Abstract
In an effort to correlate the likelihood of in utero transmission of
Johne’s disease is a bacterial disease of ruminants that is most commonly transmitted via the fecal–oral route, though in utero and milk-borne infection with the causal organism
The current management recommendation for herds trying to minimize Johne’s disease is to avoid feeding milk and colostrum from test-positive cows, but recommendations regarding how to manage the calves born to those cows are vague. Typically, “retaining calves from known positive cows as herd replacements is discouraged.” 2 Unfortunately, such a recommendation does not provide clear guidance for decision making about whether or not to keep a calf born to a cow with a specific category of test results. The project described herein was performed in an effort to determine the likelihood of in utero infection of calves born to cows with a wide range of Johne’s disease test results.
The calves used in the current study were male Jersey calves born on a dairy that had been participating in the National Johne’s Disease Demonstration Herd Project since 2003. Cows in the herd had been routinely tested by fecal culture and serum ELISA at dry off, and historical test results were available. The herd had a fecal culture–positive rate of approximately 7.7% (Table 1). Over a 1-year period, tissues were collected from 49 calves and tested for the presence of MAP by culture or polymerase chain reaction (PCR). Animals of interest were male calves born to cows that had previously had serum ELISA a results >0.25 and/or positive fecal culture b and bull calves born on the same day to test-negative cows. Study personnel were notified by the dairy when appropriate calves were born, and the calves were collected for transportation to the California Animal Health and Food Safety Laboratory (CAHFS; Tulare, CA) within 24 hr of birth. The calves were euthanized with barbiturate solution upon arrival at CAHFS (UC Davis animal use protocol no. 07-12847). Because the removal of calves from the cow and the maternity pens immediately after birth could not be guaranteed, the abomasal and ruminal contents of the calves were evaluated grossly for the presence of milk or milk curd in order to verify the animals had not suckled. If any volume of white fluid or milk curd was found, the animal was removed from the study.
Test results from 1 full year of dry cow testing for
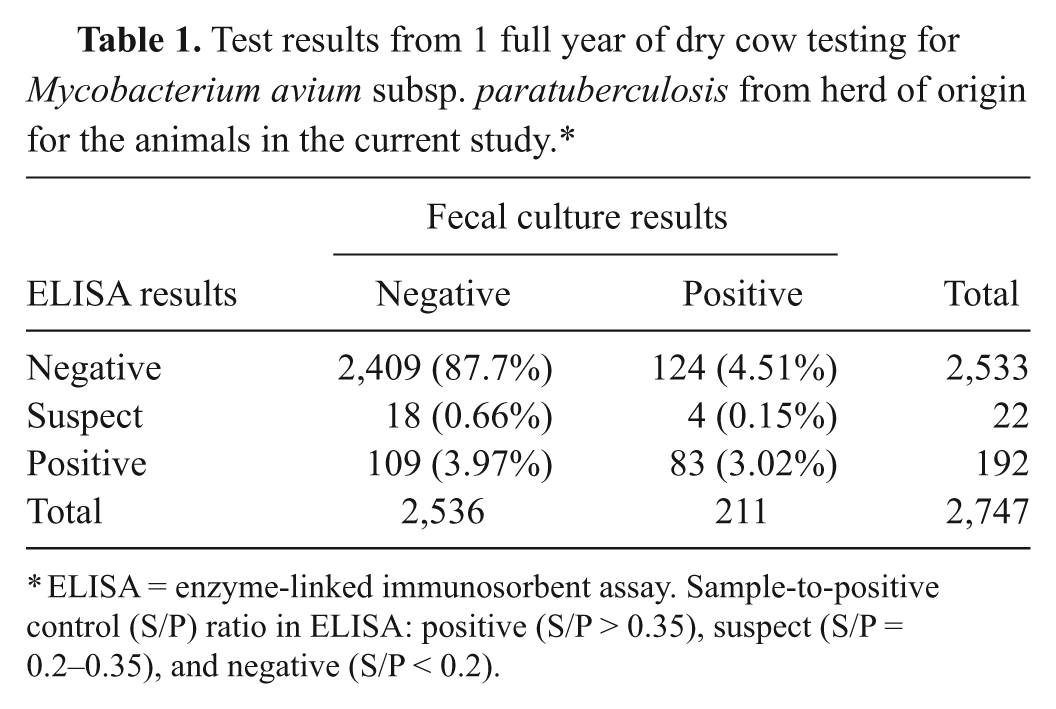
ELISA = enzyme-linked immunosorbent assay. Sample-to-positive control (S/P) ratio in ELISA: positive (S/P > 0.35), suspect (S/P = 0.2–0.35), and negative (S/P < 0.2).
After euthanasia, liver, kidney, spleen, ileocecal valve, ileocecal lymph node, 3 pools of mesenteric lymph nodes (proximal, middle, and distal), and 3 separate ileum samples, for a total of 11 samples, were harvested from each calf. Organ samples were collected first and intestinal samples last to minimize the chances of contamination by intestinal content. The samples were placed on ice immediately, and, when tissue collection had been completed, were frozen at −70°C until testing of the samples could be performed. In addition, postpartum milk, feces, and serum were collected from dams of calves included in the study. Samples were collected from the cows (within 14 days of calving on average), transported to CAHFS, and frozen at −70°C until testing of the samples could be performed.
Serum ELISA testing was performed according to the manufacturer’s instructions. a Serum samples (20 µl) were diluted with the sample diluent (380 µl) and were allowed to incubate at room temperature for 30 min. Diluted serum samples were placed into 2 individual wells of antigen-coated MAP antibody test kit plates and incubated for 30 min at room temperature. The liquid was removed from the wells, and the wells were then washed 4 times with phosphate buffered saline. Following the last wash, the plates were inverted and gently tapped on a paper towel in order to remove excess residual fluid. Horseradish peroxidase conjugate (100 µl) was placed into each well and incubated at room temperature for 30 min. The wash step was repeated. Tetramethylbenzidine substrate solution (100 µl) was placed in each well and incubated for 15 min at room temperature, after which time 100 µl of stop solution was added to each of the wells. The absorbance was measured at 620 nm, and the sample-to-positive control (S/P) ratio was calculated according to manufacturer’s instructions using the mean sample A620.
For fecal cultures, 2 g of feces was mixed with 35 ml of triple deionized water in a sterile 50-ml centrifuge tube and mixed on an orbital shaker at 150 rpm for 30 min. The tube was allowed to stand undisturbed at room temperature for a minimum of 30 min. Using a sterile transfer pipette or autopipettor, a 5-ml aliquot of sample was removed from the top portion of supernatant and added to a tube of half-strength brain heart infusion (BHI) broth with 0.9% cetylpyridinium chloride. The contents were mixed and allowed to stand undisturbed and upright for 18–24 hr in a 35–37°C incubator. The following day, the tube was centrifuged at 3,000 ×
Cows from which calves were collected were classified based on historical test results and test results from samples collected after freshening (Table 2). Twenty-one out of 49 were positive by both fecal culture and serum ELISA, 2 were fecal culture positive and suspect on serum ELISA, 5 were positive by fecal culture alone, 1 was positive by serum ELISA alone, and 20 were negative on both test methods. Of the 5 cows that were fecal culture positive but ELISA negative, all had days to positive of 42 or more. Of the 28 fecal culture–positive animals, 6 would have been considered high fecal shedders (<21 days to positive), 4 would have been considered moderate fecal shedders (21–28 days to positive), and the remaining 18 were low or very low fecal shedders (>28 days to positive). The single cow that was ELISA positive but fecal culture negative had an S/P ratio of 0.781 on one sample date, but 2 subsequent ELISA values were 0.168 and 0.058.
Distribution of cumulative testing results for
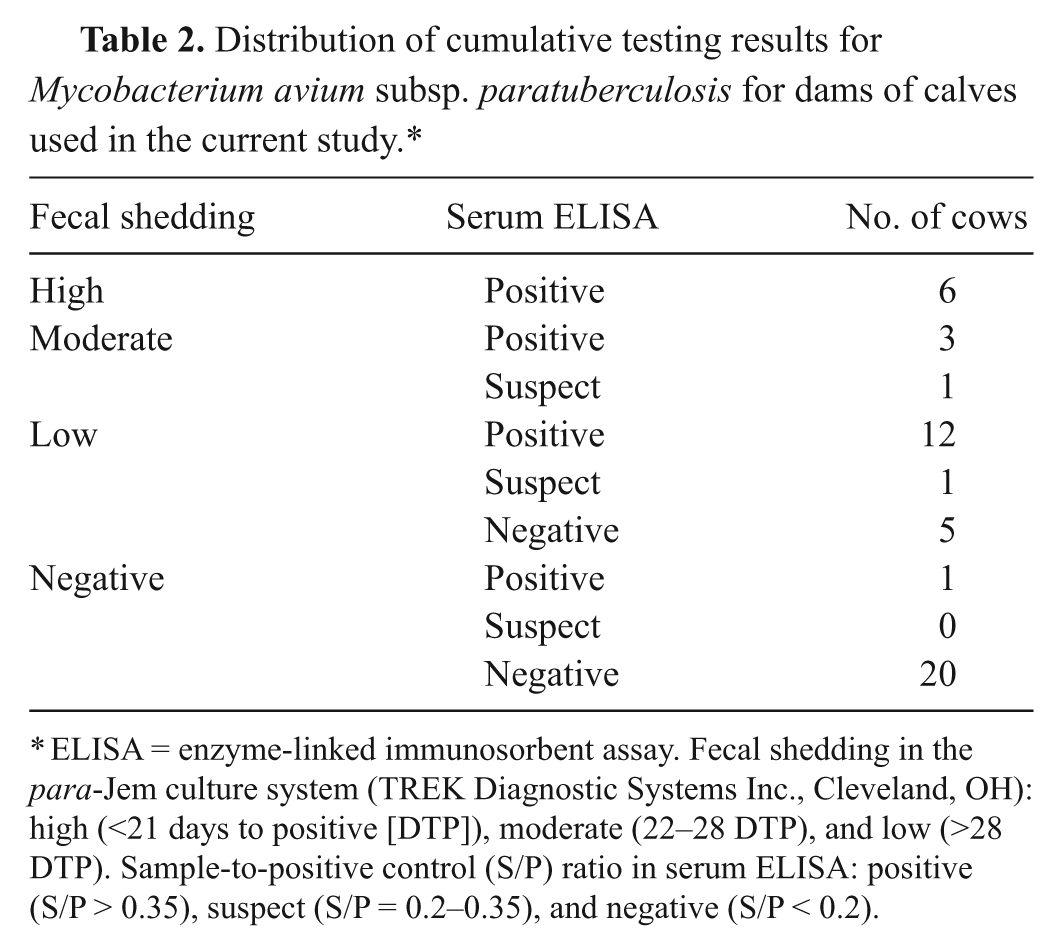
ELISA = enzyme-linked immunosorbent assay. Fecal shedding in the
Testing of calf tissues by culture was performed by the Johne’s Research Laboratory at the University of Pennsylvania (New Bolton Center, Kennett Square, PA). Tissue cultures were performed using methods similar to those described previously.
4
Briefly, 2 g of tissue were added to 25 ml of 0.7% hexadecylpyridinium chloride and homogenized with a stomacher for 2 min. The material was poured into a sterile 50-ml screw-top falcon tube and were left to stand at room temperature for 30 min. Fifteen milliliters of supernatant was removed from the top of the fluid phase, placed in a second falcon tube, and left to stand at room temperature for 3 hr. The sample was centrifuged at 900 ×
Only bull calves were used in the current study, but it is considered unlikely that there was any effect of calf gender on the frequency of in utero infection; therefore, any findings in the male calves in the present study should be equally applicable to female calves. A more significant opportunity for misclassification of the calves in the current study could be attributable to the test methods used to find MAP in the calf tissues. Tissues from known infected calves were used as controls; however, the sensitivity of the tissue culture assays is not known, and it is possible more calves were infected with MAP than were detected.
Footnotes
a.
HerdChek
b.
c.
BACTEC MGIT 960 Mycobacterial Detection System, BD Diagnostic Systems, Sparks, MD.
d.
Tetracore Inc., Rockville, MD.
The author(s) declare they do not have any conflict of interest with respect to the research, authorship, and/or publication of this article.
The authors received funding from the Center for Food Animal Health (CFAH), School of Veterinary Medicine, UC Davis, and the Johne’s Disease Integrated Program (USDA-CSREES-NRI 2008-55620-18710).
