Abstract
The
Eosinophilia develops in reactive diseases such as allergic and parasitic diseases, some malignant diseases, and in idiopathic hypereosinophilic syndrome (HES). 1,11,14 The eosinophilia is accompanied by a wide variety of inflammatory lesions because toxic inflammatory mediators are released from eosinophils. 1,11,14 In particular, systemic and chronic hypereosinophilia, as in HES, is known to result in severe organ damage. 1,14 HES is a systemic disorder defined as chronic hypereosinophilia accompanied by significant organ system dysfunction without apparent etiology. It has been reported in humans, ferrets, cats, horses, and dogs. 1,3–6,9,10,14 However, details of the pathology and pathogenesis of HES are unclear.
In 1999, Matsumoto et al. found mutant rats with spontaneous eosinophilia that were derived from pregnant specific pathogen free Slc:Sprague-Dawley (SD) rats. This colony was maintained by brother-sister matings and was named the Matsumoto Eosinophilia Shinshu (
In this study, 38 male and 38 female
In the clinical pathology examinations, apparent increases in blood eosinophils were found at 8 weeks of age (Table 1). Subsequently, numbers of blood eosinophils increased with age, and the degree of increase in females was greater than that in males. The nuclei of eosinophils were generally coiled to form a ringlike or figure-eight pattern, and the granules were normal and acidophilic. Blasts and abnormal eosinophils were not detected on blood films. There were no differences in blood IgE levels between the
Blood eosinophils and eosinophilic lesions in
Mean values.
Percent eosinophils.
Not found.
No. rats with lesions.
Histopathologically, eosinophilic proliferation and infiltration in the bone marrow, gastrointestinal tract, mesenteric lymph node, and spleen was found in some rats initially at 8 weeks of age. After 10 weeks of age, eosinophilic proliferation in the bone marrow was found in all rats. The bone marrow was hypercellular primarily because of eosinophilic proliferation. All maturation stages were present, but mature eosinophils predominated. Infiltration of mature eosinophils was found severely in the red pulp of the spleen; the lamina propria, submucosa, and muscularis of the gastrointestinal tract; and the medulla of the mesenteric lymph nodes in most rats. It was also found in the lung, thymus, liver, and uterus of some rats. After 12 weeks of age, proliferations of erythroblasts and immature eosinophils in the red pulp of the spleen were found in many rats, and the incidence and severity of these findings increased age dependently. HE staining revealed normal eosinophils were typical morphology in each stage.
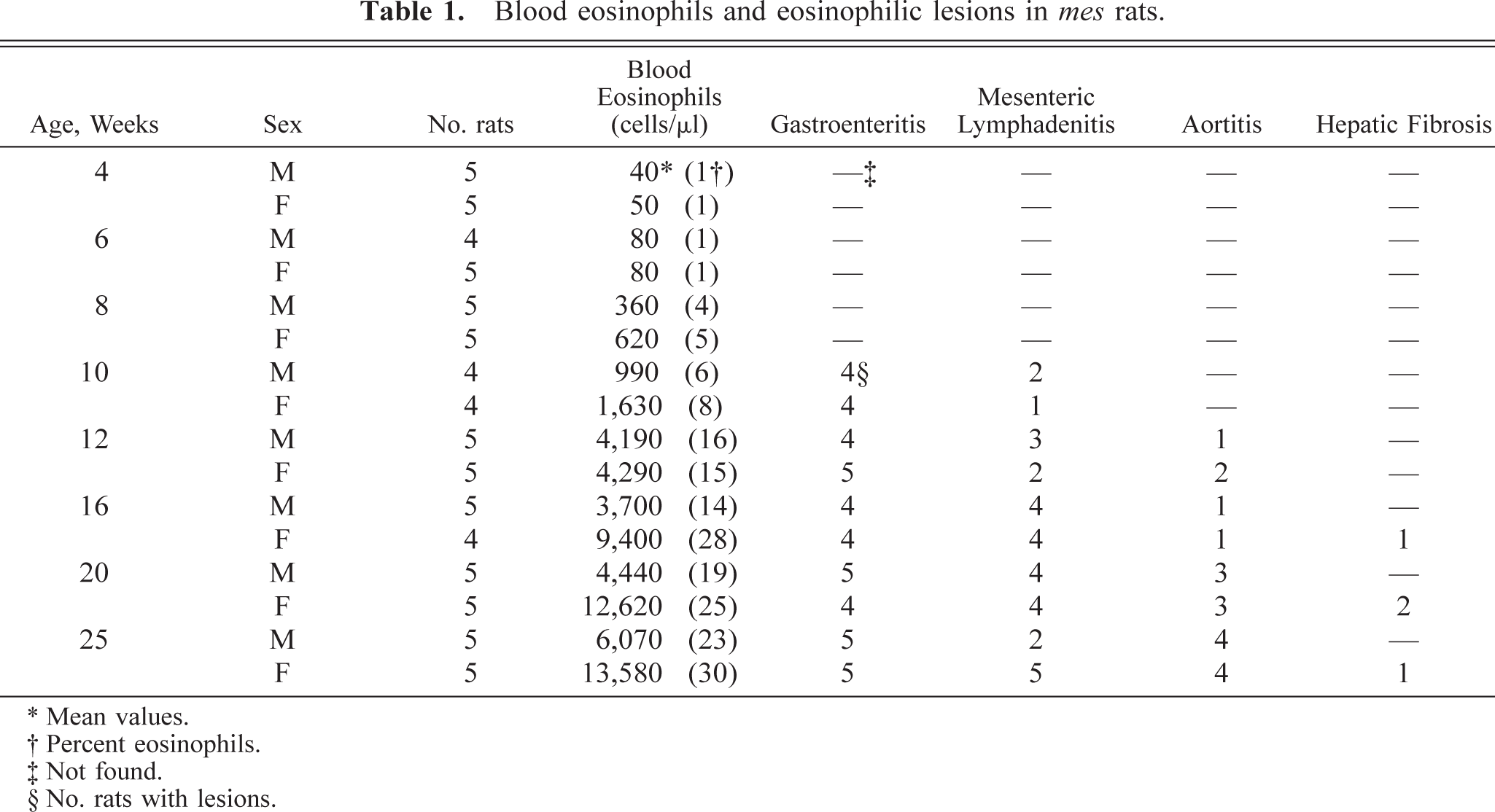
In the gastrointestinal tract, diffuse fibrosis with inflammatory response was found in the lamina propria, submucosa, and muscularis in 44 of 47 rats after 10 weeks of age (Table 1, Fig. 1); it was prominent in the lamina propria of the stomach and the muscularis in the jejunum and ileum. Fibrosis was accompanied by infiltration of numerous eosinophils and, occasionally, deposition of granular eosinophilic material associated with macrophages. Additionally, focal granulomas in the lamina propria and submucosa of the stomach were found in 8 of 47 rats (Fig. 1). The granulomas were composed of degenerated eosinophils and homogeneous or granular eosinophilic material surrounded by macrophages. Eosinophilic infiltration often disrupted the normal architecture, atrophy, and compensatory metaplasia of the gastric glands, and ulceration was confirmed in the more severe cases (Fig. 1).
Glandular stomach; 20-week-old male rat. Fibrosis and granuloma consisting of eosinophils and eosinophilic materials in the lamina propria and submucosa; ulceration and eosinophil infiltration in the lamina propria and submucosa. HE stain. Bar = 60 µm.
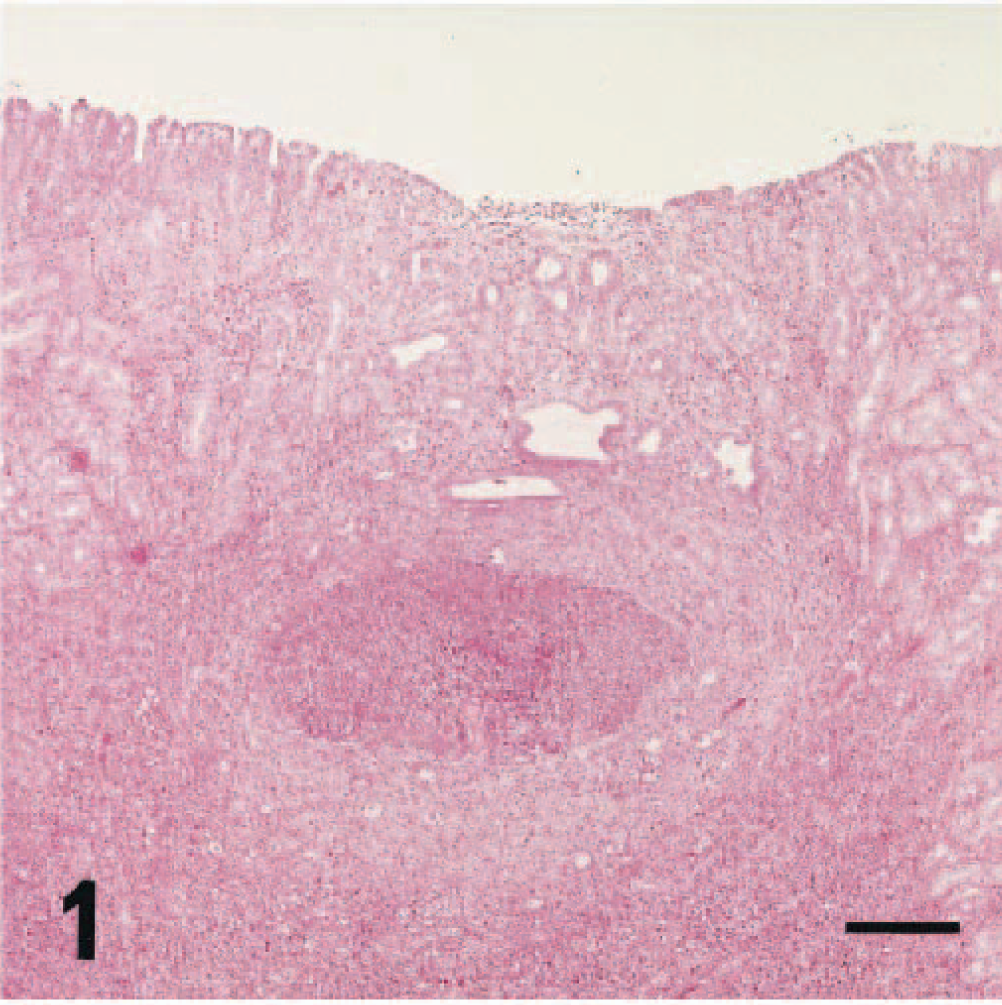
In the mesenteric lymph nodes, multifocal granulomas in the medulla were found in 31 of 47 rats after 10 weeks of age (Table 1, Fig. 2). The granulomas were composed of degenerated eosinophils and homogeneous or granular eosinophilic materials surrounded by macrophages and multinucleated giant cells. Additionally, infiltrations of numerous plasmacytes and macrophages in the paracortex were found in most rats after 8 weeks of age, and the severity correlated with the degree of macroscopic enlargement.
Mesenteric lymph node; 20-week-old male rat. Granuloma consisting of eosinophils and multinucleated giant cells in the medulla. HE stain. Bar = 10 µm.
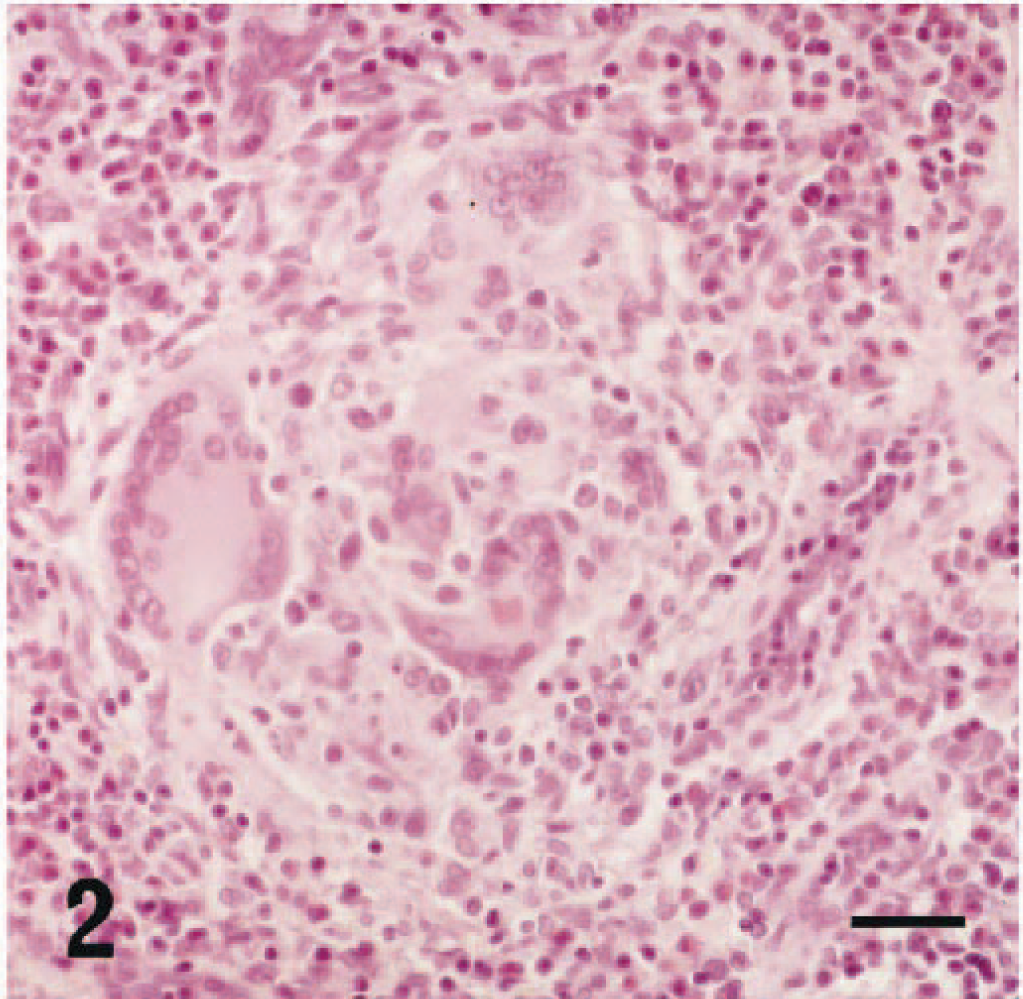
In the thoracic aorta and pulmonary artery, inflammation characterized by thickening of the intima was found in 19 of 39 rats after 12 weeks of age (Table 1, Fig. 3), and the incidence and severity of these findings increased age dependently. Eosinophilic infiltration was limited to both the intima and adventitia. Furthermore, in the thoracic aorta, deposition of bandlike eosinophilic material associated with macrophages and multinucleated giant cells accumulated between the intima and media (Fig. 3). In the liver, centrilobular fibrosis and periportal eosinophil infiltration were found sporadically in four rats that exhibited severe eosinophilia and anemia (Table 1, Fig. 4). Centrilobular eosinophil infiltration was not present. In the lung and thymus, granulomas similar to those described in the mesenteric lymph nodes were found sporadically in some rats.
Thoracic aorta; 25-week-old male rat. Thickening of the intima, eosinophil infiltration at both the intima and adventitia, and deposition of eosinophilic material between the intima and media. HE stain. Bar = 25 µm.
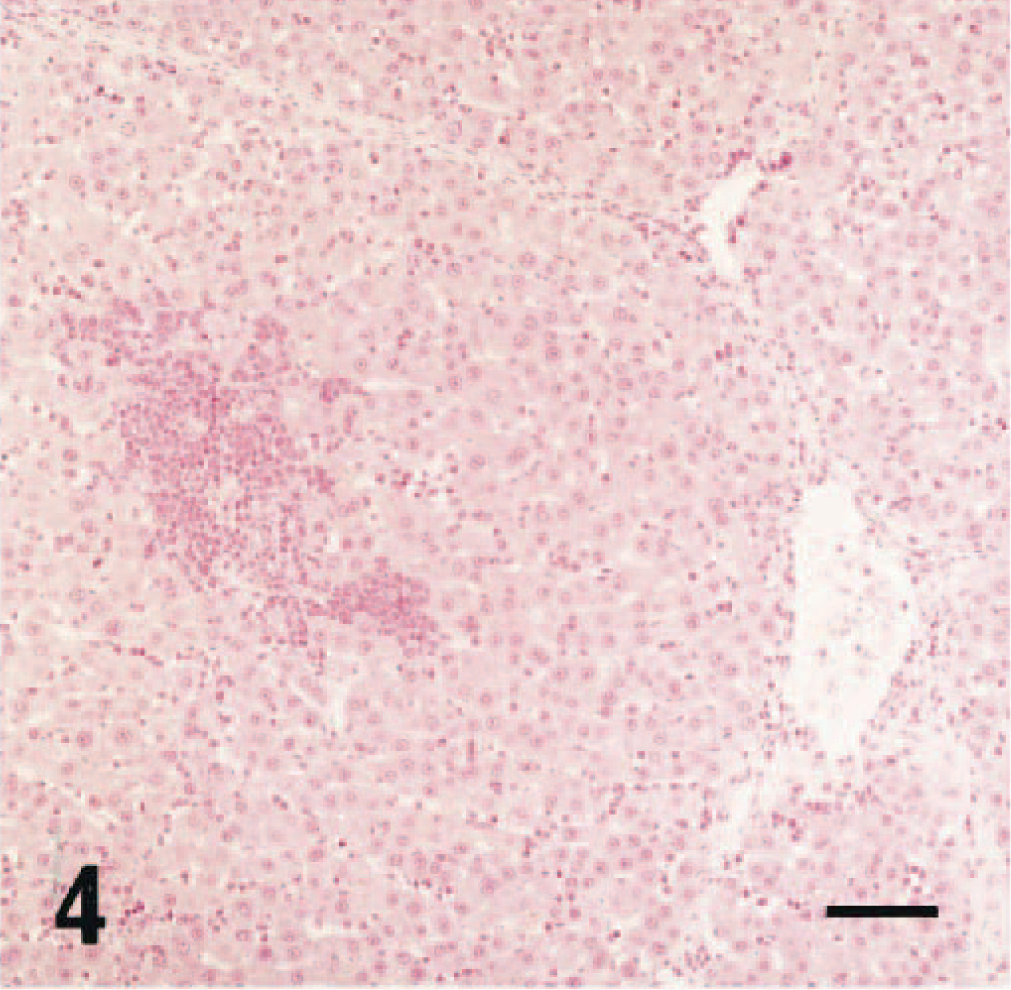
Liver; 20-week-old female rat. Centrilobular fibrosis and periportal eosinophil infiltration. HE stain. Bar = 25 µm.
When sections of the gastrointestinal tract, mesenteric lymph nodes, and thoracic aorta were stained with Giemsa stain and PAS stain, the eosinophilic material was pink with Giemsa stain and reacted positively with PAS stain. Organisms were not detected in Gram, Giemsa, and PAS staining.
The onset of systemic eosinophilia in the
Gastroenteritis, mesenteric lymphadenitis, and aortitis are thought to be eosinophil-derived inflammatory lesions because deposition of eosinophilic material was found in the lesions. This eosinophilic material is thought to be Splendore-Hoeppli material because the characteristics are similar to those described in ferrets. 3 It is suggested that inflammation is caused by the cytotoxicity of mediators such as the eosinophilic granule proteins that are released from the eosinophils. 11,14 In particular, the diffuse forms of gastroenteritis and mesenteric lymphadenitis are similar to the lesions described in ferrets, cats, horses, and dogs. 3,4,9,10
Many eosinophilic inflammatory lesions were found in the
The relationship between the hepatic fibrosis and the eosinophilia is unclear because the fibrous lesion was not correlated with eosinophil infiltration. Hepatic fibrosis may be induced by anemia because it was only found in rats showing severe anemia. These specific increases of blood eosinophils and mesenteric lymphocytosis and plasmacytosis suggest the participation of interleukin-5 (IL-5) in the pathogenesis of the eosinophilia in
From these results, it is concluded that
Footnotes
Acknowledgements
We thank M. Uehara and K. Matsumoto for their technical assistance.
