Abstract
Pathologic changes were studied in 27 interferon-gamma gene knockout mice 34–54 days after being fed graded doses of Sarcocystis neurona sporocysts derived from a naturally infected opossum. The target tissue for S. neurona infection was the central nervous system. Characteristic histopathologic changes present in all mice consisted of an inflammatory infiltrate consisting of mostly neutrophils and macrophages, fewer eosinophils, and rare multinucleated giant cells. Intralesional protozoa and scattered subacute perivascular cuffs were present. Where the infiltrates were extensive, neuropil rarefaction was frequent. Pathologic changes were much more frequent and severe in the caudal portion of the brain, especially in the cerebellum, than in the middle and cranial portions. Changes were present in all spinal cords examined (10 of 10). Lesions were equally distributed in white and gray matter of the brain and spinal cord and their meningeal linings.
Keywords
Equine protozoal myeloencephalitis (EPM) is a serious and progressive disease of the equine central nervous system (CNS). 8 Sarcocystis neurona is the most common cause of EPM. The complete life cycle of S. neurona is not known. Opossums (Didelphis virginiana, D. albiventris) are known to be definitive hosts, and become infected by ingesting S. neurona sarcocysts from muscles of intermediate hosts. Horses are aberrant hosts. 8 Only schizonts and merozoites, but no sarcocysts, are found in horses. Histopathologic changes similar to those reported in horses with EPM have been seen in cats, mink, raccoons, skunks, Pacific harbor seals, and sea otters with S. neurona infections. 8
Pathologic changes in horses have been seen only in the CNS and involve both the white and gray matter. 1,2,4,5 Most of what is known about the disease has been based on natural infections. 8 Attempts to induce EPM by infecting horses has resulted in only a small percentage developing clinical signs, and the parasite has not been demonstrated in lesions induced by feeding sporocysts. 8 For this reason, a small animal model for the disease has been developed. Interferon-gamma (IFN-γ) gene knockout (KO) mice develop encephalitis after ingesting sporocysts of S. neurona. 7 In the present report, we characterized the pathologic changes in KO mice infected by the natural route (oral) and by using the natural infective stage (sporocyst).
Sporocysts were collected from the intestine of naturally infected opossums, identified in bioassays as S. neurona, suspended in antibiotic saline, and stored at 4 C for 2–96 days before being fed to the mice as previously described. 3 Graded doses of sporocysts were fed to 27 KO mice as previously described. 3
IFN-γ gene KO mice (BALB/c-IfngtmlTs) were bred from parents obtained from Jackson Laboratories (Bar Harbor, ME) and raised in-house (USDA, Animal and Natural Resources Institute, Beltsville, MD) in microisolator cages. Mice were predominantly males and were 5–8 weeks old. Sporocysts were fed to 27 mice and 34–54 days after infection they were killed either by CO2 asphyxiation or cervical dislocation. Five age-matched, uninfected IFN-γ gene KO mice were similarly killed during the study to serve as controls. At necropsy, representative samples of all organs were taken, fixed in formalin, and processed routinely for histopathology. Immunohistochemical procedures were performed as previously described. 3 Briefly, rabbit S. neurona polyclonal antiserum was used in conjunction with an Elite rabbit immunoglobulin G avidin–biotin complex kit (Vector Laboratories, Burlingame, CA). Sarcocystis speeri-infected mouse liver and skeletal muscle served as the negative controls. For transmission electron microscopy (TEM), selected samples of fresh brain were immersed in a mixed aldehyde fixative and processed routinely for TEM as previously described. 9
The target for S. neurona infection was the CNS, particularly the cerebellum. The typical finding in the CNS was an asymmetric, multifocal inflammatory cell infiltrate of neutrophils, macrophages, and fewer eosinophils (Figs. 1–3) with rare multinucleated giant cells. These infiltrates were present in all mice equally in white and gray matter of the brain and spinal cord, and in the meninges (Fig. 4). Other findings included intralesional protozoa, perivascular cuffs, reactive glial cells, neuropil rarefaction (Fig. 2) with neuronal degeneration, and necrosis. Lesions were much more frequent and severe in the caudal portion of the brain, especially in the cerebellum (Fig. 5), than in the middle and cranial portions. In the 10 mice from which spinal cords were available for examination, pathologic changes qualitatively similar to those described for the brain were present in all mice. Lesions in the spinal cord were much milder than in the brain.
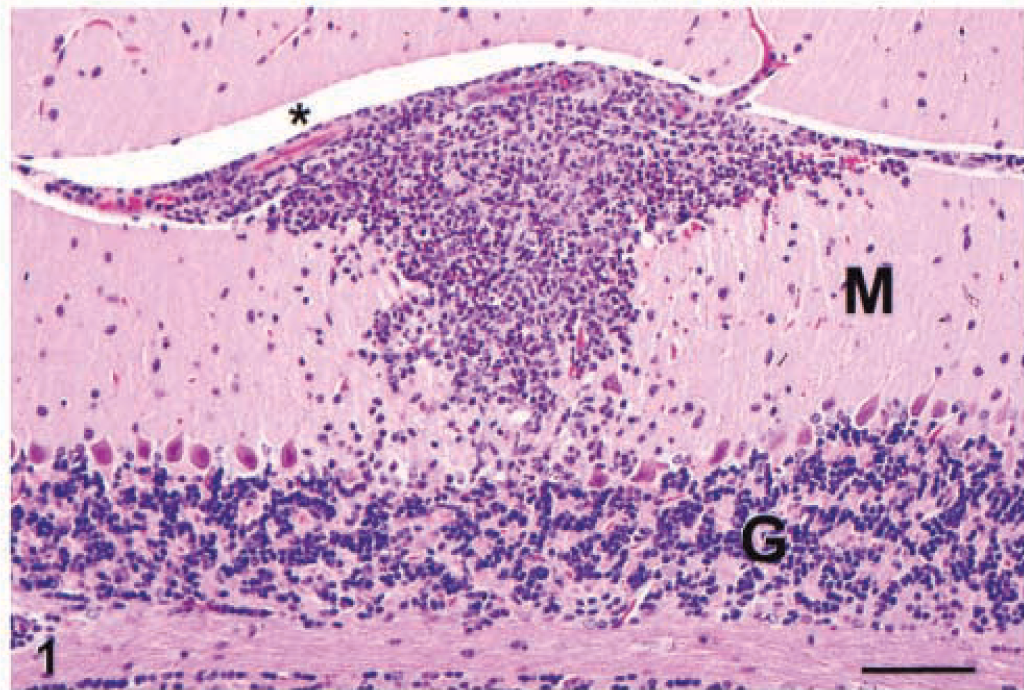
Cerebellum; IFN-γ gene KO mouse. The inflammatory cell infiltrate extends from the pia mater in the intrafolial sulcus (asterisk) into the molecular layer (M). The granular layer is labeled (G). HE. Bar = 80 µm.
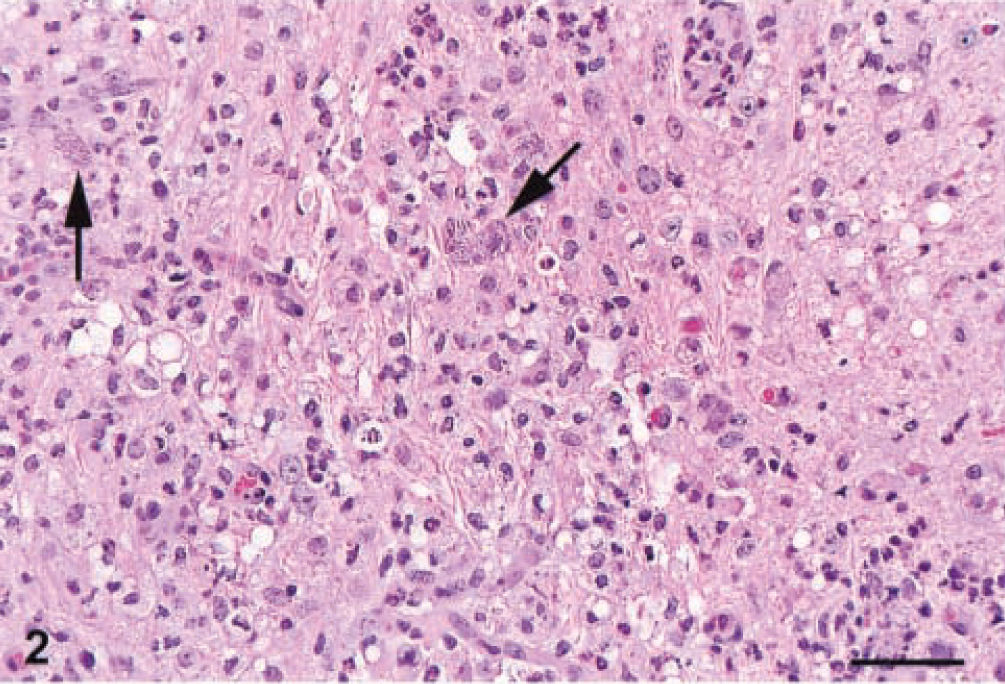
Cerebrum; IFN-γ gene KO mouse. An infiltrate of neutrophils, macrophages, and few eosinophils accompanied by mild spongiosis. Two unidentified infected cells (arrows) contain schizonts. HE. Bar = 30 µm.
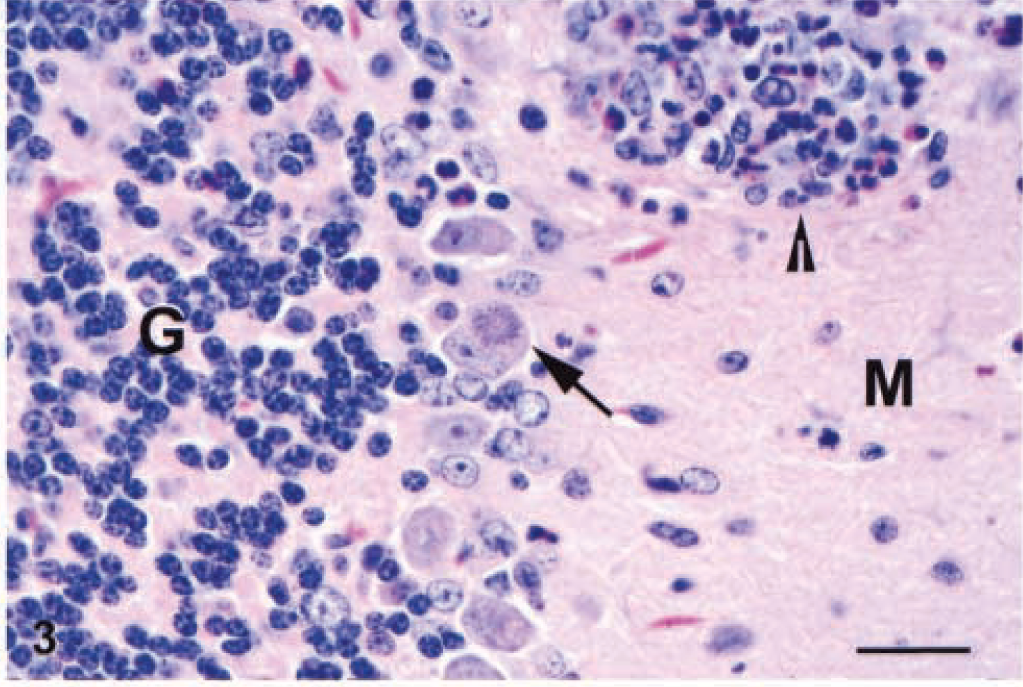
Cerebellum; IFN-γ gene KO mouse. A Purkinje cell contains a schizont with radially arranged merozoites (arrow). An inflammatory cell aggregate at the top of the field contains numerous eosinophils (arrowhead). The granular (G) and molecular (M) layers are labeled. HE. Bar = 40 µm.
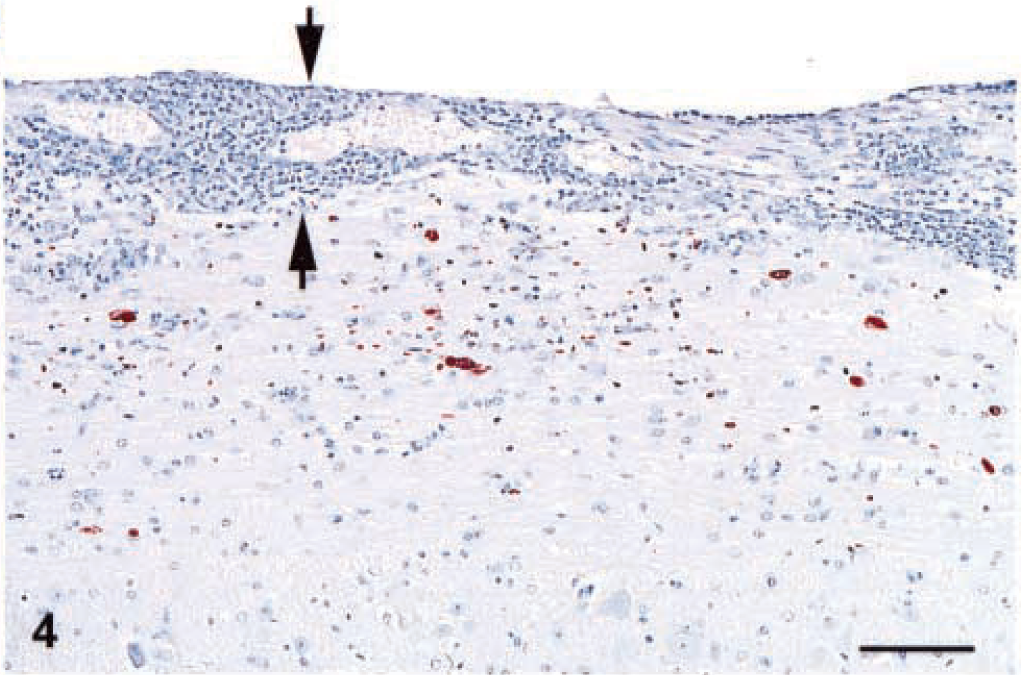
Spinal cord; IFN-γ gene KO mouse. The brown aggregates are immunoreactive S. neurona. Note that the inflammatory cells infiltrate in the meninges (between arrows) and neuropil. Envision™ system, rabbit anti-S. neurona serum. Mayer's hematoxylin counterstain. Bar = 80 µm.
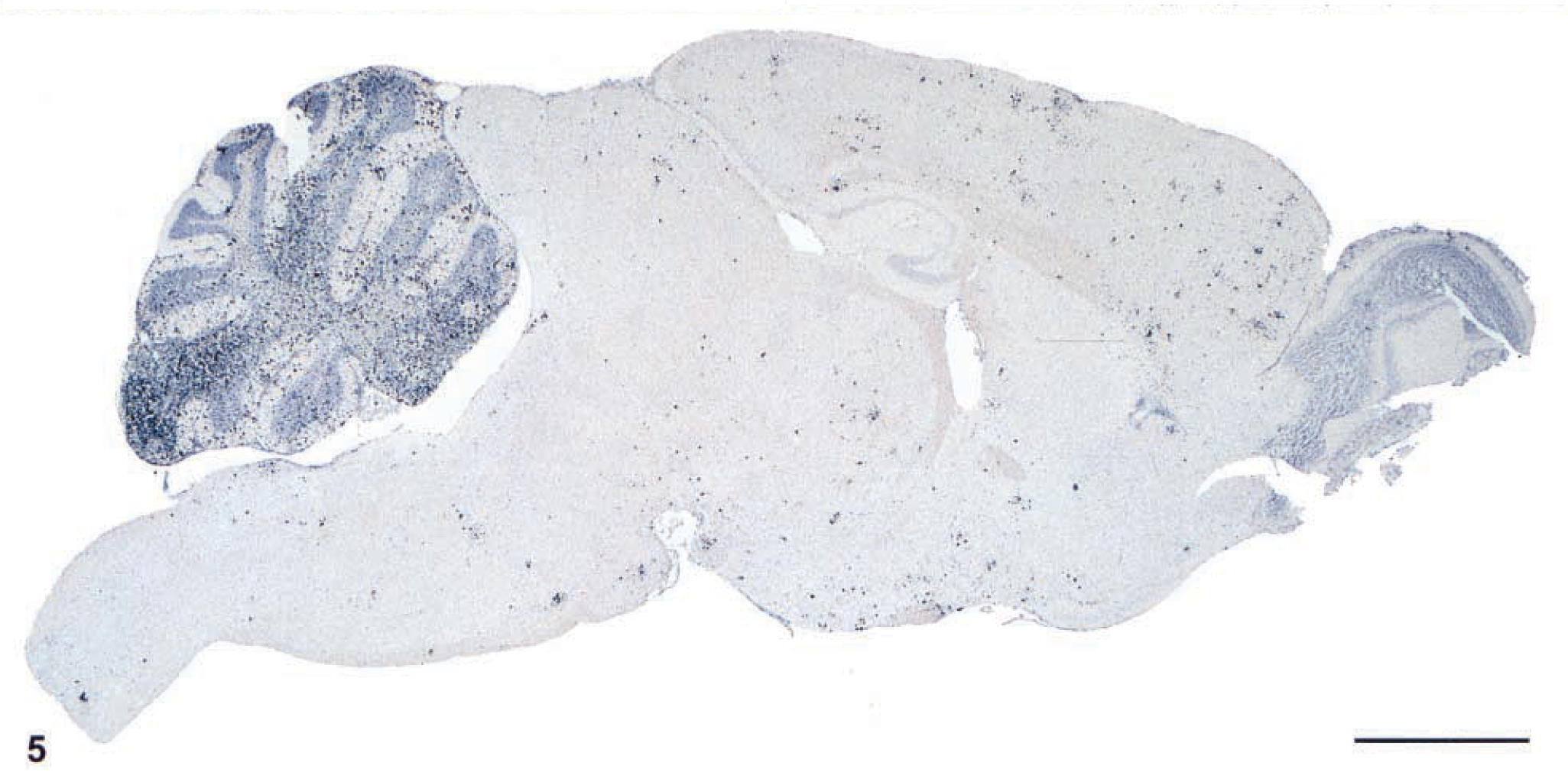
Brain; IFN-γ gene KO mouse. Note the concentration of immunoreactivity in the cerebellum as compared with other areas of the brain. Envision™ system, rabbit anti-S. neurona serum. Mayer's hematoxylin counterstain. Bar = 100 µm.
Schizonts and merozoites were usually present in macrophages or neutrophils and less frequently in glial cells and neurons (Fig. 3). Within the cerebellum, infected cells were most frequently seen within the granular layer. Schizonts and merozoites were evident in the long cell processes in the molecular layer as well. We presumed that many of these infected cells were the small neurons of the molecular layer (stellate cells). Purkinje cells were only occasionally infected (Fig. 3).
Histopathologic changes related to S. neurona infection were also noted in tissues other than the CNS. In the lung, mild granulomatous pneumonia and subacute perivasculitis were present in 14 of 21 mice examined. A very mild infiltrate of neutrophils was present in the subretinal choroid of the eye of seven mice (of 21 examined). Moderate lymphoid hyperplasia and mild lymphocytolysis (presumed to be apoptosis) was present in the splenic white pulp of all 21 mice examined. Finally, disseminated small aggregates of macrophages were present in the livers of 21 of 21 mice examined, but protozoa were never identified. In the five uninfected control mice, the only histopathologic changes present included bronchiolar-associated lymphoid tissue in two mice, subacute portal hepatitis in four mice, and subacute otitis externa in one mouse.
Immunoreactive schizonts and merozoites were present throughout the brain (Fig. 5) and spinal cord (Fig. 4) and usually were associated with the inflammatory cell infiltrates. Scattered infected host cells were often seen in otherwise normal areas of brain and spinal cord. Immunoreactive schizonts and merozoites were also present within the granulomatous inflammation in the lung. Finally, rare immunoreactive schizonts and merozoites were present focally in the retinas of several mice.
By TEM, merozoites were identified mostly within the cytoplasm of neutrophils and macrophages and less frequently in eosinophils (Fig. 6), oligodendroglial cells, astrocytes, and neurons. In neurons, merozoites were most frequently seen in the perikaryon but only occasionally in axons (Fig. 7). Protozoa were always free within the cytoplasm of the host cell. In addition to merozoites, schizonts in all stages of development were identified.
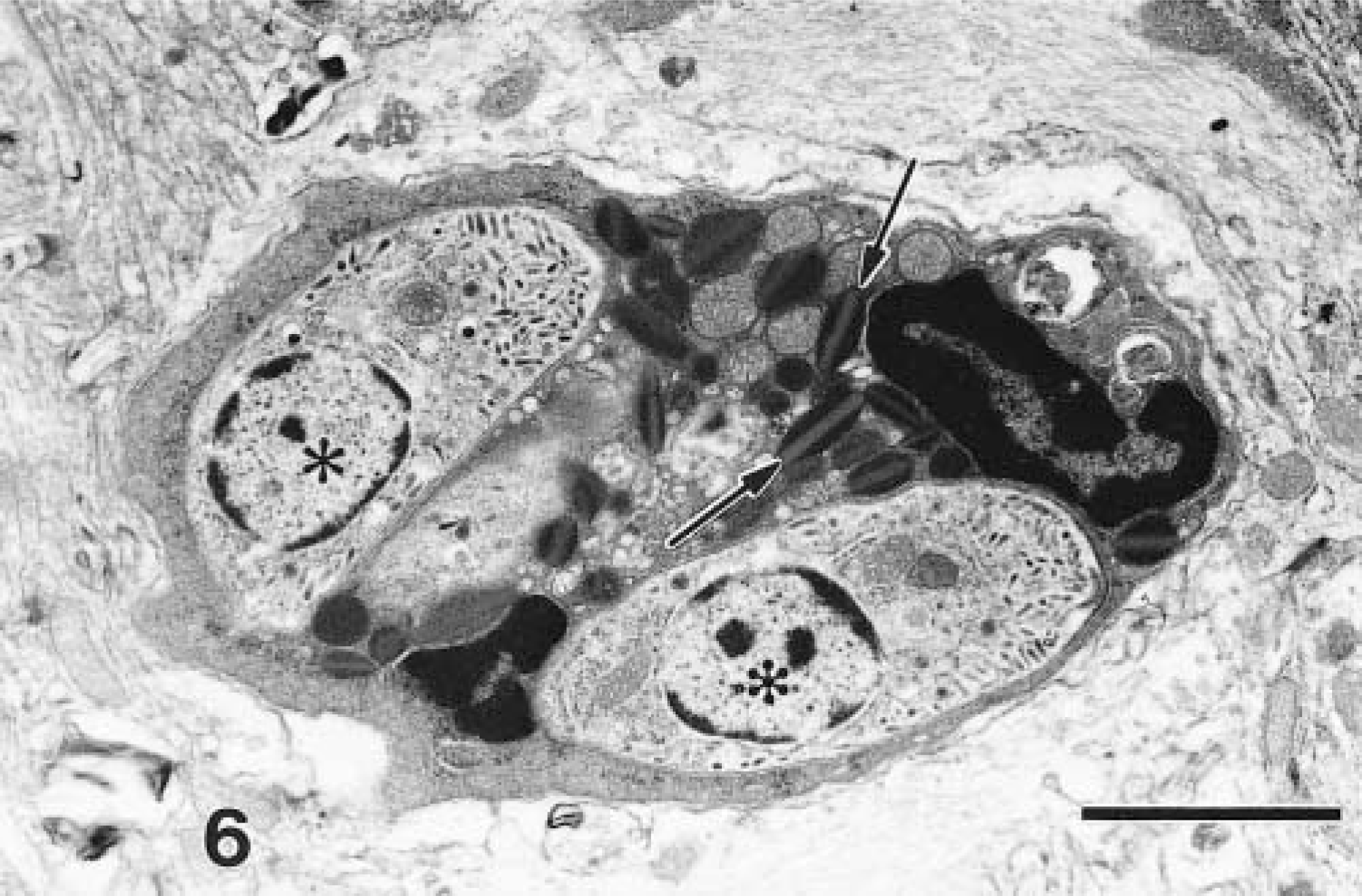
Cerebrum; IFN-γ gene KO mouse. An eosinophil with characteristic granules (arrows) contains two merozoites (asterisks). Lead citrate, uranyl acetate poststain. Bar = 2 µm.
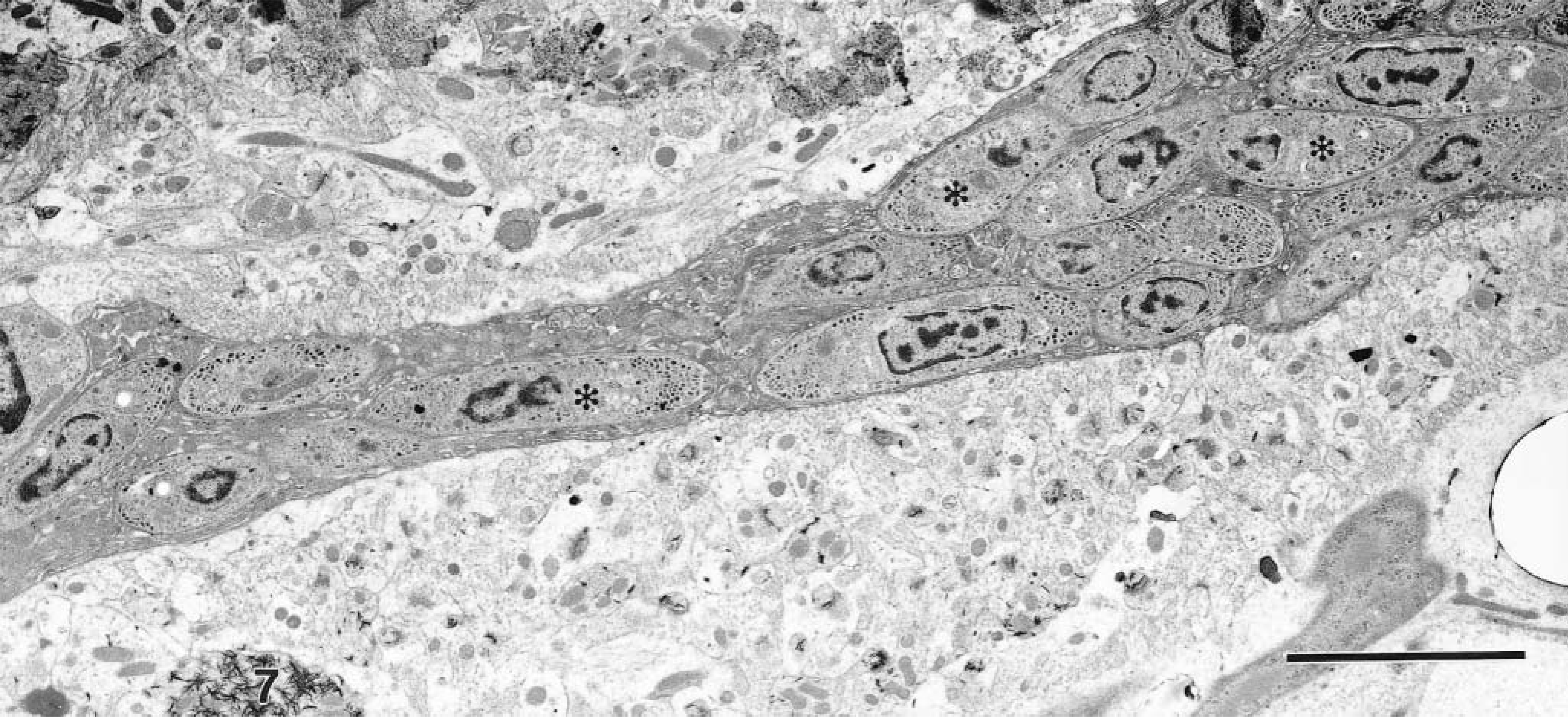
Cerebellum; IFN-γ gene KO mouse. The axoplasm of this unmyelinated nerve axon is completely filled by numerous merozoites (asterisks). Lead citrate, uranyl acetate poststain. Bar = 5 µm.
The extent of the S. neurona-related lesions in the lungs of some of the mice was somewhat surprising, but organisms were clearly immunolabeled in these areas of granulomatous inflammation. Because the mice were given the sporocysts by stomach tube, it is not likely that the organisms could have been aspirated.
Histopathologic changes in the spleen were probably reactive changes in response to the infection. The changes in the liver were only slightly more extensive than similar lesions present in control mice, but these changes were probably also reactive. Immunohistochemistry showed no protozoa in spleens or livers of any of the mice. However, this may be a time-related phenomenon because spleens and livers are infected during the first 2 weeks after feeding sporocysts. 3
Although the IFN-γ gene KO mouse is probably ideally suited for a variety of studies such as the one we describe, not all aspects of the disease processes of EPM in horses are the same as in KO mice. At the present time, EPM cannot be reliably produced in any other hosts including equids. 8 The IFN-γ gene KO mouse has proven to be an exceedingly useful model for EPM with regard to testing therapeutics 6 and will possibly further our knowledge about host response to this protozoal agent.
Footnotes
Acknowledgements
We thank Lynda Miller, Neil Davis, Jeff Brubaker, Kathy Kuehl, D'Angelo Austin, and Larry Ostby for technical support. Finally, we also thank Drs. Kelly Davis, Mark Martinez, and Nate Powell, and Ms. Katheryn Kenyon for their critical review of this manuscript. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the U.S. Army.
Research was conducted in compliance with the Animal Welfare Act and other Federal statutes and regulations relating to animals and experiments involving animals and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, 1996. The facility where this research was conducted is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
