Abstract
Formalin-fixed paraffin-embedded archival tissues of seven adult cats of both sexes
that died after being experimentally infected with
Keywords
In this study, archival tissues from seven cats with experimentally induced fatal plague
were used to characterize the histopathology in affected organs. These seven cats were
selected from 20 cats exposed to
Materials and Methods
Bacterial strain and exposure
All cats and mice were infected with virulent
Animals and tissues
In part of a previous series of experiments, 20 adult cats, both males and females,
were divided into two experimental groups (P. W. Gasper, unpublished material). There
were originally 16 cats in group I, of which six died (38% mortality), and four cats
in group II, of which two died (50% mortality). Gross and histopathologic examination
and bacteriology results revealed that all deaths were a result of the
The animals were obtained from the City of Denver, Animal Control Section, and were
housed in the Laboratory Animal Research Center at Colorado State University. All
animals were handled according to the guidelines and protocols established by the
Colorado State University Animal Care and Use Committee. At the Center, cats were
held for quarantine and conditioning according to standard operating procedures of
the center. On receipt, any cat found to be palpably pregnant or to have a
significant health problem was rejected. Cats that were not pregnant and that were
healthy or had minor treatable problems were held in a limited access room in
individual cages and treated appropriately. All cats retained were vaccinated with a
feline viral rhinotracheitis–calici virus–feline panleukopenia vaccine (FVR-CP,
Pitman-Moore, Lincoln, NE) according to standard protocol. Each cat was treated with
niclosamide (for tapeworm), metronidazole (for
Histopathology
Tissues were fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Blocks were sectioned at 5 µm and routinely stained with hematoxylin and eosin (HE). One infected lymph node section from cat No. 4 was additionally stained with Brown and Hopps Gram stain. Samples from a complete set of tissues from each animal were examined by light microscopy.
Immunohistochemistry
Tissues from two representative cats (No. 2 from group I and No. 7 from group II) and
infected lymph nodes from cat No. 4 were examined immunohistochemically.
Five-micrometer-thick paraffin-embedded tissue sections were prepared on positively
charged microscopic slides (Superfrost/Plus, Fisher Scientific, Pittsburgh, PA). The
sections were deparaffinized and rehydrated. For tissues from cat Nos. 2 and 7, the
staining series following deparaffinization was carried out using an automated cell
staining machine (Version 2.0 P.D. Optimax Automated Cell Staining System Plus,
Biogenex, San Ramon, CA). Tissues were washed between each step using
phosphate-buffered saline and deionized water. All incubations were done at room
temperature. Following incubation with a peroxidase inhibitor and a protein block
(Biogenex), a polyclonal antibody raised against the fraction 1 capsular antigen of
Results
Table 1 provides a summary of the histologic findings of the seven cats that died of experimentally induced plague.
Summary of histopathologic findings∗ in seven cats with experimentally induced plague.
+ = present; 0 = not present; 0 = tissue not examined histologically.
Gross pathology
All orally challenged cats (group I) exhibited marked enlargement (approximately 7–10-fold increase in size) and abscessation of the medial retropharyngeal lymph node. Less consistently involved were the tonsils and submandibular and cranial thoracic lymph nodes. Cat No. 4 had similar changes in the ileocecal lymph node. There was dark reddish discoloration of tissues within 7–20 mm of affected nodes. Focal areas of petechiation were noted throughout all tissues, with larger (5–10 mm) ecchymoses present in the lungs and spleens of these cats.
Enlarged lymph nodes were not palpable in the animals that died following
subcutaneous challenge (group II), although
Histopathology
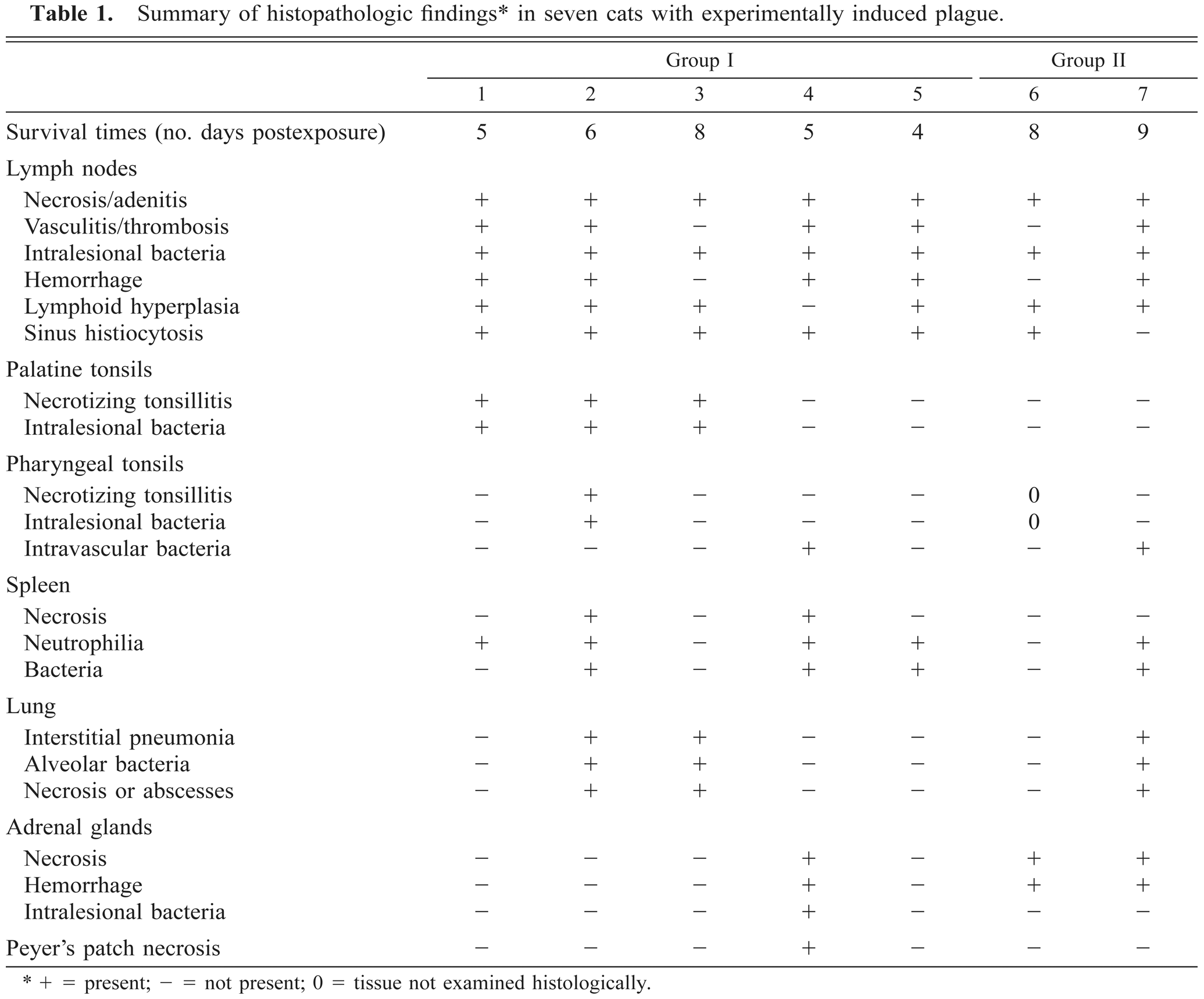
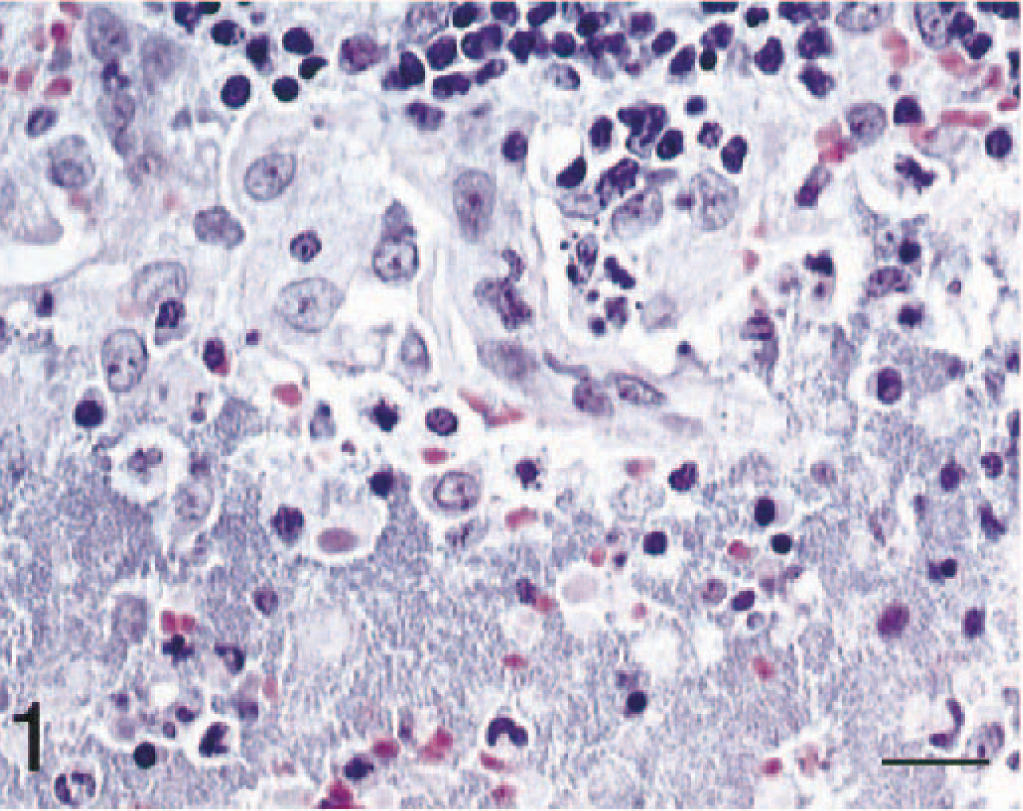
The most consistent changes were in the anterior cervical, medial retropharyngeal, submandibular, and cranial thoracic lymph nodes. All cats had lesions in at least one of these nodes. In most cats, lymph node architecture was markedly altered by acute necrotizing inflammation. Lymph nodes were variably effaced by hemorrhage, edema, and fibrin admixed with eosinophilic cellular and karyorrhectic debris. Numerous viable and degenerate neutrophils and large aggregates of 1–3-µm gram-negative bacilli were present (Figs. 1, 2). Hemorrhage, inflammatory debris, and bacteria often extended beyond the lymph node capsule and infiltrated adjacent fibroadipose tissue. The vascular media of intranodal arterioles frequently was infiltrated by neutrophils and less often was necrotic. There was widespread endothelial necrosis and loss within both venules and arterioles in these lymph nodes. Numerous small venules and fewer arterioles contained fibrinocellular thrombi.
Lymph node; cat No. 4. Massive numbers of bacteria, admixed with macrophages and degenerate and necrotic neutrophils, expand the medullary sinus. HE. Bar = 25 µm.
Lymph node; cat No. 4. Same lymph node as in Fig. 1. Numerous immunolabeled bacteria are present. Streptavidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
Mesenteric lymph nodes in two cats (Nos. 2 and 5) also were acutely inflamed but to a lesser degree than those in anterior cervical areas, and they generally contained fewer bacteria. Often lymph nodes exhibited moderate to marked lymphoid hyperplasia and varying degrees of sinus histiocytosis and draining hemorrhage.
Significant pulmonary lesions were found in three cats, two from group I and one from group II. In the most severely affected lung (cat No. 2), there was a diffuse interstitial pneumonia. Edema, strands of fibrin, and numerous neutrophils and macrophages expanded the alveolar septa, frequently filling alveolar lumina. Alveolar macrophages sometimes contained phagocytosed erythrocytes, necrotic cellular debris, and rarely bacteria. Large aggregates of bacilli were randomly distributed in interstitial spaces and within alveoli (Figs. 3, 4). Multifocally within the lung parenchyma there were coalescing areas of necrosis that obliterated the normal architecture and in some areas formed large abscesses. Abscesses were characterized by a central area of lytic necrosis and degenerate neutrophils, which blended peripherally with numerous bacteria entwined in fibrin and hemorrhage. Within the more severely affected portions of lung, bronchiolar walls were necrotic, and bronchiolar lumina contained hemorrhage, fibrin, necrotic cellular debris, and bacteria. A section of trachea contained hemorrhage and numerous bacteria. Lungs of less severely affected cats had a few scattered focal necrotic areas containing degenerate neutrophils, fewer macrophages, and few bacteria.
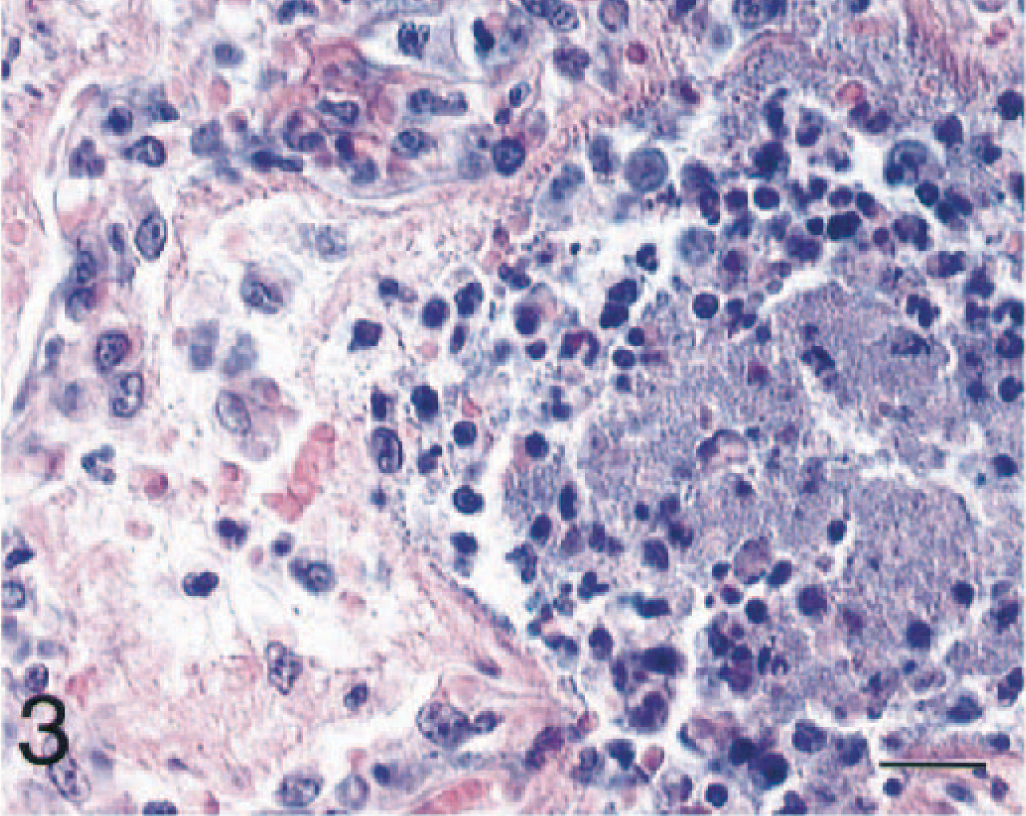
Lung; cat No. 2. The alveolus is filled with numerous bacteria, degenerate and necrotic neutrophils, fewer macrophages, and fibrin. Note expanded alveolar wall (upper left). HE. Bar = 25 µm.
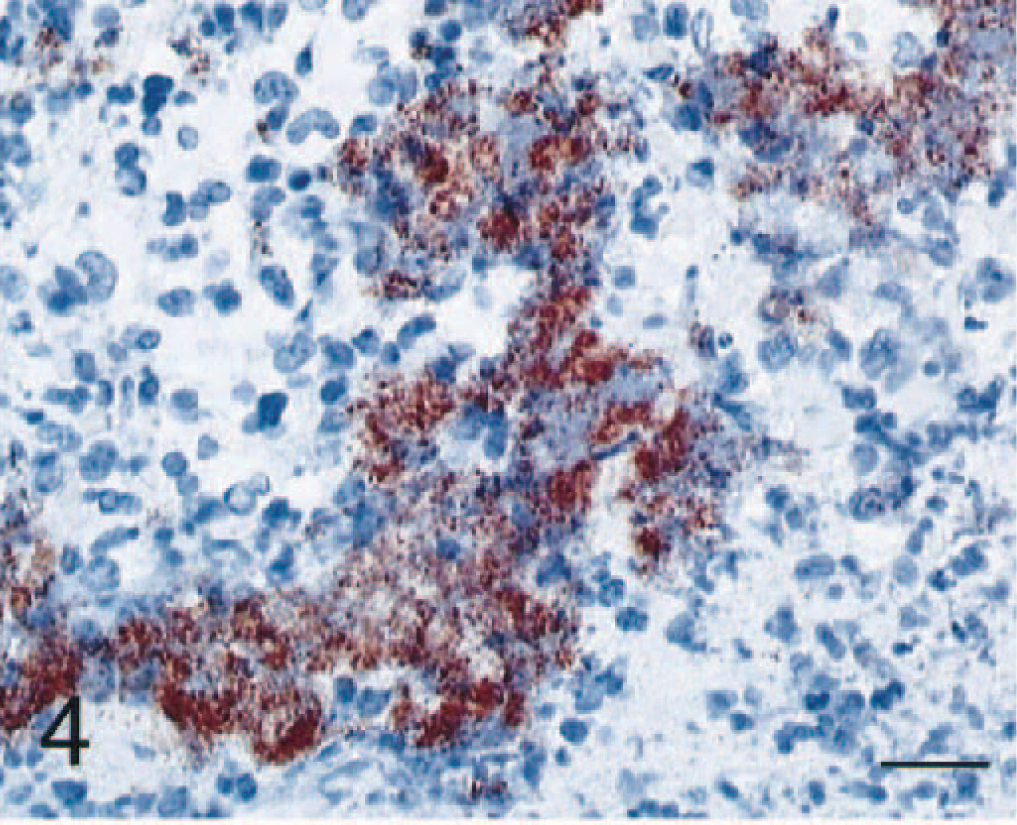
Lung; cat No. 2. Note aggregates of immunolabeled bacteria. Streptavidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
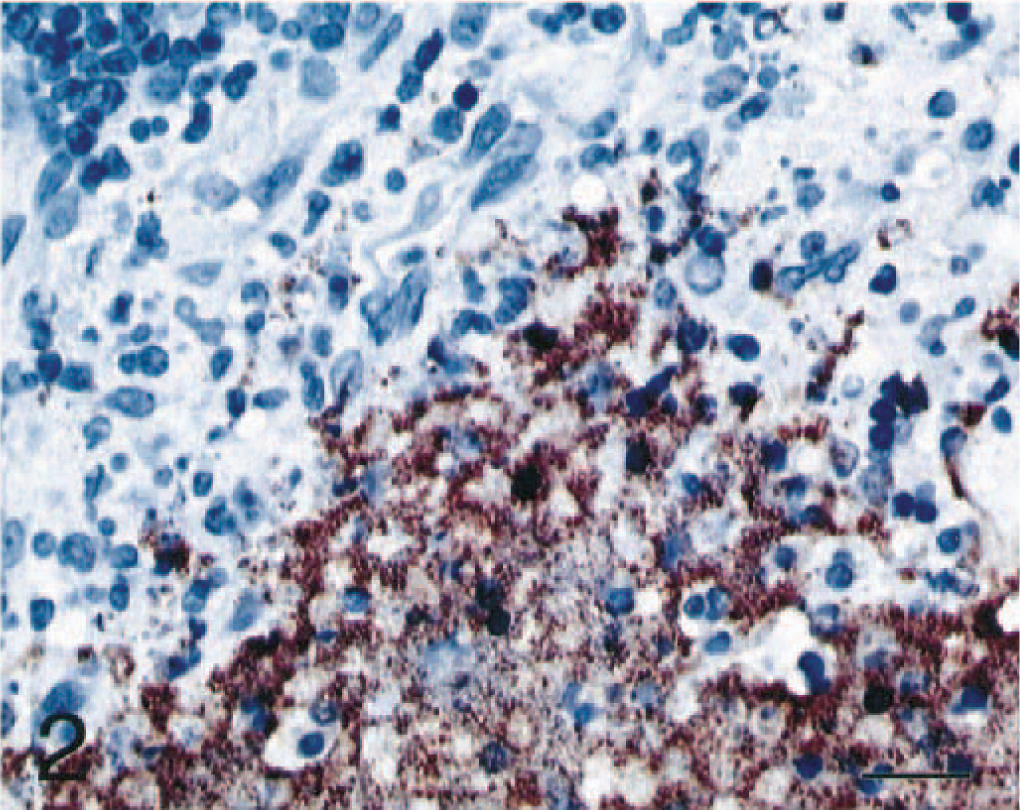
In three cats (Nos. 1–3), severe acute inflammation, widespread necrosis, and large aggregates of bacteria were present in palatine tonsils and, in one cat (cat No. 2), in the pharyngeal tonsils (Figs. 5, 6). Peritonsillar soft tissue was expanded by edema, fibrin, and hemorrhage, and bacteria were present within the perimyseal fascia of adjacent facial skeletal muscle bundles.
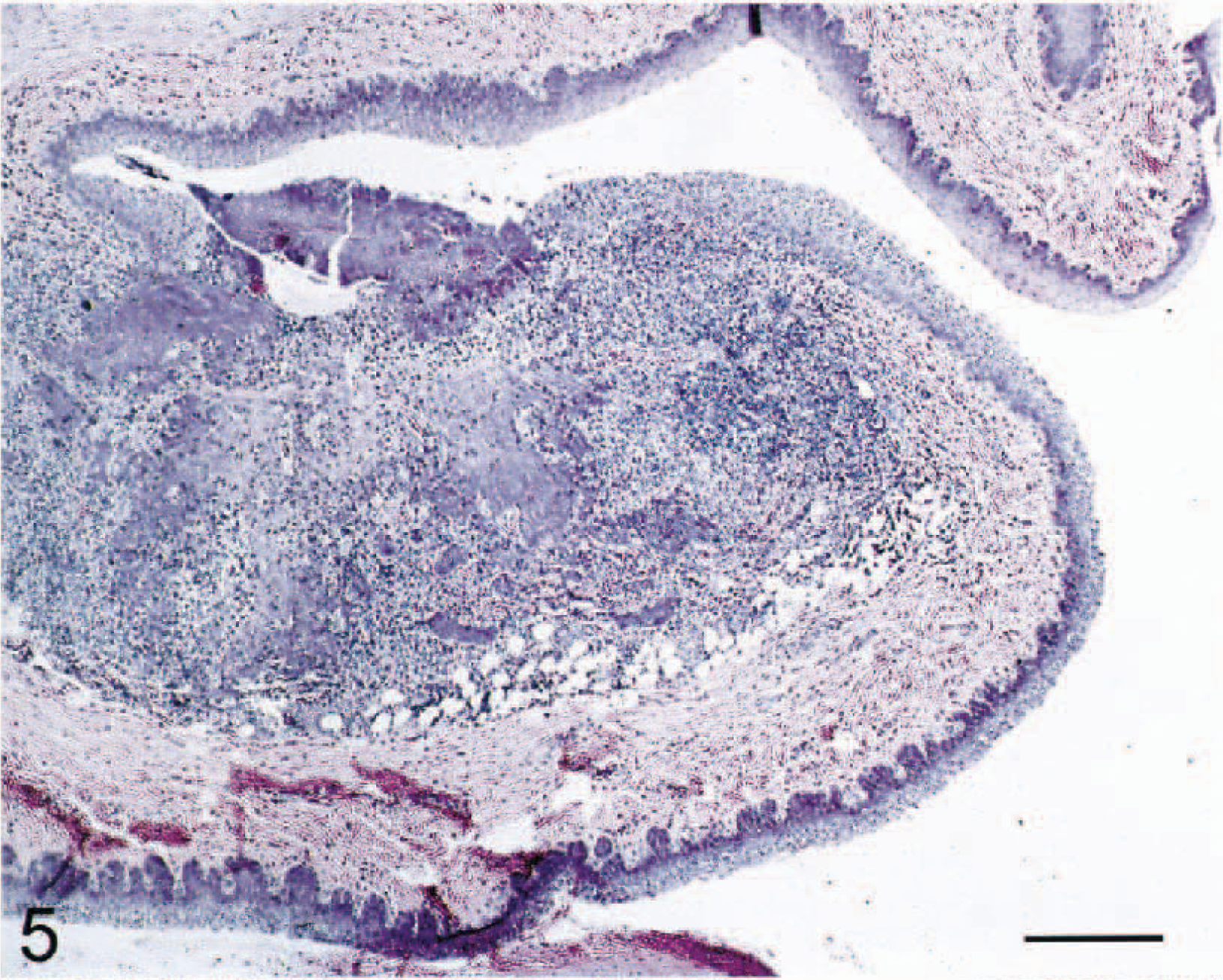
Pharyngeal tonsil; cat No. 2. There is necrosis of lymphoid tissue and disruption of the overlying tonsillar epithelium. HE. Bar = 210 µm.
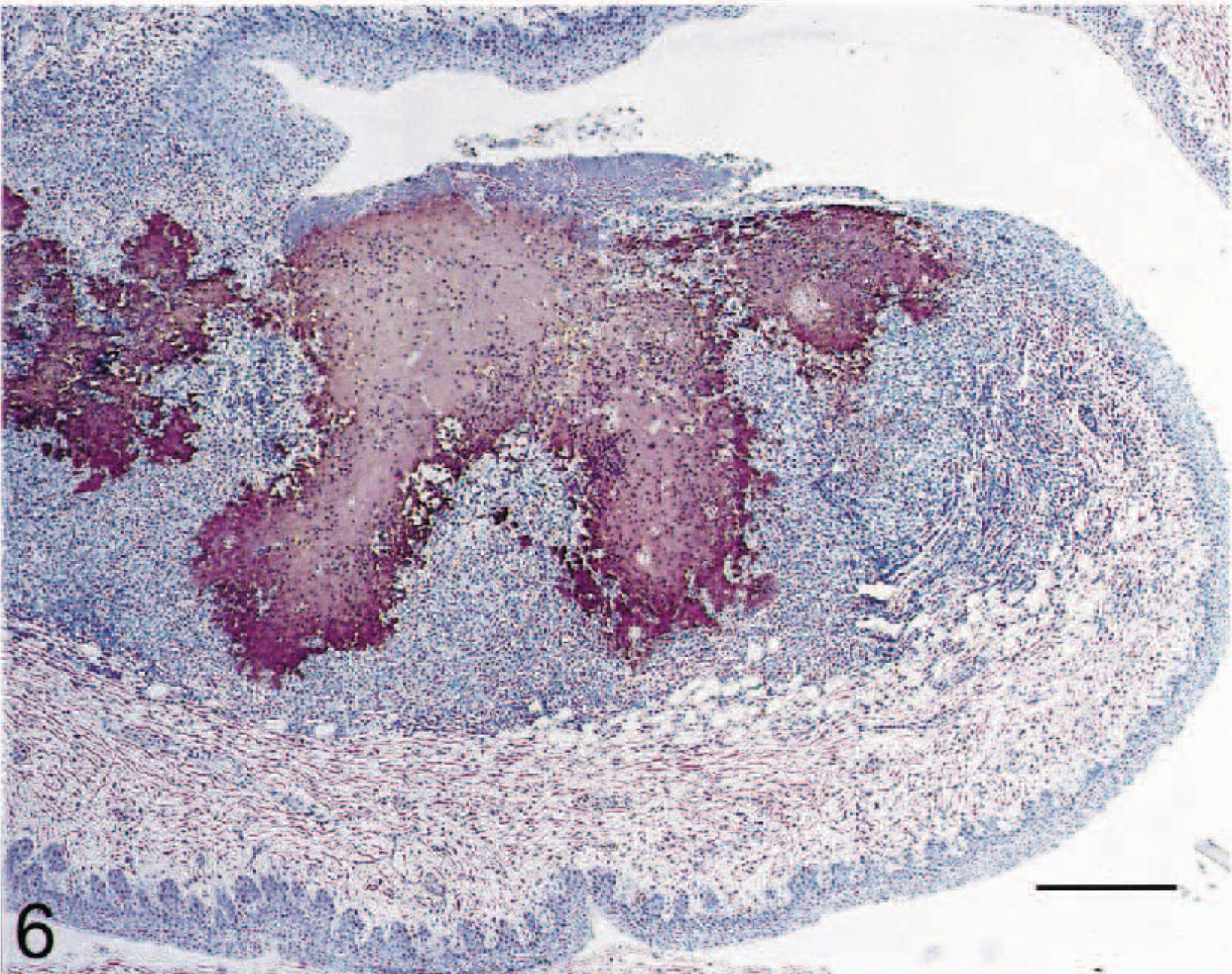
Pharyngeal tonsil; cat No. 2. Same tonsil as in Fig. 5. There is intense immunolabeling of necrotic lymphoid areas. Streptavidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 210 µm.
Large numbers of bacteria were found in the lumina of blood and lymphatic vessels both within and near affected lymph nodes. Two cats (Nos. 2 and 4) had acute fasciitis with few bacteria present adjacent to a large peripheral nerve. Spleens of four cats (Nos. 2, 4, 5, and 7) contained few to many bacteria and suppuration to a degree that approximately paralleled the number of bacteria present.
Adrenal glands in four cats (group I: Nos. 4 and 5; group II, Nos. 6 and 7) had moderate to marked multifocal hemorrhage within the cortex. Small focal accumulations of necrotic cells were present in some areas of hemorrhage, and in one cat few bacteria were present in these lesions. An additional cat in group I (cat No. 5) had hemorrhage and fibrin adjacent to the adrenal gland capsule and extending slightly into the surrounding fibroadipose tissue.
One cat from group I (cat No. 4) had focally extensive necrosis of lymphoid cells in the gut-associated lymphoid tissue of the ileum. The Peyer's patch was replaced by eosinophilic cellular debris that contained numerous 3–5-µm bacilli, whose morphology was consistent with enteric bacterial flora. This cat and one other in the same group (No. 5) had mild to moderate hemorrhage multifocally within the small intestinal submucosa.
Sections of thymus were normal except in one cat (No. 1). In this cat, the thymus contained multiple randomly placed aggregates of foamy macrophages, often accompanied by acicular (cholesterol) clefts. Similar lesions were not found in any other organs of this cat or in any of the other cats, and the significance of this finding was not determined. In the perithymic adipose tissue there was moderate acute necrotizing steatitis, and there was vascular necrosis and thrombosis of nearby small-caliber blood vessels.
No significant lesions were found in any other organs examined histologically.
Immunohistochemistry
Immunohistochemical procedures demonstrated that the bacteria visualized with HE
staining were
Positive staining also was present in some areas where fewer bacteria were aggregated
and were difficult to discern with HE staining alone (e.g., within intramuscular
fascia). One section of subcutaneous tissue from a group II cat (No. 7) stained
positively for
Discussion
Although the idiosyncratic susceptibility that felines have for
Plague is described as an endemic disease of rodents, in which certain species and their
fleas are believed to be the natural reservoir for the bacterium. There appears to be a
schism between the few descriptions of the lesions of rodents with natural infections
and those from other mammals that die from
Almost all carnivores that have been tested, including black bears, raccoons, badgers,
and dogs, either experimentally or in surveys for natural exposure in endemic areas
mount a robust immune response that prevents the development of clinical signs or
pathologic changes.
4–6,
13
17
20
28
34
Cats and probably black-footed ferrets are an exception in plague susceptibility
among carnivores.
15
26
33
Bubonic, pneumonic, and septicemic plague are technically morphologic diagnoses
of
Plague lesions∗ in carnivores following experimental challenge with Yersinia pestis compared with human lesions.

+ = occurs; - = does not occur.
The presence of numerous intralesional bacteria is a consistent finding in fatal plague.
Each of the affected cats exhibited enlargement and abscessation of various lymph nodes,
with disseminated petechiation and ecchymosis as the principal pathologic manifestation.
Grossly, the infection appeared to be contained within the nodes, which were swollen and
hemorrhagic. However, microscopic examination revealed that the massive numbers of
There was widespread endothelial necrosis and loss within both venules and arterioles, especially in lymph nodes, with many small venules and fewer arterioles partially or completely occluded by fibrinocellular thrombi. These lesions were noted in all seven cats and are consistent with a fatal gram-negative sepsis and disseminated intravascular coagulopathy.
Because of the severity of the lesions described here, future investigations should
focus on the pathogenetic mechanisms that underlie the unique susceptibility of cats to
subacute fatal infections by
Footnotes
Acknowledgements
We gratefully acknowledge the technical assistance and skills of S. Kodsi of The Armed Forces Institute of Pathology and Specialist J. Hart in the Veterinary Immunohistochemistry Laboratory at the Walter Reed Army Institute of Research. We also thank D. Miles of The Walter Reed Army Institute of Research for the photography of pathology specimens and Drs. A. Barnes and T. J. Quan for conceptualizing, facilitating, and conducting the experimental studies that provided the archival tissues for this study.
