Abstract
A 6.5-year-old male rhesus macaque (Macaca mulatta) was presented with acute paraplegia following a surgical procedure. During surgery, he experienced an acute hypotensive episode that required 3 hours of supportive therapy before hemodynamic stability was restored. Paraplegia remained unchanged until euthanasia 72 hours later. At necropsy, spinal cord segments T10-S3 had acute, massive panmedullary necrosis, which involved most of the central and middle spinal cord, sparing the peripheral white matter. Additional lesions included arterial border zone necrosis of the brain, centrilobular hepatic necrosis, and proximal renal tubular necrosis. The histologic findings of central spinal cord necrosis with a preserved rim of white matter is consistent with severely decreased flow in the anterior spinal artery. Such lesions can result from episodes of generalized hypoperfusion. Possible factors contributing to the hypotensive episode in this case included the addition of xylazine to ketamine and atropine in the preanesthetic medication, positioning during anesthesia, and decreased blood pressure during surgery.
The junction of two or more arterial territories within a tissue is referred to as an arterial border zone. Because the tissue in such regions is supplied by blood with the lowest regional oxygen tension, this tissue is most susceptible to hypoxic damage during hypotensive episodes. 2 4 The arterial anatomy of the spinal cord creates border zones in both transverse and longitudinal planes. 9 Systemic hypoperfusion associated with hemodynamic shock or cardiorespiratory arrest may result in spinal cord ischemia, necrosis, and acute onset paraplegia. 5 7–10 Here, we describe acute onset spinal cord necrosis associated with a surgical procedure in a rhesus macaque (Macaca mulatta).
A 6.5-year-old male rhesus macaque was presented to the necropsy service of the Section of Comparative Medicine, Yale School of Medicine. He had undergone an experimental intracranial procedure lasting 6 hours, during which he was placed in sternal recumbancy with the head raised and fixed in a stereotactic device. The hind limbs were placed at a 90 degree angle to the rest of the body, with the right side down. Xylazine, ketamine, and atropine were administered as preanesthetic agents. During surgery, he received 500 ml of lactated Ringer's solution intravenously and was maintained on isoflorane at 1.5–2%. Heart rate, respiratory rate, and temperature were normal for the first 6 hours of surgery. After 6 hours, he experienced abrupt bradycardia and dyspnea, and his mucous membranes became pale grey. He was given prednisolone sodium succinate, intravenous fluids, doxapram, yohimbine, dexamethasone, and enrofloxacin. He experienced increased awareness to surroundings after 3 hours but was unable to move his lower limbs, maintain an upright position, or urinate. Palpation detected a large bladder and a tender abdomen. There was no response to stimuli of the hind limbs, and the patellar reflex could not be elicited. Supportive treatment including cystocentesis, frequent turning, subcutaneous and intravenous fluids, antibiotics, steroids, and analgesics was given throughout the next 2 days with no improvement. The animal was euthanatized 3 days after surgery and perfused by the investigator, who also removed the brain before submitting the carcass for diagnostic pathology.
Gross examination revealed severe hemorrhage and necrosis of the sublumbar, epaxial, caudal thigh, quadriceps, gluteus, and adductor muscles of the right hind limb and back. The left hind limb appeared normal. Blood, presumably from repeated cystocentesis, covered the entire ventral surface of the bladder, and fibrinous adhesion of the bladder wall to ventral abdominal musculature was present. The cervical and midthoracic portions of the spinal cord were normal. From T9 caudally, the cord became progressively swollen and hemorrhagic. This change was moderate in the terminal thoracic segments and most severe in lumbar and sacral segments (Fig. 1). On cross section, hemorrhage and myelomalacia was present involving the central regions of the affected spinal cord segments.
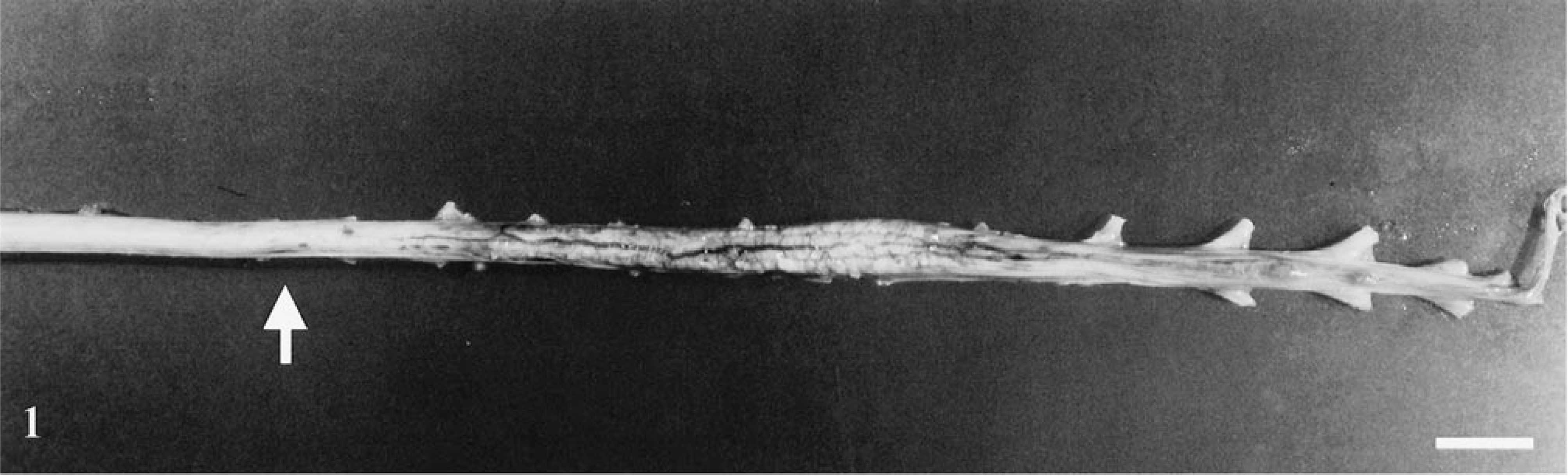
Spinal cord; rhesus macaque. There is swelling, congestion and hemorrhage of the terminal thoracic, lumbar, and sacral spinal cord. Hemorrhage was initially visible at T9 (arrow). Bar = 1.5 cm.
Spinal cord segments C1–T6 were histologically normal. In more distal segments (T7–T8), radial pools of hemorrhage dissected between fibers of the white matter tracts. Lesions in this region were limited to central chromatolysis in occasional neurons. Lesions at the junction of grossly normal and affected cord (T9; Fig. 1, arrow) were predominantly vascular. There was variable transmural necrosis of thin-walled intramedullary vessels, with extravasation of blood, fibrin, and platelet aggregates. Spinal cord segments T10–S3 had acute, massive panmedullary necrosis, with sparing of the peripheral white matter (Fig. 2, arrowhead). In this region, most neurons displayed abnormalities ranging from shrunken angular and acidophilic cell bodies typical of ischemic change to complete loss of nuclei, dispersal of Nissl substance, and fragmentation of the cell body. Some neurons appeared intact and relatively normal. In the central and midperipheral white matter, myelin was fragmented and dispersed, appearing as disorganized coils and ribbons of lamellar material. Most axons were absent, but occasional swollen axons were suspended within the debris. Dorsal ganglia were acutely necrotic, but dorsal and ventral nerves were intact. All intramedullary vessels had transmural necrosis and were often plugged with platelets, fibrin, and erythrocyte ghosts. Transmural extravasation of blood was common in affected vessels (Fig. 3), and pools of blood were present throughout the white and gray matter. Small numbers of neutrophils were dispersed throughout necrotic spinal cord parenchyma. Extramedullary blood vessels were normal, and no thrombi were seen. Macrophages with intracytoplasmic lipid material were present throughout the white matter. Astrocytes retained normal nuclear features in all but the most severely damaged regions, where they appeared shrunken and pycnotic.
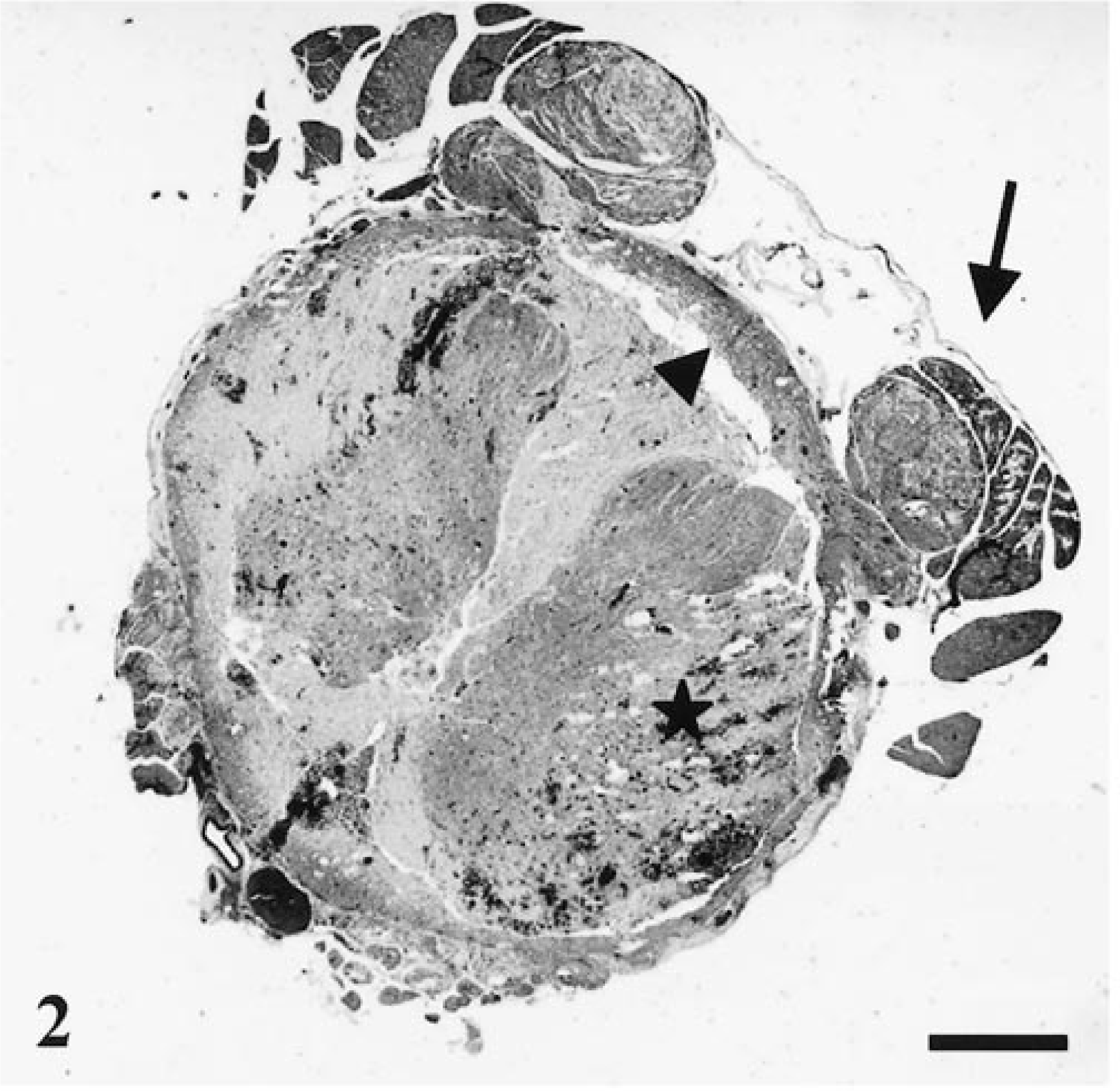
Spinal cord (S1); rhesus macaque. There is acute necrosis, hemorrhage, and disintegration of central and midperipheral spinal cord elements (asterisk), with separation of necrotic tissue from a peripheral rim of intact white matter (arrowhead). The proximal portions of dorsal roots are necrotic and edematous (arrow). HE. Bar = 250 µm.
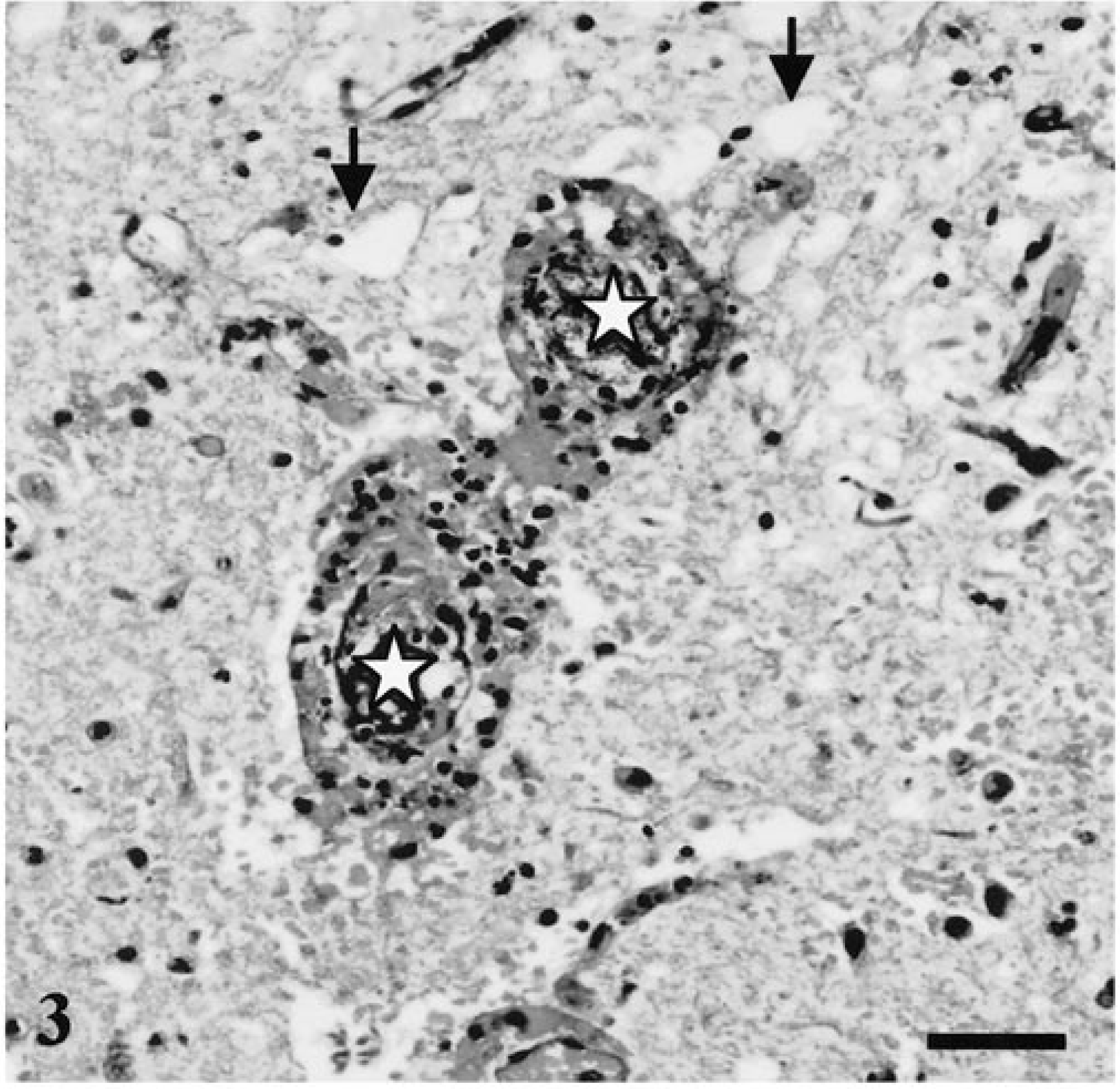
Spinal cord (S1); rhesus macaque. Higher magnification of blood vessels located in the region of the asterisk in Fig. 2 (midperipheral white matter). The lumens of two vessels are indicated by white asterisks. Vessels are occluded and surrounded by blood and nuclear debris. There is disintegration and edema (arrows) of surrounding parenchyma. HE. Bar = 50 µm.
Additional lesions were present in cerebellum, liver, kidney, and skeletal muscle. Bilaterally symmetrical acute necrosis of cerebellar folia was apparent (Fig. 4). These lesions involved the depths of the cortex adjacent to the white matter. Ischemic change and loss of Purkinje cells accompanied by loss of basket cell soma and their descending axons (“baskets”) were frequently seen in the sulci of cerebellar folia (not shown). Muscles of the right hind limb displayed acute necrosis and marked hemorrhage. Histologically, lesions were characterized by loss of myobrillar detail, myofiber disintegration, and macrophage infiltration. Muscles of the contralateral region were histologically normal. The liver had diffuse centrilobular coagulative necrosis, and renal changes included proximal renal tubular necrosis with cellular and granular casts.
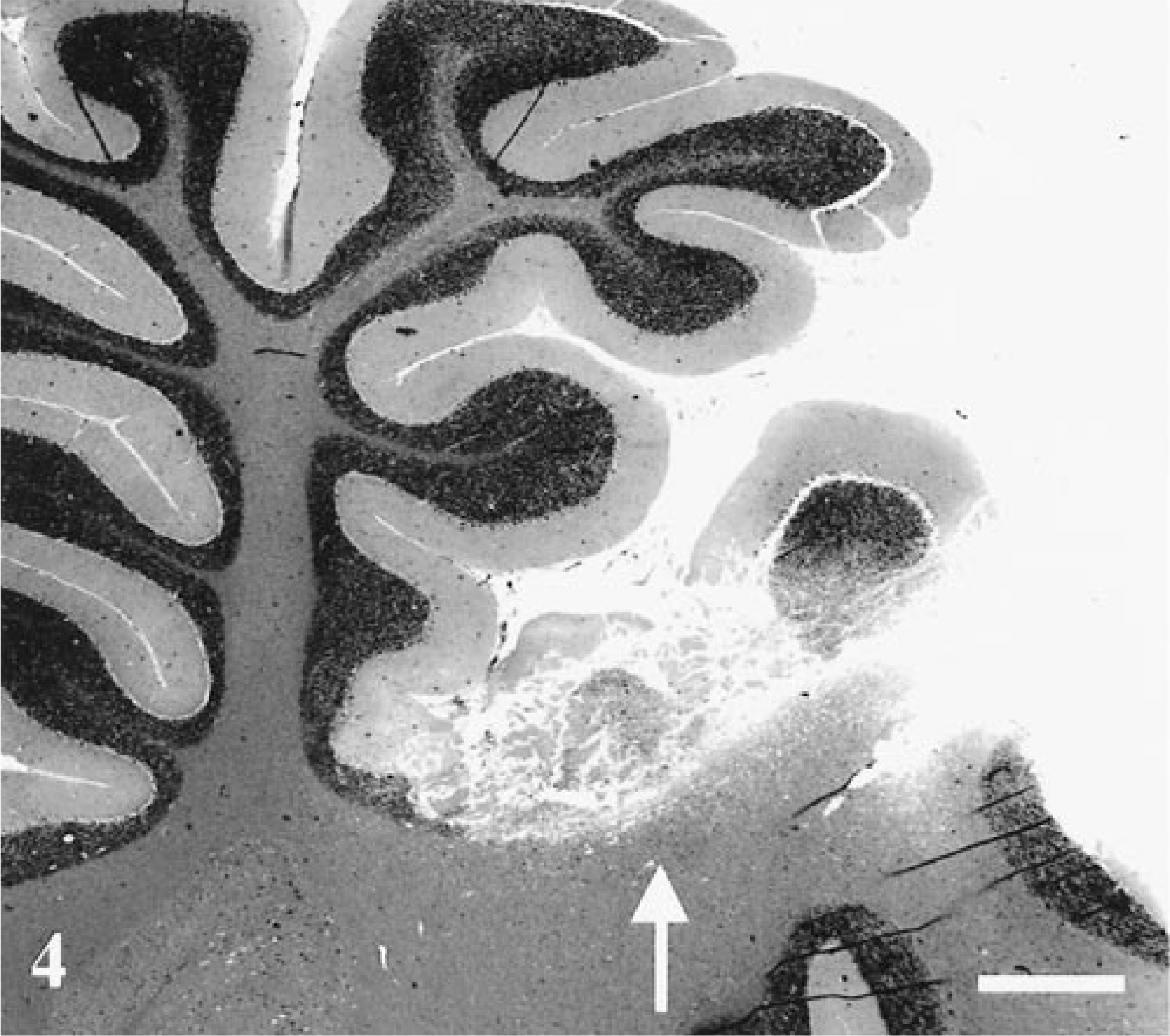
Cerebellum; rhesus macaque. There is acute necrosis of cerebellar folia (arrow). A bilaterally symmetrical lesion was present on the contralateral side of the cerebellum. HE. Bar = 250 µm.
Global ischemia of the brain and spinal cord due to arterial hypotension results in bilaterally symmetrical necrosis of boundary regions between major arterial territories. 2 4 In the cerebellum, such lesions are located at the boundary zone between the superior and posterior inferior cerebellar arteries and typically occur in the depths of the sulci. 4 The distribution of spinal cord lesions in this macaque coincided with the territories supplied by spinal cord vasculature (Fig. 5). A cross section of the cord reveals three extrinsic longitudinal arteries: a single midline anterior spinal artery and two posterior spinal arteries just medial to the dorsal roots. Both anterior and posterior spinal arteries originate at the level of the foramen magnum from the union of the vertebral arteries. These longitudinal arteries are reinforced by the segmental arteries that pass through vertebral foramina and anastamose with anterior and posterior spinal arteries via medullary branches. The lumbar region is supplied by a large medullary artery, the artery of Adamkiewicz in humans. Similar arteries are present in cats, dogs, and monkeys. 1 6 Typically, this vessel arises in the low thoracic or upper lumbar region and provides the major blood supply to the lumbar cord. The bulk of the gray matter is supplied by a branch of the anterior spinal artery that lies within the ventral median sulcus. The peripheral white matter is supplied by the vasacorona, a vascular ring formed by branches of both the anterior and posterior spinal arteries. The vasacorona encircles the cord and supplies its periphery by means of short penetrating vessels. In the longitudinal axis, the distribution of the segmental arteries results in an arterial border zone in the midthoracic region. In the tranverse plane, the border zone is located at the periphery of the cord at the junction of the vessels arising from the anterior spinal artery and the penetrating vessels of the vasocorona. Experimental cord ischemia in rabbits has demonstrated that distribution and severity of infarction is proportional to the extent of ischemia. 1 In the longitudinal plane, the rostral border of the lesion originates in the thoracolumbar region and extends posteriorly according to the extent of ischemia. In the transverse plane, initially the ventral and intermediate horns become necrotic, followed by centrifugal extension of necrosis with increasing ischemia. The spinal cord lesions in this macaque, i.e., central necrosis from the midthoracic region caudally, reflect the combined effects of reduced perfusion of arterial border zones in both longitudinal and transverse planes of the spinal cord.
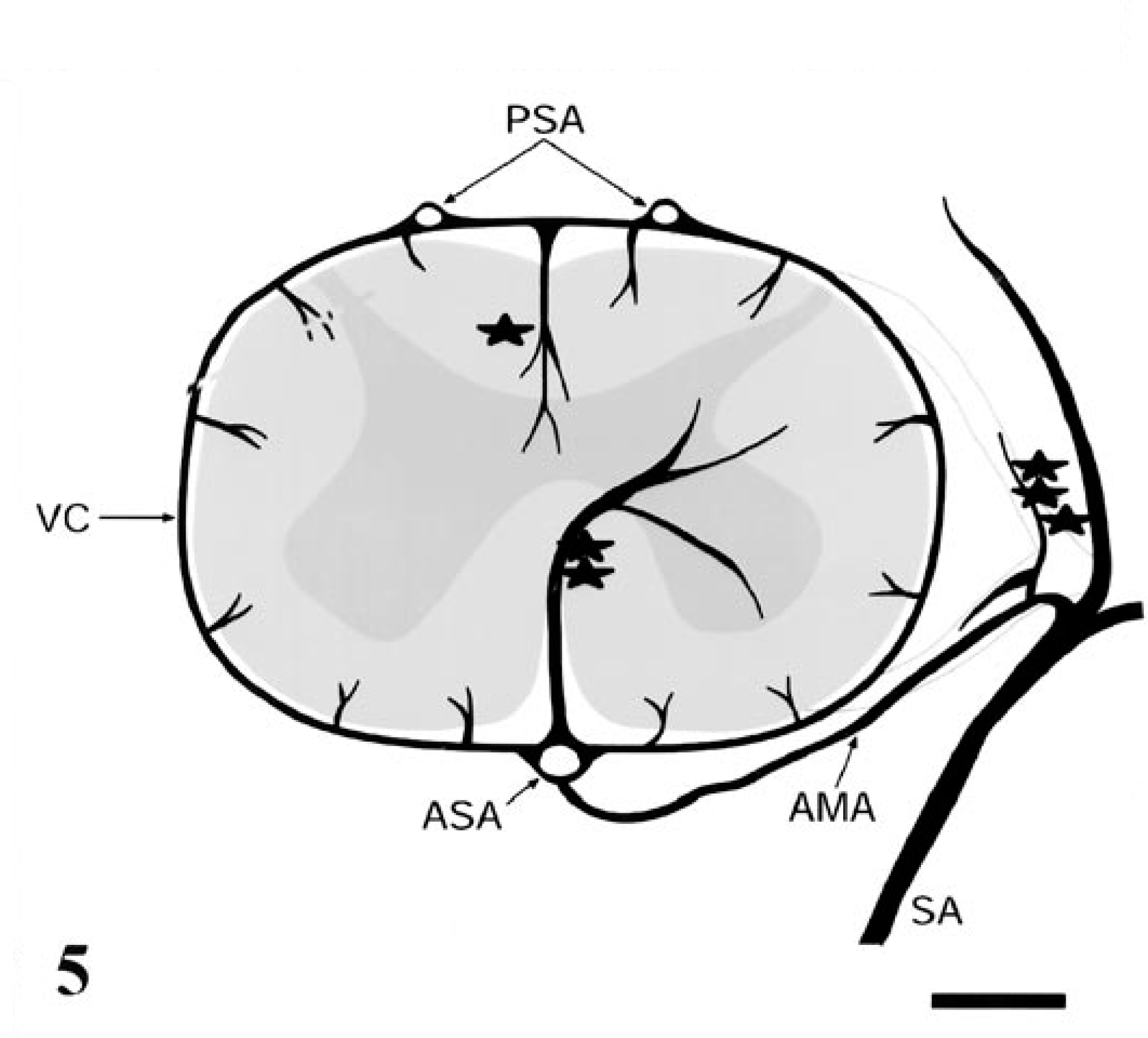
Vasculature of the primate spinal cord. The three longitudinal arteries of the spinal cord, the anterior spinal artery (ASA) and posterior spinal arteries (PSA), are reinforced by the segmental arteries (SA). The SAs arise from the aorta and supply the ASA via the anterior medullary artery (AMA). The arterial vasocorona (VC) encircles the spinal cord to supply its periphery. Minor branches include the artery of the posterior median septum (one asterisk), the central sulcal artery (two asterisks), and the radicular arteries (three asterisks). Figure adapted. 6 9 .
Central spinal cord necrosis with a preserved rim of white matter is typical of lesions resulting from obstruction or severe decreased flow of the anterior spinal artery. In humans, such lesions are most common in obstruction or traumatic avulsion of the medullary arteries 10 and severe hypotension due to ruptured aortic aneurysm 5 10 and less frequently in hypotension associated with surgery. 7 8 Abrupt onset of lower motor neuron paraplegia accompanied by loss of bladder and bowel control is the typical clinical presentation. In patients who survive, recovery of motor function is minimal. 8 In the case reported here, the animal had a history of prolonged hypotension during surgery and displayed lesions secondary to systemic hypoperfusion (central spinal cord necrosis, border zone necrosis of the brain, centrilobular necrosis of the liver, and proximal renal tubular necrosis). Restitution of normal hemodynamic status with reperfusion of the ischemic cord would have contributed to the vascular leakage, hemorrhage, and necrosis seen histologically. Several possible mechanisms for these changes have been proposed. Endothelial swelling and microvascular sludging are two of the earliest lesions seen in experimental hypoxic brain damage, 3 and these lesions further compromise parenchymal circulation. Mechanisms of hypoxic damage include accumulation of lactic acid and excitatory neurotransmitters (particularly glutamate), elevated intracellular Ca2++ levels, and free radical generation. Reperfusion of hypoxic tissue results in additional injury due to increased formation of oxygen derived radicals, synthesis of prostaglandins, lactic acid formation, and edema. 2 4
The precise extent of ischemia needed to produce the lesions identified in this macaque is unclear. Fifteen minutes to 1 hour of aortic occlusion causes irreversible spinal cord damage in experimental rabbits. 1 In humans with hypotension-associated cord necrosis, hypotensive periods ranged from 20 minutes at systolic pressure 60 mm Hg to 2.5 hours at 47/25 mm Hg and 10 minutes during cardiorespiratiory arrest. 8 In this macaque, hypotension was assessed by the more subjective parameter of poor capillary refill time, and the animal was treated for shock for approximately 3 hours before it was considered stable. The addition of the alpha2 adrenergic agonist xylazine to the premedication regimen may have predisposed the macaque to hypotension. In addition, the animal's position during surgery may have worsened lumbar perfusion by creating a 90° twist at the waist. The period of presumed spinal ischemia in this animal is consistent with those reported in humans with similar lesions, and restitution of perfusion after this ischemic period is likely to have contributed to significant reperfusion injury. Prevention of hypotension and frequent repositioning during the long surgical procedure may have prevented spinal cord infarction in this case.
