Abstract
We studied the distribution of bovine respiratory syncytial virus (BRSV) RNA in lungs of experimentally inoculated lambs by in situ hybridization at different times postinoculation. The probe used for in situ hybridization was prepared by reverse transcription of BRSV RNA, followed by polymerase chain reaction (PCR) amplification of the cDNA. Twenty-five Merino lambs of both sexes with a live weight of 17 ± 3 kg received an intratracheal inoculation of 20 ml saline solution containing 1.26 × 106 TCID50 BRSV (strain NMK7)/ml. Lambs were slaughtered 1, 3, 7, 11, and 15 days postinoculation (PID). Bronchial and bronchiolar epithelial cells were positive for BRSV nucleic acid by ISH at 1, 3, 7, and 11 PID. However, alveolar epithelial cells contained positive cells at 1, 3, and 7 PID. Cells containing viral RNA were detected from 1 to 11 PID in exudate within bronchial and bronchiolar lumina and from 3 to 7 PID in alveolar exudates. Positive hybridization signals were identified in interstitial mononuclear cells and in bronchi-associated lymphoid tissue from 3 to 11 PID. Mononuclear cells were located in peribronchiolar tissue and interalveolar septa. The highest signal intensity in positive cells was observed at 3 and 7 PID, coinciding with the most important histopathological findings.
Respiratory syncytial virus (RSV) is a respiratory tract pathogen of both humans and cattle. Human RSV is the most important cause of viral lower respiratory tract disease in infants and young children. 26 Bovine and human RSV strains (enveloped RNA virus) are pneumoviruses of the Paramyxoviridae family and are currently considered to be antigenically related. 5 Although greater suceptibility has been reported in cattle, 6 7 9 11–13, 17 27 bovine respiratory synctial virus (BRSV) may also affect others domestic species, particularly small ruminants. 1–4, 8 20–24, 32
The RSV fusion protein causes fusion of viral and host cellular membranes and cell-to-cell fusion. 19 This protein is synthetisized as an F0 precursor, which is proteolytically cleaved into two disulfide-linked polypeptide subunits, F1 (48 kDa) and F2 (20 kDa), that form the active fusion protein. 19 25 28 29 37 38 The BRSV F mRNA is 1,899 nucleotides in length. 19 The nucleotide sequence encoding the F protein is highly conserved (> 95%) among BRSV strains. 28 This identity is lower between F mRNAs of RSV of bovine and human origin (71.5%). 19
A digoxigenin-labeled DNA probe corresponding to the part of the genome that codes for bp 736–1522 of the BRSV protein gene was developed for localization of virus in lung sections.
The aim of the present study is to analyze the pathology of lambs experimentally inoculated with BRSV and to determine specific hybridization signals for this virus in lung samples.
Materials and Methods
Animals and BRSV inoculum
Twenty-five gnobiotic Merino lambs of both sexes with a live weight of 17 ± 3 kg each received an intratracheal inoculation of 20 ml of the NMK7 strain of BRSV (supplied by Dr. Gómez-Tejedor, Instituto Nacional de Investigaciones Agrarias, Madrid, Spain) diluted to a concentration of 1.26 × 106 TCID50/ml. The origin and characterization of the NMK7 isolate of BRSV has been previously reported by other authors. 4 The lambs were housed in an isolation barn. Lambs were slaughtered 1, 3, 7, 11, and 15 days postinoculation (PID).
Virus inoculum was prepared using primary bovine fetal kidney (BFK) cell cultures inoculated with a BRSV stock strain NMK7, to which minimum essential medium (Sigma, Madrid, Spain) with 15% bovine fetal serum (Sigma) was added; the final cultures were incubated at 37 C in a CO2 atmosphere. Ten control animals were inoculated with an identical volume of uninoculated BFK cell culture. They were kept in another isolation box.
Aliquots of virus were cultured and were determined to be free of aerobic bacteria and mycoplasma by standard techniques. Viral inoculum was free of aerobic bacteria, mycoplasma, and BVD virus.
Samples of lung were collected for microbiological studies and virus isolation. Samples of lung were fixed in 10% neutral buffered formalin and processed by routine paraffin-embedding methods for histopathological technics and in situ hybrydization.
Histopathology
Sections 3 µm thick were cut and stained with hematoxylin and eosin for routine morphological studies.
Immunoperoxidase staining procedure for detection of BRSV antigens
An avidin-biotin-peroxidase complex was carried out on deparaffined and trypsinized lung samples. Sections were incubated in diluted (1:50) normal swine serum (Dakopatt, Spain) for 15 minutes to reduce background and were incubated in diluted (1:1,000) rabbit anti-RSV (Dakopatt) for 3 hours at 20 C. Diluted (1:500) biotinylated swine anti-rabbit IgG (Dakopatt) was placed on the sections for 30 minutes, and sections were then incubated with diluted (1:50) ABPC reagent (Dakopatt) for 1 hour. Sections were incubated in diaminobenzidine solution for 5 minutes and were counterstained lightly with Mayer's hematoxylin. The positive controls included BRSV-infected BFK cell cultures (the cytopathic effect was evident in the formation of numerous syncytia) and noninoculated BFK cell cultures. Test control sections were also stained using nonimmune rabbit serum as the first layer.
Microbiological investigations
Samples of lung tissue were homogenized in phosphate-buffered saline. Serial 10-fold dilutions were made up to 104 and 0.1 ml of each dilution was spread onto half a sheep's blood agar plate. Duplicate plates were used, one incubated aerobically and the other anaerobically under H2 containing 10% CO2. Colonies were identified using the methods described by Cowan. 16 Bronchial swabs and lung samples were also cultured for mycoplasma in modified Hayflick's broth, and colonies were identified on agar plates.
Nasal swab specimens and samples of lung were used to detect BRSV by virus isolation in BFK cells following the technique described previously by other authors. 13 Isolates were confirmed as BRSV by indirect immunofluorescence. 21
Extraction of genomic RNA
BT cells (75-cm2 flasks) were inoculated with BRSV strain 375 18 27 (provided by Dr. Kelling, Department of Veterinary and Biomedical Sciences, University of Nebraska, Lincoln, NE, USA). This is a BRSV strain plaque purified more than three times that was originally obtained from cattle and is bovine diarrhea virus free. 27 Cell cultures were incubated at 33 C in high glucose Dulbecco modified Eagle medium (DMEM) supplemented with 2% horse serum. Viruses were allowed to replicate and form syncytia (cytopathic effect), at which point the RNA was extracted from infected cells. RNA was isolated with the Trizol LS Reagent (Gibco, NY, USA) following the manufacturer's instructions. RNA concentration of the extracts was estimated by measuring the A260 with a Beckman DU-64 Spectrophotometer.
Real-time polymerase chain reaction
An amplified product of 786 bp length encoding for bp 736-1522 of the BRSV protein F gene was obtained by nested real-time polymerase chain reaction (RT-PCR). Primer 1555 (5′ TTTCCTACATCTACACTG 3′) was used to obtain cDNA and primers 736 (5′ TGGTATTACCACACCCCTCAG 3′) and 1522 (5′ TCAGATCGACGTATGAAAGCC 3′) were used for polymerase chain reaction (PCR). The PCR was performed in 35 cycles of 1 minute at 95 C, 1 minute at 55 C, and 1 minute at 72 C.
Cloning
The amplified product was cloned in the pCR 2.1 plasmid vector (Invitrogen, CA, USA). The correct sequence was confirmed by sequencing.
Labeling of the probe
The probe was prepared by removing the DNA insert by digestion with restriction enzyme EcoRI. The DNA was submitted to gel electrophoresis and the fragment extracted using Quiaex II (Quiagen, Hilden, Germany). DNA fragment was digoxigenin labeled by ramdom priming (Boehringer Mannheim Biochemicals, Mannheim, Germany). The specificity of the probe was checked by dot blot and Southern blot onto nylon membranes.
Preparation of samples for ISH
BRSV-inoculated and uninoculated cell cultures that served as negative controls were used for optimization of ISH. BT cells were inoculated and grown, as has been described before for RNA extraction. When the cultures showed cytopathic effect, the cells were trypsinizated and washed twice with DMEM, the first time with the DMEM containing 5% equine serum and the second time containing as serum. After counting, the harvested cells were applied to ProbeOn Plus microscope slides (Fisher Scientific, Pittsburgh, PA, USA) by centrifugation at 1,500 rpm for 3 minute with a cytocentrifuge Shandon Cytospin 2 at a density of 2 × 105 cells per spot. Slides were fixed in 4% paraformaldehyde and dehydrated through an ethanol series. The fixed slides were kept at 41 C until use.
Three-micrometer-thick lung sections were deparaffinized with xylene and rehydrated through decreasing concentrations of ethanol. All the samples (slides containing cells or sections) were equilibrate in phosphate-buffered saline (PBS). Permeabilization to allow the penetration of the probe was carried out by treatment with 0.2 N HCl for 20 minute at room temperature and digestion with 5 µg/ml of proteinase K (Promega, Madison, WI, USA) for cells and 30 µg/ml for tissues in PBS for 20 minute at 37 C. Some slides were treated with 50 µg/ml of RNase (Boehringer Mannheim Biochemicals) in PBS for 60 minute at 37 C in a wet chamber, to be used like specificity control. Next, slides were refixed in 5% paraformaldehyde and followed by two rinses in PBS. Slides were acetylated in 100 mM triethanolamine with 0.25% of acetic anhydre for 10 minute to neutralize positive charges and thus reduce nonspecific electrostatic binding of the probe. Slides were then washed twice in 2× SSC (1× SSC is 15 mM of sodium citrate plus 0.15 M NaCl, pH 7.0).
Prehybridization
Slides were prehybridized at 70 C for 60 minute in a humidified chamber by adding 200 µl of hybridization solution containing 4× SSC, 2 mM EDTA, 10% dextran sulfate, 50% deionized formamide, 500 µg of salmon sperm DNA, and 1× Denhardt solution (0.02% polyvinylpyrrolidone, 0.02% bovine serum albumin, and 0.02% Ficoll 400) per milliliter without cDNA probe.
Hybridization
The ISH conditions were as described elsewhere, 34 with minor modifications.
Results
Clinical findings
Compared with the control group, 21 of 25 lambs inoculated with BRSV developed a mild clinical response characterized by depression between PID 1 and 7. Increased respiratory rate, febrile response, coughing, a very slight increase in serous nasal discharge, and mild conjunctivitis were the principal clinical features observed.
Gross pathology and histopathology
Similar gross lesions were found in all animals inoculated with virus, the only variations being observed in the degree to which the lobes were affected. Affected lobes contained consolidated and emphysematous areas. The extent of these lesions in different parts of the lungs are shown in Table 1.
Consolidated and emphysematous areas in the lung of experimentally BRSV-inoculated lambs. Mean of percentages affected lobe's surface.∗
A = caudal lobe of the right lung; B = cranial portion of the right cranial lobe; CR = caudal portion of the right cranial lobe; D = intermediate lobe of the right lung; CL = caudal lobe of the left lung; F = cranial portion of the left cranial lobe; G = caudal portion of the left cranial lobe; H = accessory lobe of the right lung; PID = postinoculation day; C = consolidated areas (%); E = emphysematous areas (%).
The histopathological study has been previously reported. 23 24 Catarrhal bronchiolitis observed on 1 PID was associated with neutrophil infiltration of the bronchiolar lumen. The interalveolar septa were thickened, with pronounced interstitial edema and moderate cell reaction.
Alveolar exudate in animals slaughtered 3 and 7 PID consisted of neutrophils, lymphocytes, and multinucleate giant cells, which formed the syncytia. There was considerable thickening of the interalveolar septa due to the presence of edema and neutrophil and monocyte infiltration. Bronchiolitis was accompanied by epithelial cell necrosis. Hyperplasia of bronchiolar epithelial cells was conspicuous, and early stages of re-epithelization were apparent. Exudate in the bronchiolar lumina consisted of desquamated cells, necrotic debris, and syncytial cells.
Severe bronchial, bronchiolar, and alveolar damage was visible by 11 PID. Lung parenchyma had clear focal areas of consolidation due to bronchiolitis obliterans and the alveolar collapse caused by infiltration of macrophages, lymphocytes, and syncytial cells into lumina.
Animals slaughtered at 15 PID presented a marked interstitial inflammatory reaction, with considerable septal thickening. Intense bronchiolar and/or alveolar hyperplasia was also observed. Lung consolidation was less marked than in animals slaughtered earlier.
Immunohistochemistry signals
The immunohistochemistry (IHC) study has been previously published. 21 24 BRSV antigen was detected in bronchial and bronchiolar epithelial cells, in bronchial mucous cells, and in alveolar epithelial cells at 3 and 7 PID. Intense staining was also observed in alveolar macrophages, interstitial mononuclear cells, and syncytial cells from 3 to 15 PID. Antigen was commonly detected in exudate within bronchiolar, bronchial, and alveolar lumina. Specific staining was absent in the negative control (data not shown).
Microbiological findings
y-three of the lambs inoculated with BRSV and nine of the control animals were bacteriologically negative at the conclusion of the experiment. Contaminative species of bacteria were Escherichia coli and Bacillus spp. Mycoplasma was not isolated from the upper or lower respiratory tract of any lamb.
ISH signals
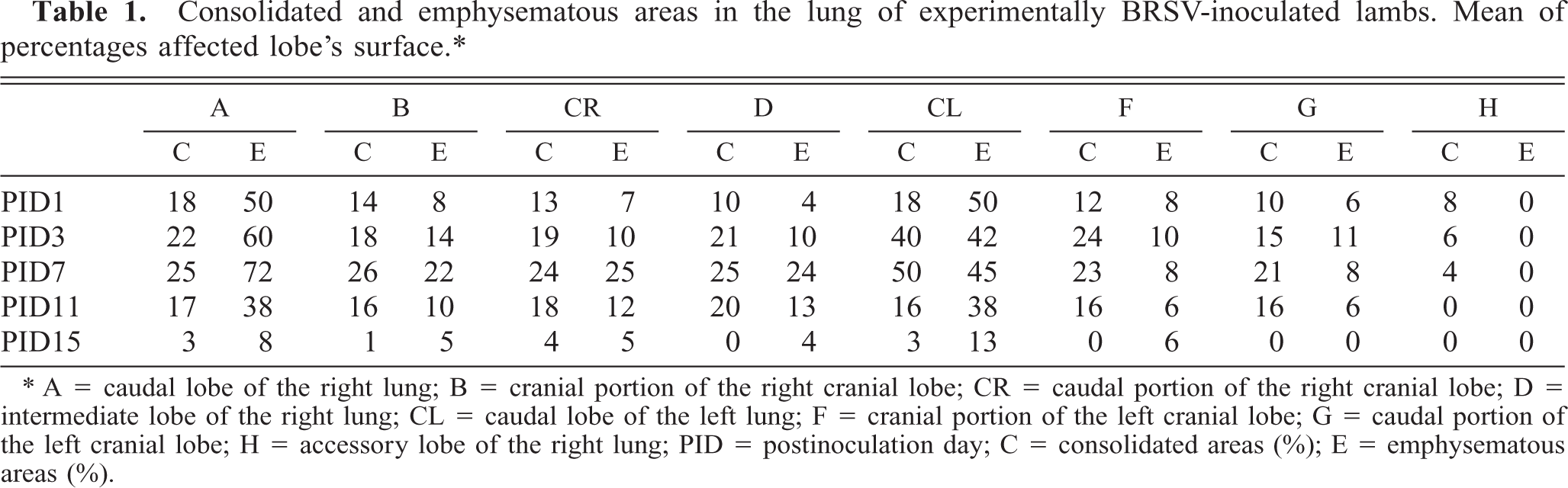
At all PID, the specificity of the signal reaction was confirmed by the rigorous observance of specific controls (Fig. 1). BRSV Nucleic acid signal was specific because this was completely absent from noninfected cells. There was no detectable ISH signal of BRSV RNA in the lungs of the control lambs.
Bovine respiratory syncytial virus (BRSV)-inoculated BT cells hybridized with a DIG-labeled cDNA probe. A positive ISH signal in the cytoplasm of cells is shown. Methyl green counterstain. Bar = 120 µm.
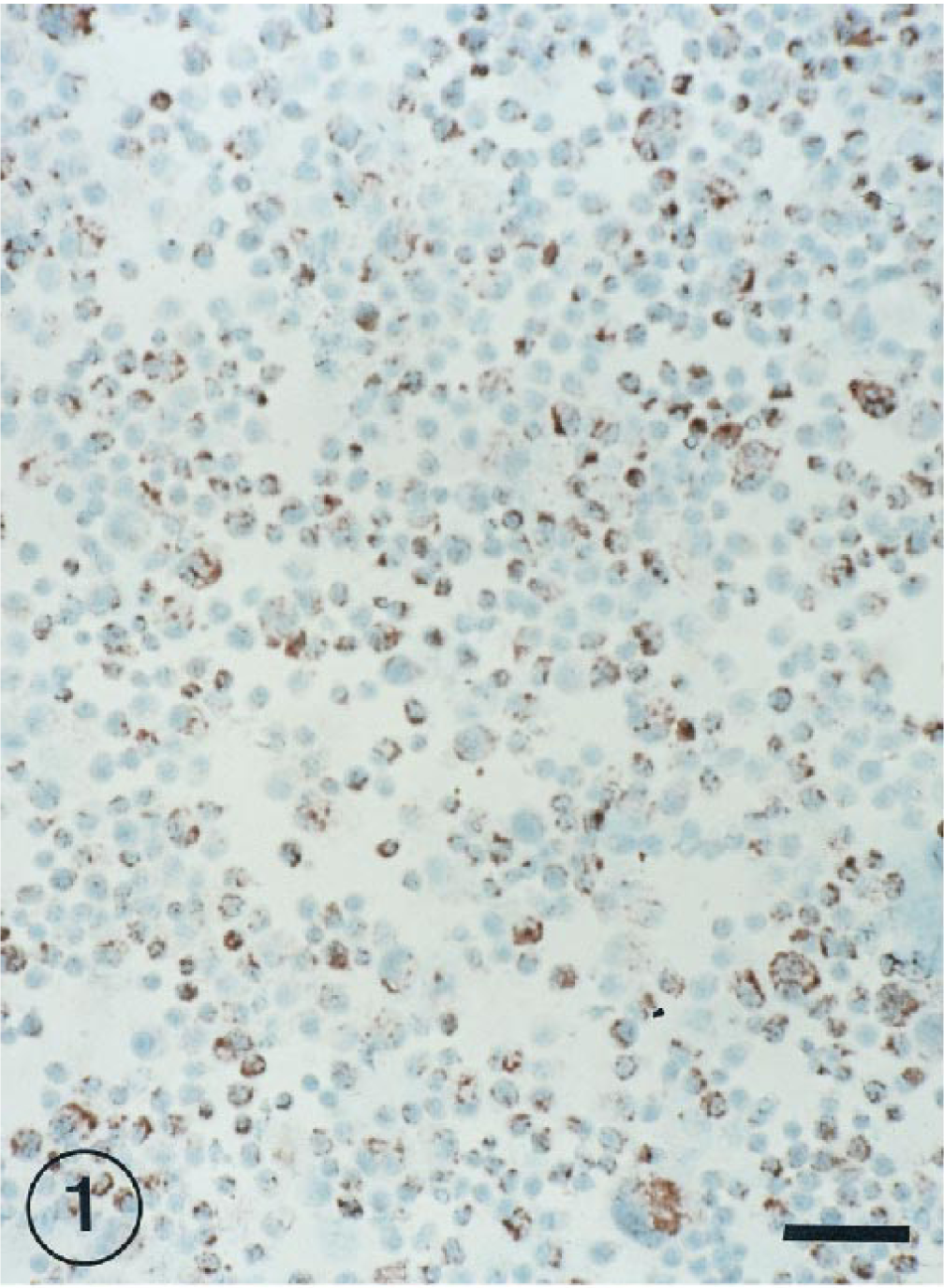
Hybridization signals specific for BRSV RNA were detected in bronchial and bronchiolar epitelial cells at 1, 3, 7, and 11 PID (Fig. 2), but they were not observed at 15 PID.
Lung of lamb BRSV infected at 7 days postinoculation. ISH-positive signal in bronchiolar epithelial cells. ISH counterstained with methyl green. Bar = 60 µm.
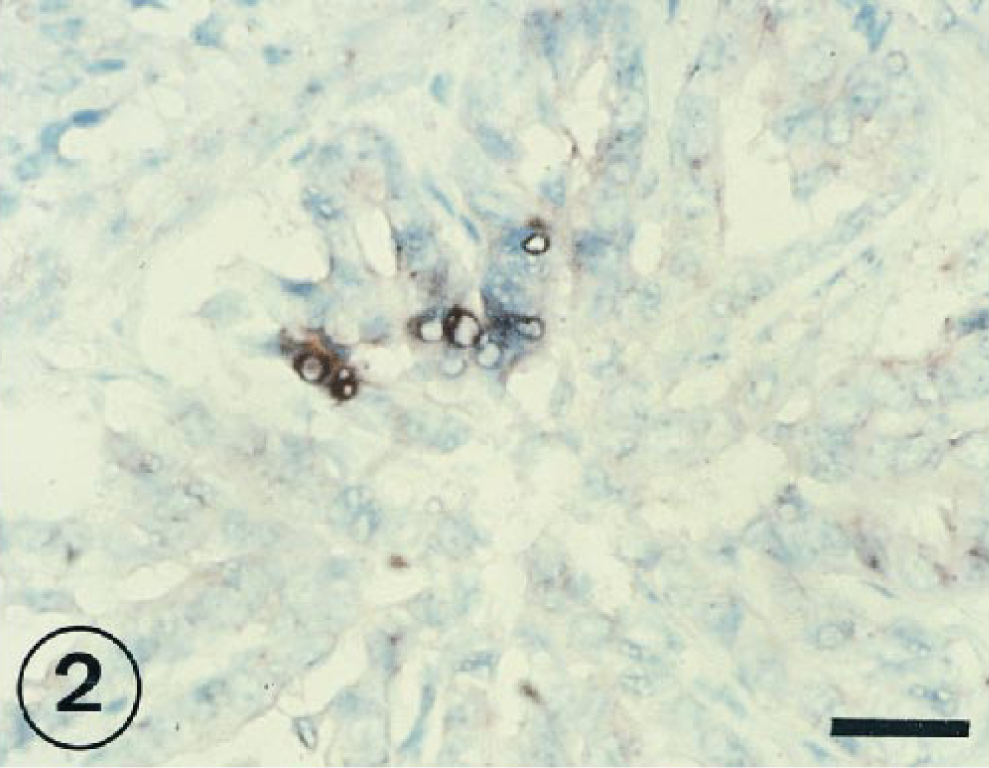
BRSV-positive cells were commonly detected in exudate within bronchial and bronchiolar lumina from 1 to 11 PID (Fig. 3).
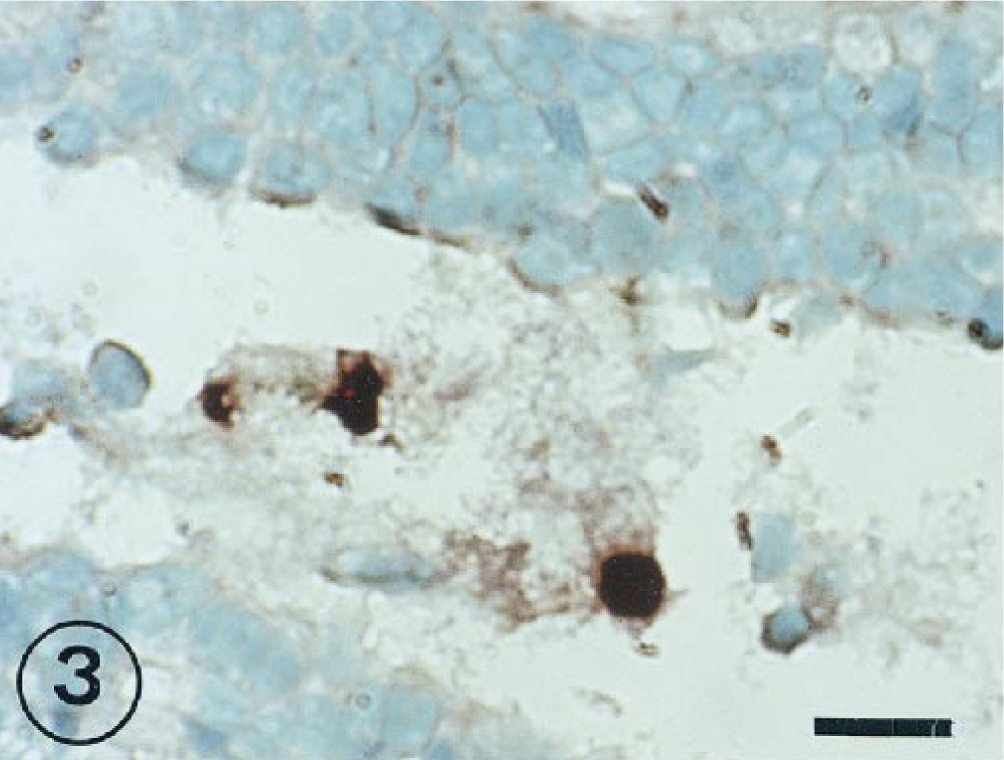
Lung; lambs inoculated intratracheally 7 days previously with BRSV. BRSV-RNA-infected cells located in bronchiolar exudations. ISH counterstained with methyl green. Bar = 30 µm.
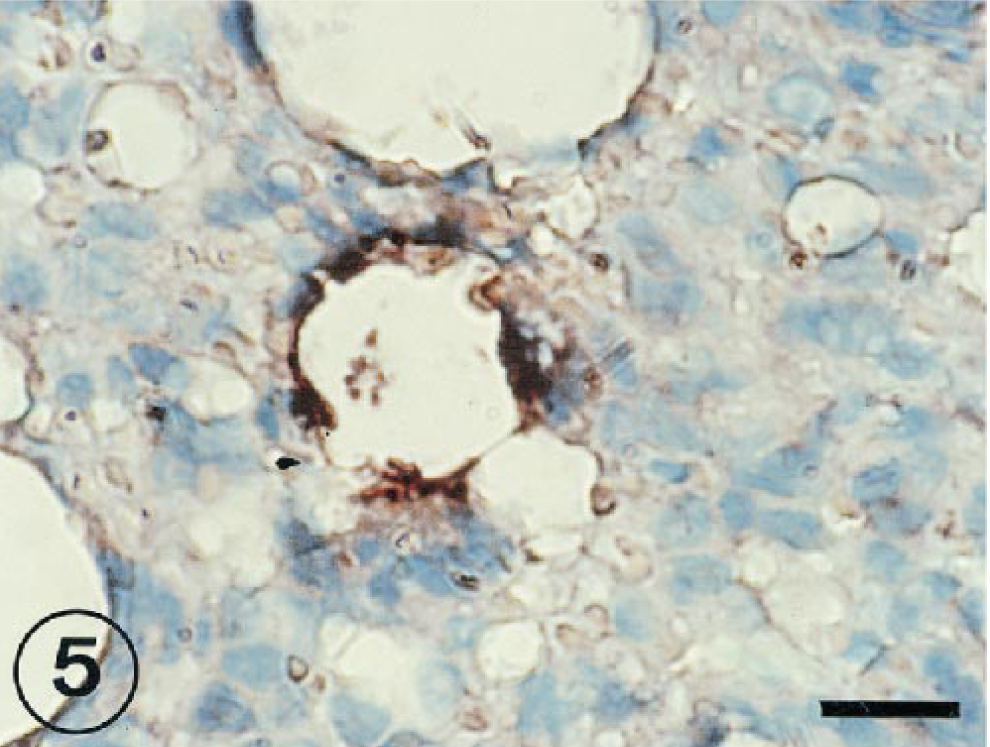
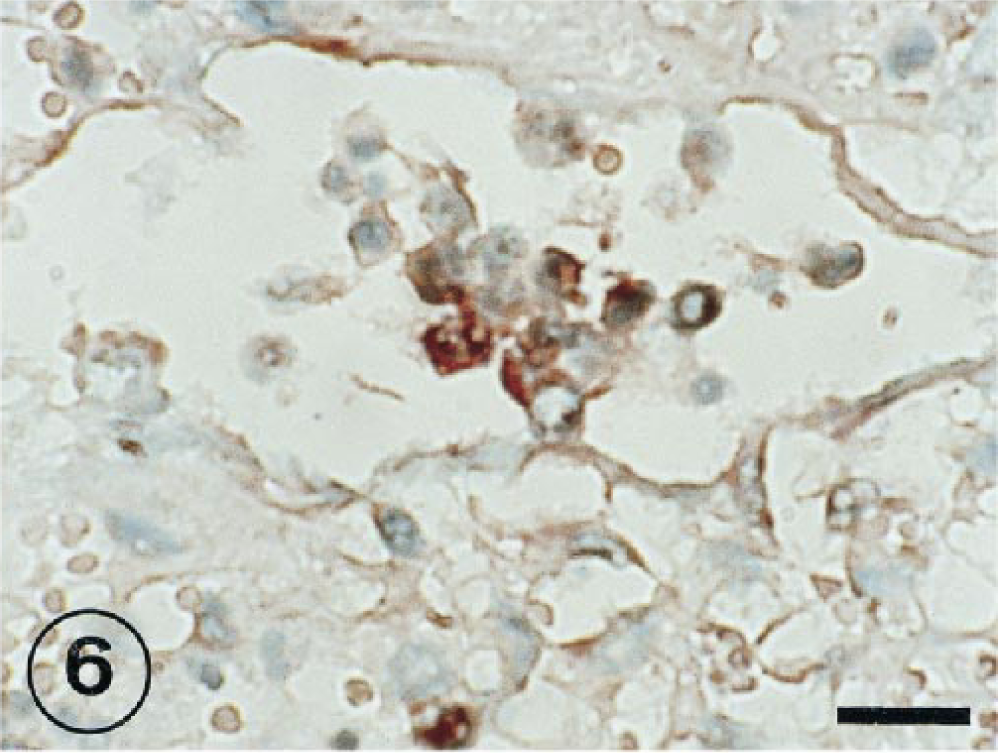
The positive IHS signal was observed in alveolar epithelial cells at 1, 3, and 7 PID (Figs. 4, 5). In several cases, the signal of the RNA virus also coincided with a cell morphology and location that would suggest the cell could be classified as a type II pneumocyte. The RNA genome was visible in free cells in alveolar space (Fig. 6) and syncitial alveolar cells (Fig. 7) at 3 and 7 PID.
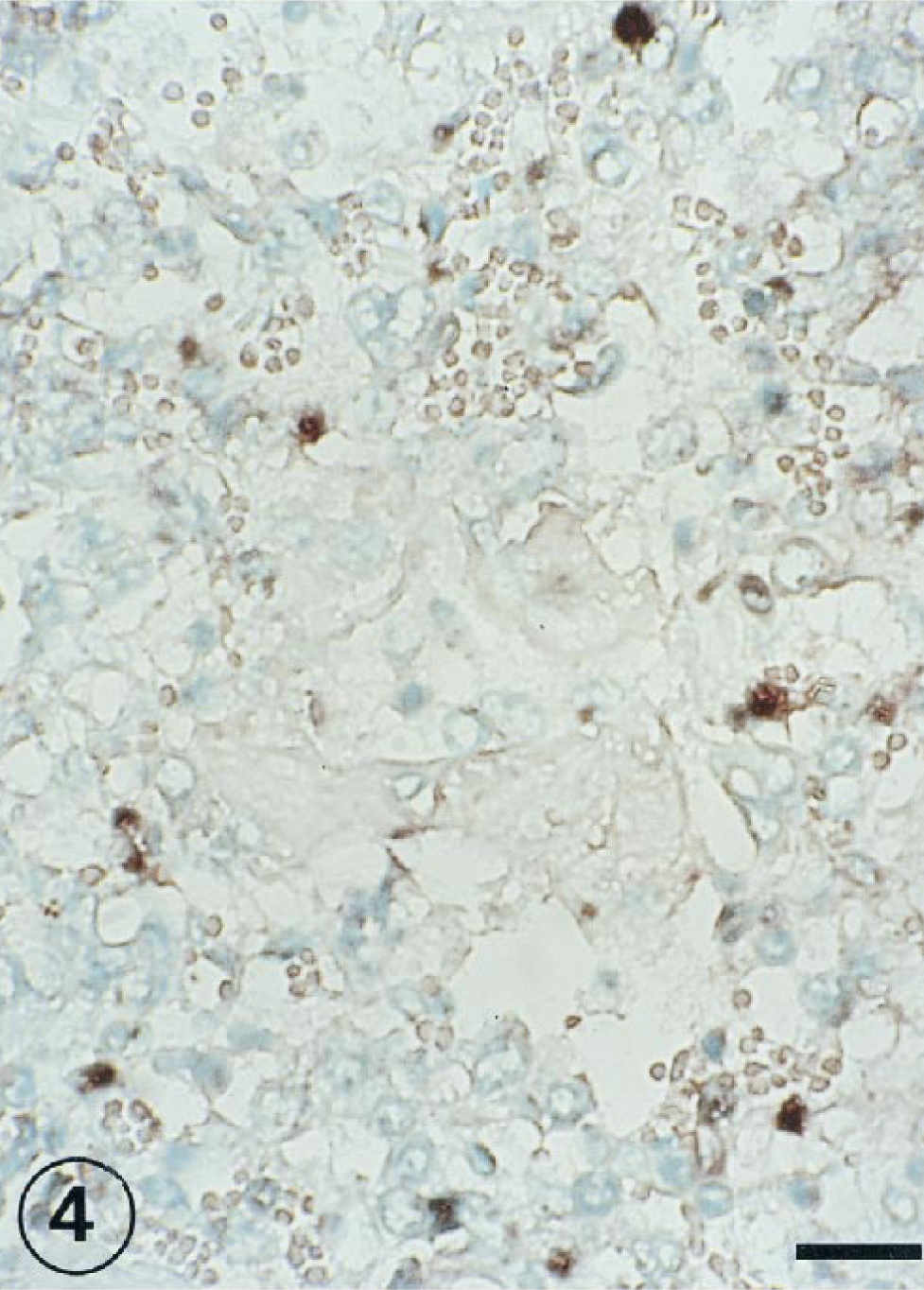
Lung; lambs inoculated intratracheally 1 day previously with BRSV. BRSV nucleic acid detected in alveolar epithelial cells. ISH counterstained with methyl green. Bar = 120 µm.
Lung; lambs inoculated intratracheally 7 days previously with BRSV. BRSV-infected cells located in alveolar epithelium. ISH counterstained with methyl green. Bar = 60 µm.
BRSV-inoculated lung tissue taken from an inoculated lamb at 3 PID. Large numbers of positive cells free in alveolar space. ISH counterstained with methyl green. Bar = 60 µm.
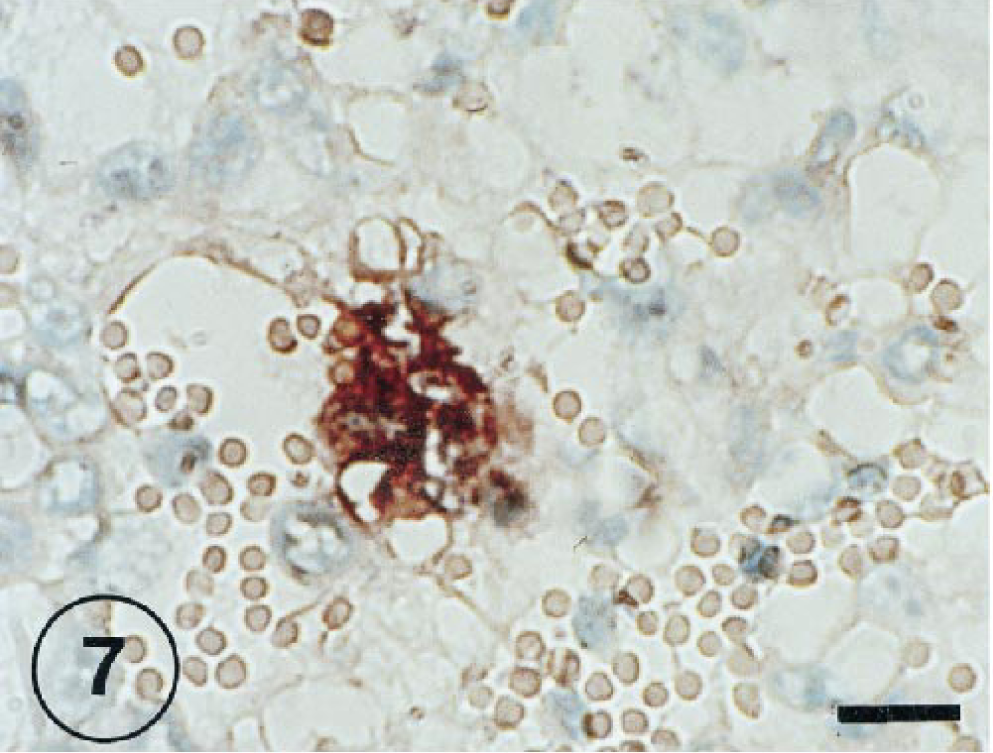
BRSV-infected lung tissue taken from an inoculated lamb at 7 PID. Syncytia containing BRSV-RNA-positive cells. ISH counterstained with methyl green. Bar = 60 µm.
Hybridization signal specific for BRSV RNA was observed in the peribronchiolar area (Fig. 8), in bronchus-associated lymphoid tissue (Fig. 9), and in mononuclear cells of interalveolar septum (Fig. 10) from 3 to 11 PID.
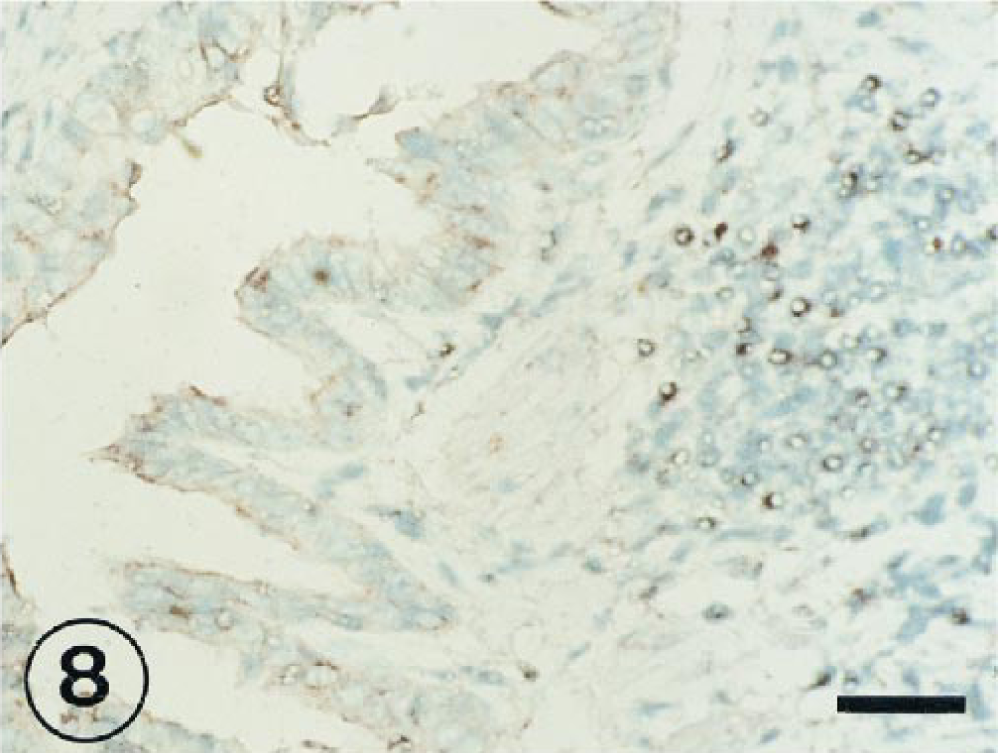
Positive signal in the peribronchiolar area of the lung of a lamb at 3 PID with BRSV. ISH counterstained with methyl green. Bar = 60 µm.
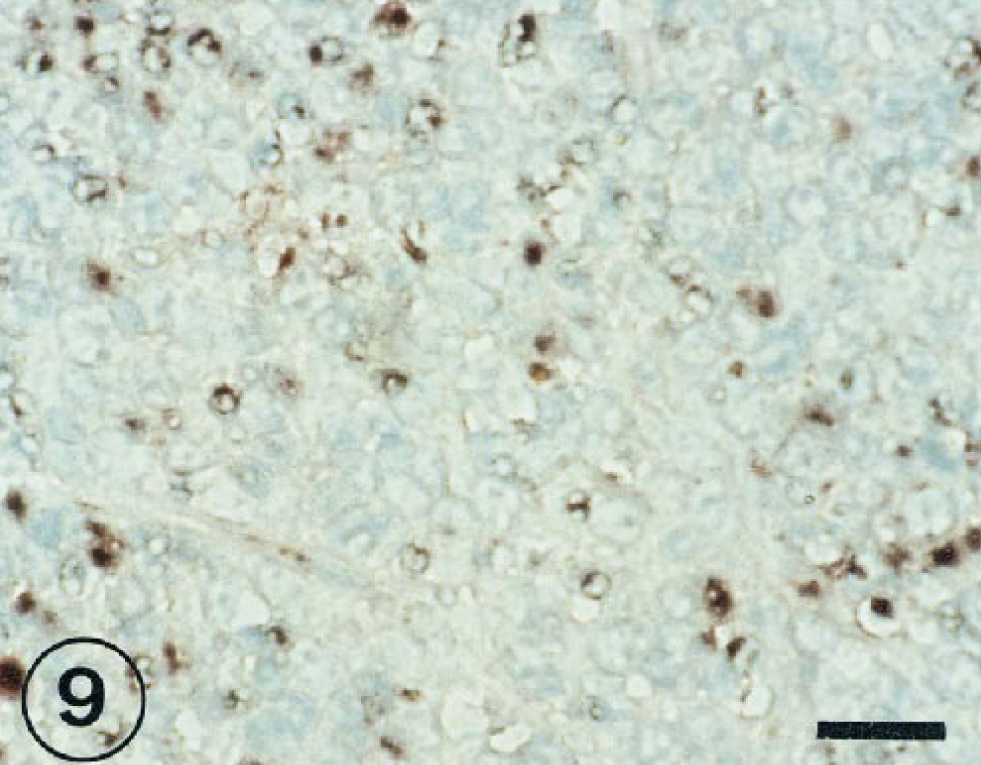
BRSV-inoculated lung tissue taken from an inoculated lambs at 7 PID. Large numbers of BRSV-positive cells in the bronchus-associated lymphoid tissue. ISH counterstained with methyl green. Bar = 30 µm.
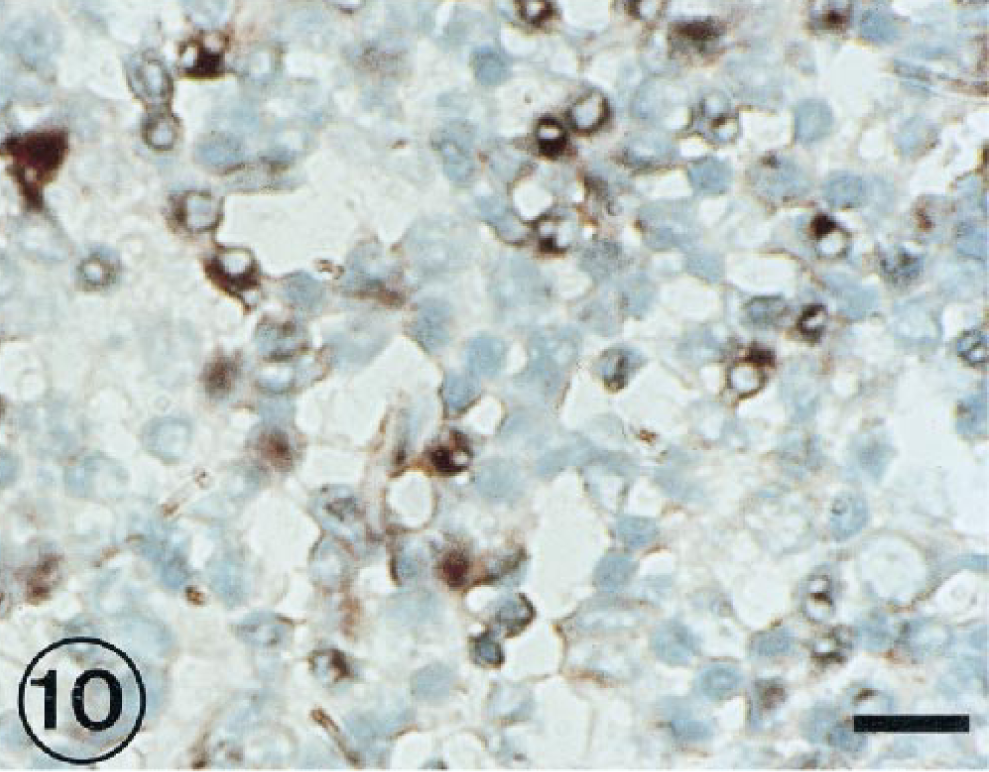
Lung; lambs inoculated with BRSV and slaughtered at 11 PID. BRSV-RNA-positive cells in interalveolar septa. ISH counterstained with methyl green. Bar = 60 µm.
Viral isolation
Results of BRSV isolation attempts from lung homogenates or nasal secretions are presented in Table 2.
Results of BRSV isolation from nasal secretions and lung homogenates.∗

+ = lamb positive by viral isolation or ISH signal; ± = weak ISH signal; − = negative.
Discussion
signal in bronchi and bronchioles is indicative of the direct pathogenic action of BRSV at these levels 36 inducing necrosis and desquamation of bronchial and bronchiolar epithelia. Necrosis of bronchial and/or bronchiolar epithelia has been described elsewhere as a limitation of pulmonary antibacterial defenses with impaired mucociliary clearance and as a predisposition to secondary bacterial infection. 1–3, 12 13 20–24, 30 36
Cells obstructing bronchial and bronchiolar lumina tend to form syncytia, 4 6 9 10 15 20–24 which are usually located in bronchioles and are rarely observed in bronchi. This finding has previously been reported in studies of IHC 23 24 or in in situ hybridization. 35 The possible involvement of type I and II pneumocytes in BRSV replication in sheep lung has been detected by IHC. 21 23 24 Extensive hypertrophy and hyperplasia of type II pneumocytes are also known to occur during acute BRSV infection in the lung. 20 22–24 In the present study, results with IHS bears out the role of type II pneumocytes in BRSV replication since the lung section had cells with positive IHS signals and that were morphologically and topographically consistent with type II pneumocytes. In this connection, other authors 35 suggest that the replication of BRSV in alveolar type II cells may lead to a change in the quantity and/or quality of surfactant. A deficiency of surfactant caused by BRSV replication in the alveolar type II cells could contribute to the collapse of alveoli seen in the lambs described in the present study. In order to confirm that type II pneumocytes represent a specific BSRV replication site, transmission electron microscopy should be performed on the same sections as those used for IHS.
In this experiment, a positive IHS signal for BRSV in alveolar macrophages could not be established. 36 These results agree with previous reports 33 suggesting that in vitro bovine alveolar macrophages (BAMs) exhibit a high intrinsic resistance for infection with BRSV and that bovine alveolar macrophages do not appear to be important for replication of BRSV. However, large numbers of BAMs may harbor virus antigen, even for several days, that may influence the function of these cells. 33 However, replication of human respiratory syncytial virus in alveolar macrophages has been shown. 14
A large number of alveolar syncytia containing BRSV-RNA were detected. The location of these syncytia coincided with the positive hybridization signals reported by Viuff et al. 36 and with the site of BRSV antigen detected elsewhere by IHC. 7 21 23 24 However, the reason for syncytia formation remains an enigma. Probably syncytia are a result of fusion of cells and a direct spread of virus from cell to cell. 36
In the present study, positive IHS signals were observed in mononuclear cells in the interstitial (interalveolar and peribronchial) space and in BALT of damaged areas from 3 to 11 DPI. The distribution and topography of specific hybridization signals in the interstitium was very similar to the location of BRSV antigen detected by IHC. 7 21 23 24 However, the site of BRSV–RNA positive cells in BALT was a differentiating feature since similar findings have not been reported elsewhere. 36
Interstitial cells expressing BRSV-RNA may carry virus particles to other organs, as described elsewhere. 19 31
The results obtained showed that more intense positive hybridization signals were detected at 3 and 7 DPI. It was precisely on these days that lesions were most intense. 20–24
In previously published experiments using parallel sections for IHC and IHS, 36 authors were able to show, through IHC, that sites containing viral antigen corresponded to virus replication sites.
It is a well-known fact that immunodetection requires a large number of molecules in order to obtain positive reactions. In prolonged postinoculation periods, the number of positive cells detected by immunohistochemistry decreased; it was also sometimes difficult to differentiate between positive and negative signals, especially with high levels of background noise. IHC therefore has more limitations than IHS; with impaired antigen production in infected cells, viral RNA in cells persists. 33
In the present experiment, BRSV was detected using ISH in several areas were IHC was negative. Further studies of comparison of consecutive serial sections are necessary to establish cell-to-cell correlation between the signal of both techniques.
Footnotes
Acknowledgements
A. J. Masot received a fellowship from the Dirección General de Investigación Científica y Técnica (Ministerio de Educación y Ciencia) of Spain.
The paper was supported by Hoeschst Roussel Agri-Vet Company, USDA Animal Health Funds, the Nebraska Agricultural Experiment Station.
