by mild perivascular cuffing and gliosis within dorsal and ventral horns of cervical cord in two animals (dog Nos. 3 and 5). No abnormalities were detected in thoracic cord or lumbar cord, or in ocular tissue (including optic nerve).
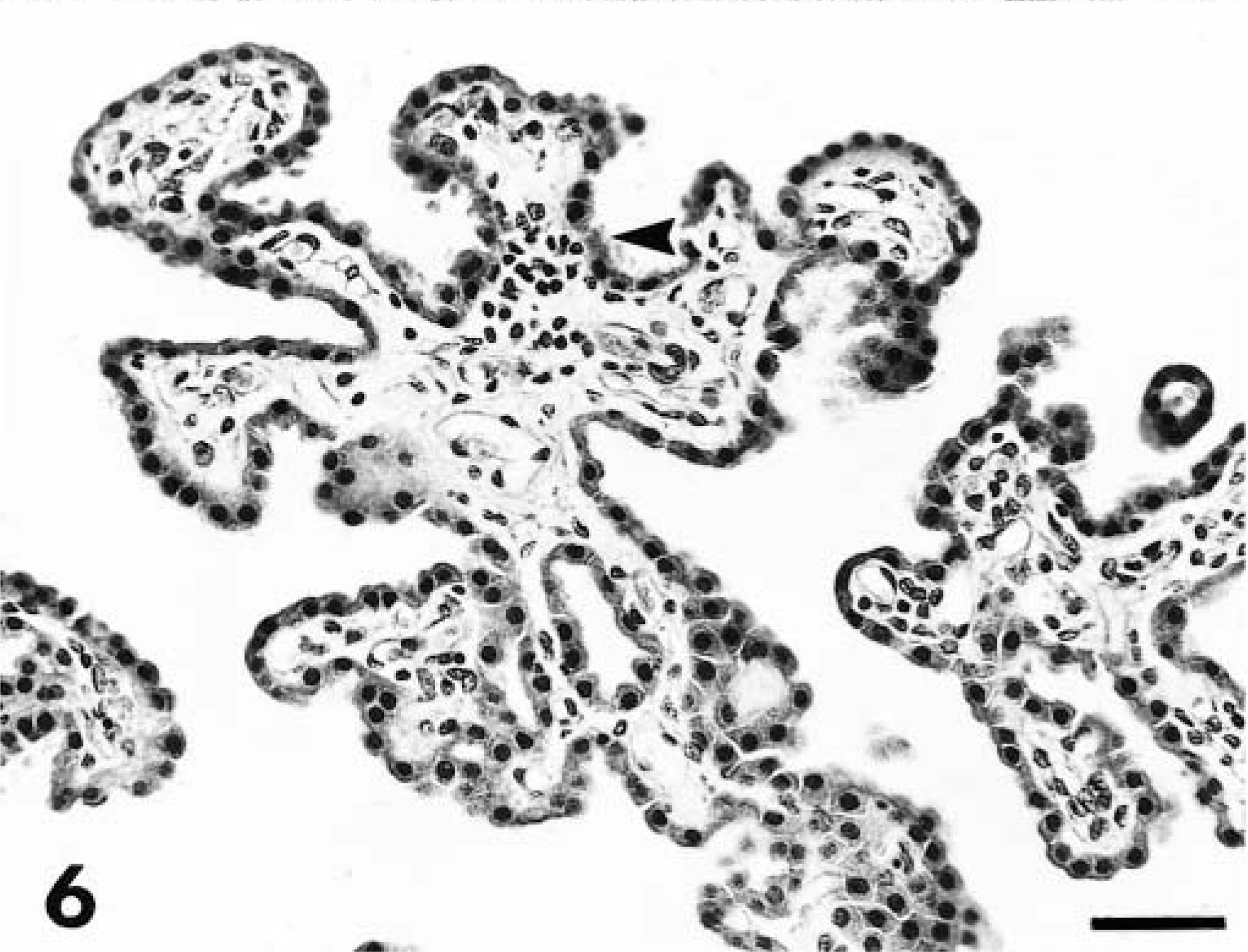
Choroid plexus; dog No. 3. Focal aggregations of lymphocytes (arrowhead) within the choroid plexus of the fourth ventricle. HE. Bar = 57 µm.
Cerebellum; dog No. 5. Focal perivascular cuffing and gliosis within the molecular layer accompanied by an overlying meningeal mononuclear cell infiltration. HE. Bar = 114 µm.
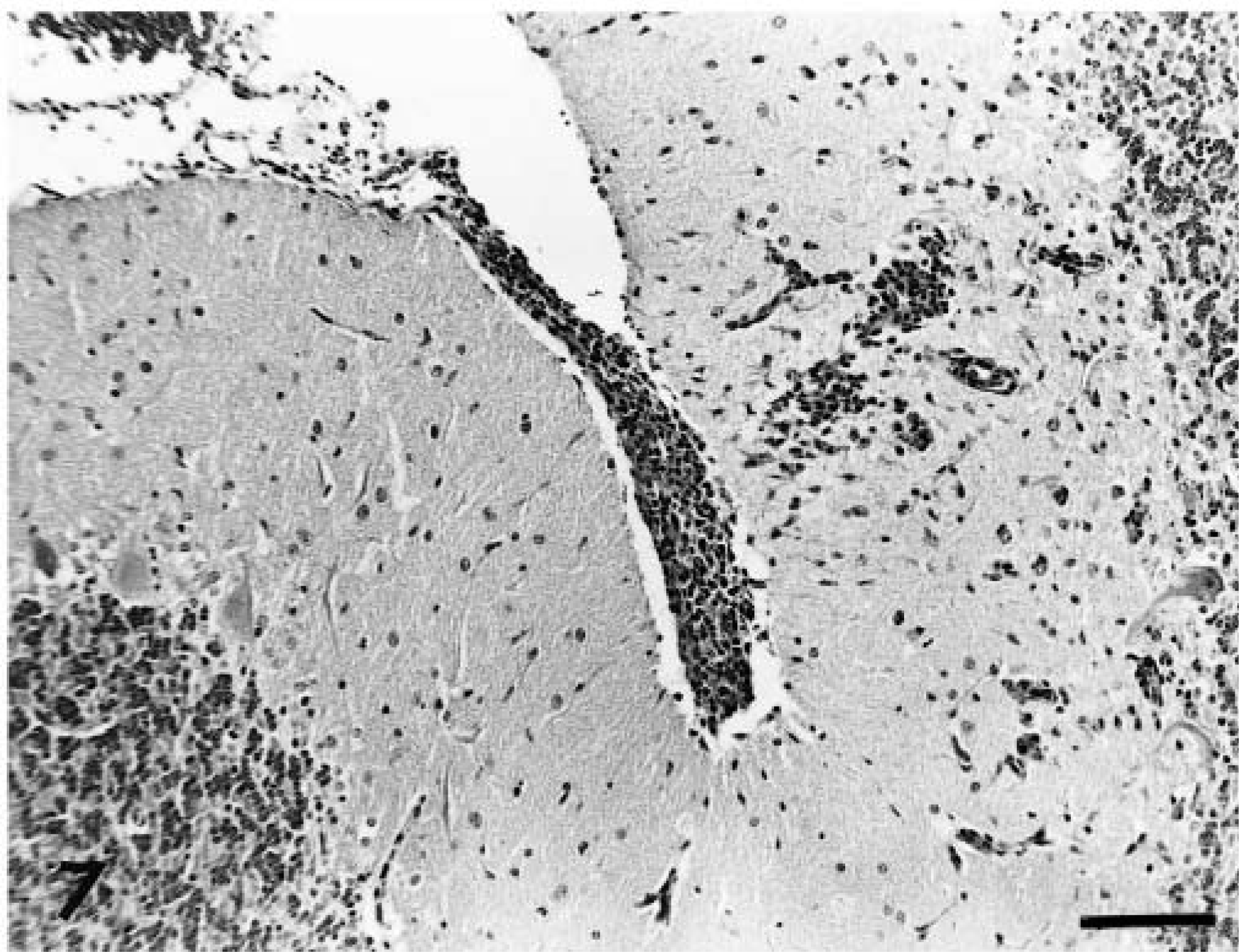
Inflammation within the brain parenchyma was accompanied by lymphocytic and plasma cell meningeal infiltration with prominent perivascular infiltrates within Virchow-Robin spaces. This lesion was of greatest severity over the cerebrum (Fig. 1). A similar inflammatory cell infiltrate was observed over cerebellum adjacent to infiltrates within the molecular layer (Fig. 7). Minimal meningeal infiltrates were also noted in regions covering the brainstem and cranial spinal cord.
Demyelination, neuropil necrosis, neuronophagia, or vasculitis were not significant features. No inclusion bodies, protozoal cysts, or fungal agents were observed.
Chronic cases
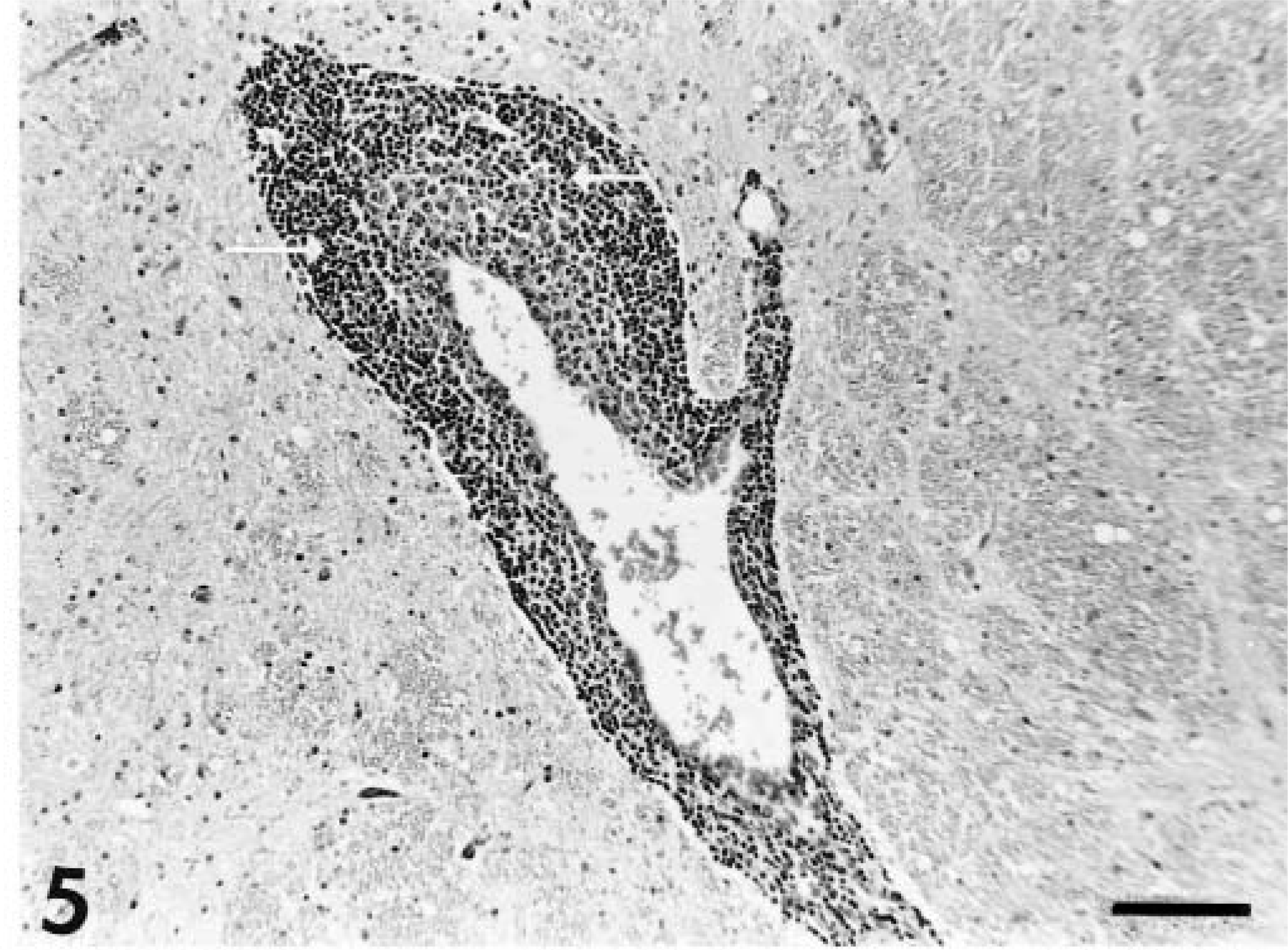
A similar pattern and character of inflammation was noted in the three chronically affected animals (dog Nos. 12–14). Diffuse and nodular gliosis, gemistocytosis, and perivascular cuffing were observed in the gray matter of the cerebrum, midbrain, pons, and medulla. These lesions were accompanied by meningeal lymphocyte and plasma cell infiltrations. In contrast to the acute cases, the lesions in cerebrum, cerebellum, and midbrain were in general milder, but occasionally cerebral tissue contained solitary lesions of similar severity to those of the acute cases. Such a lesion was also accompanied by rarefaction of the parenchyma in one animal (dog No. 13; Fig. 8). In addition, the optic chiasms of these three animals contained diffuse lymphocyte infiltrations within the meninges. Meningeal infiltrates were of milder severity than those noted in the acute cases. No abnormalities were detected in the spinal cords of dog Nos. 12 and 13. Mild cervical spinal cord meningeal infiltrations were noted in dog No. 14. Results of examination of ocular tissue were unremarkable in all three animals.
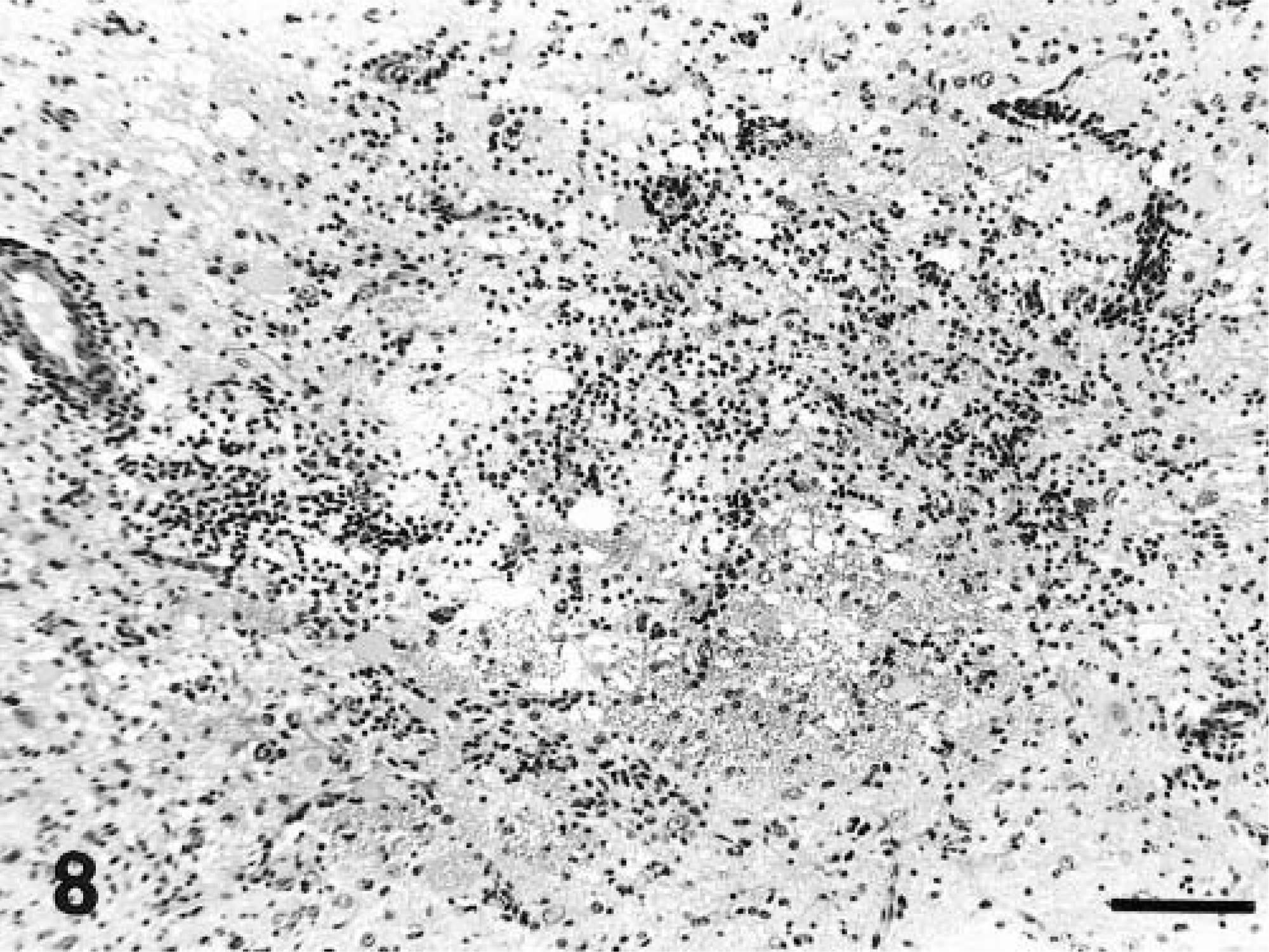
Cerebrum; dog No. 13. Diffuse gliosis with rarefaction of the neuropil adjacent to the lateral ventricle. HE. Bar = 114 µm.
Non-neural tissues
Examination of tonsillar tissue from dog No. 2 confirmed reactive lymphoid tissue covered by a serocellular crust. Focal epithelial necrosis and neutrophil exocytosis were present. No significant abnormalities were detected in nonneural tissues in all Greyhounds that received postmortem examinations, including hepatic (
Serology
Serology was performed on six animals (dog Nos. 1–5 and 7), and results are summarized in Table 2. Two to five animals were tested for antibodies to CDV, louping ill virus, porcine herpesvirus I,
Immunohistochemistry
Immunohistochemical examinations on selected sections of brain tissue from dog Nos. 2, 3, and 4 failed to reveal any evidence of infection with CDV, CHV, European tick-borne encephalitis virus, louping ill virus, Borna disease virus,
In situ hybridization
No evidence was found by in situ hybridization methods to suggest involvement of CDV.
Discussion
This paper presents a novel neurologic syndrome in the Greyhound breed. In both presentation and pathologic features, this syndrome seemed different from known nonsuppurative meningoencephalitic conditions in the dog. Affected dogs were obtained from three of the major racing and breeding Greyhound kennels in Ireland. Animals with neurologic signs were rarely observed in these kennels before 1997. Since 1997, the identification of this problem in groups of young Greyhounds and among siblings suggested that this was an emerging disease rather than a previously unconfirmed existing condition. Clinical and historical evidence indicated that many more suspect cases were observed (more than 28 in total) in the 2-year period covered by this paper (unpublished observations). These additional cases involved three other kennels and were not confirmed by histopathology.
The lesions were similar in all animals, chiefly affecting the gray matter of the brain and characterized by a mixed mononuclear cell inflammation, suggesting a viral meningoencephalitis. This finding was consistently of greatest severity in the cerebral cortex and in the caudate nucleus. Lesions of reduced severity were observed in the midbrain, pons, and medulla and occasionally in the cranial spinal cord. Occasional gliosis was noted in the molecular layer of the cerebellum.
Specific nonsuppurative meningoencephalitic syndromes recognized in the dog in Ireland and the adjacent UK are produced by infection with CDV,
29
louping ill virus,
19
protozoal (
Canine distemper encephalitis may present in many forms and may manifest as primarily a gray matter disease in which the classical respiratory and intestinal clinical signs are mild and go unnoticed. 26,27 In addition, a study by Raw and colleagues 22 highlighted the need to suspect distemper in vaccinated young adult dogs that suddenly develop nervous signs such as ataxia and paraplegia. All of the Greyhounds in the present study had been vaccinated against CDV. Three different commercial vaccines had been used. These factors would tend to eliminate CDV or any viral contaminant within a vaccine as a potential agent responsible for the meningoencephalitis observed in this study. 6 No clinical or histologic evidence was found of gastrointestinal or respiratory tract involvement. Histopathologic examination failed to reveal any inclusion bodies, and brain pathology did not include demyelination. In addition, viral antigen was not found in the CNS by either immunohistochemical or in situ hybridization techniques. 3,20
Louping ill infection is endemic in sheep in certain parts of Ireland, and dogs can be infected with this virus. 13,19 Histopathologic findings did not correlate with those previously reported in the dog. 19 In particular, neuronal degeneration was not a feature and, although glial nodules were noted within the cerebellar molecular layer, Purkinje cell degeneration was not a feature noted. A lack of serologic evidence and an absence of viral antigen also supported these conclusions, although the latter findings could also be explained by the rapid clearance of virus in flavivirus-induced encephalitis. 17
Protozoal encephalitis associated with
Canine adenoviral and CHV infections are also associated with encephalitis in the dog. 26 In contrast to the present series of cases, canine adenoviral encephalitis is essentially a vasculitis, and intranuclear inclusions are observed in vascular endothelial cells. CHV has been reported in many breeds of dogs, including Greyhounds, where it is a disease of very young dogs and causes multiorgan necrosis and hemorrhage, encephalitis, and retinitis. 18 Unlike the present series of cases, the diffuse meningoencephalitis associated with CHV is often accompanied by cerebellar inflammation, segmental cerebellar necrosis, spinal cord inflammation, and ganglionitis. 21 CHV antigen was not detected by immunohistochemistry in the three dogs tested from this series of cases.
Canine Aujeszky's disease, caused by porcine herpesvirus I, is noted sporadically in Ireland and the UK, particularly in dog kennels that use food that has not been commercially prepared (E. Weavers, personal communications). 8 Clinical features in the cases in the present study were not like those associated with canine Aujeszky's disease (peracute onset of respiratory and alimentary clinical signs or intense pruritus), and histopathologic features of neuronal degeneration, ganglionitis, and the presence of inclusion bodies were not observed. Antibody titers were not observed in the two animals that were tested.
In mainland Europe, rabies, European tick-borne, and Borna virus encephalitis have been recognized in the dog. 26,32,33 Histopathologic features, in particular the lack of involvement of the spinal cord and dorsal root ganglia and the absence of intraneuronal inclusion bodies, were not characteristic for rabies. This observation was supported by both immunohistochemical findings and the clinical presentation. European tick-borne encephalitis, caused by a virus closely related to louping ill virus, has been documented in dogs. 32 Histopathologic features of this condition are similar to those of louping ill virus infection and include prominent neuron necrosis, neuronophagia, and cerebellar molecular layer gliosis. Neuronal necrosis was not observed in the present series of cases and virus was not found by immunohistochemical evaluations. Borna disease virus was documented in a dog in Austria that had nonsuppurative meningoencephalitis. 33 Lesions were of greatest severity within the cerebrum, with minor lesions noted in the cerebellum and brainstem. In the present series of cases, Borna disease viral antigen was not detected by immunohistochemical techniques and the intranuclear Joest-Degen inclusions typically observed in this condition were not seen.
The association with Texas through transportation of animals between racing and breeding establishments introduced the possibility of involvement of rickettsial agents responsible for Rocky Mountain spotted fever (RMSF) and ehrlichiosis. 9 Neuropathology, favoring a similar distribution and cell makeup as that of the present series of cases, has been recognized in up to 25% of cases of canine ehrlichiosis. 12 However, both ehrlichiosis and RMSF are characterized by multisystemic signs, including ocular nasal discharges, fever, and lymphadenopathy accompanied by thrombocytopenia. In RMSF, vasculitis is the most prominent pathology affecting many organs, including the brain. Such features were not observed in the present series of cases and this was supported by serologic evaluation on two animals.
Reports of the involvement of
Although GME has not been documented in Ireland, the dogs in the present study, if assessed on an individual basis without knowledge of the multiple presentations, would be suggestive of such a syndrome because of acute onset neurologic signs in vaccinated young adult dogs. Such features would be supported by histopathologic examinations revealing moderate lymphocyte-rich and plasma cell–rich mononuclear perivascular cuffing consistent with GME. 28 In two of the dogs in the present series, perivascular cuffs were noted with prominent aggregations of histiocytes. However, many features were not typical of GME. First, the nature of the outbreak revealed that the condition was not sporadic but was clustered in certain kennels and affected many siblings in one litter. This is in contrast to GME, which is sporadic, with limited documentation of familial predispositions. 7,11 Second, GME has not been documented in the Greyhound. Third, histopathologic findings were not typical of GME in that histiocytes did not predominate in the pervascular mononuclear cell infiltrations. In addition, the infiltrates in the dogs in the present study were consistently moderate and diffuse, with lesions favoring cerebral gray matter rather than white matter and having limited involvement of the brainstem or spinal cord. 10,28 However, because the cause of GME remains unknown and variants of histopathologic presentations exist, 28 a possibility exists that novel presentation of this condition may evolve.
Over the past 20 years, breed-associated nonsuppurative meningoencephalitis conditions have been observed in the Pug, Maltese, and Yorkshire Terrier breeds. 5,24,30 Similar to Greyhound meningoencephalitis, lesions in both the Pug and Maltese are predominantly confined to the cerebral hemispheres, but, in contrast to the Greyhound, the lesions have a prominent necrotizing component. The lesion in the Yorkshire Terrier, although also having a prominent necrotizing component, favors brain stem white matter distribution, in which microcavitations and parenchymal necrosis are major features.
Nonsuppurative meningoencephalitis is not recognized as a pathologic feature with genetic origins, although the nature of the present outbreak involving a single breed and variable numbers of siblings within one litter may be suggestive of a genetically linked problem. In contrast to developmental or degenerative CNS disorders, which may be related to primary genetic defects, the present series of cases more likely reflects a genetic predisposition to an as yet unknown infectious agent. 5 A second issue requiring consideration relates to the possibility that the lesions described in the present cases reflect a postinfectious encephalitis. This syndrome is recognized in humans and is postulated to be an autoimmune disease triggered by a spectrum of infections. 14 Lesions in the present study were not typical of postinfectious encephalitis, which usually involves white matter and results in both inflammation and demyelination.
In summary, a specific neurologic syndrome has become apparent in the Greyhound breed in Ireland. The nature of the outbreak from both clinical presentation and histopathologic findings would suggest an infectious agent. Whether this represents the presence of a new agent, presumably a virus, within this population or whether this outbreak represents a novel presentation within this breed of a known virus remains to be determined.
Footnotes
Acknowledgements
We acknowledge the assistance of Ms. S. Worrell, J. Brady, Ms. M. McCullogh, S. Hogan, M. Garrett, M. O'Connor, and B. Cloak. Photographs were prepared by C. King. The advice and opinions of Professor B. Sheahan, Professor B. Jones, Dr. H. Larkin, Dr. C. Payne-Johnson, Dr. A. deLahunta, and Dr. H. Thompson are gratefully acknowledged. We are indebted to P. Dalton, Ms. A. Quinlan, S. Burke, The Greyhound Owners Association, and Bord na gCon.
