Abstract
Expression of p27Kip1 was identified in feline lymphoid tissues by immunohistochemistry. In normal lymphoid tissues, p27Kip1 was detected as a distinct nuclear stain in lymphocytes of the follicular mantle zone and interfollicular small lymphocytes, whereas activated lymphoblasts in the germinal center were negative. Lymphoid hyperplasia was similarly immunolabeled but with an expanded mantle zone and marginal zone of p27Kip1-reactive lymphocytes. Both T- and B-cell lymphomas lacked p27Kip1 immunolabel and were determined to be proliferative based on immunohistochemical detection of the Ki-67 antigen. Scattered p27Kip1- immunolabeled lymphocytes were detected throughout the lamina propria of most specimens characterized as lymphoplasmacytic enteritis. The results of this study suggest that the antiproliferative effect of the cell cycle regulator p27Kip1 is abrogated in feline lymphoma, presumably allowing cells to bypass the G1-S checkpoint of the cell cycle.
Keywords
Cell cycle progression is a complex process governed by cyclin-dependent kinases (CDKs) that are regulated by several mechanisms, including binding to regulatory subunits (cyclins), phosphorylation and dephosphorylation, and binding to CDK inhibitors (CDKIs). 2,12,15 Aberrant expression of cell cycle regulators has been frequently identified in human neoplasms. Regulators with high expression in cancer that promote proliferation include G1 cyclins and cdc25 phosphatases, whereas negative regulators of cell cycle progression are frequently functionally inactivated by mutations or deletions or reduction of protein products. 18 The CDKI protein p27Kip1 regulates the G1-S transition of the cell cycle. 7,17 p27Kip1 is present in large amounts in quiescent cells and in response to differentiation signals, and the levels decline when cells proliferate in response to mitogenic signals. 13 P27Kip1 expression is regulated primarily at the posttranscriptional level; p27Kip1 mRNA levels are constant during the cell cycle, but the protein is degraded rapidly in resting cells by the ubiquitin–proteasome pathway. 17
G1-S transition defects in the cell cycle are considered critical in tumor development; loss of p27Kip1 does not appear to be a primary cause of cancer, but reduced expression probably accelerates cancer progression. 9,12 p27Kip1 is expressed in quiescent lymphocytes within lymphoid tissues and peripheral blood, whereas activated lymphocytes are negative. 13 In human non-Hodgkin's lymphoma, there is an inverse relationship between p27Kip1 expression and cellular proliferation. Further, p27Kip1 expression has independent prognostic implications in non-Hodgkin's lymphoma. 5 Because p27Kip1 has been considered a tumor suppressor gene, we studied its expression by immunohistochemistry in feline lymphoid tissues and lymphomas.
Materials and Methods
Tissues
Paraffin-embedded tissues were obtained from the archives of the pathology laboratory of the University of California Veterinary Medical Teaching Hospital. Tissues selected represented normal (healthy) lymphoid tissues, including lymph node and gut-associated lymphoid tissue (GALT), reactive lymphoid hyperplasia associated with both infectious and immunologically mediated processes, intestinal biopsy specimens representing lymphoplasmacytic enteritis, and lymphoma. Lymphoma specimens included lymph nodes and tissues from a variety of sites infiltrated with neoplastic lymphocytes.
Immunohistochemistry
P27Kip1 expression was determined by immunohistochemistry. Tissue sections (5 µm) were deparaffinized and cleared, and endogenous peroxidase activity was blocked with a 3% solution of hydrogen peroxide and methanol. Sections were then hydrated in graded alcohols to distilled water. Microwave-induced (100 C) antigen retrieval was done in citrate-buffered solution for a total of 4 minutes (two 2-minute cycles). Slides were cooled to room temperature and placed in a humidified chamber. A solution of 10% normal horse serum was applied for 20 minutes. After blotting, the primary antibody p27 (clone DCS72, Oncogene Science, Cambridge, MA) was applied at a dilution of 1:400 and incubated overnight at 4 C. After rinsing in phosphate buffered saline (PBS), horse anti-mouse biotinylated antibody (Vector Laboratories, Burlingame, CA) was applied at 1:800 dilution and incubated for 60 minutes at room temperature. Two PBS rinses were followed by a 30-minute incubation with avidin–biotin complex (Vector Laboratories). Color development was achieved by application of the chromagen diaminobenzidene (Vector Laboratories), and brown stain was visualized after 2–3 minutes by light microscopy. Slides were rinsed in tap water, counterstained in Mayer's hematoxylin, dehydrated, cleared, and coverslipped. Negative controls were prepared on duplicate sections from each tissue by following all immunostaining procedures except that the primary antibody was omitted and replaced with a purified murine serum IgG, diluted in the same way as the primary correlate. Positive controls were paraffin-embedded sections of a human p27-reactive lymph node. Specimens were considered abnormal if more than 40% of cells showed loss of nuclear staining.
Immunohistochemistry for detection of T lymphocytes (CD3), B lymphocytes (CD79), and Ki-67 antigen was done on formalin-fixed, paraffin-embedded tissues using methods similar to those described for p27Kip1. For lymphocyte markers, the sections were immunostained with primary antibody (monoclonal rat CD3, Vector Laboratories; HM57, Dako Corp., Carpinteria, CA), biotinylated secondary antibody, streptavidin–peroxidase reagent, and 3-amino-9-ethylcarbazol as chromagen. For Ki-67, the primary antibody was MIB-1 (Oncogene Science), biotinylated secondary antibody, alkaline phosphatase–labeled streptavidin, and red chromagen (Fast Red, Biogenex, San Ramon, CA).
Results
Peripheral and mesenteric lymph nodes from four normal (healthy) cats were examined. The p27Kip1 immunolabeling was easily visualized as a distinct nuclear stain in lymphocytes. In lymph nodes, there was staining of lymphocytes in the follicular mantle zone and in interfollicular small lymphocytes, whereas proliferating activated lymphoblasts in germinal centers were negative (Fig. 1). There were occasional scattered positive cells within the germinal centers. CD3 stains of adjacent sections suggested that these cells were predominately T lymphocytes. The pattern of expression was opposite that seen with the MIB-1 antibody (Fig. 2). There was diffuse staining of interfollicular lymphocytes for CD3, corresponding to p27Kip1-immunolabeled regions of the lymph nodes.
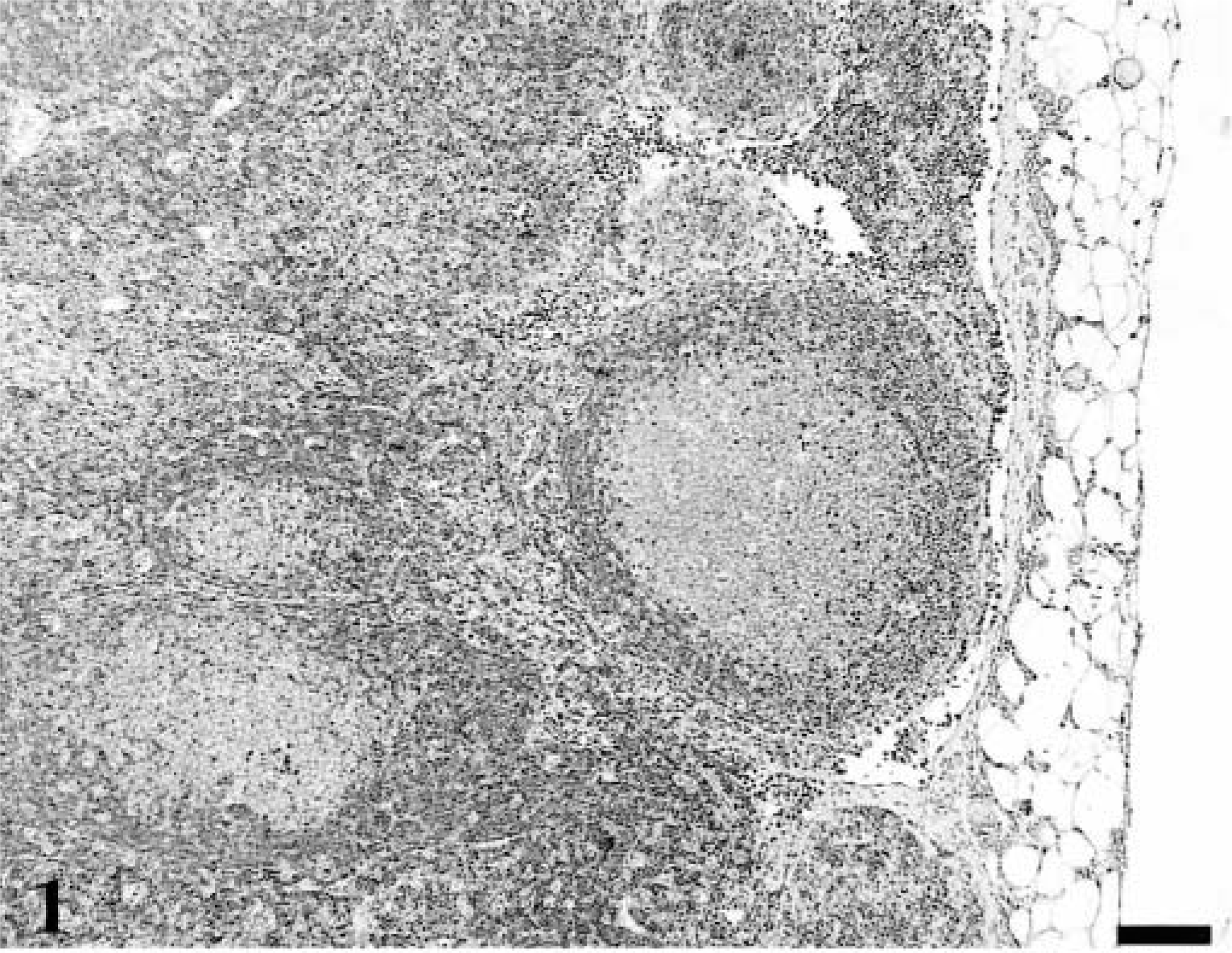
Lymph node; healthy adult cat. P27Kip1-immunolabeled lymphocytes are in the follicular mantle zone and in interfollicular small lymphocytes, whereas cells of the germinal center are negative. Avidin–biotin complex/diaminobenzidene–peroxidase, hematoxylin counterstain. Bar = 100 µm.
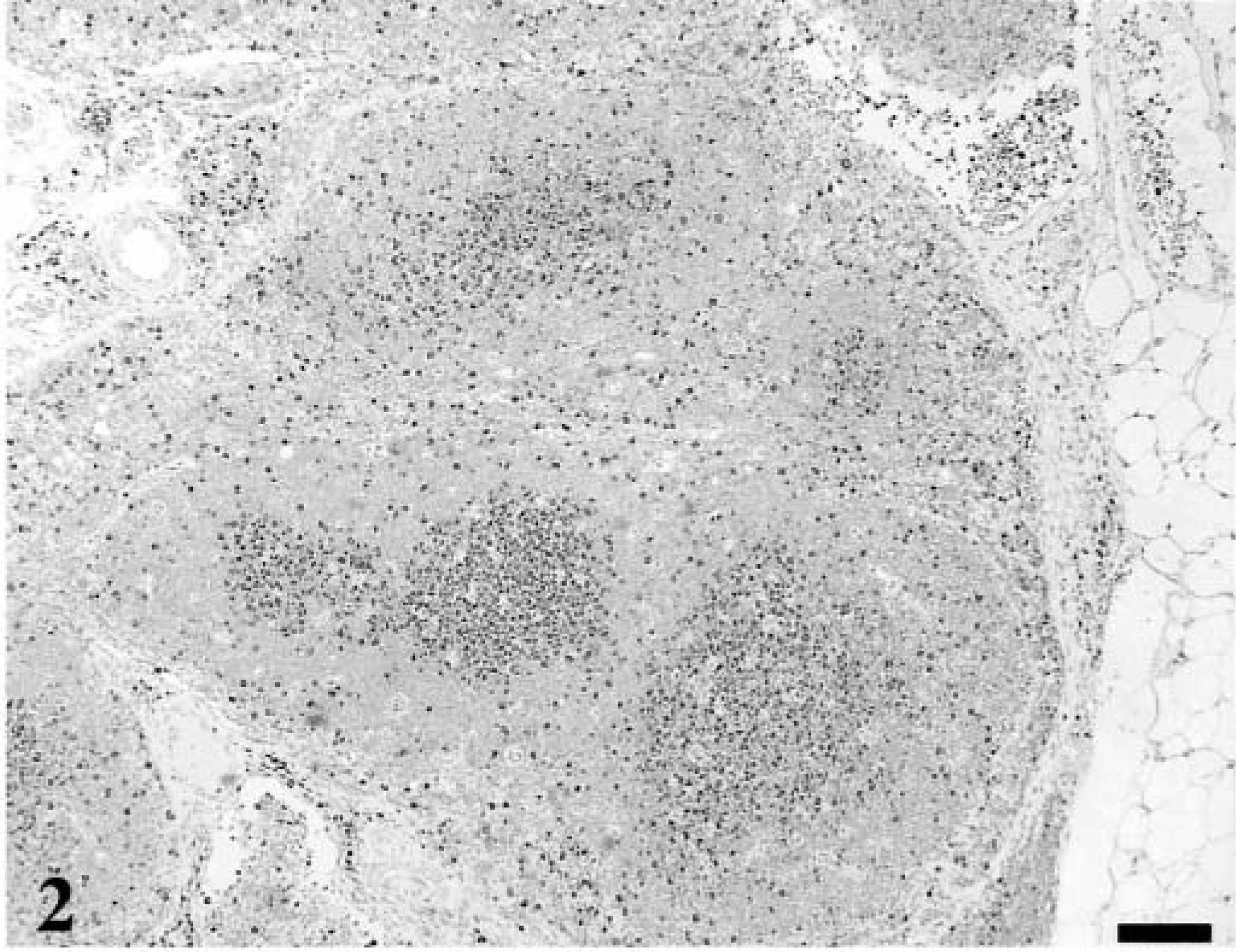
Lymph node; healthy adult cat. A section adjacent to that depicted in Fig. 1 stained for the proliferation antigen Ki-67 with MIB-1 antibody. The pattern of Ki-67 immunoreactivity was opposite that seen with the p27Kip1 stain. Biotin–alkaline phosphatase–labeled streptavidin/fast red, hematoxylin counterstain. Bar = 100 µm.
Eleven lymph node specimens representing hyperplasia from 11 separate cats were examined. The reactivity of lymphoid components in hyperplastic nodes from all cats was similar to that described for the normal lymph node but with expanded mantle and marginal zone of p27Kip1-reactive lymphocytes and MIB-1–reactive cells in germinal centers.
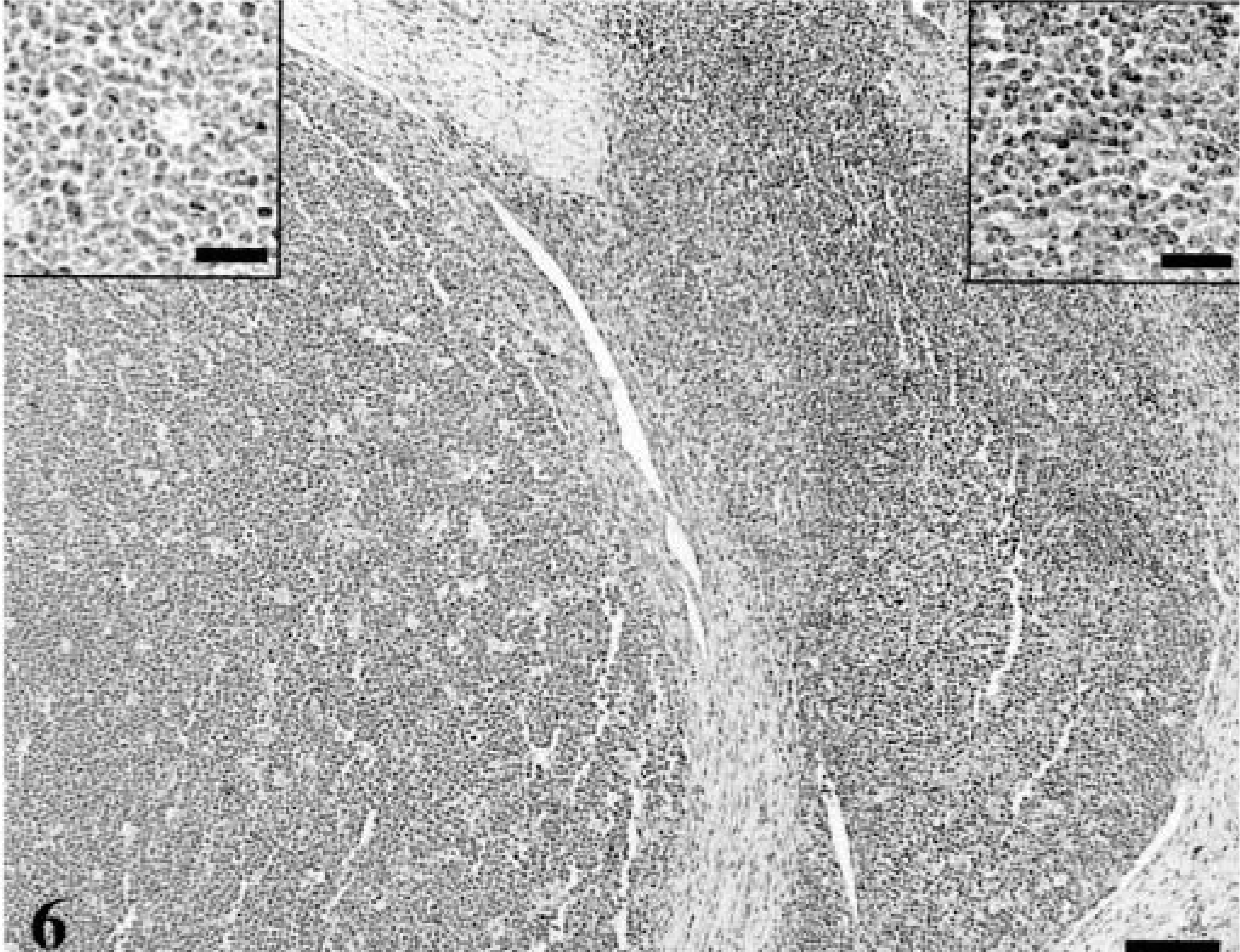
Forty lymphomas were examined. Of 39 tumors immunophenotyped, 15 were of T-cell origin and 24 were derived from B cells. Feline lymphoma cells, both T-cell and B-cell variants, were almost universally negative for p27Kip1, although there were occasional intermingled cells that were immunolabeled; these were small lymphocytes, whereas the large lymphoid cells were devoid of immunoreactivity. In each case, there was high MIB-1 labeling index (Figs. 3–5). In many of the gastrointestinal specimens, normal GALT or reactive lymphoid hyperplasia was visualized adjacent to sheets of neoplastic lymphoid cells (Fig. 6). In those specimens, reactive lymphoid hyperplasia contained the same follicular architecture with strong p27Kip1 immunolabel in the folliclular mantle and perifollicular lymphocytes but no p27Kip1 immunolabel in germinal centers of reactive follicles or within the sheets of neoplastic lymphoid cells (Figs. 7, 8).
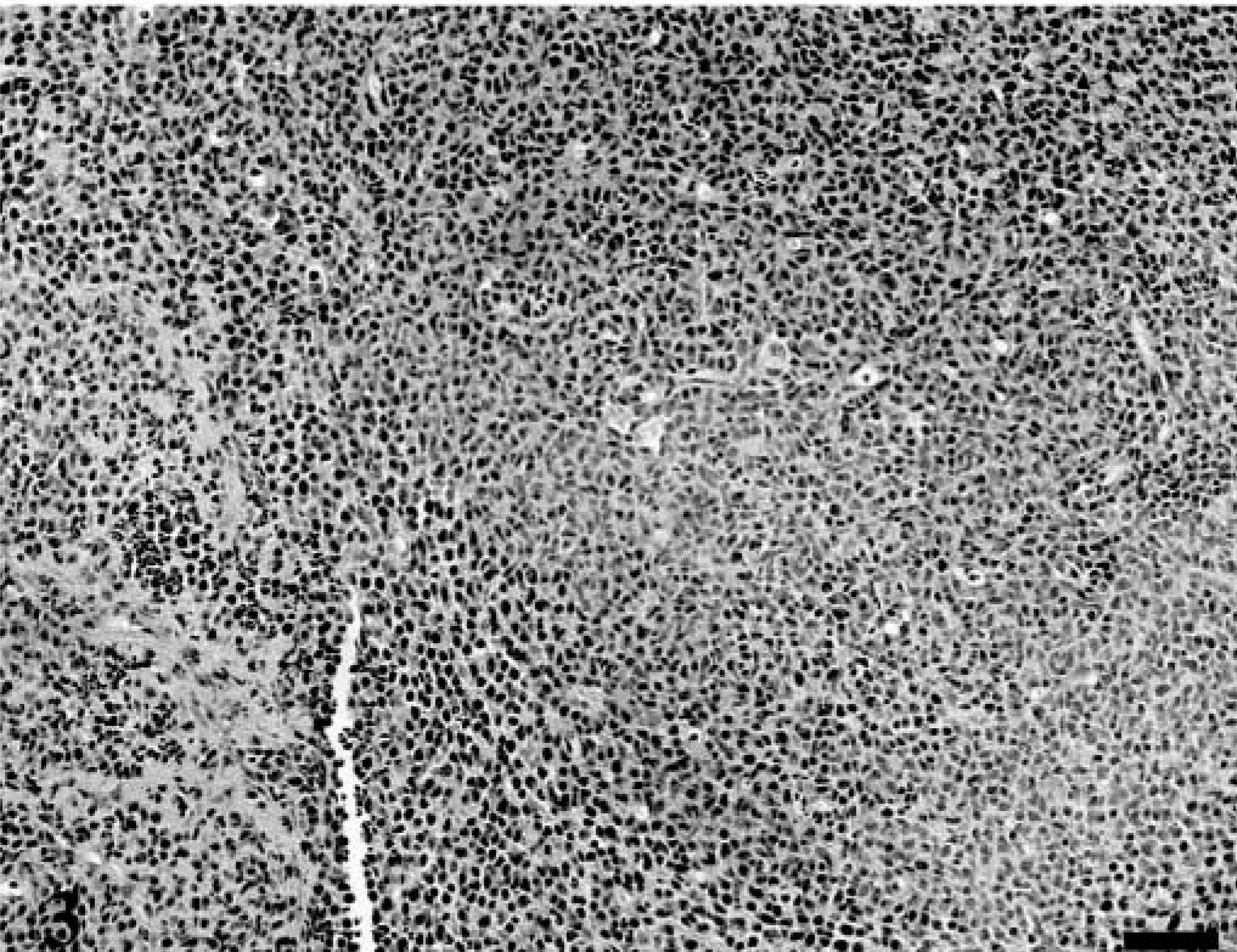
Mandibular lymph node; 15-year-old cat with lymphoma. The lymph node architecture is effaced by sheets of large lymphocytes. HE. Bar = 200 µm.
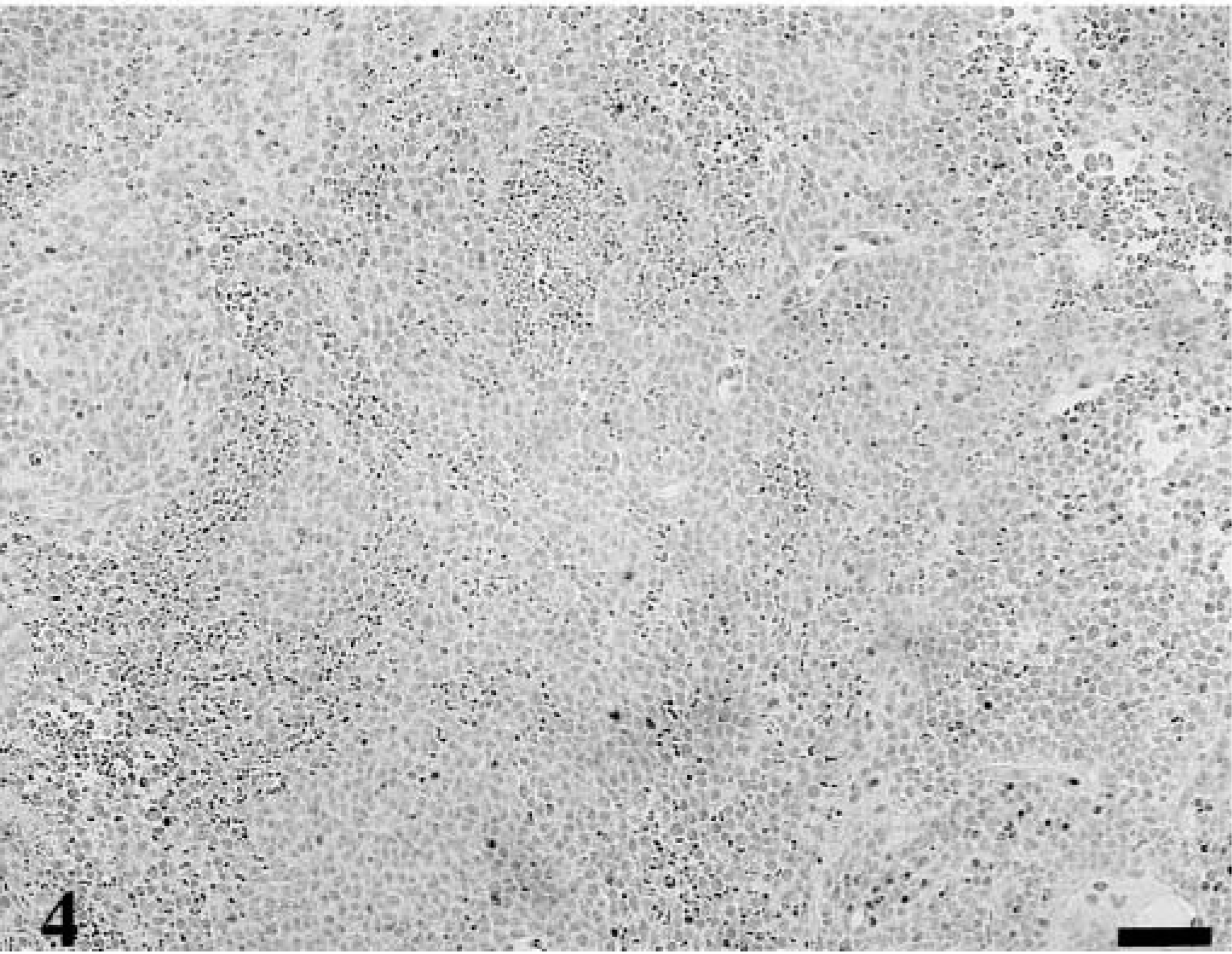
Mandibular lymph node; 15-year-old cat with lymphoma. A section adjacent to that depicted in Fig. 3 stained for p27Kip1. Tumor cells are negative for immunolabel, but stained for B-cell markers HM57 and B222 (other markers not shown). Avidin–biotin complex/diaminobenzidene–peroxidase, hematoxylin counterstain. Bar = 200 µm.
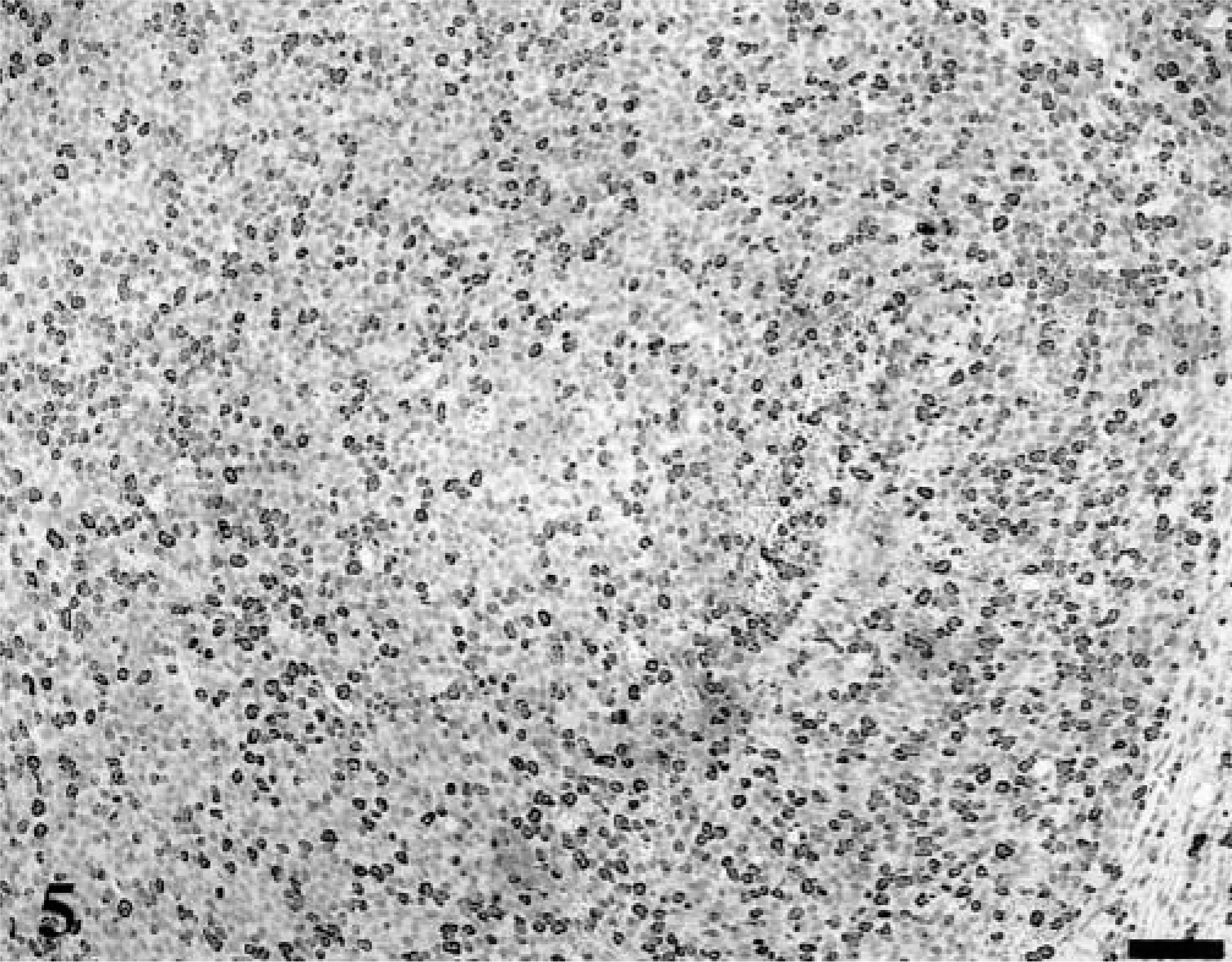
Intestine; 3-year-old cat with lymphoma. The ileum is massively infiltrated with neoplastic lymphocytes (left) that lack p27Kip1 immunolabel, whereas primary follicles adjacent to the tumor (right) are labeled. Avidin–biotin complex/diaminobenzidine–peroxidase, hematoxylin counterstain. Bar = 100 µm. Insets: Left. There is no p27Kip1 immunolabel in neoplastic lymphocytes. Bar = 25 µm. Right. Reactive lymphocytes show immunolabel. Bar = 25 µm.
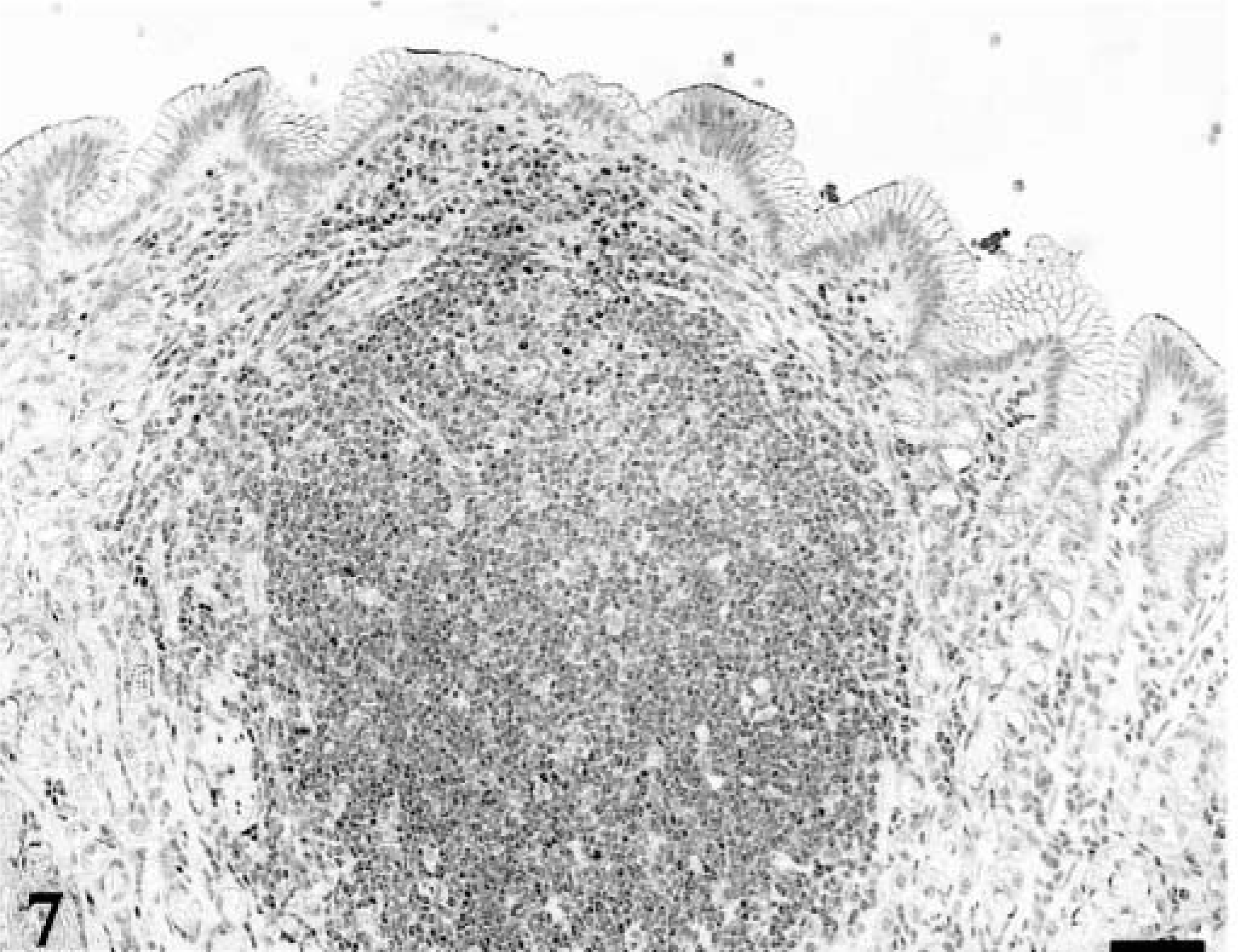
Stomach; 9-year-old cat with gastric lymphoma. This lymphoid follicle from nonneoplastic area represents lymphoid hyperplasia, showing immunostain for p27Kip1. Avidin–biotin complex/diaminobenzidene–peroxidase, hematoxylin counterstain. Bar = 200 µm.
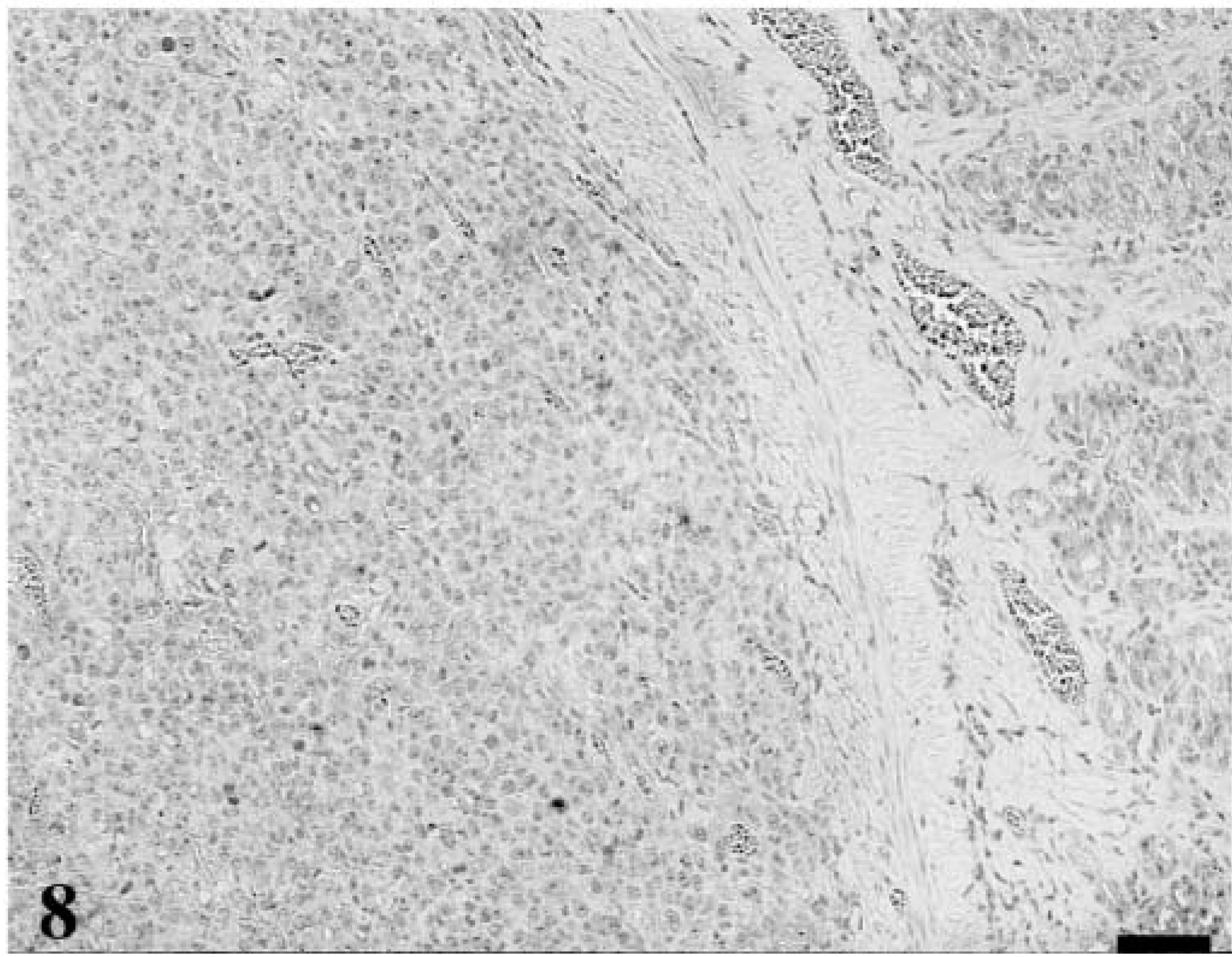
Stomach; 9-year-old cat with lymphoma. This section is adjacent to that depicted in Fig. 7. The neoplastic lymphocytes lack p27Kip1 immunolabel. Avidin–biotin complex/diaminobenzidene–peroxidase, hematoxylin counterstain. Bar = 200 µm.
Ten specimens characterized as enteritis or colitis (lymphoplasmacytic) by examination of hematoxylin and eosin (HE)-stained tissue sections generally showed p27Kip1-immunolabeled lymphocytes diffusely scattered throughout the intestinal villi, lamina propria, and submucosa. Associated lymphoid structures showed p27Kip1 immunolabel similar to that of reactive lymphoid hyperplasia. However, in two specimens representing enteritis, infiltrating lymphocytes were devoid of p27Kip1 immunoreactivity, but organized lymphoid tissues within these specimens showed characteristic immunolabel within follicular mantle zones.
Discussion
The results of this study suggest that reduced expression of p27Kip1 is a characteristic feature of feline lymphoma, regardless of site of origin. Tissues characterized histologically as lymphoma, of either B- or T-cell origin, were almost universally negative for p27Kip1, and those cells immunoreactive for p27Kip1 within lymphoma tissues were small lymphocytes that stained for CD3 on adjacent tissue sections. MIB-1 immunolabeling confirmed that cells lacking p27Kip1 were able to proliferate, and most lymphomas examined showed high MIB-1 labeling index. In contrast, most tissues characterized histologically as lymphoplasmacytic enteritis showed diffusely scattered p27Kip1-stained lymphocytes as part of the inflammatory millieu within the lamina propria, suggesting that immunoreactivity for p27Kip1 might allow distinction in some cases between intestinal lymphoma and inflammatory bowel disease. This distinction was not absolute, however; several intestinal specimens considered to represent lymphoplasmacytic enteritis, based on microscopic features of the HE-stained sections, were devoid of p27Kip1 immunolabel.
Neoplastic growth is the result of cell production minus cell loss. Degradation of p27Kip1 expression may be an important event in the pathogenesis of feline lymphoma because it impairs the normal control of cell proliferation, thus enhancing growth and tumor development. P27Kip1 is one of the inhibitor genes within the Cip/Kip family. It is regulated by specific growth factors, such as transforming growth factor-beta. 14 In addition to its role as a CDKI, p27Kip1 is a putative tumor suppressor gene. Loss of p27Kip1 allows tumor cells to bypass the G1-S checkpoint by removing inhibition of G1 cyclin–CDK complexes. 13 P27Kip1 and other Cip/Kip family proteins are considered universal CDKIs because they interact with various CDK complexes, with cyclins A, E, D1, D2, and D3, and with CDKs. 9 In humans, p27Kip1 protein expression decreases during tumor development and progression in some epithelial, lymphoid, and endocrine tissues. The decreased expression occurs mainly at the posttranslational level, with protein degradation by the ubiquitin–proteasome pathway. 9 Genomic alterations of p27Kip1 are extremely rare in human cancers, 12 and no p27Kip1 mutations were detected in a variety of feline tumors, although lymphoma was not represented by those feline tumors examined. 11
The role of other cell cycle regulatory proteins in the pathogenesis of feline lymphoma is unclear. The bcl-2 protein is expressed in some feline lymphomas, suggesting that dysregulation of cell death may also play a role in lymphoma pathogenesis. 10 In those tumors that show bcl-2 expression, the protein is presumed to block apoptosis and contribute to tumor growth by prolonging cell survival. 6 It may also render those cells resistant to chemotherapy. 16 The complexity of these relationships is underscored further by the observation that p27Kip1 promotes apoptosis. 8
Therapeutic strategies for cats with lymphoma might emerge from more precise characterization of cell cycle events. For example, there is strong upregulation of p27Kip1 in Epstein-Barr virus immortalized (human) lymphoblastoid cell lines that are exposed to retinoids; that upregulation has an antiproliferative effect that is not dependent upon terminal differentiation of the immortalized lymphocytes. 3 Glucocorticoids inhibit proliferation by inducing cell cycle lengthening, and this inhibition may be mediated in part by increased p27Kip1 expression. 1 Interferon-alpha, which has antitumor activity in T cell malignancies, abrogates mitogen-induced reduction of p27Kip1 levels in vitro by maintaining the stability of the p27Kip1 protein. 4
Footnotes
Acknowledgements
This study was supported by a DuBee Cancer Award, administered by the Center for Companion Animal Health, School of Veterinary Medicine, University of California, Davis.
