Abstract
A 4-year-old spayed female ferret
Keywords
With the increased popularity of ferrets (
A 4-year-old spayed female ferret was clinically evaluated for a small, 6-mm subcutaneous firm mass with a 1-month duration and located in the dorsal lumbar area. The animal was otherwise asymptomatic. The cutaneous mass was completely excised, fixed in 10% neutral-buffered formalin, and submitted to the Veterinary Diagnostic Laboratory at the University of Illinois for histopathologic evaluation. Grossly, the mass was a well-demarcated and expansive, solid round nodule located in the panniculus. On cut section the nodule was firm, pale gray-white, and with a shiny surface (Fig. 1). Small multifocal to coalescing areas of dark brown-black discoloration were present predominantly in the central portions of the mass. The tissue was embedded in paraffin; sectioned at 3 µm; and stained with hematoxylin and eosin (HE), Warthin-Starry and Fontana-Masson silver stains, and with antibodies directed against S100, cytokeratin, vimentin, and Melan A proteins. In addition, samples of formalin-fixed tumor tissue were postfixed in osmium tetroxide, stained with uranyl acetate (en bloc), embedded in epoxy resin (Lx 112), and thin sectioned for transmission electron microscopy.
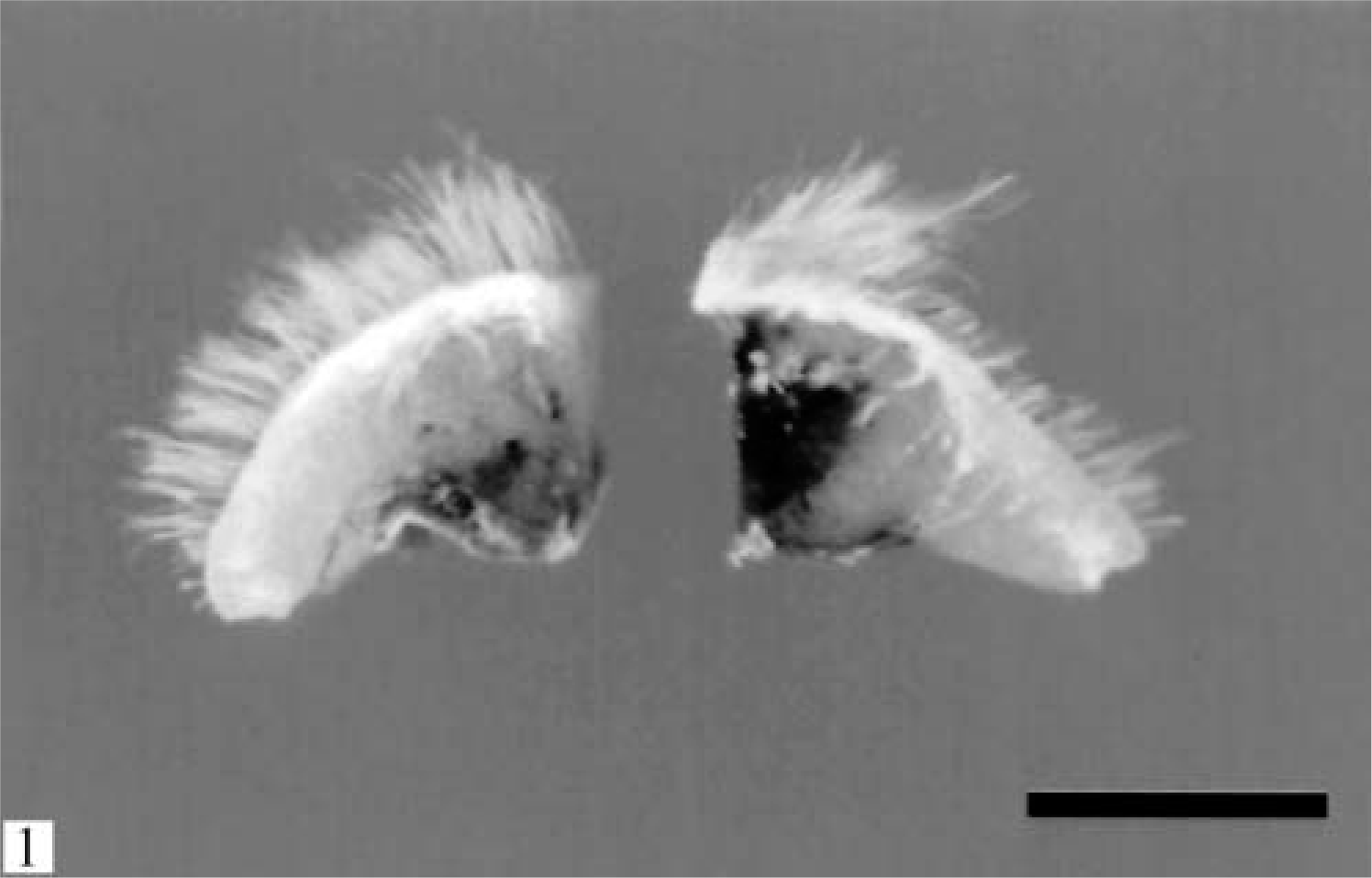
Skin; ferret. A round, well-demarcated, 6-mm nodular mass is present in the panniculus. Multifocal to coalescing areas of black pigmentation are evident within the central portion of the mass. Bar = 5 mm.
Microscopically, the mass was well demarcated, nonencapsulated, focally expanding the subcutaneous fat, compressing the underlying subcutaneous skeletal muscles, and elevating the overlying dermis. The epidermis was intact. The mass was composed of closely packeted large, polygonal to spindle-shaped cells arranged in sheets, poorly formed nests, and short bundles contained within a scant intervening fibrous stroma (Fig. 2). The cells had indistinct cytoplasmic margins and abundant, finely fibrillar eosinophilic cytoplasm. Few (less than 5%) cells contained variable amounts of brown to black intracytoplasmic pigment. The pigmentation varied from fine dusting to moderate quantities of granular to coarse material. A rare cell was completely filled with pigment. Nuclei varied considerably in size. Many nuclei were large, round to oval, and with a vesicular pattern of chromatin dispersion. The nuclear chromatin was coarsely clumped and often marginated. Most nuclei contained one and rarely two large and round magenta nucleoli. Mitotic figures averaged one per ten 400× magnification fields with occasional bizarre, asymmetric mitotic spindles seen. Scattered apoptotic cells were rarely present throughout the mass. A single large area of necrosis and hemorrhage was present in the center of the mass. Cells along the superficial margins of the mass abutted and occasionally surrounded the bulbs of the deeper hair follicles. Both Warthin-Starry and Fontana-Masson stains demonstrated variable numbers of fine, silver-impregnated black intracytoplasmic granules present in the majority of the neoplastic cells that appeared nonpigmented on HE. The atypical cells of the mass stained strongly positively for vimentin and S100 protein and negatively for cytokeratin and Melan A.
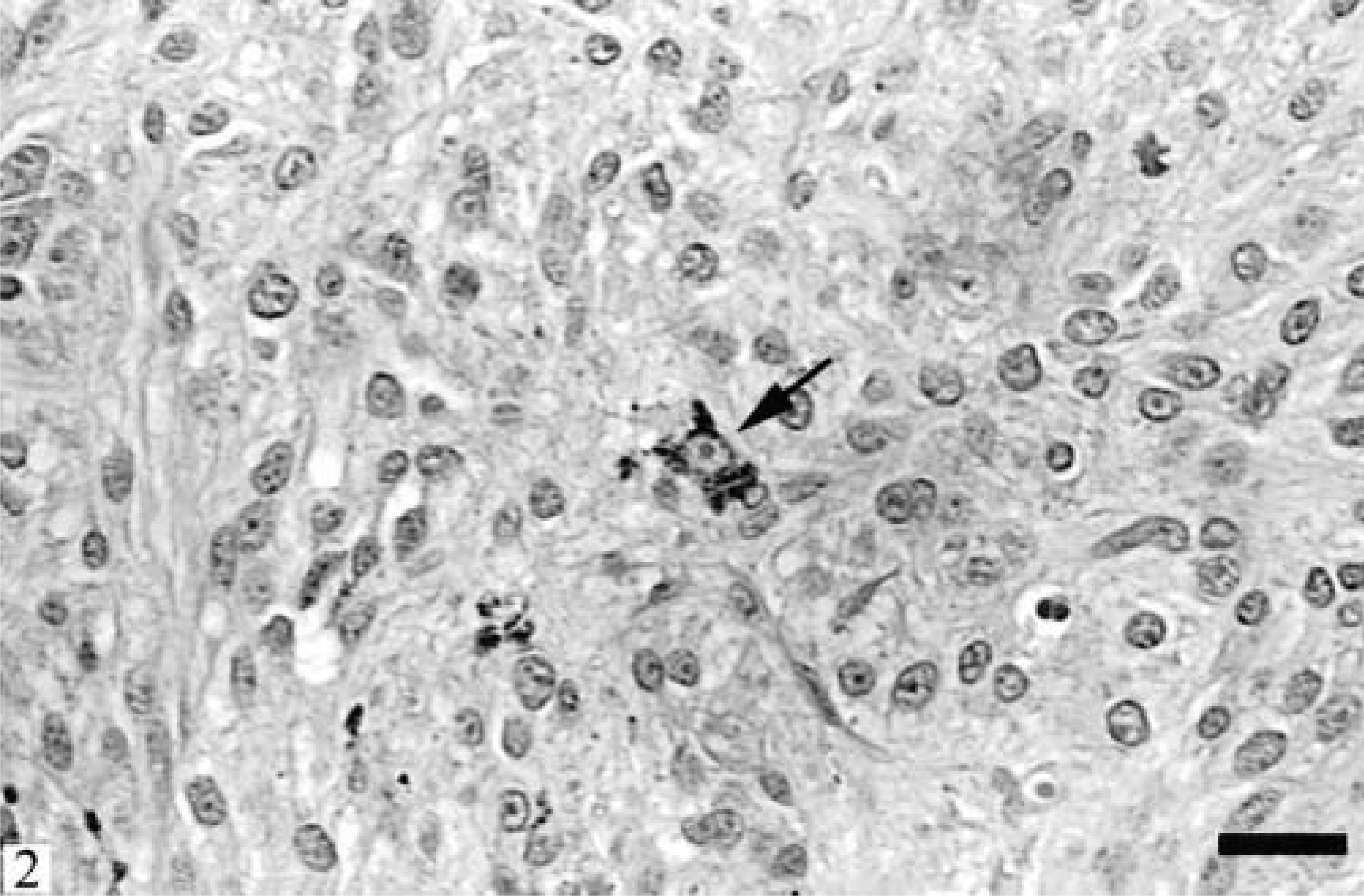
Skin; ferret. The mass consists of closely packeted large, polygonal to spindle-shaped cells arranged in sheets and short bundles and contained within scant fibrous stroma. Single cells contain intracytoplasmic granular pigment (arrow). HE. Bar = 20 µm.
Ultrastructurally, the mass was composed of large neoplastic cells with irregular borders and frequent blunt cytoplasmic processes. The cells had abundant cytoplasm containing copious rough endoplasmic reticulum and variable numbers of melanosomes in different stages of development (Fig. 3). Most melanosomes were in stage IV; occasional stage III melanosomes and rarely a stage II aberrant melanosome were identified. Generally, the melanosomes were oval and occasionally elongated measuring between 0.3 and 0.6 µm. Melanosomes in all stages had a single limiting membrane. Stage II melanosomes appeared as nonpigmented helical structures containing fibrilar material. Occasionally, fibrils were oriented perpendicularly to the limiting membrane. Stage III melanosomes were also helical in shape and contained longitudinally oriented electron-dense fibers and moderate quantities of pigment. Stage IV melanosomes were typically round, fully melanized, and devoid of internal structure. No compound melanosomes were present in the examined tissue specimens.
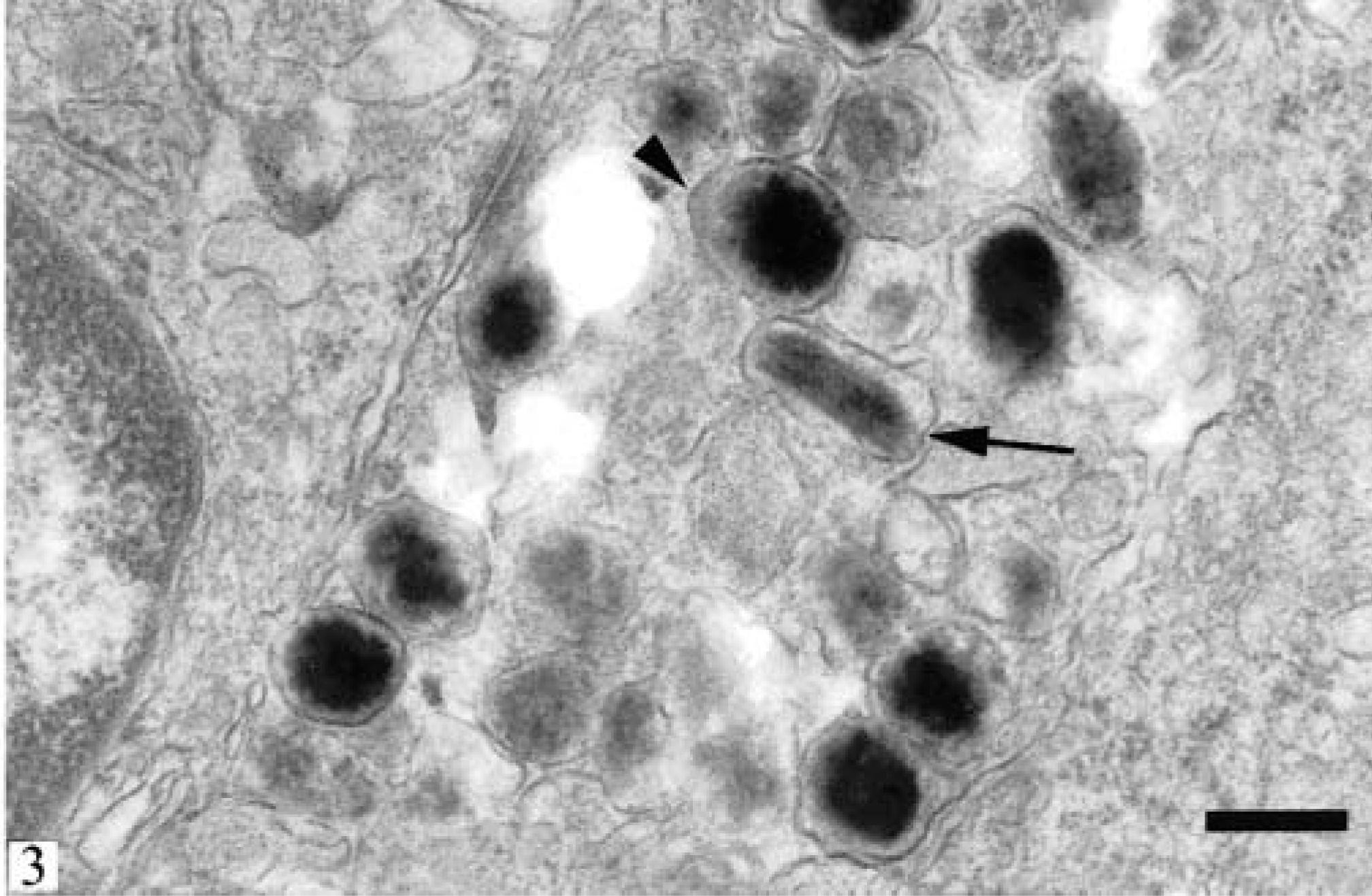
Skin; ferret. Electron micrograph of a neoplastic cell. Stage III (arrow) and stage IV (arrowhead) melanosomes are present. Lack of compound melanosomes and presence of solitary melanosomes in consecutive stages identifies the neoplastic cells as melanocytes. Bar = 0.5 µm.
Dermal melanomas have been reported in a variety of domestic and wild animals. A survey of the literature has found no documented evidence of cutaneous melanomas in ferrets. A retrospective study of 574 ferrets with neoplastic diseases reported 81 skin tumors, 26 of which were not identified or specified. 4 The diagnosis of poorly pigmented and amelanotic melanomas is a challenge to pathologists because of the varied cytomorphology of the neoplastic melanocytes. 2 The utilization of histochemical silver stains (Fontana-Masson and Warthin-Starry) for melanin and immunohistochemical demonstration of the presence of vimentin and S100 protein 7 aids the microscopic diagnosis of poorly pigmented melanocytic neoplasms. However, these stains are nonspecific and should be interpreted in conjunction with the histologic appearance of the tumor. Antibodies to tyrosinase, melanosomal proteins (HMB-45), and specific melanocyte antigens (Melan A) are routinely used for immunohistochemical diagnosis of human melanocytic tumors. Among these antigens, Melan A has been demonstrated in more than 80% of primary or metastatic canine oral melanomas. 6 The tumor described in this report was negative for Melan A. Positive confirmation of the neoplastic cell type was established by transmission electron microscopic demonstration of melanosomes in different stages of development. 2,3 No clinical evidence of recurrence or metastasis of this tumor was found during a 1-year postsurgical monitoring period, suggesting benign biological behavior.
