Abstract
The role of γδ T cells in the bovine immune response to Mycobacterium avium subsp. paratuberculosis (M. paratuberculosis) infection is poorly understood. Accordingly, using BALB/c mice that are innately susceptible to M. paratuberculosis, we compared wild-type and γδ T cell knockout BALB/c mice to study the protective roles of γδ T cells in M. paratuberculosis infection. Ten-week-old mice were inoculated intraperitoneally with either a low dose (4 × 106 colony-forming units [CFU]/mouse) or a high dose (4 × 109 CFU/mouse) of M. paratuberculosis strain ATCC 19698. Histopathologic and morphometric examinations showed reductions in the number and area of granulomatous lesions in the liver of the knockout mice at 18 weeks after inoculation with either the low or the high dose of the mycobacteria. Furthermore, at 18 weeks after inoculation, the bacterial load in the spleens of the knockout mice inoculated with the high dose was significantly lower than that of wild-type mice. No differences were found in bacterial load between the knockout and the wild-type mice in the low-dose groups. In contrast, in the livers of wild-type mice inoculated with either the low or high mycobacterial dose, increased areas of epithelioid granulomata were observed and the granulomata became disseminated widely during the experimental period. These findings in model mice suggest that γδ T cells, rather than restricting mycobacterial growth, may play a crucial role in development of epithelioid granulomata similar to those seen consistently in bovine paratuberculosis. The results of this study may have relevance to our understanding of the pathogenesis of paratuberculosis in ruminants, in which a prominent number of γδ T cells exist in the lymphoid system.
Mycobacterium avium subsp. paratuberculosis (M. paratuberculosis) causes a chronic infectious disease of domesticated and wild ruminants in many parts of the world. The disease, paratuberculosis, is characterized clinically by chronic diarrhea and progressive emaciation, and pathologically by diffuse granulomatous enteritis and lymphadenitis. 7 To date, it has been established that protective cell-mediated immune response against M. paratuberculosis and other mycobacteria species is mediated mainly by αβ T cells including CD4+ T cells 31 and CD8+ T cells. 6 Thus, γδ T cells have been suggested to play a protective role in the early infectious stage of diseases caused by mycobacteria. 25 27 and other intracellular bacteria. 10 29 Proliferation and activation of γδ T cells at the site of infection are induced by 70-kd mycobacterial heat shock protein 5 and cytokines. 18 28 36 Some investigators have demonstrated that activated γδ T cells produce proinflammatory cytokines, 2 15 34 and generate immunoreactive macrophages, αβ T cells, and natural killer (NK) cells. 3 Therefore, γδ T cells are thought to bridge the gap between the phagocytic system and the highly evolved type of immune response mediated via αβ T cells in host defense against microbial pathogens. 11 However, some investigators have called into question the inhibitory role of γδ T cells in mycobacterial growth in vivo. 9 33 Moreover, other studies suggested that γδ T cells are involved only in recruitment of inflammatory cells to the site of infection. 19 35
In ruminants, the actual functions of γδ T cells as immune regulatory cells in mycobacterial infection have not been clarified. This is largely due to the difficulties associated with the execution of in vivo experiments in the natural host animals. These problems may be overcome by selection of appropriate experimental animal models. We previously reported that wild-type BALB/c mice inoculated with M. paratuberculosis had increased numbers and size of granulomata similar to those seen consistently in bovine paratuberculosis, and that the mycobacterial load in the liver and spleen was significantly increased compared to C3H/HeJ mice. 37 Therefore, we proposed that innately susceptible BALB/c mice might be useful models for bovine paratuberculosis. To clarify the role of γδ T cells in M. paratuberculosis infection, we carried out a comparative study between wild-type BALB/c mice and γδ T cell knockout BALB/c mice by evaluation of granuloma formation and the mycobacterial growth. We report here that γδ T cells may be important for the development of epithelioid granulomata in M. paratuberculosis infection but that γδ T cells do not seem to be necessary for elimination of the mycobacteria.
Materials and Methods
Mice
Female TcR-δ−/− mutant BALB/c mice (γδ T cell knockout mice group) and TcR-δ+/+ wild-type BALB/c mice (wild-type mice group) were used in the present experiments. Thirty mice from each group were used beginning at 10 weeks of age. The knockout mice, which lack the TcR-δ gene, have been described previously. 16 Briefly, chimeric mice were produced by injecting ES clones into wild-type BALB/c mice. A homogeneous TcR-δ+/− population was established by back-crossing δ heterozygotes to BALB/c mice more than five times. The resultant heterozygotes (TcR-δ+/−) were bred to obtain the TcR-δ−/− homozygotes. The γδ T cells were not detected in any of the lymphoid or epithelial organs from the knockout mice by flow cytometry. The wild-type BALB/c mice used in the present experiment were purchased from a commercial supplier (JSLC Co., Hamamatsu, Japan). Mice were maintained under specific-pathogen-free conditions and offered feed and water ad libitum.
Experimental design
Twenty mice of each group were inoculated intraperitoneally with either a low dose (4 × 106 colony-forming units [CFU]/mouse) or a high dose (4 × 109 CFU/mouse) of M. paratuberculosis strain ATCC 19698 (American Type Culture Collection, Rockville, MD). 21 As noninfected controls, the remaining 10 mice of each group were inoculated with 0.5 ml of sterile physiologic saline (control mice group). Five mice from each group were sacrificed at 6 and 18 weeks after inoculation via cervical dislocation. From each animal, the liver was collected for histologic and morphometric examination. The spleens were assayed for bacterial growth, and all other abdominal and thoracic organs were used for histopathologic examination. Except for the spleens, the organs were fixed in 10% formalin in 0.1 M phosphate buffer (pH 7.2) and processed for paraffin wax embedding. Five-micrometer-thick sections were stained with hematoxylin and eosin (HE), and by the Ziehl–Neelsen method.
Morphometric assay
Four sections stained with HE prepared from the liver of each mouse were used for morphometric experiments. The method for measurement of granuloma area in the livers has been described previously. 37 Granuloma formation was evaluated by the number and size of granulomata, with a granuloma being defined as a collection of >10 macrophages. The total number and area of granulomata in 25 randomized microscopic fields at 400× magnification in each mouse were counted or measured with an image analyzer (Luzex F, Nireco Co., Tokyo, Japan). Thereafter, the mean number and area of granulomata per mm2 were calculated.
Bacterial growth
At 6 and 18 weeks after inoculation, the whole spleen from each mouse was removed and homogenized. M. paratuberculosis was grown on modified Herrold's egg yolk medium. 23 CFU of M. paratuberculosis, per spleen were determined by plating 10-fold serial dilutions of organ homogenate from each mouse. CFU were expressed as the mean ± SD of three experiments.
Statistical analysis
Quantitative data are expressed as mean ± SD of five mice per group. The statistical significance of the data was determined by analysis of variance using Sheffe's F-test at 0.5% of significant level.
Results
Histologic studies
In livers of wild-type BALB/c mice inoculated with the high dose of M. paratuberculosis, small multifocal granulomata composed predominantly of macrophages and epithelioid cells with a few lymphocytes were found in the periportal and midzone of the hepatic lobule at 6 weeks after inoculation (Fig. 1). At 18 weeks after the high-dose inoculation, the granulomata in the wild-type mice were larger, caused by an increase in numbers of macrophages and epithelioid cells with occasional multinuclear giant cells. Furthermore, some nodular granulomata seemed to adhere together, forming larger nodular epithelioid granulomata throughout the hepatic parenchyma (Fig. 2). Therefore, the area of each granuloma was greater and the number of granulomata per microscopic field was lower than that on week 6 after inoculation. Similar epithelioid granulomata were also observed in the low-dose wild-type mice group, but the number of component cells was smaller than that of the high-dose group at 6 and 18 weeks after inoculation. Frequently, those epithelioid granulomata contained many acid-fast bacteria without apparent signs of central necrosis and caseation in the wild-type mice inoculated with either the low or high dose. Only a few acid-fast bacteria were found at sites other than in the epithelioid granulomata. On the other hand, in livers of the knockout BALB/c mice inoculated with the high dose of the mycobacteria, nodular granulomata composed primarily of macrophages with many lymphocytes and a few neutrophils were observed on week 6 after inoculation (Fig. 3). At 18 weeks after the high-dose inoculation, the granulomatous lesions that were composed of macrophages and lymphocytes without epithelioid cells and multinuclear giant cells were noticeably reduced with respect to the number of component cells (Fig. 4). Nevertheless, phagocytized acid-fast bacteria were detected widely in the macrophages and Kupffer cells throughout the liver. In livers of the knockout mice inoculated with the low dose, the granulomatous lesions were minimal and acid-fast bacteria in those lesions were rare at 6 and 18 weeks after inoculation.
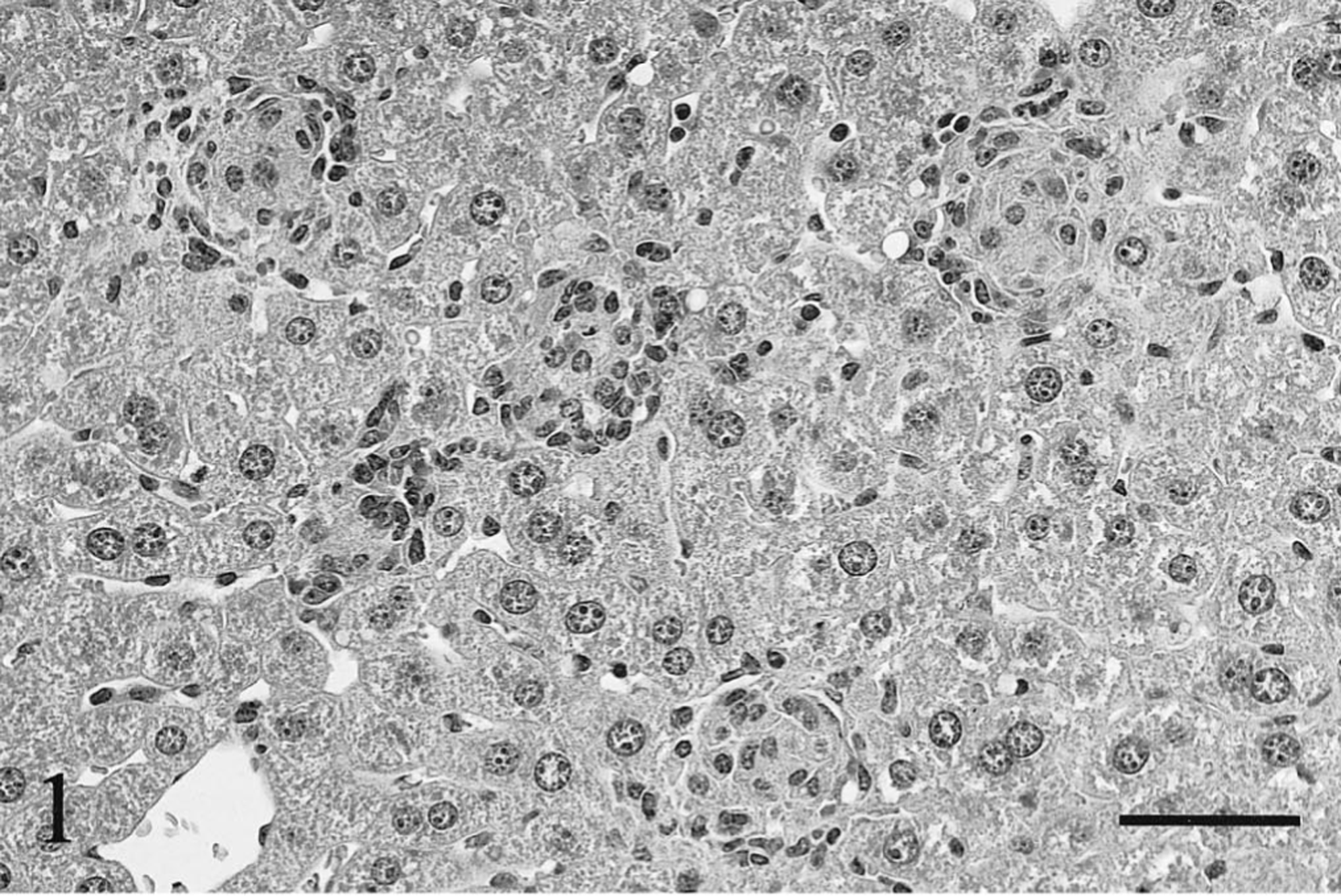
Liver; wild-type BALB/c mouse, 6 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. The granulomata mainly consist of epithelioid cells enclosed by lymphocytes. HE. Bar = 50 μm.
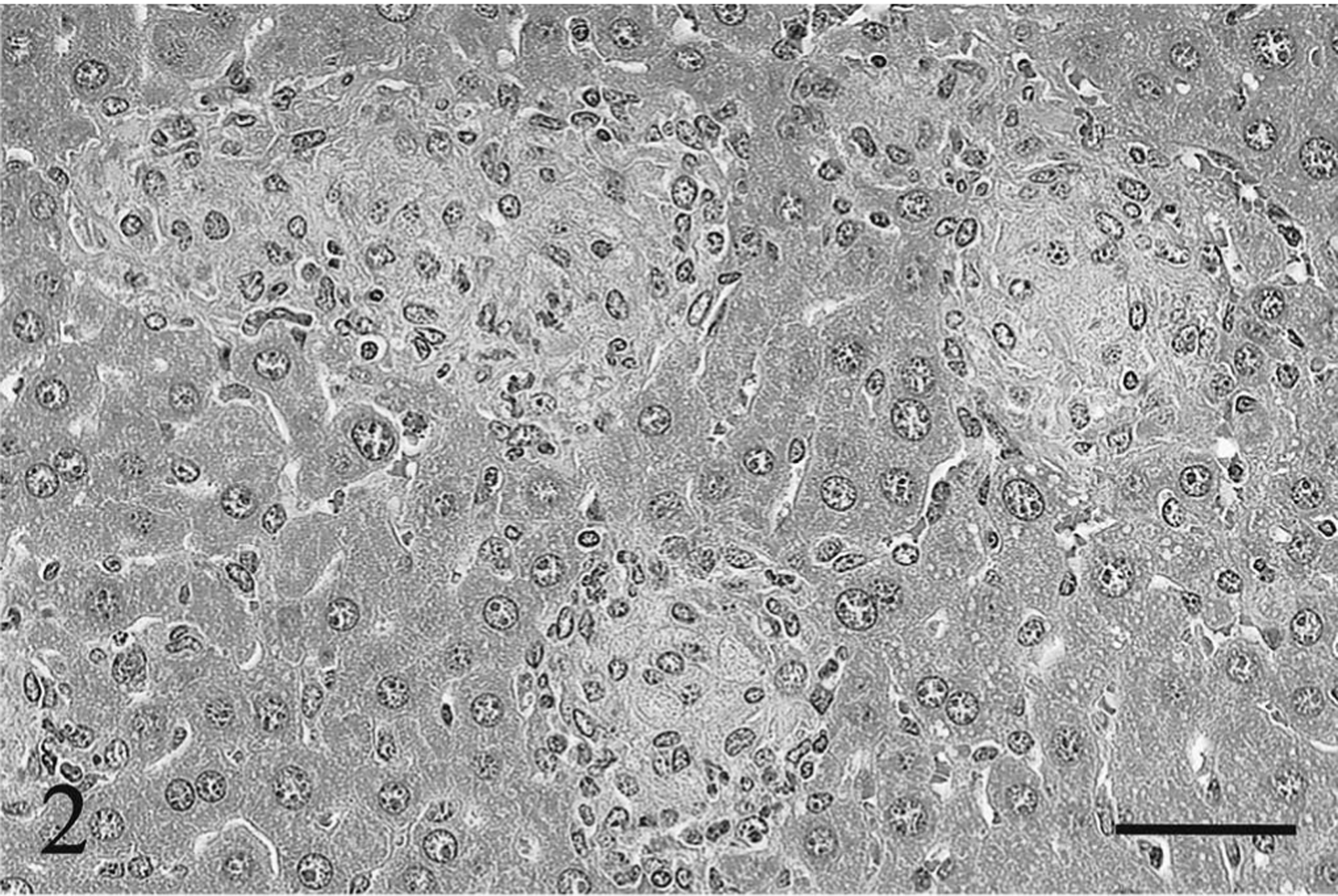
Liver; wild-type BALB/c mouse, 18 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. Nodular epithelioid granulomata without other inflammatory cell accumulation are observed. HE. Bar = 50 μm.
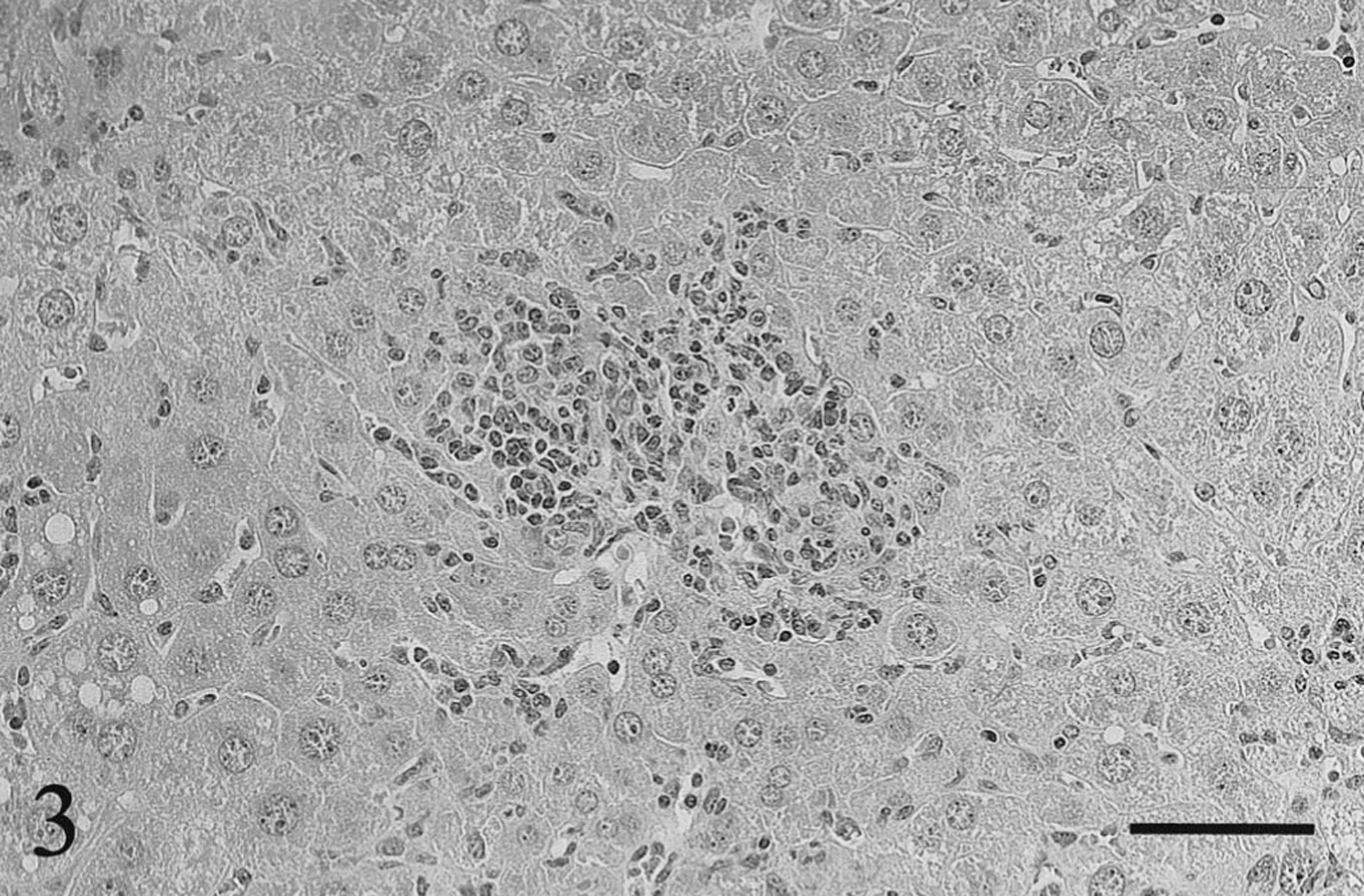
Liver; γδ T cell knockout BALB/c mouse, 6 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. A lymphocytic granuloma is found, yet epithelioid cells are few. HE. Bar = 50 μm.
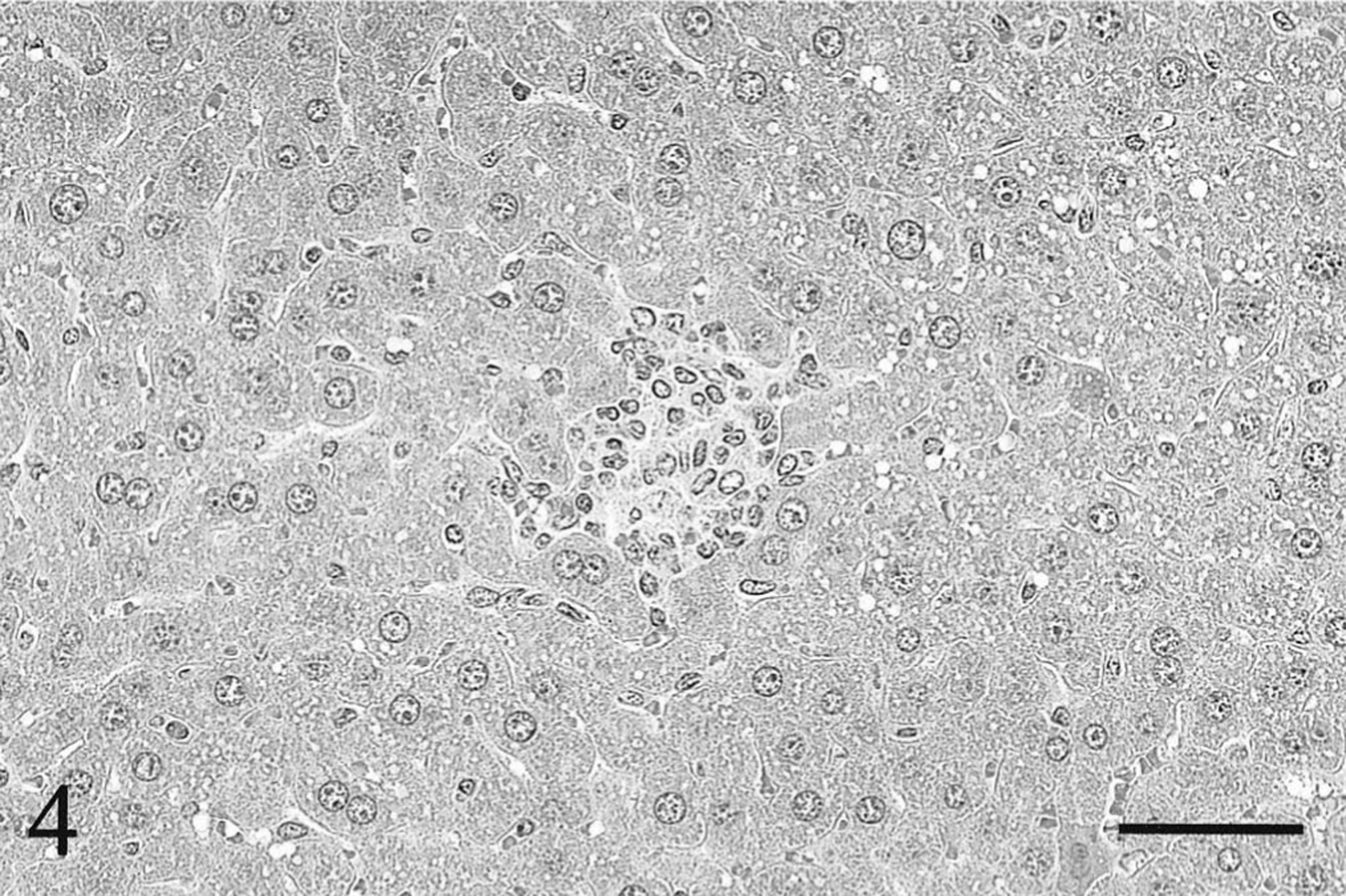
Liver; γδ T cell knockout BALB/c mouse, 18 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. The number and size of granulomata are decreased compared with that seen at 6 weeks in Fig 3. HE. Bar = 50 μm.
In the other organs, a few acid-fast bacteria phagocytized by macrophages were found in the cortex of mesenteric lymph nodes, the intestinal serosa, the serosa of urinary bladder and uterus, and the pleura of the lungs in both the wild-type and knockout mice inoculated with the high dose of the mycobacteria at 6 and 18 weeks after inoculation. Additionally, only in the wild-type mice at 18 weeks in the high-dose group, nodular granulomata containing acid-fast bacteria were detected in both the cortex and medulla of the thymus as well as in the lymphoid follicles of Peyer's patches (Figs. 5, 6).
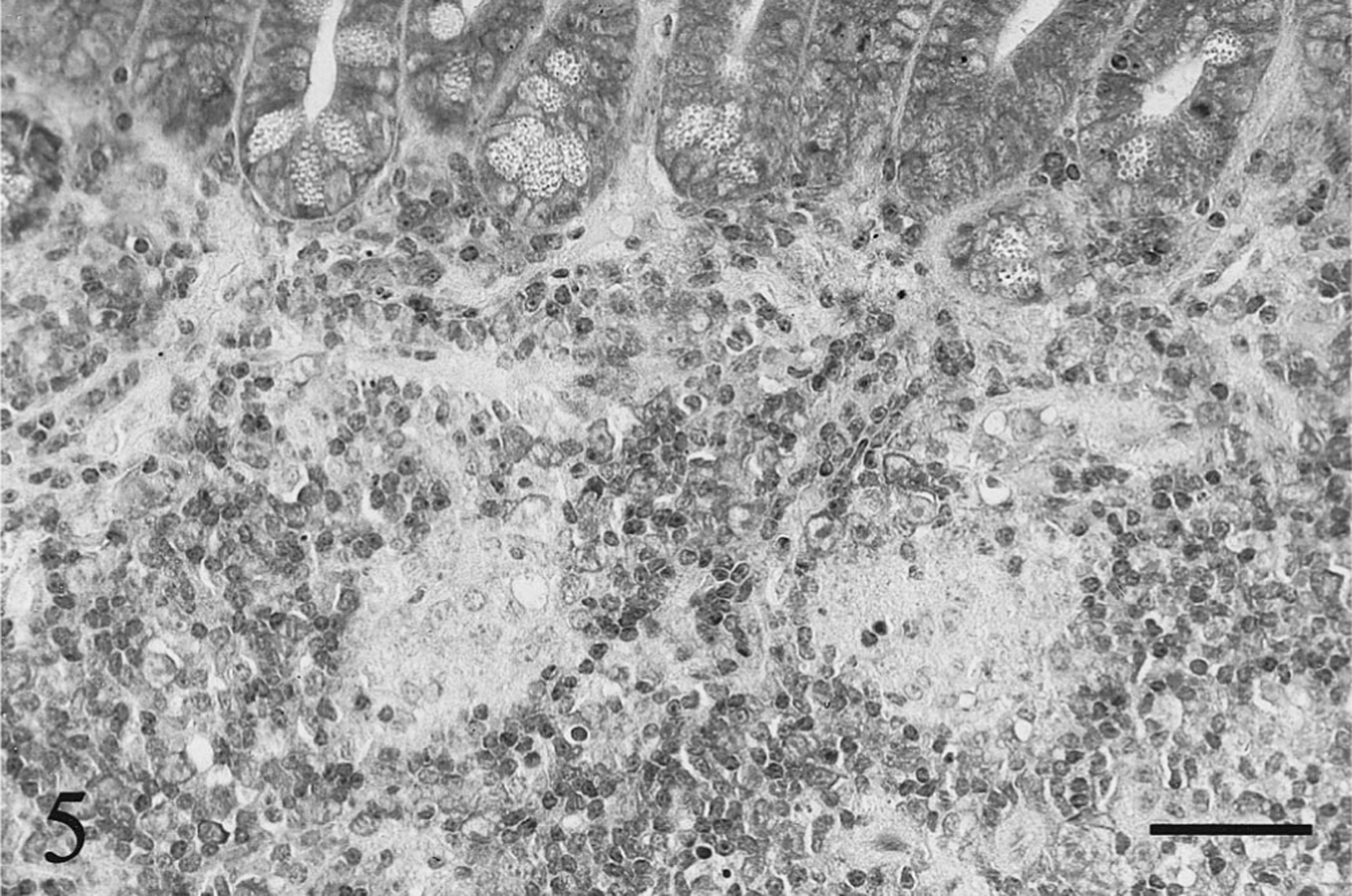
Small intestine; wild-type BALB/c mouse, 18 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. Nodular epithelioid granulomata are found in a lymphoid follicle of Peyer's patch. HE. Bar = 40 μm.
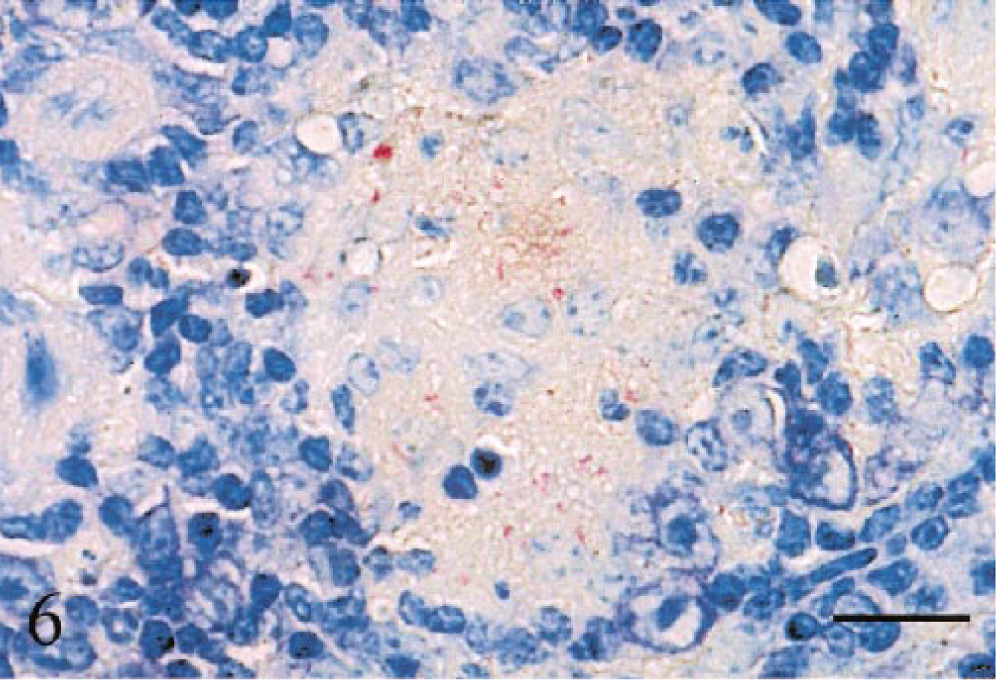
Small intestine; wild-type BALB/c mouse, 18 weeks after inoculation with the high dose of Mycobacterium paratuberculosis. Notice many acid-fast bacteria detected within the granuloma. Ziehl–Neelsen method. Bar = 20 μm.
In control mice groups, no lesions were detected in either the wild-type or the knockout mice at 6 and 18 weeks after inoculation.
Morphometric analysis of hepatic granuloma formation
The mean number and area of granulomata in the liver of the γδ T cell knockout BALB/c mice and the wild-type BALB/c mice inoculated with either the low or the high dose of M. paratuberculosis are shown in Table 1. The mean number of granulomata in the knockout mice tended to decrease at 18 weeks after inoculation, especially in the high-dose groups (P < 0.005). In contrast, in the wild-type mice, no significant differences were found in the mean number of granulomata between 6 and 18 weeks after inoculation with the low or the high dose. Significantly greater numbers of granulomata were observed in the wild-type than in the knockout mice at 18 weeks after high-dose inoculation (P < 0.05). No significant differences were found in the mean area of granulomata between the wild-type and knockout mice in both the low- and the high-dose groups on week 6 after inoculation. At 18 weeks after either low- or high-dose inoculation, significantly larger granulomatous areas were found in the wild-type than in the knockout mice (P < 0.05). Thus, the mean area of granulomata measured in the wild-type mice demonstrated a 3.3-fold increase over in the low-dose inoculation and a 6.4-fold increase over in the high-dose inoculation compared with the knockout mice at the same time point.
Mean number and area of granulomata in the liver of γδ T cell knockout BALB/c mice and wild-type BALB/c mice (n = 5 mice/group).

CFU = colony-forming units.
P < 0.05 compared with the knockout mice group at 18 weeks after the low-dose inoculation.
P < 0.005 compared with the knockout mice group at 6 weeks after the high-dose inoculation.
P < 0.05 compared with the knockout mice group at 18 weeks after the high-dose inoculation.
Mycobacterial growth in spleens
Table 2 shows the growth of M. paratuberculosis in the spleens of γδ T cell knockout BALB/c mice and wild-type BALB/c mice at 6 and 18 weeks after inoculation. Significantly higher growth of M. paratuberculosis was observed in both the wild-type mice and the knockout mice at 18 weeks after either the low- or high-dose inoculation compared with that on week 6. No significant differences were found in the bacterial load between the wild-type mice and the knockout mice in the low-dose groups at 6 and 18 weeks after inoculation. However, in the high-dose groups, the bacterial load in wild-type mice was significantly higher (P < 0.05) than that in knockout mice on week 18. No significant differences were evident at 6 weeks after inoculation.
Growth of Mycobacterium paratuberculosis in the spleens of γδ T cell knockout BALB/c mice and wild-type BALB/c mice (n = 5 mice/group).
CFU = colony-forming units.
P < 0.0001 compared with the knockout mice group at 6 weeks after the low-dose inoculation.
P < 0.001 compared with the wild-type mice group at 6 weeks after the low-dose inoculation.
P < 0.0001 compared with the knockout mice group at 6 weeks after the high-dose inoculation.
P < 0.0001 compared with the wild-type mice group at 6 weeks after the high-dose inoculation.
P < 0.05 compared with the knockout mice group at 18 weeks after the high-dose inoculation.
Discussion
To clarify the roles of γδ T cells in the pathogenesis of paratuberculosis, we performed a comparative study between wild-type BALB/c mice that are innately susceptible to M. paratuberculosis and γδ T cell knockout BALB/c mice as assessed by granuloma formation and bacterial load after M. paratuberculosis infection. In the livers of the wild-type mice inoculated with either a low dose (4 × 106 CFU/mouse) or a high dose (4 × 109 CFU/mouse) of the mycobacteria, an increased area of epithelioid granulomata was observed in a dose-dependent and time-dependent fashion. Furthermore, significantly greater numbers and a larger area of granuloma formation were found in livers of the wild-type mice than in the knockout mice at 18 weeks after inoculation, especially in the high-dose group. The development of mature, organized epithelioid granulomata is presumed to be important in restricting the spread of obligatory intracellular pathogens, 17 yet the formation of granulomata is unlikely to kill or restrict the intracellular growth of ingested M. paratuberculosis. We detected numerous acid-fast bacteria within the epithelioid granulomata as sites of mycobacterial growth as well as in some nongranulomatous areas. Furthermore, the bacterial load in spleens of the wild-type mice was significantly increased at 18 weeks after inoculation. Notably, in the knockout mice, prominent participation of lymphocytes in granulomata was observed at 6 weeks. However, the lesions were resolved at 18 weeks after inoculation with either the low or the high mycobacterial dose. Although the lesions in both the knockout mice and the wild-type mice are categorized to some variation of granuloma, obvious differences exist in morphologic features of the lesions between the two mice groups. In the wild-type mice, macrophages transformed into epithelioid cells or multinuclear giant cells were observed frequently, and lymphocytes and granulocytes participating in the lesions were not conspicuous. However, in the knockout mice, the transformed macrophages were rare, and lymphocytes and nontransformed macrophages were predominant in the lesions. Therefore, the lesions in the knockout mice could be considered as immature granulomata or aberrant inflammatory lesions. Consequently, accumulation of lymphocytes and newly infiltrated macrophages within granulomata of the knockout mice conceivably may contribute to resolution of the lesions. A similar reduction of granuloma formation also has been reported in γδ T cell–depleted mice infected with other obligatory intracellular pathogens such as Mycobacterium tuberculosis, 19 Mycobacterium avium, 33 and Listeria monocytogenes. 8 The acid-fast bacteria in the livers of the knockout mice were detected infrequently within and adjacent to the granulomatous lesions. Moreover, the bacterial load in the spleens of the knockout mice inoculated with the high dose was significantly lower than that in the wild-type mice, which may be due to the reduction of granulomata that provide an intracellular niche for the growth of M. paratuberculosis. An alternative possibility is that an impaired capacity of macrophages in γδ T cell knockout mice for tumor necrosis factor (TNF)-α production may relate to resistance against lethal infection, as has been reported in Salmonella choleraesuis infection. 12 Other studies in γδ T cell knockout mice infected with M. tuberculosis have suggested that this cell population contributes to granuloma architecture and containment of bacterial dissemination. 9 19 A more recent study in bovine severe combined immunodeficient xenochimeric mice infected with M. tuberculosis has demonstrated that bovine γδ T cells may play a role in recruitment of various cell types to sites of infection. 35
Hitherto, two distinct subsets of bovine γδ T cells have been reported. 20 One population expresses the WC1 antigen and is negative for CD2, CD4, and CD8. The other population expresses CD2 and CD8 but not WC1. Although the functions of the two subsets of bovine γδ T cells remain unknown, bovine γδ T cells as well as mice and human γδ T cells have the ability to produce Th1-type cytokines such as interferon (IFN)-γ, TNF-α, and IL-2. 38 IFN-γ and TNF-α are known to act synergistically in granuloma formation. 13 IL-2 induces proliferation of T cells, B cells, macrophages, and NK cells, 1 32 and enhances IFN-γ and TNF-α secretion from CD4+ T cells. 24 Although accumulation of macrophages at the site of M. paratuberculosis infection could occur even in severe combined immunodeficient beige (SCID bg) mice, 26 which lack functional T and B lymphocytes, and have a decreased NK cell activity, it also has been reported that γδ T cells may produce the macrophage chemokine, MCP-1, to regulate the inflammatory process. 8 Thus, the γδ T cells seem to have an auxiliary function for macrophages, NK cells, and αβ T cells relevant to development of mature granulomata.
The γδ T cells are present in higher numbers in ruminants compared to in humans and mice. 14 Prominence of γδ T cells in young calf gut–associated lymphoid tissue (GALT) 30 may contribute to high susceptibility for M. paratuberculosis infection, and may cause a predominance of granuloma formation in the tissue during the course of infection. Histamine, cytotoxin, and pyrogen cause diarrhea, emaciation, and anemia in bovine paratuberculosis. 22 In addition, the uncontrolled development of granulomata similar to those seen consistently in sarcoidosis 4 can contribute to the destruction of normal intestinal tissue and may partially explain the clinical signs in paratuberculosis.
In summary, γδ T cells may play an important role in the regulation of cell-mediated immunity and development of epithelioid granulomata in M. paratuberculosis infection rather than providing protection against mycobacterial infection. Furthermore, the functions of γδ T cells associated with granuloma formation may be involved in the pathogenesis and clinical manifestations in paratuberculosis.
Footnotes
Acknowledgements
We give special thanks to Dr. Elliott Kagan, Department of Pathology, Uniformed Services University of the Health Sciences, Bethesda, MD, USA, for useful suggestions and editorial assistance.
