Abstract
Gross and histopathologic examinations were performed on 70 North American bison (
Paratuberculosis (Johne's disease), a chronic infectious disease of ruminants, has been described throughout the world since its discovery in 1895. The causative agent is
To our knowledge, paratuberculosis has been reported only recently in the North American bison (
Materials and Methods
Approximately 2,800 bison are grazed on 100,000 acres of a ranch in the foothills of the northern United States. The bison were originally acquired from various sources and were introduced to the ranch in 1989. The herd started with 100 bulls and 577 cows. Cattle occupied the ranch before the introduction of the first bison. Over the past 5 years, individual bison were noticed losing weight and failing to shed winter hair. The poor body condition was attributed to inadequate nutrition, endoparasites, and stress from herd social behavior. Management made adjustments to overcome the condition deficits, but these were to no avail. In July 1997, a thin adult female bison was subjected to a field necropsy and showed gross findings of mucosal corrugation in the distal small intestine and of enlarged mesenteric lymph nodes. These findings suggested Johne's disease. Histopathologic examination yielded a tentative diagnosis of Johne's disease. The bison tested negative on fecal culture for
For the histologic Johne's disease assessment, multiple tissues were examined independently by pathologists from five different institutions. Each pathologist examined tissues of only a subset of the animals under study.
Tissues from four animals that died in the field were randomly selected from a field necropsy, including the index case mentioned above (pilot animals 1–4, Group A), and were submitted to the Montana State Diagnostic Laboratory for a microscopic examination. A second group of tissues from 26 sick or dead bison (animals 1–26, Group B) were subjected to a complete field necropsy and were collected for various analyses, including microscopic examination. Tissue sections for microscopic examination included three sites in the jejunum, one site in the ileum, one site of jejunal and ileocecal lymph nodes, cecum, colon, heart, lung, spleen, kidney, liver, and abomasum. Alimentary tract tissues from Group B were microscopically examined by pathologists at the Montana State Diagnostic Laboratory, Cornell University, and the University of Wisconsin. A third group of 25 bison (animals 27–51, Group C) was sent to slaughter because of poor body condition and for Johne's disease surveillance. For these animals, a protocol was established to selectively take specimens from the lower jejunum, midjejunum, upper jejunum, associated mesenteric lymph nodes, ileum, ileocecal lymph node, cecum, colon, and other organs, as deemed necessary. Specimens from the Group C animals were microscopically assessed at the University of Florida and the University of Pennsylvania. Last, a subset of 15 bison (animals 52–66, Group D) was culled for reproductive failure, poor conformation, bad temperament, or Johne's disease surveillance. Ileum, mesenteric lymph nodes, and ileocecal lymph nodes were collected at slaughter. These tissues were microscopically examined at the University of Florida.
Tissues collected for histopathologic examination were fixed in 10% neutral buffered formalin, embedded in paraffin, and sectioned at 6 μm. All sections were stained with hematoxylin and eosin, and selected sections were stained with an acid-fast Ziehl-Neelsen or a Brown-Brenn Gram stain.
Histomorphologic criteria were used to categorize each animal as positive, suspicious, or negative for paratuberculosis. The criteria for a definitive histomorphologic diagnosis of paratuberculosis required the presence of noncaseating granulomatous inflammation composed of Langhans' type giant cells and epithelioid macrophages and the identification of at least one intracellular small acid-fast rod compatible with
The severity of the inflammation and the semiquantification of identifiable acid-fast bacilli were summarized by one investigator on the basis of the information rendered by other pathologists participating in the project. Lesions defined as noncaseating granulomatous inflammation were scored semiquantitatively by severity using a mild (1), moderate (2), and severe (3) scale of assessment. The category of acid-fast bacilli was semiquantitatively scored as paucibacillary or multibacillary. The bacterial score was subjectively compared with the inflammatory score.
The methodology of the cultural, serologic, and polymerase chain reaction (PCR) tests has been previously described. 28 Briefly, fecal and tissue cultures were cultured on Herrold's Egg Yolk, PCR was conducted by using primers of the IS900 sequence, and serum was tested on agar gel immunodiffusion (AGID) and with the CSL/IDEXX enzyme-linked immunosorbent assay (ELISA) Johne's kit.
Results
The cohort of animals, the necropsy and microscopic findings regarding paratuberculosis, the bacillary status based on the number of acid-fast bacilli stained, the host inflammatory response to the assumed presence of
Signalment and summary of test results in paratuberculosis-positive bison.∗
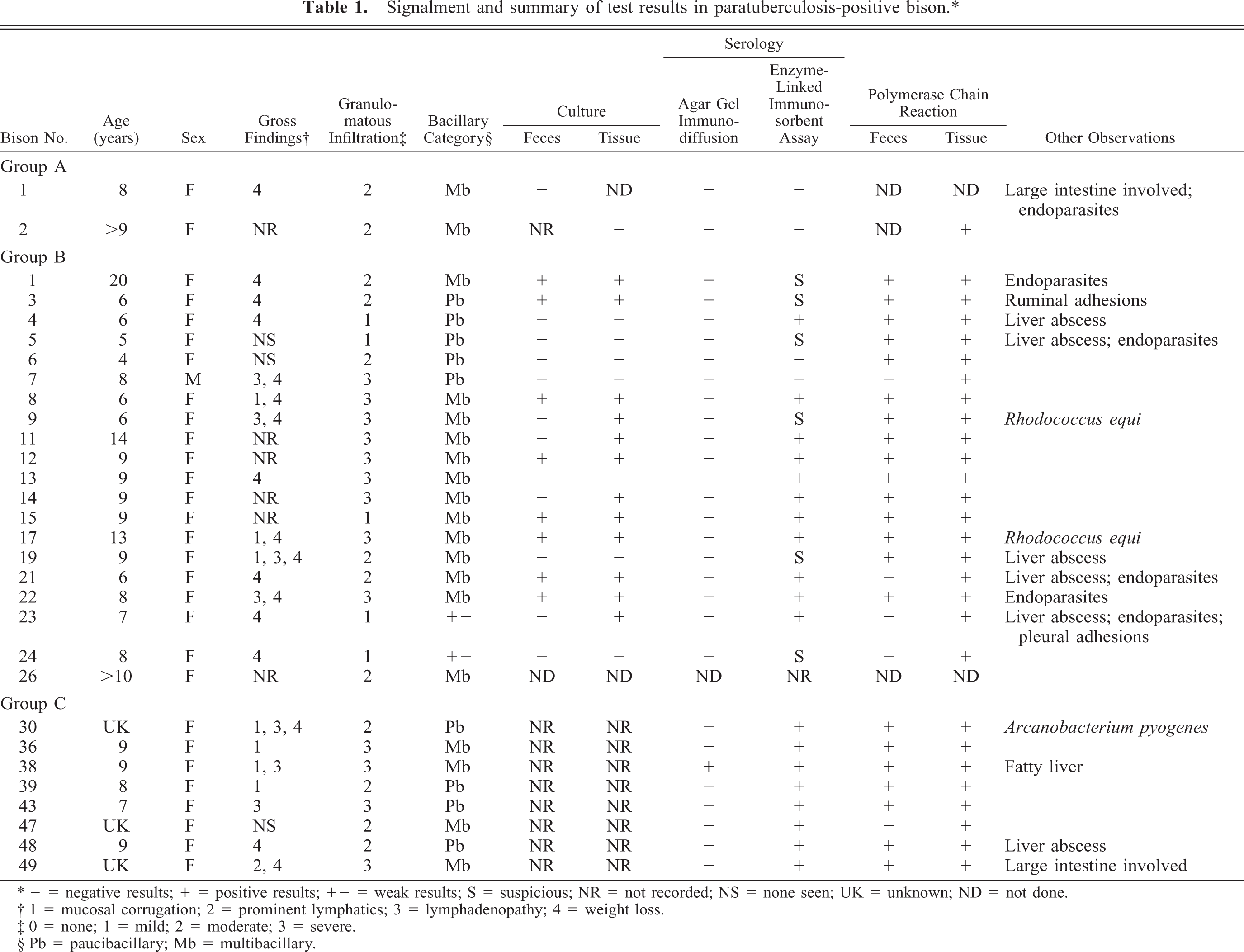
- = negative results; + = positive results; +- = weak results; S = suspicious; NR = not recorded; NS = none seen; UK = unknown; ND = not done.
1 = mucosal corrugation; 2 = prominent lymphatics; 3 = lymphadenopathy; 4 = weight loss.
0 = none; 1 = mild; 2 = moderate; 3 = severe.
Pb = paucibacillary; Mb = multibacillary.
Signalment and summary of test results in paratuberculosis-suspicious bison.∗
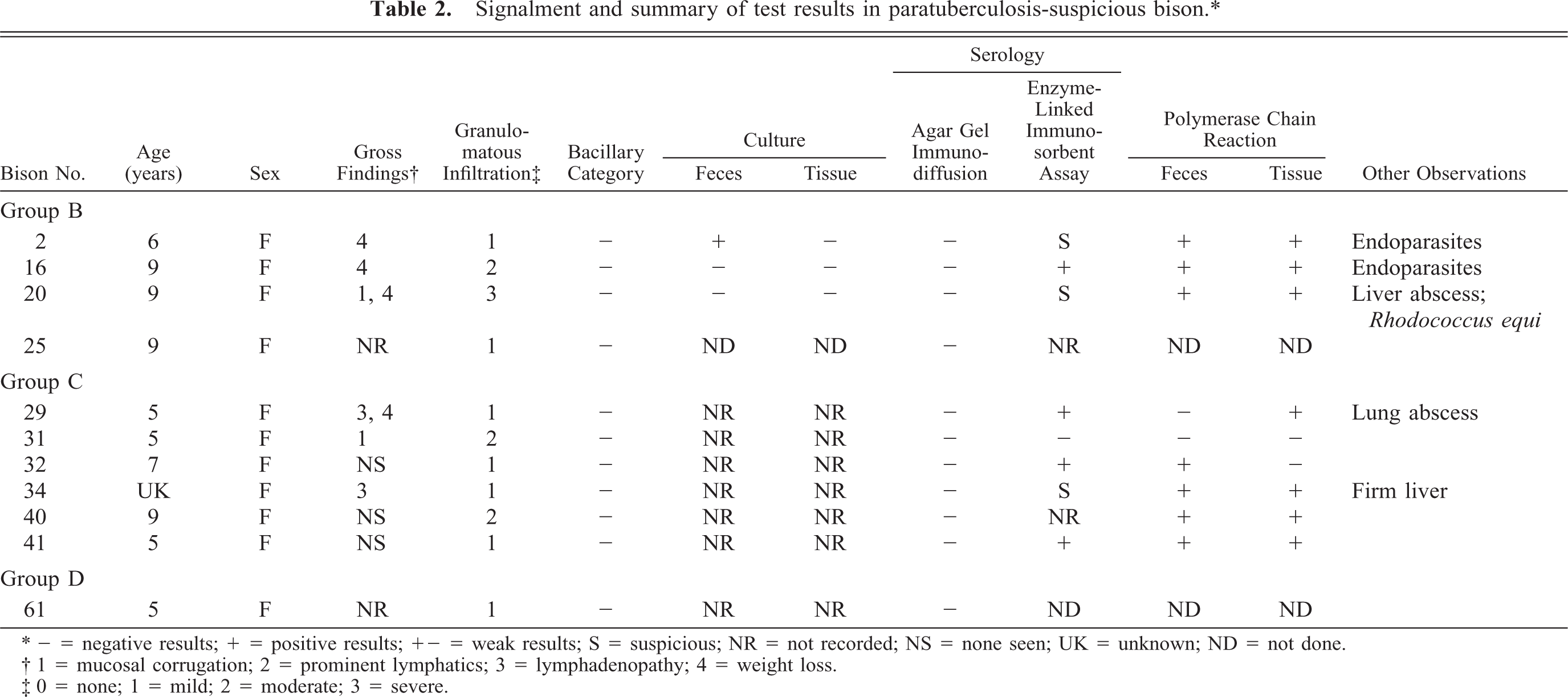
- = negative results; + = positive results; +- = weak results; S = suspicious; NR = not recorded; NS = none seen; UK = unknown; ND = not done.
1 = mucosal corrugation; 2 = prominent lymphatics; 3 = lymphadenopathy; 4 = weight loss.
0 = none; 1 = mild; 2 = moderate; 3 = severe.
Signalment and summary of test results in paratuberculosis-negative bison.∗
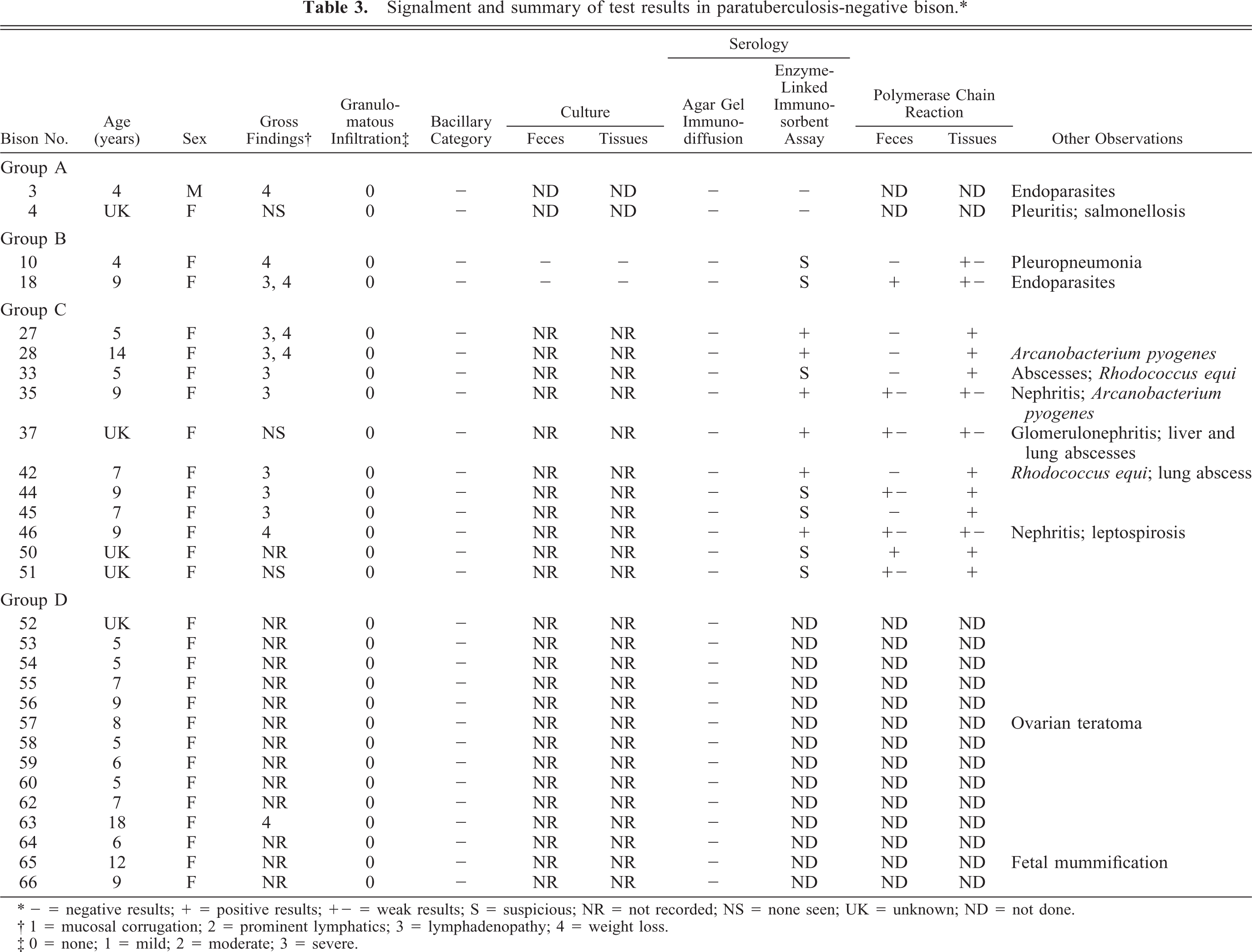
- = negative results; + = positive results; +- = weak results; S = suspicious; NR = not recorded; NS = none seen; UK = unknown; ND = not done.
1 = mucosal corrugation; 2 = prominent lymphatics; 3 = lymphadenopathy; 4 = weight loss.
0 = none; 1 = mild; 2 = moderate; 3 = severe.
Most (68 in 70; 97%) of the animals examined were female. Only two males were included in the collection. The mean age of all bison sacrificed was 7.9 years (range 4–20 years). The mean age of the 30 bison considered positive for paratuberculosis was 8.2 years (range 4–20 years).
Of the examined bison, the gross changes consistent with paratuberculosis included weight loss due to skeletal muscle mass reduction, atrophy of body fat, or both, in 28 of 70 (40%) animals; thickening of the mucosa of the small intestine in 8 of 70 (11%) animals; and prominent mesenteric or ileocecal lymph nodes in 16 of 70 (23%) animals. One animal had distended serosal lymphatics.
In four of the bison with gross findings suggestive of paratuberculosis, three bison had intestinal ulcers covered by a laminated caseating exudate in the mucosa of the small intestine. The fourth bison had caseating necrosis in mesenteric lymph nodes. Culture of the intestinal and lymph glandular exudates resulted in the isolation of
Thirty of 70 (43%) bison were considered positive for Johne's disease on the basis of established histomorphologic criteria. A suspicious diagnosis was obtained in 11 of 70 (16%) bison. Twenty-nine of 70 (41%) animals were assessed as histologically paratuberculosis free. The majority of the paratuberculosis-positive animals were from Group B; no animal was diagnosed as positive from Group D. Granulomatous infiltrations in the positive and suspicious paratuberculosis categories were graded as mild (1) in 12 of 70 (17%) bison, moderate (2) in 15 of 70 (21%) bison, and severe (3) in 14 of 70 (20%) bison. When there was a discrepancy of scoring between tissues from the same animal, the highest grade obtained from one tissue was applied to the final score.
Twelve (17%) of the 70 bison had mild cellular changes suggestive of paratuberculosis, but only two of these clearly had acid-fast bacilli indicative of
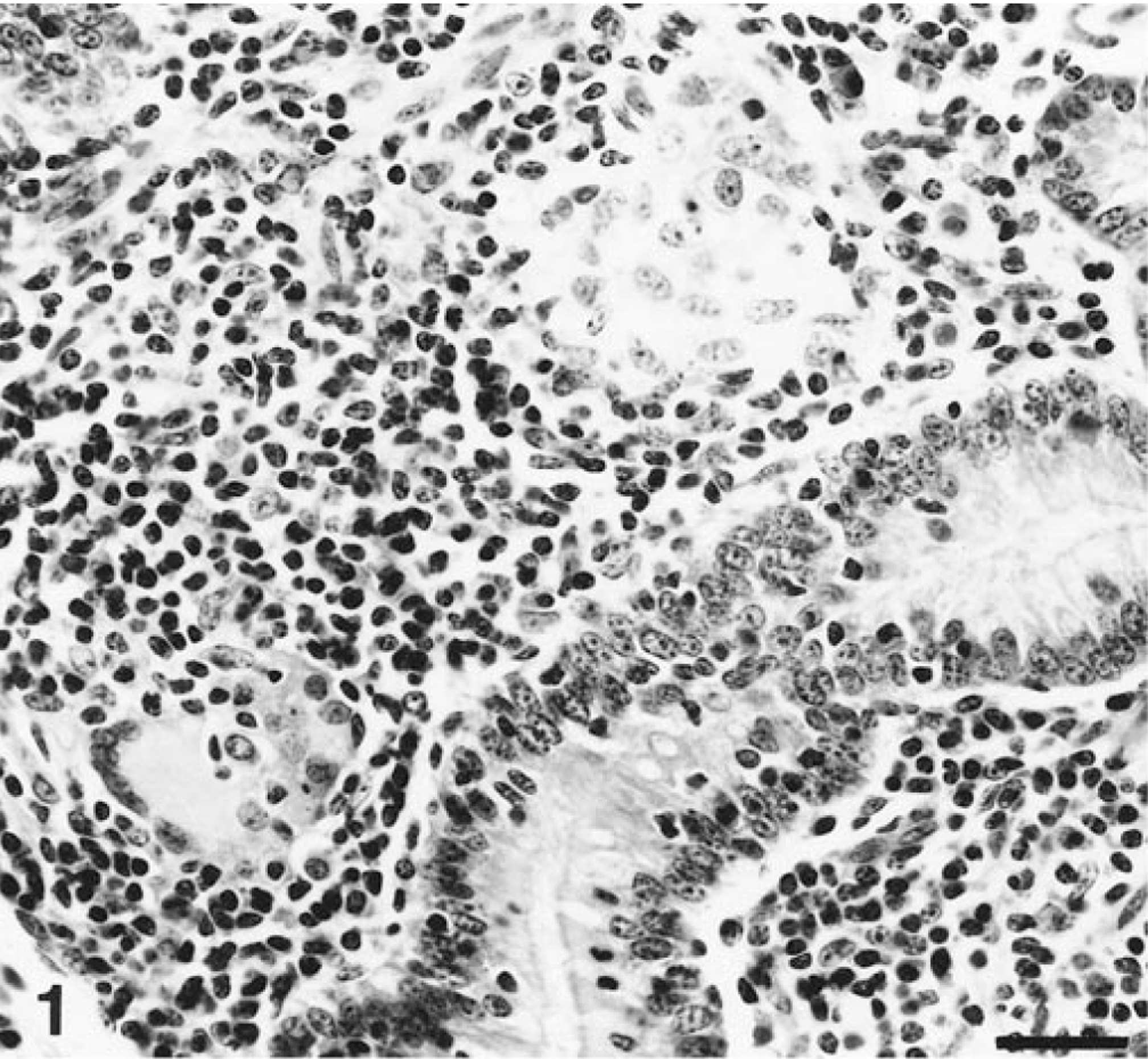
Upper jejunum; bison 40. Individual Langhans' type giant cell adjacent to mucosal crypt is embedded among resident lymphocytes of the lamina propria. HE. Bar = 25 μm.
Bison with moderate histologic lesions (15 of 70, 21%) had several small groups of epithelioid macrophages, often with several Langhans' type giant cells that were limited to the lamina propria of intestinal villi or within the paracortical zone of regional mesenteric lymph nodes. In eight of these bison, epithelioid macrophages were more numerous than giant cells; in four bison, the proportion of macrophages to giant cells favored giant cells; and in three other bison, only epithelioid macrophages were noted (Fig. 2). When specific sites of the lymph node selection were known (Group C), moderate granulomatous inflammation was present in the midjejunal, upper jejunal, and ileocecal lymph nodes, with the midjejunal lymph nodes showing characteristic changes in most of the cases.
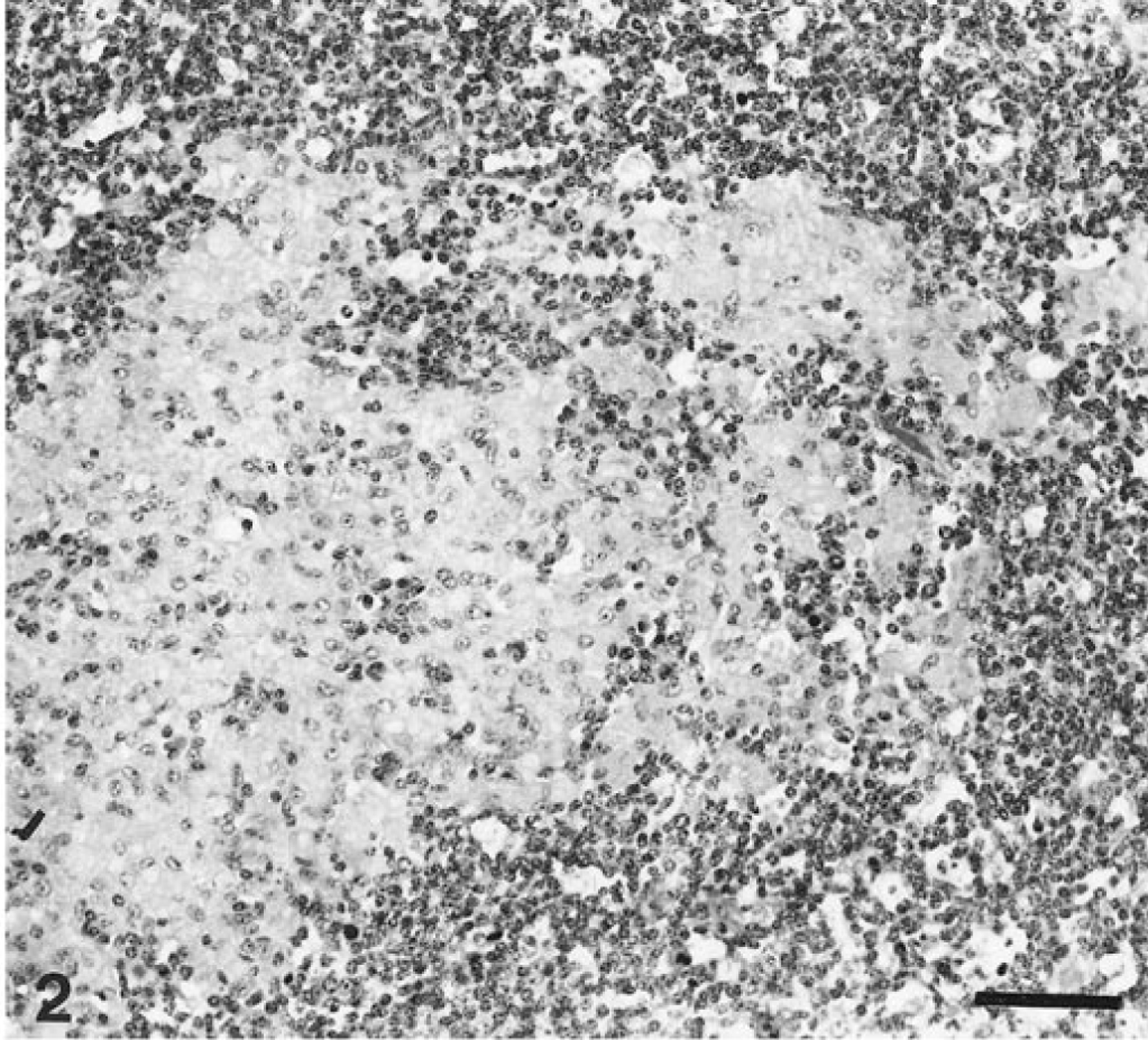
Mesenteric lymph node; bison 41. Moderate Johne's disease. Cluster of epithelioid macrophages present within the paracortex of the mesenteric lymph node. HE. Bar = 50 μm.
Many macrophages and lesser numbers of Langhans' type giant cells had infiltrated the lamina propria and submucosa of various segments of the small intestine in bison with severe lesions (14 of 70; 20%) of paratuberculosis (Fig. 3). Some of these inflammatory cells had invaded the tunica muscularis and tunica serosa as well. Granulomatous cell infiltrates cuffed intramural intestinal lymphatics. Intestinal villi were short and distorted. Villous lacteals were prominent because of dilatation. The granulomatous inflammatory response extended to the adjacent mesenteric lymph nodes. The lymphoid cells within the paracortical zone were largely replaced by epithelioid macrophages and giant cells. Lymphoid follicles were spared. In two animals (I and 49), the cecum and spiral colon also had granulomatous inflammation. Seven bison in this group had inflammation composed exclusively of epithelioid macrophages, whereas one bison had predominantly giant cells present. Six other bison had a mixed granulomatous inflammation with primarily epithelioid macrophages.
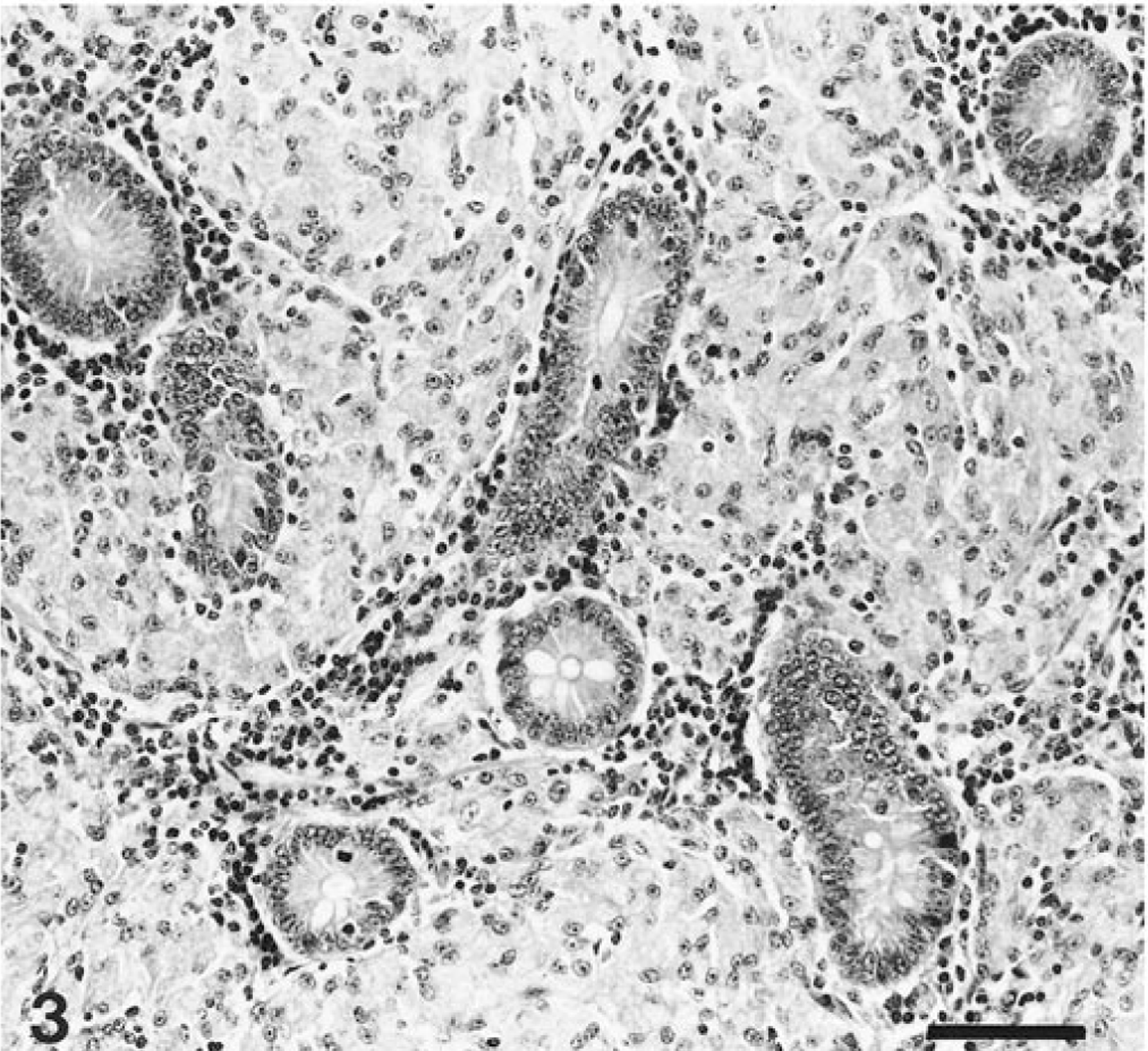
Small intestine; bison 50. Advanced Johne's disease. Epithelioid macrophages and individual Langhans' type giant cells have infiltrated the lamina propria and have replaced mucosal crypts. HE. Bar = 50 μm.
All bison with paratuberculous granulomatous inflammation had uniformly formed, well-differentiated epithelioid macrophages with dense eosinophilic cytoplasm, distinct cell boundaries, eccentric oval hyperchromatic nuclei, and several nucleoli of various sizes.
In the animal cells that stained positive with Ziehl-Neelsen stain, nine had a few (usually 1–3) acid-fast–positive bacilli within giant cells. This group of animals would fit the category of paucibacillary infection. Nineteen bison had a plethora of acid-fast bacilli, mainly within macrophages and typically arranged in clusters (Fig. 4). These animals would fit the category of multibacillary infection.
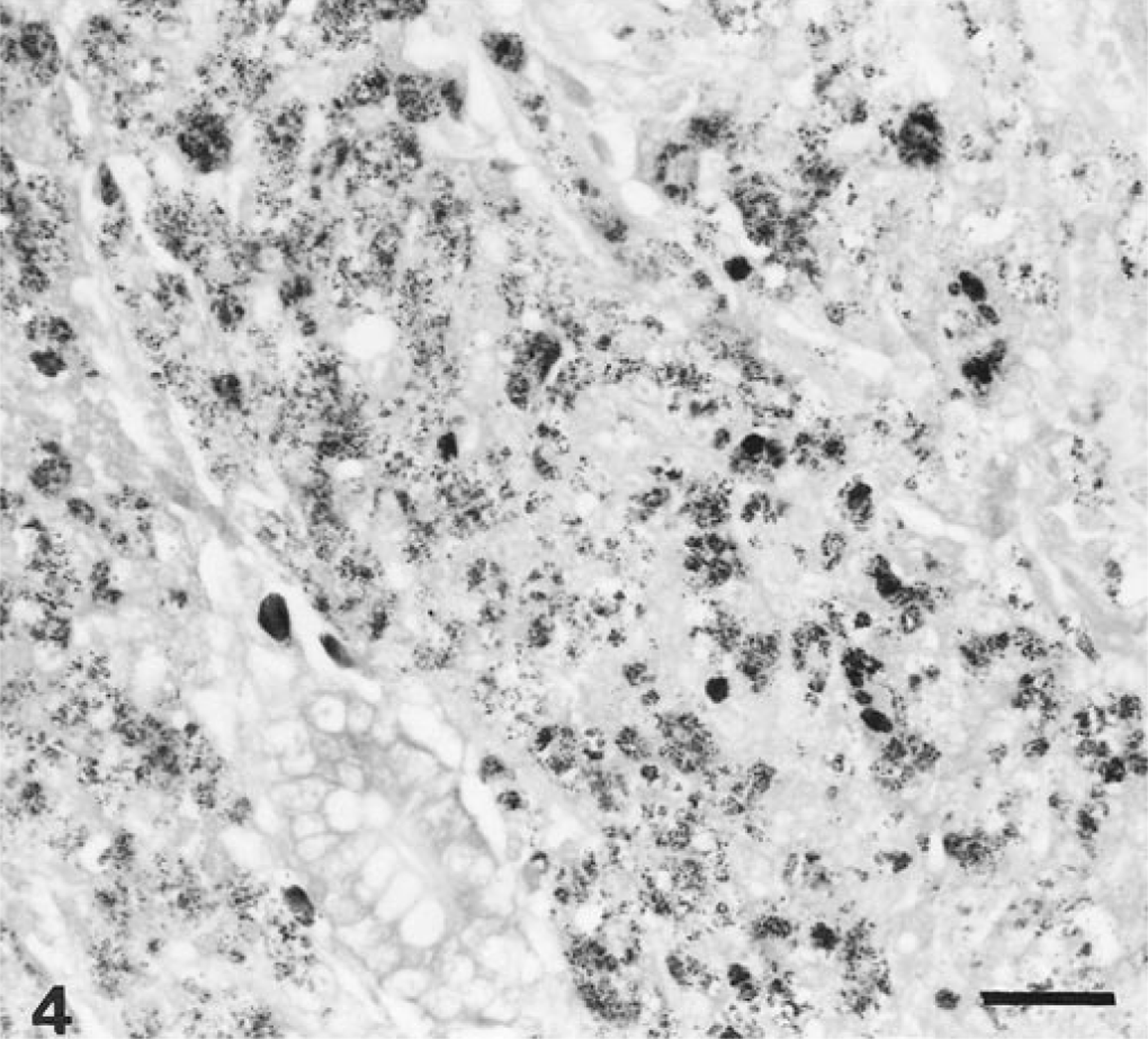
Small intestine; bison 50. Clusters of
In four animals (9, 17, 20, and 42), the histologic changes of paratuberculosis were accompanied by necrotizing pyogranulomatous changes due to
The antemortem tests showed one (3%) of the 30 bison morphologically positive on AGID; 19 (63%) bison were positive on ELISA. With the fecal culture system, 8 of 20 morphologically positive bison were identified as shedders, whereas in the same group, 12 animals had a positive tissue culture. Fecal PCR identified 22 of 30 (73%) correctly but also had eight additional bison as positive; tissue PCR identified 100% of the morphologically positive animals but also 15 of the suspicious and negative bison.
Discussion
The distribution and character of pathologic changes of spontaneous bison paratuberculosis were similar to the lesions of bovine paratuberculosis. 3 27 There was protracted weight loss leading to a state of emaciation. Gross intestinal lesions were most easily seen in the distal segments of the small intestine and there was gross evidence of enlargement of mesenteric lymph nodes of up to 3–4 times the normal size. Histologically, advanced cases of paratuberculosis had diffuse, severe, noncaseating granulomatous inflammation of the distal small intestine and associated mesenteric lymph nodes. Histologic changes of subclinical paratuberculosis were rather mild.
The granulomatous inflammation was noncaseating and composed of epithelioid macrophages with varying numbers of intermingled multinucleate Langhans' type giant cells. Animals with paucibacillary and multibacillary paratuberculosis were identified, the latter accompanied by severe granulomatous inflammation. Other animals had no visible acid-fast bacilli but had mild changes with individual giant cells, small clusters of epithelioid macrophages, or both, best seen in mesenteric lymph nodes.
The absence of demonstrable acid-fast bacilli in the inflammatory giant cells and epithelioid macrophages has prompted the creation of a morphologic category of paratuberculosis-suspicious animals if epithelioid macrophages, but, more typically, giant cells, are detected in target tissues despite the animal's exhibiting clinical signs suggestive of Johne's disease and its origin from a herd of established paratuberculosis-positive status. By applying this criterion to our case material, 11 bison would have been classified as suspicious.
The Langhans' type giant cells may be the first to form within the lamina propria or, together with small clusters of epithelioid macrophages, within the paracortex of the mesenteric lymph nodes in the cases of mild multifocal lesions. Langhans's type giant cells are thought to arise by fusion of mature macrophages. 1
Epithelioid cells have been described as the most differentiated and the end stage of stimulated phagocytic macrophages. They are derived from circulating monocytes of bone marrow origin.
1
Macrophages turn into epithelioid cells when they become immobilized at the site of inflammation. Epithelioid cells are active in the phagocytosis and degradation of ingested bacilli. They have a secretory function, which may partially explain the calcification of cardiac endocardium and the aorta in some bovine animals infected with
The epithelioid macrophages of mild paratuberculosis should not be confused with enlarged resident macrophages of the intestinal tract and associated lymph nodes that contain yellow granules or crystalline material in their cytoplasm. 18 The latter are readily distinguished from epithelioid cells and giant cells because of the pigment and because of the observation that they have not been demonstrated to contain acid-fast organisms. The cytoplasm of these resident macrophages constantly stained deep blue with acid-fast stain, suggesting metabolic activity. 18
It is probably the combined capability of activated epithelioid macrophages and giant cells that destroys phagocytized mycobacterial microbes in the early phases of infection with
In four animals, the demonstration of gram-positive intracellular small coccoid rods suggested the etiologic diagnosis of
Infection with lungworms (
The bison with confirmed paratuberculosis originated from other paratuberculosis-positive bison herds, so it is likely that the disease was introduced to the ranch as a preexisting, subclinical condition at the time of purchase of the original bison rather than from cattle previously inhabiting and grazing the ranch or from free-range wildlife. At least six different bison herds supplied original animals to the ranch. A strain fingerprinting by restriction-length fragment polymorphisms (RLFP) analysis resulted in the identification of B-C1,CC-1, and CC-18 strain types in the bison herd as evidence of multiple strains having infected the herd.
17
28
These strains have also been identified in cattle.
17
The fingerprinting method has been proposed to identify the identity of
A restraining aspect for Johne's disease control of the herd was the dilemma associated with the diagnostic deficiency of the tests applied to the bison. 28 The 70 bison of the different subsets were subjected to one or several different Johne's disease tests (fecal and tissue culture methods, AGID, ELISA, tissue, and fecal PCR) commonly used for cattle.
The AGID test identified only one (3%) of the 30 mophologically positive bison, whereas the ELISA test, after some modification, identified 19 (63%) of the morphologically positive bison. Only 25 bison were subjected to fecal culture, and 8 of the 20 (40%) of the morphologically positive bison were correctly identified. One bison morphologically classified as suspicious had a positive fecal culture. The same 25 bison were also subjected to tissue culture, and 12 of 20 (60%) of them were correctly identified. Finally, 49 bison were subjected to a PCR test on feces, and 50 were subjected to a tissue PCR test. The fecal PCR identified 22 of 30 (73%) of the morphologically positive bison correctly, but it also identified eight additional bison as positive when the other tests, including the histologic tests, were negative in these animals. The tissue PCR identified 100% of the positive bison, but it also identified 15 of the suspicious and negative bison as positive.
From the comparison of these results, we concluded that the incidence of paratuberculosis infection in bison was higher when using PCR testing than histopathology or other tests. Some of the tests correlated with each other, whereas others did not. In some circumstances, acid-fast bacteria were microscopically detected in the tissue of bison, but the organisms could not be cultured from feces or tissues, and specific antibodies could not be detected from serum samples. This suggests that tests that work for cattle need to be modified to make them work reliably for bison. The discrepancy between tests prompted the overall conclusion that the results of attempts to diagnose paratuberculosis in bison with the test methods usually applied to cattle do not follow the pattern of sensitivity and specificity expected from these tests in cattle.
Prevention of paratuberculosis infection and the establishment of paratuberculosis-free populations is a management goal. This goal is extremely important when livestock mix with free-range wildlife or when one species of free-range wildlife mixes with another. 15 There are presently no treatment modalities or effective vaccines available for paratuberculosis control. Control of infection and disease is recommended through test-and-cull procedures combined with management and hygiene adjustments. 7 Vigorous removal of animals with poor body condition should be practiced with routine monitoring for Johne's disease by gross examination and histopathology at the time of culling. Of the 28 bison with poor body condition, 17 (61%) had a final positive diagnosis of paratuberculosis after microscopic examination. The observation of failing body condition created a simple, inexpensive, presumptive first antemortem test for paratuberculosis infection on the ranch once the presence of Johne's disease was originally established. Animals identified in such a way were then subjected to various Johne's disease tests.
Finally, it is very important to establish and adhere to morphologic criteria for the confirmatory diagnosis of paratuberculosis, as histologic evaluation, next to fecal or tissue culture, is presently considered the gold standard for paratuberculosis diagnosis. 27 Comparison among the various institutions involved in the histologic assessment of tissue material obtained from the bison originally showed disparity for the diagnosis of paratuberculosis status in a few animals among pathologists because of different morphologic definitions used. The disparity was resolved, and reclassification was done by adhering to a uniform definition of a confirmatory positive diagnosis for paratuberculosis if the histologic findings revealed acid-fast rods together with Langhans' type giant cells and epithelioid macrophages, and by adhering to the uniform definition of a suspicious diagnosis if the findings showed giant cells and/or epithelioid macrophages but no acid-fast rods.
Pathologists should not be divided as to morphologic criteria used for a positive diagnosis of paratuberculosis. Only in some countries, such as Australia, have standard recommendations for histopathologic assessment and reporting of results been implemented as a quality assurance program. 4 8 Specific recommendations have been drafted, including how many tissues, which specific tissues, how many sections, and for what time duration these sections should be examined under the microscope.
There should be global uniformity of interpreting and reporting pathologic findings in paratuberculosis. This uniformity will ultimately benefit the efficacy of herd surveillance programs for Johne's disease control and eradication.
Footnotes
Acknowledgements
We thank Mrs. Glenda Eldred, Mrs. Betty Hall, and Ms. Sherron Muzquiz for excellent technical help in the preparation of the glass slides.
