Abstract
Gastric mucosal hypertrophy/nodular hyperplasia occurs as a consequence of Helicobacter infection in mice and humans. The pathogenesis of this hyperplastic response is not understood. To determine the role of host cellular immunity in gastric mucosal hypertrophy/hyperplasia, 6-8-week-old female euthymic BALB/c (n = 30) or NIH athymic nude (n = 40) mice were inoculated with H. heilmannii. Equal numbers of uninoculated mice of each strain served as controls. Mice from each group were euthanatized at 0.5, 6, 12, and 18 months postinoculation (PI). Lymphoplasmacytic gastritis and lymphoid follicle development were less severe in nude mice than in euthymic mice at < 6 months PI. The prevalence of gastritis at 0.5, 6, 12, and 18 months PI was 0%, 17%, 67%, and 88%, respectively, in infected nude mice and 33%, 83%, 71%, and 100%, respectively, in infected BALB/c mice. CD4+ T cells in infected nude mice were evident at ≥ 6 months PI but were less numerous than in infected BALB/c mice at comparable time intervals. However, numbers of CD4+ T cells increased substantially throughout the experiment in infected BALB/c mice. The prevalence of nodular mucosal hyperplasia at 0.5, 6, 12, and 18 months PI was 0%, 0%, 33%, and 20%, respectively, in infected nude mice and 0%, 33%, 80%, and 80%, respectively, in infected BALB/c mice. Nodular hyperplasia occurred in association with the appearance of chronic lymphoplasmacytic inflammation and CD4+ T cells at 12 and 18 months PI in nude mice. H. heilmannii-associated gastritis and mucosal remodeling is reduced in immunodeficient mouse strains lacking normal CD4+ T cell numbers as compared with the response in immunocompetent mice. Additionally, CD4 immunocompetence is an integral aspect of the mucosal hypertrophy/nodular hyperplasia in experimental H. heilmannii-associated disease of mice.
Helicobacter heilmannii are tightly coiled spiral bacteria approximately 7–10 µm in length. These bacteria reside in the lumina of gastric glands, in pits of the gastric antrum and fundus, and within parietal cell canaliculi. They can be isolated as parietal cell preparations, which are infectious for BALB/c and nude mice and cause gastritis, gastric epithelial proliferation, and gastric ulcers. 3–5, 8 32 These gastric lesions mimic many morphologic aspects of H. pylori-related gastric disease in humans. 3–5 Thus, H. heilmannii infection of mice has become an accepted model for H. pylori-associated gastric disease of humans. Differences between H. pylori and H. heilmannii include distribution and localization of bacterial colonization within the stomach. H. pylori are extracellular organisms that attach to epithelial cells of the glands and pits of the pyloric region. Unlike H. heilmannii, H. pylori does not infect the oxyntic mucosa and is not found in intracellular sites. 4 5 14 26 36 38
Persistent infection with certain Helicobacter species causes chronic active gastritis and gastric epithelial hyperplasia in both mice and humans. 1–7, 9 10 12–15, 22 23 26 H. heilmannii-associated gastritis in mice is characterized by inflammatory cell infiltrates ranging from predominantly lymphoplasmacytic to predominantly neutrophilic depending upon the strain of mouse. 5 12 In chronic stages of the disease, these inflammatory infiltrates are associated with mucosal hypertrophy characterized by nodular or diffuse mucosal thickening. 3–5 This mucosal hypertrophy is attributed to epithelial hyperplasia, stromal restructuring, and inflammatory cell accumulation. Gastric mucosal hypertrophy can be exacerbated by exogenous nitroso compounds, and this mucosal proliferation or hypertrophy may be a contributory factor in Helicobacter-associated carcinogenesis. 9 31 Chronic gastric Helicobacter infection in humans has been associated with epithelial proliferation, but mucosal hypertrophy is rarely evident. 1 2 11 13 23 26 The pathogenesis of Helicobacter associated gastric nodular mucosal hypertrophy is not known, but reports in the literature indicate that host rather than bacterial factors may play a role. 4 26 In mice, gastric nodular hyperplasia has been induced experimentally by several Helicobacter species, including H. heilmannii, and occurs only with development of severe gastritis. 3–5, 12 22 27 In other host species, H. heilmannii causes little or no disease. 28
Immunodeficient nude mice were used in this study to compare the development of nodular mucosal hyperplasia with that in an immunocompetent strain (BALB/c). The whn gene is mutated in nude mice and rats, resulting in disrupted thymus development and hair growth. The gene encodes a winged-helix transcription factor with a DNA-binding region, 25 and its lack prevents thymic T-cell development and results in immunodeficiency. Nude mice are generally T-cell deficient early in life but develop T cells by extrathymic means with age and prolonged pathogen exposure. 17–19, 21 29 30 34
The goal of this study was to investigate the relationship of T-cell immunocompetence to gastric epithelial proliferation by comparing disease development in immunocompetent (BALB/c) and immunodeficient (nude) mouse strains inoculated with H. heilmannii and examined at intervals up to 18 months after inoculation.
Materials and Methods
Animals and microbiology
H. heilmannii-infected parietal cells from 10 infected athymic mice were isolated and pooled. H. heilmannii infection in pooled samples was verified by light microscopy. Bacteria isolated in association with parietal cells are infectious for mice and cause gastritis. 8 Mice were inoculated with 1 × 107 infected parietal cells in 0.2 ml of sterile Brucella broth. Control mice were not inoculated because previous studies have indicated that BALB/c mice inoculated with sterile Brucella broth, gastric homogenates from uninfected mice, or filter-sterilized (0.22 µm) gastric homogenate from infected mice do not develop gastritis, mucosal ulceration, or mucosal hypertrophy up to 12 months postinoculation (PI). 5 Other Helicobacter species that infect the stomach or large intestine were not detected by light microscopy of Warthin–Starry (WS)-stained intestine and gastric sections or bacterial culture of mucosal sections from mice used for inoculum or experimental mice.
Sixty 6–8-week-old female BALB/c (euthymic) and 80 6–8-week-old female NIH nude (athymic) mice (Harlan Sprague-Dawley, Indianapolis, IN) were randomly assigned to two groups for each respective strain. Mice were housed in filtertop cages in HEPA-filtered barrier units, fed sterile rodent chow (Harlan-TekLad rodent diet, Harlan-TekLad, Inc., Madison, WI), and given sterile water adlibitum. Thirty BALB/c and 40 nude mice were inoculated by orogastric intubation, and 30 BALB/c and 40 nude mice served as controls. Representative mice from each group were euthanatized at established intervals of 0.5, 6, 12, and 18 months PI (Table 1). Early deaths accounted in part for inconsistencies in group number in the final evaluations. Additionally, fewer BALB/c mice were assigned to the 0.5-month interval because fewer lesions were expected. At scheduled intervals, all mice were given 5 µg of 5-bromo 2′-deoxyuridine (BrdU, Sigma Chemical Co., St. Louis, MO) by intraperitoneal injection and euthanatized 2 hours later by intraperitoneal injection of 0.05 ml sodium pentobarbital. The stomach of each mouse was removed, opened along the greater curvature, and examined with a dissecting microscope for the presence of gross lesions. The stomach was then cut into 1–2-mm-wide longitudinal sections that included all anatomic regions (i.e., forestomach, fundus, and pylorus). Two representative sections of each stomach and a section of spleen were immersion-fixed in formalin, and representative samples of the same tissues were embedded in OCT compound (Sakura Finetek, Torrance, CA), snap frozen, and stored at −70 C. All procedures involving animals were approved by the Ohio State University Institutional Laboratory Animal Care and Use Committee.
Number of mice euthanatized at scheduled intervals.
Inoculated with Helicobacter heilmannii.
Histology and immunohistochemistry
Formalin-fixed, paraffin-embedded tissues were sectioned at 5 µm and stained with hematoxylin and eosin (HE) and WS stain. Additional sections were immunohistochemically stained with antibodies directed against IgA+, IgG+, IgM+, and BrdU. Frozen tissue was sectioned at 5 µm and stained immunohistochemically to detect CD4 (T-helper lymphocyte), CD8 (cytotoxic T lymphocyte), Mac-1 (granulocyte, monocyte, and macrophage), and B220 (B lymphocyte) antigens.
Frozen sections were thawed, fixed in cold acetone for 10 minutes, and treated with dithiothreitol (160 µg/ml) for 5 minutes to remove gastric mucus. Paraffin-embedded sections were deparaffinized and treated with Protease VIII (0.5 mg/ml; Sigma) at 37 C for 5 minutes for antigen retrieval. After blocking with normal goat serum, sections were incubated with primary antibodies, including monoclonal rat anti-mouse CD4, CD8α (Gibco-BRL, Grand Island, NY), Mac-1, B220 (Caltag, South San Francisco, CA), and polyclonal goat anti-mouse IgA, IgG, and IgM (Zymed, South San Francisco, CA), overnight at 4 C. Slides were washed, treated with secondary antibody (biotinylated anti-rat, Vector Laboratories, Burlingame, CA) for 30 minutes at room temperature, washed again, and treated with an avidin-biotin peroxidase complex method (ABC) (Vector Laboratories). Endogenous peroxidase was quenched by incubation for 30 minutes in 10% H2O2. Diaminobenzidine was utilized as the chromogen, and sections were counterstained with hematoxylin. For negative controls, primary antibody was replaced with 1% bovine serum albumin in phosphate-buffered saline (PBS), pH 7.2. All slides also contained splenic tissue from the same animal to serve as an internal positive control for CD4, CD8, Mac-1, B220, and the immunoglobulin/immunohistochemical staining procedures. BrdU staining was performed as previously described. 5
All sections were examined in a blinded fashion without knowledge of treatment group or euthanasia interval. One slide per animal was evaluated. The gastric glandular mucosa in HE-stained sections was evaluated for epithelial nodular hyperplasia, percentage of fields with mononuclear inflammatory infiltrates, neutrophilic inflammatory infiltrates, mucosal metaplasia, and mucosal lymphoid follicles. The extent of the histologic changes was assessed by determining the percentage of microscopic fields with positive criteria for lesions. 7 All microscopic fields (200× magnification) of each stomach section were evaluated. Criteria for a positive response in a field were 1) mononuclear cell infiltrate sufficient to deform gastric glands (gastritis), 2) mucosal neutrophilic infiltration, 3) replacement of fundic epithelium by mucus cells (metaplasia), and 4) mucosal lymphoid follicles.
Stomach sections were considered to have mononuclear gastritis if ≥10% of evaluated fields contained inflammation severe enough to deform glands (i.e., minimal = 10–20% of fields affected; mild = 21–30% of fields affected; moderate = 31–40% of fields affected; marked = >40% of fields affected). The scoring system was based upon observations that severity and distribution of inflammation, “gastritis,” are directly correlated in Helicobacter-infected mice and add an objective measurement of distribution to a subjective scoring scheme. 7
WS-stained sections of stomach of each animal were evaluated for colonization with H. heilmannii and scored as follows: 0 = no bacteria; 1 = mild colonization (rare bacteria); 2 = moderate colonization (easily identified bacteria, widely scattered); 3 = marked colonization (glandular lumina filled with bacteria).
BrdU labeling index (LI) was determined for focal areas with and without nodular hyperplasia. In each section, three nodular and/or nonnodular sites were chosen, and 100 epithelial cell nuclei were evaluated at each site. LI was determined by dividing the number of labeled nuclei by the total number of nuclei counted. The reported number is the group mean LI. The mean LI of the chosen sites for each animal was used for calculation of the group mean LI. The overall LI was the average of all individual site LIs (nodular and/or nonnodular) for each group.
CD4+, CD8+, Mac-1+, IgA+, IgG+, and IgM+ cells were enumerated in each of three nonoverlapping 40× mucosal fields and three adjacent submucosal fields chosen at random. The reported number is a group mean of the three locations of each animal. Because of the intense focal distribution of B220+ cells within mucosal lymphoid follicles, these cells were not counted.
Enzyme-linked immunosorbent assay
Serum collected at the time of euthanasia was utilized to evaluate circulating levels of anti-Helicobacter IgG using an enzyme-linked immunosorbent assay (ELISA). The ELISA protocol was performed according to a previously described method. 24 The 96-well plates were coated with 100 µl of H. felis sonicate (100 µg/ml protein in 0.1 M PBS) after blocking. Samples and positive and negative control sera (100 µl/well, 1:50 dilution) were added to wells in triplicate and incubated at 37 C for 90 minutes, washed, treated with conjugated secondary antibody 100 µl/well, 1:2,000 dilution goat anti-mouse IgG, alkaline phosphatase conjugated; Biorad, Hercules, CA at 37 C for 90 minutes, washed again, and treated with alkaline phosphatase substrate (100 µl/well; Biorad) in the dark for 30 minutes. The reaction was stopped with 0.4 M NaOH (100 µl/well). Optical density at 405 nm (OD405) was determined using a Coulter spectrophotometer/plate reader. IgG levels were considered negative at OD405 < 0.2 (i.e., twice the highest negative control OD405 value). IgG levels were considered positive at OD405 > 0.5.
H. felis sonicate was utilized as the antigen in the ELISAs based upon the inability to culture the H. heilmannii organisms and data that support reliable cross-reactivity between H. felis and H. heilmannii antigens. 6
Statistics
Values are reported as group mean ± SEM. Group means of immunohistochemical cell counts and LIs, percentage of affected mucosa (HE), and OD405 values from ELISAs were compared with the Mann–Whitney two-tailed U-test. The level for significance was set at P < 0.05. Comparisons were performed between mouse strains and between infected and control mice for all parameters at all time points. Pearson correlation coefficients (r-values, P < 0.05) were determined for the correlation of gastritis scores and BrdU LI (nodular and overall) and of CD4+ counts and BrdU LI (nodular and overall).
Results
Pathologic findings
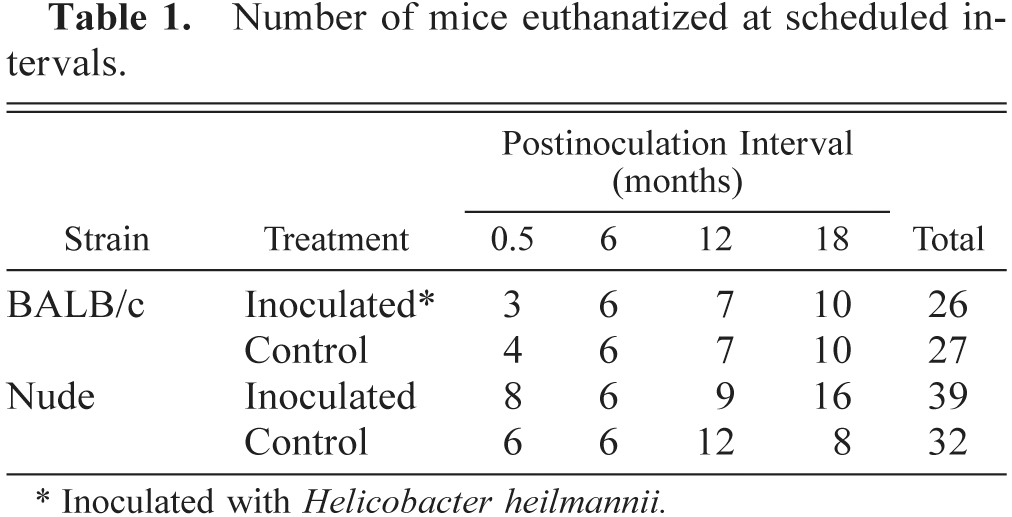
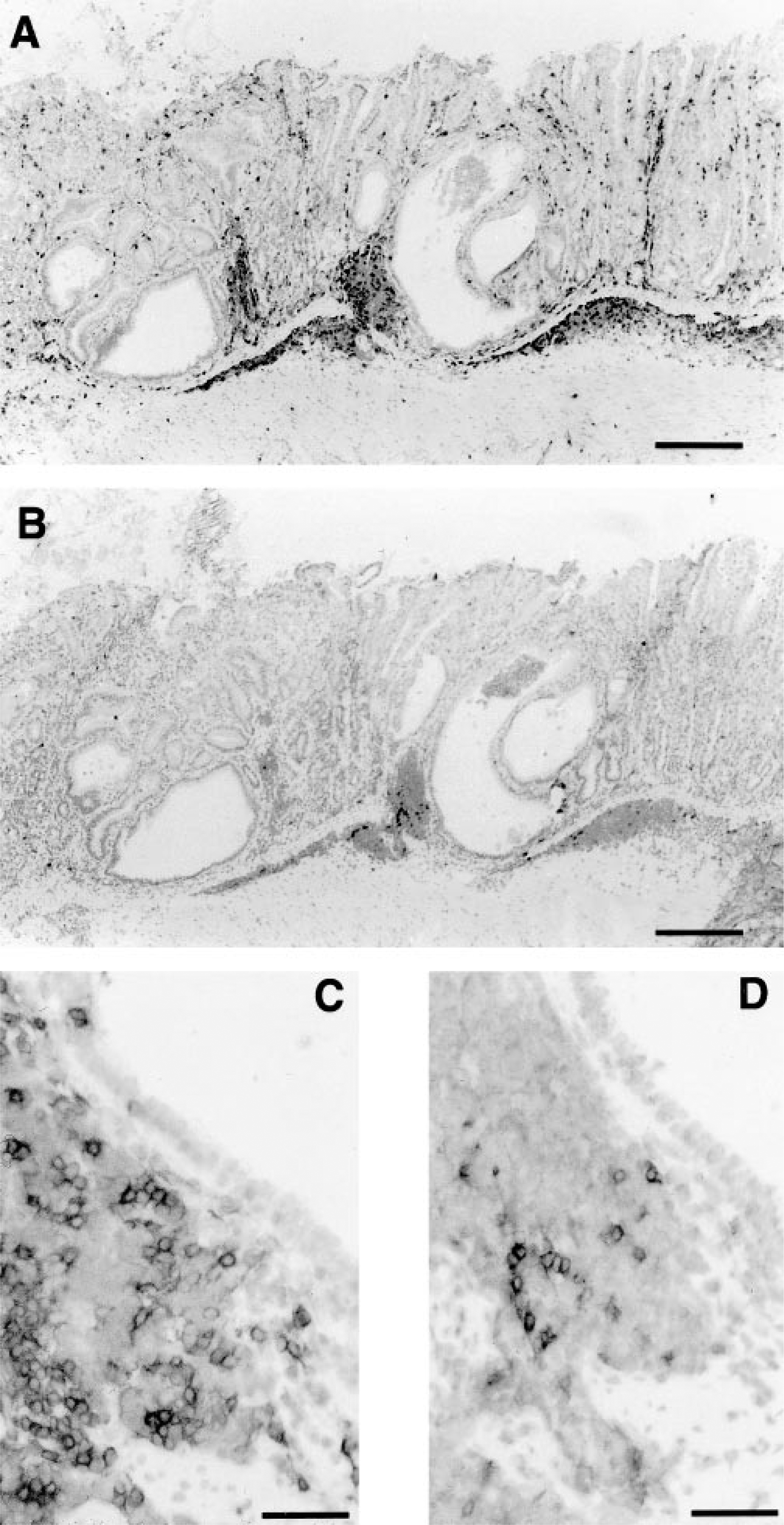
Gross multifocal nodular gastric hyperplasia was present in 80% of infected BALB/c mice and 20% of infected nude mice by 18 months PI. Nodular hyperplasia was not present in controls. Nodules were 1–2 mm in diameter, raised, and distributed mostly along the greater curvature of the stomach. Histologically, these lesions were characterized by gastric mucosal hypertrophy, metaplasia of gastric gland epithelium, mucosal lymphoid follicles, and lymphoplasmacytic gastritis (Figs. 1, 2).
Stomach; BALB/c mouse, 18 months after inoculation with H. heilmannii. Lymphoplasmacytic gastritis involves the lamina propria and submucosa and mucosal lymphoid follicles (asterisks). Note the nodular appearance (nodular mucosal hyperplasia) of the gastric surface. Nude mice 18 months postinoculation had similar histologic lesions. The white box illustrates an area of mucosal metaplasia in the deep aspect of the fundic glands. HE. Bar = 256 µm.
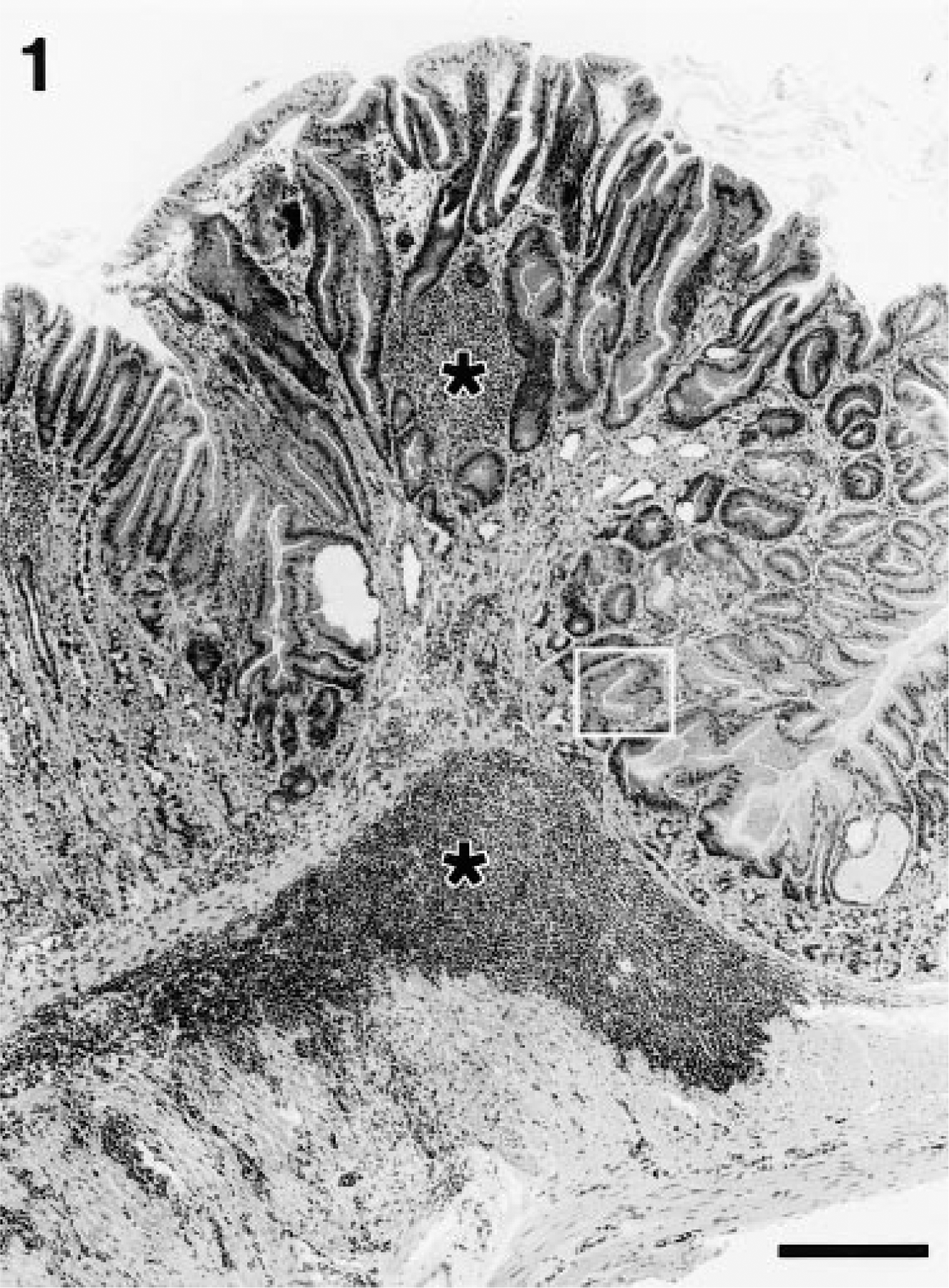
Stomach; BALB/c mouse, 18 months after inoculation with H. heilmannii. Higher magnification of section in Fig. 1. Mucosal metaplasia is characterized by replacement of normal fundic glandular epithelium with mucous-type epithelium. Note the large apical mucin vacuoles of the epithelial cells (black arrow). HE. Bar = 35 µm.
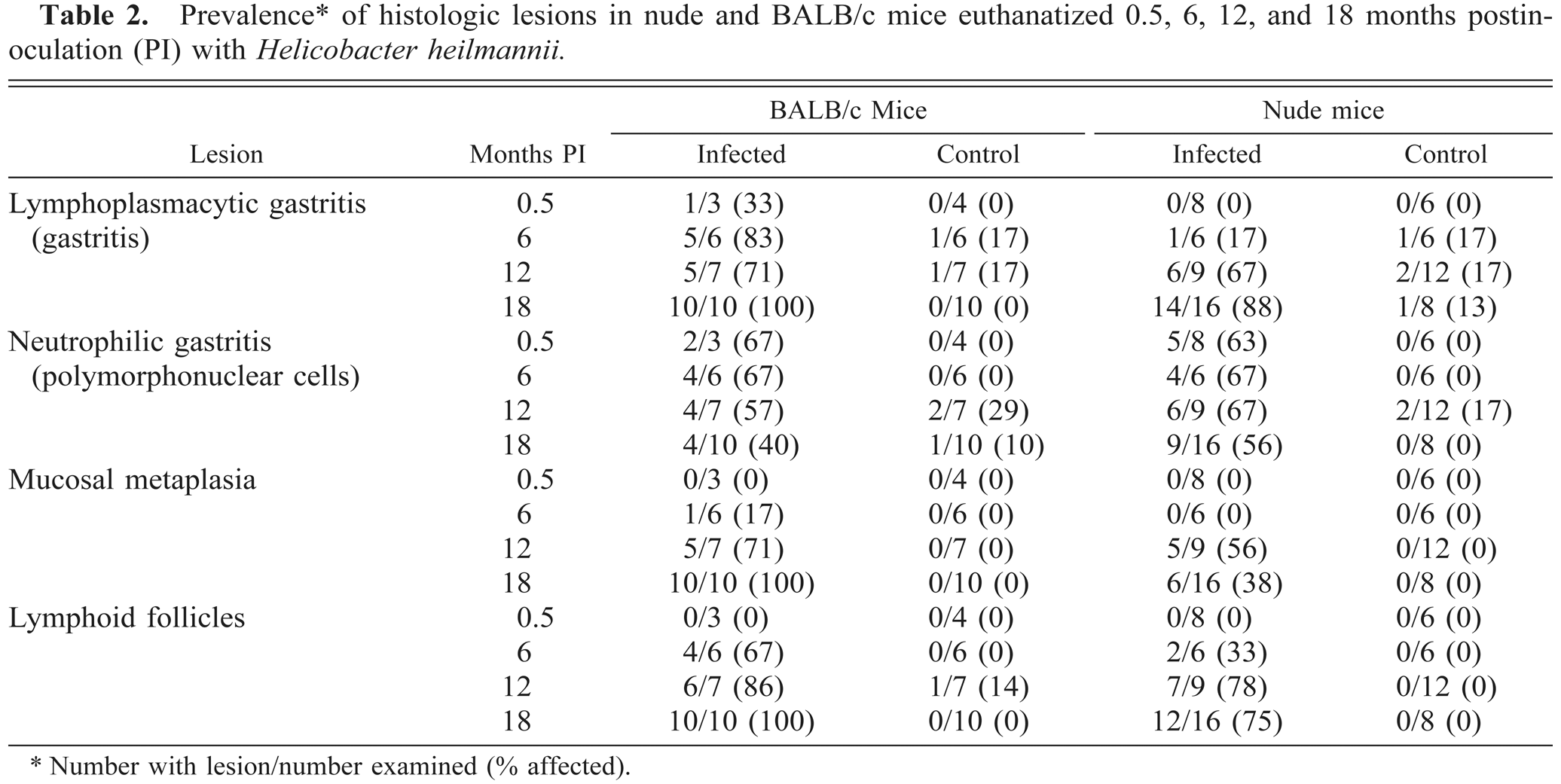
The prevalence and extent of lymphoplasmacytic gastritis was greater in infected BALB/c mice than in infected nude mice at all time points (Fig. 3, Table 2). Lymphoplasmacytic gastritis increased over time in both strains. In infected mice, gastritis was multifocal, characterized by infiltrations of lymphocytes and plasma cells in the superficial gastric mucosa. Lymphoplasmacytic gastritis was present in <20% of microscopic fields in uninfected mice of either strain and in infected nude mice euthanatized at 2 weeks or 6 months PI (Figs. 3, 4, Table 2).
Prevalence∗ of histologic lesions in nude and BALB/c mice euthanatized 0.5, 6, 12, and 18 months postinoculation (PI) with Helicobacter heilmannii.
Number with lesion/number examined (% affected).
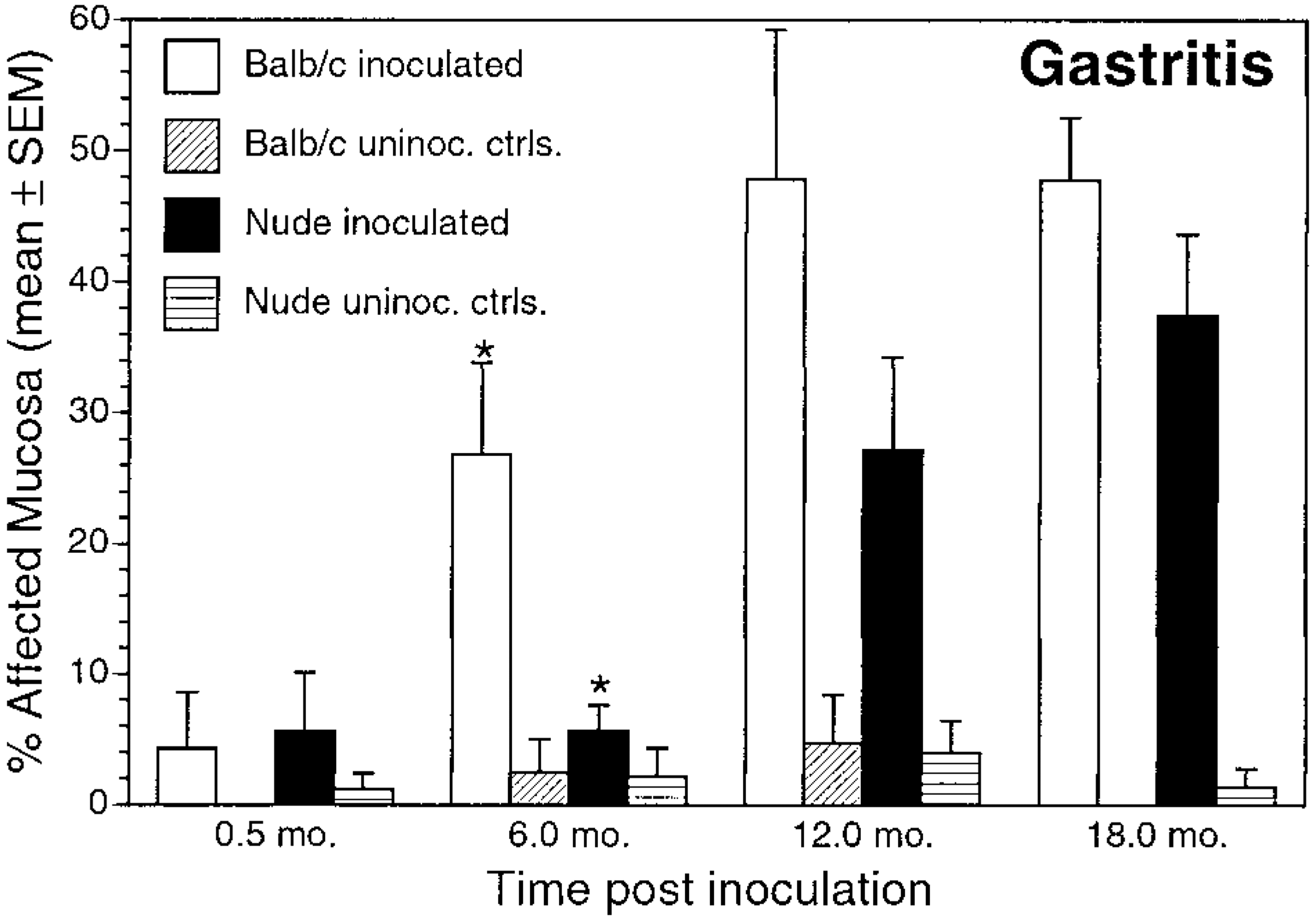
Gastritis. Mean ± SEM percentage of affected mucosa in inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
Stomach; BALB/c control mouse, 18 months after beginning of study. Note the absence of gastritis and mucosal hypertrophy/nodular hyperplasia. Uninoculated BALB/c and nude mice at all time points were similar histologically. HE. Bar = 45 µm.
The extent and character of gastritis differed between BALB/c and nude mice. In BALB/c mice, lymphoplasmacytic gastritis increased in extent until 12 months PI (Fig. 3, Table 2). In infected nude mice, a significantly increased prevalence and extent of lymphoplasmacytic gastritis over controls did not develop until 12 months PI. The pattern of neutrophilic inflammation differed between the mouse strains. In BALB/c mice, neutrophilic inflammation was present 0.5 months after inoculation but diminished over the course of the experiment (Fig. 5, Table 2). In contrast, neutrophilic inflammation in nude mice remained relatively unchanged over the course of the experiment and was more extensive than that in BALB/c mice at 12 and 18 months PI.
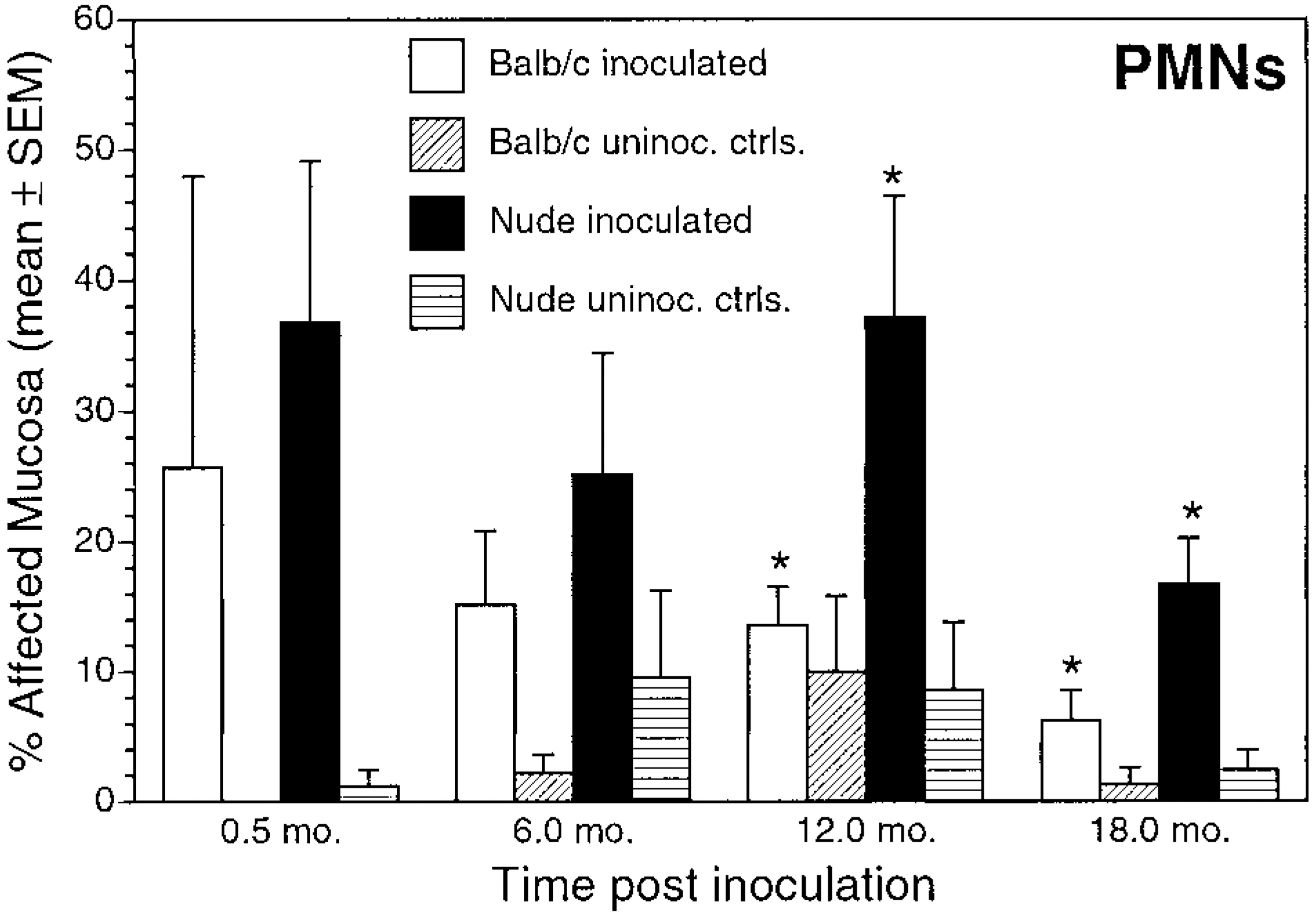
Polymorphonuclear leukocytes/neutrophils (PMNs). Mean ± SEM percentage of affected mucosa in inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
In addition to differences in gastritis, the timetable of epithelial lesion development differed between nude and BALB/c mice. Gastric epithelial metaplasia, defined as replacement of gastric gland epithelium with mucus cells, was present at 6, 12, and 18 months PI in inoculated BALB/c mice, but in nude mice metaplasia did not develop until 12 months PI. Mucosal metaplasia was significantly greater in BALB/c mice than in nude mice at 6 and 18 months PI (Fig. 6, Table 2). Mucosal metaplasia was not present in control mice. Mucosal lymphoid follicles, defined as aggregates of lymphocytes with occasional plasma cells and macrophages within the lamina propria, were present at 6, 12, and 18 months PI in inoculated BALB/c and nude mice. Mucosal lymphoid follicles were not present in control mice (Fig. 7).
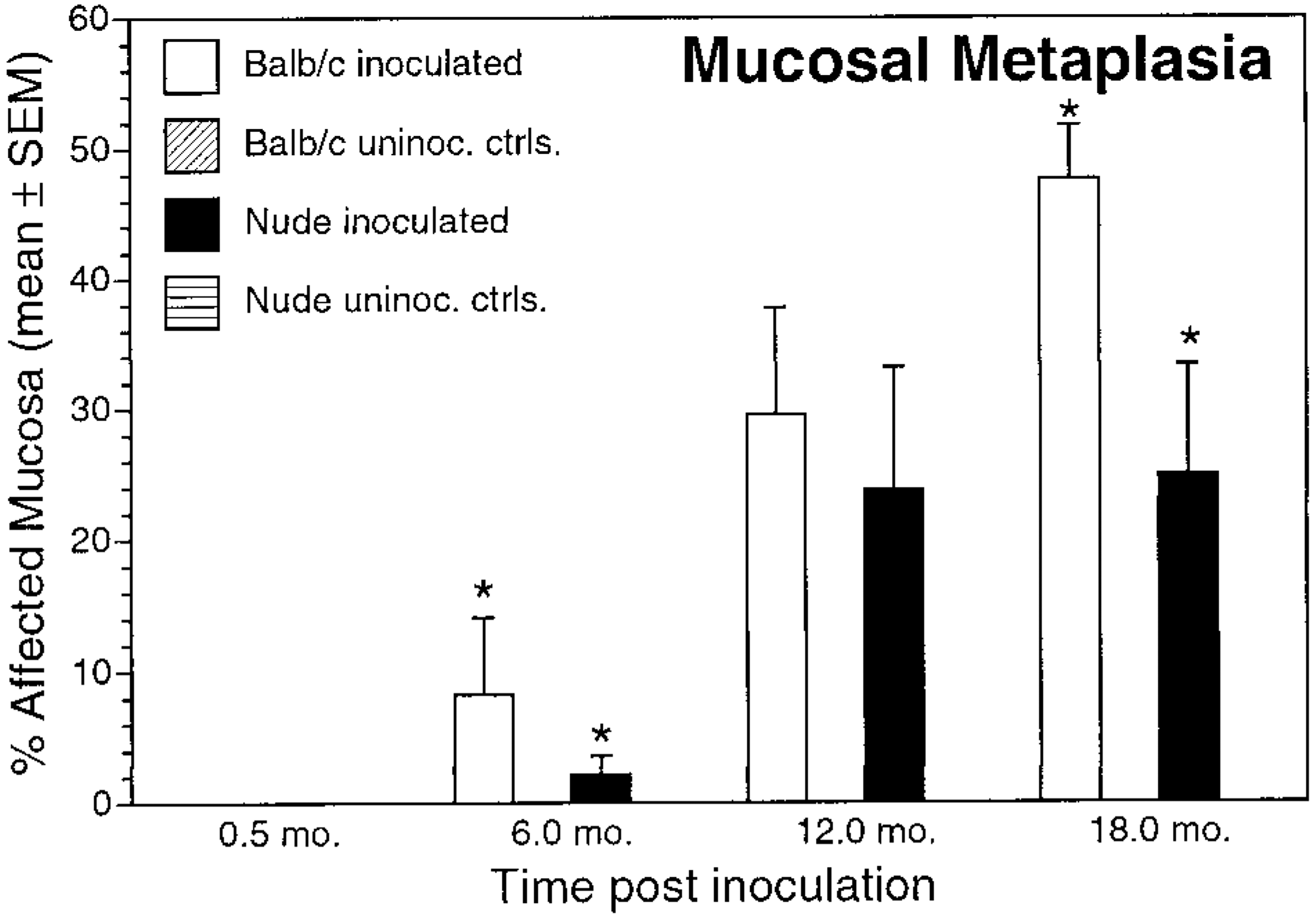
Mucosal metaplasia. Mean ± SEM percentage of affected mucosa in inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
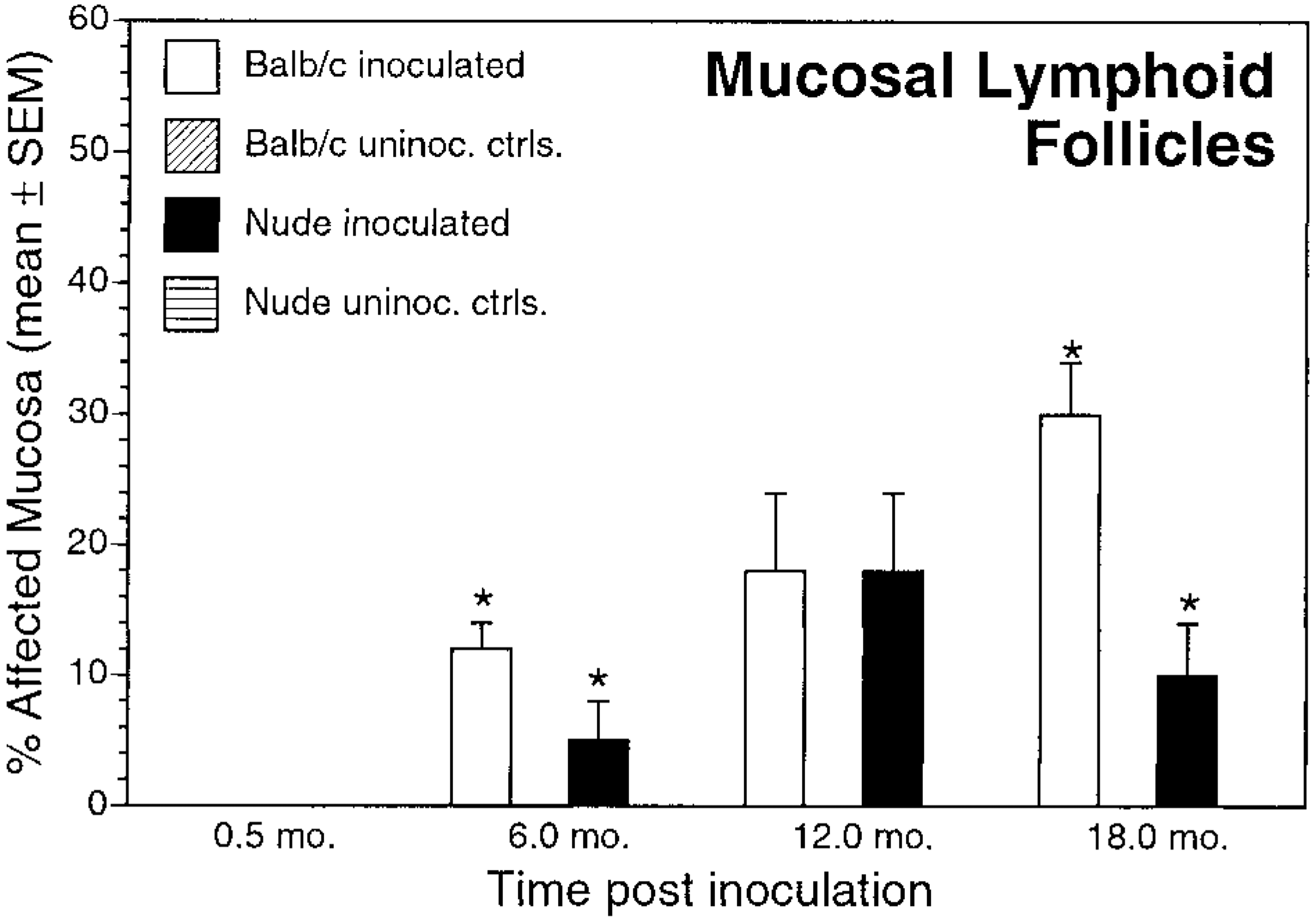
Mucosal lymphoid follicles. Mean ± SEM percentage of affected mucosa in inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
H. heilmannii was not identified microscopically in WS-stained stomach sections from control BALB/c and nude mice. Bacteria were, however, identified in all inoculated mice (Table 3). Mean bacterial colonization scores decreased significantly during the study for inoculated BALB/c mice but remained relatively stable for inoculated nude mice (Table 3).
Helibacter heilmannii colonization of stomach of nude and BALB/c mice 0.5, 6, 12, and 18 months postinoculation (PI).
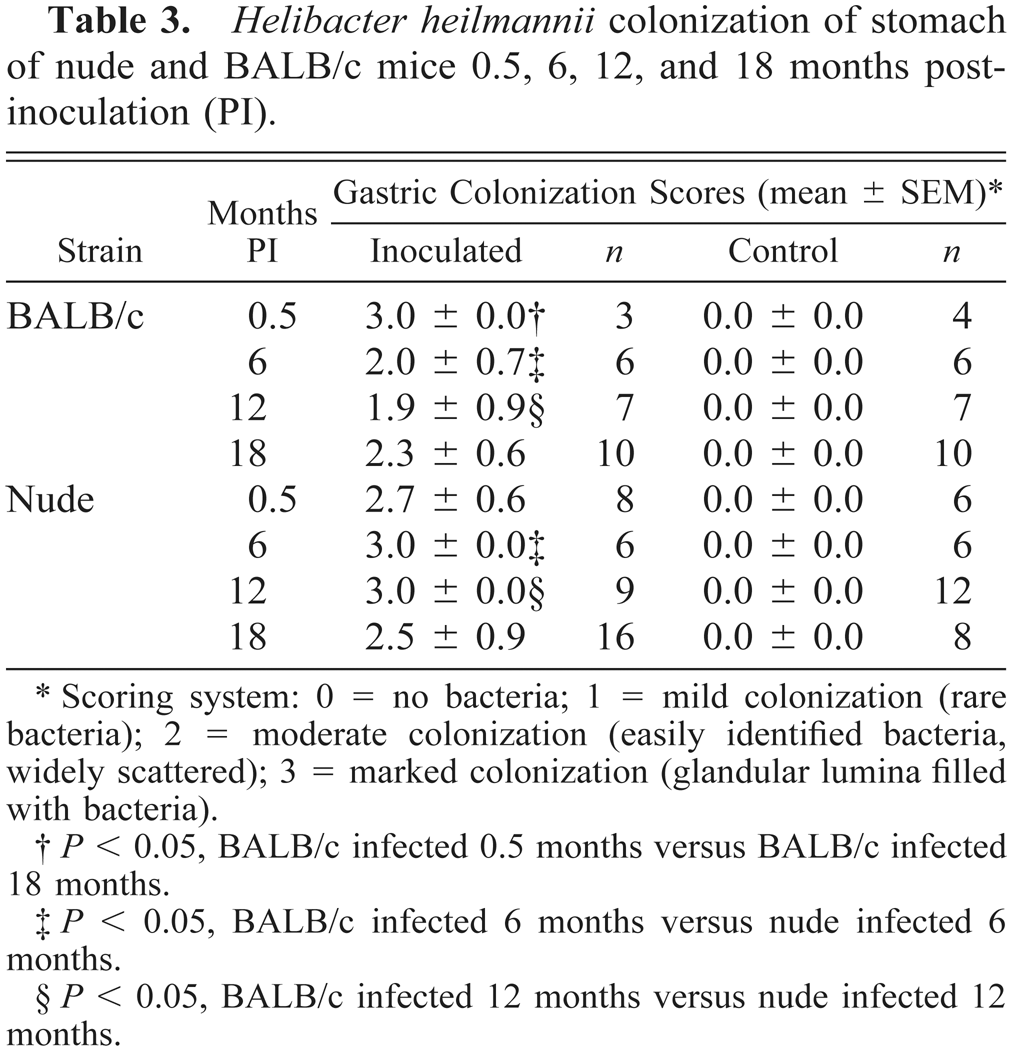
Scoring system: 0 = no bacteria; 1 = mild colonization (rare bacteria); 2 = moderate colonization (easily identified bacteria, widely scattered); 3 = marked colonization (glandular lumina filled with bacteria).
P < 0.05, BALB/c infected 0.5 months versus BALB/c infected 18 months.
P < 0.05, BALB/c infected 6 months versus nude infected 6 months.
P < 0.05, BALB/c infected 12 months versus nude infected 12 months.
Gastric nodular mucosal hyperplasia, characterized by epithelial hyperplasia, increased lamina proprial and submucosal lymphoplasmacytic inflammation as compared with nonnodular areas, and stromal remodeling was present in infected BALB/c and nude mice. Hyperplastic nodules also contained areas of gastric epithelial metaplasia and lymphoid follicles that were frequently more extensive than those in nonnodular areas. The prevalence of gastric mucosal nodular hyperplasia for infected and control mice of both strains is summarized in Table 4. Gastric mucosal nodular hyperplasia in infected BALB/c mice was first seen at 6 months PI and increased over time to include 80% of mice at 18 months PI. For infected nude mice, nodular mucosal hyperplasia was noted at 12 months PI and was less prevalent than that in BALB/c mice at all assessment intervals. Nodular mucosal hyperplasia was not present in control mice (Table 4). BrdU LI in proliferative nodules (26.5 ± 9.4) was significantly greater than LI in nonnodular epithelium (3.4 ± 3.8), whether or not inflammation was present (P < 0.05). There was little difference in LI for nonnodular mucosa of infected versus control mice. There was a positive correlation between lymphoplasmacytic gastritis scores and LI in nodular areas in inoculated BALB/c and nude mice (r = 0.659 and 0.981, respectively; P < 0.05) and between mononuclear gastritis scores and overall LI in inoculated BALB/c and nude mice (r = 0.862 and 0.841, respectively; P < 0.05).
Gastric epithelial proliferation (BrdU labeling indices [LI] and prevalence of nodular hyperplasia) in nude and BALB/c mice at 0.5, 6, 12 and 18 months postinoculation (PI) with Helicobacter heilmannii.
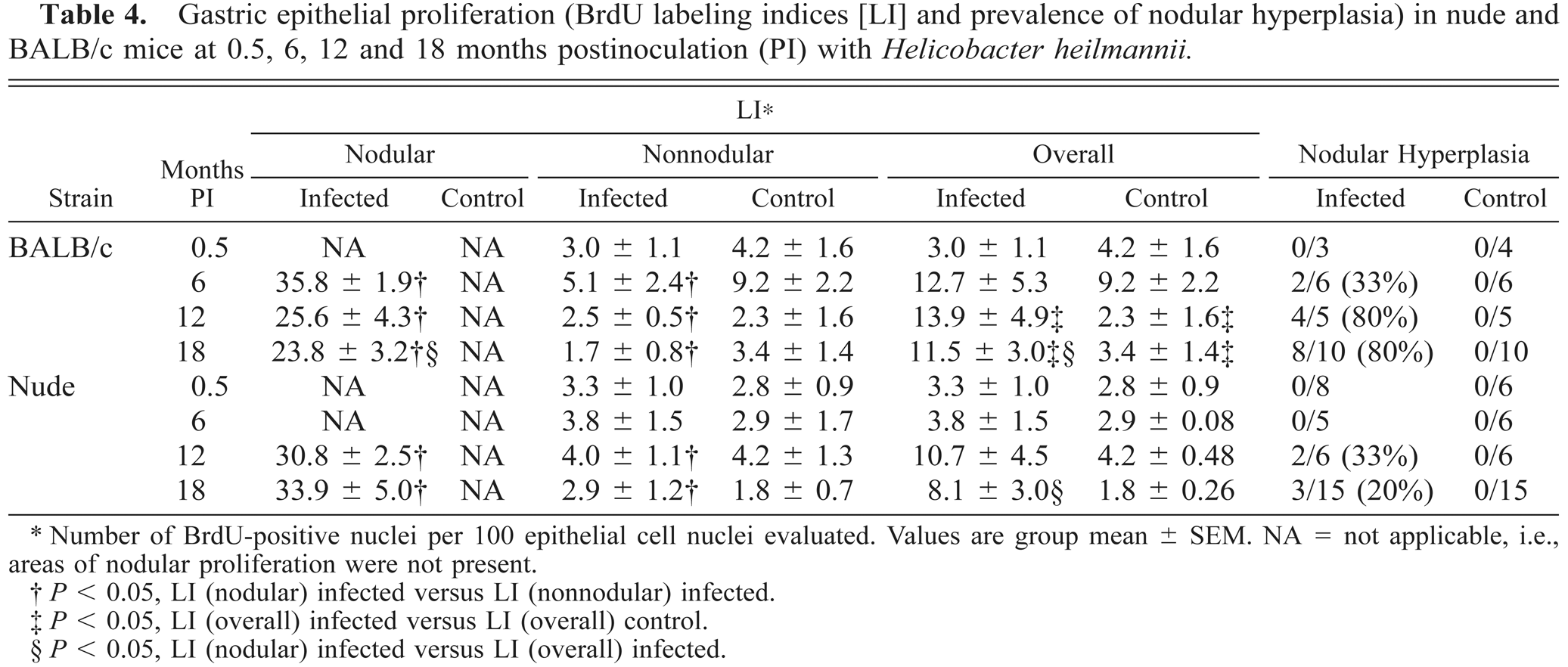
Number of BrdU-positive nuclei per 100 epithelial cell nuclei evaluated. Values are group mean ± SEM. NA = not applicable, i.e., areas of nodular proliferation were not present.
P < 0.05, LI (nodular) infected versus LI (nonnodular) infected.
P < 0.05, LI (overall) infected versus LI (overall) control.
P < 0.05, LI (nodular) infected versus LI (overall) infected.
Immunohistochemistry
At 0.5 months, CD4+ cells were present in BALB/c mice but were not evident in nude mice. In infected BALB/c mice, numbers of CD4+ T-cells increased substantially throughout the experiment (Fig. 8). CD4+ T cells in infected nude mice were evident at ≥6 months PI but were less numerous than in infected BALB/c mice at comparable time intervals (Figs. 8, 9A, C). There was a positive correlation between CD4+ cell counts and LI in nodular areas in inoculated BALB/c and nude mice (r = 0.711 and 0.514, respectively; P < 0.05) and between CD4+ cell counts and the overall LI in inoculated BALB/c mice (r = 0.856; P < 0.05).
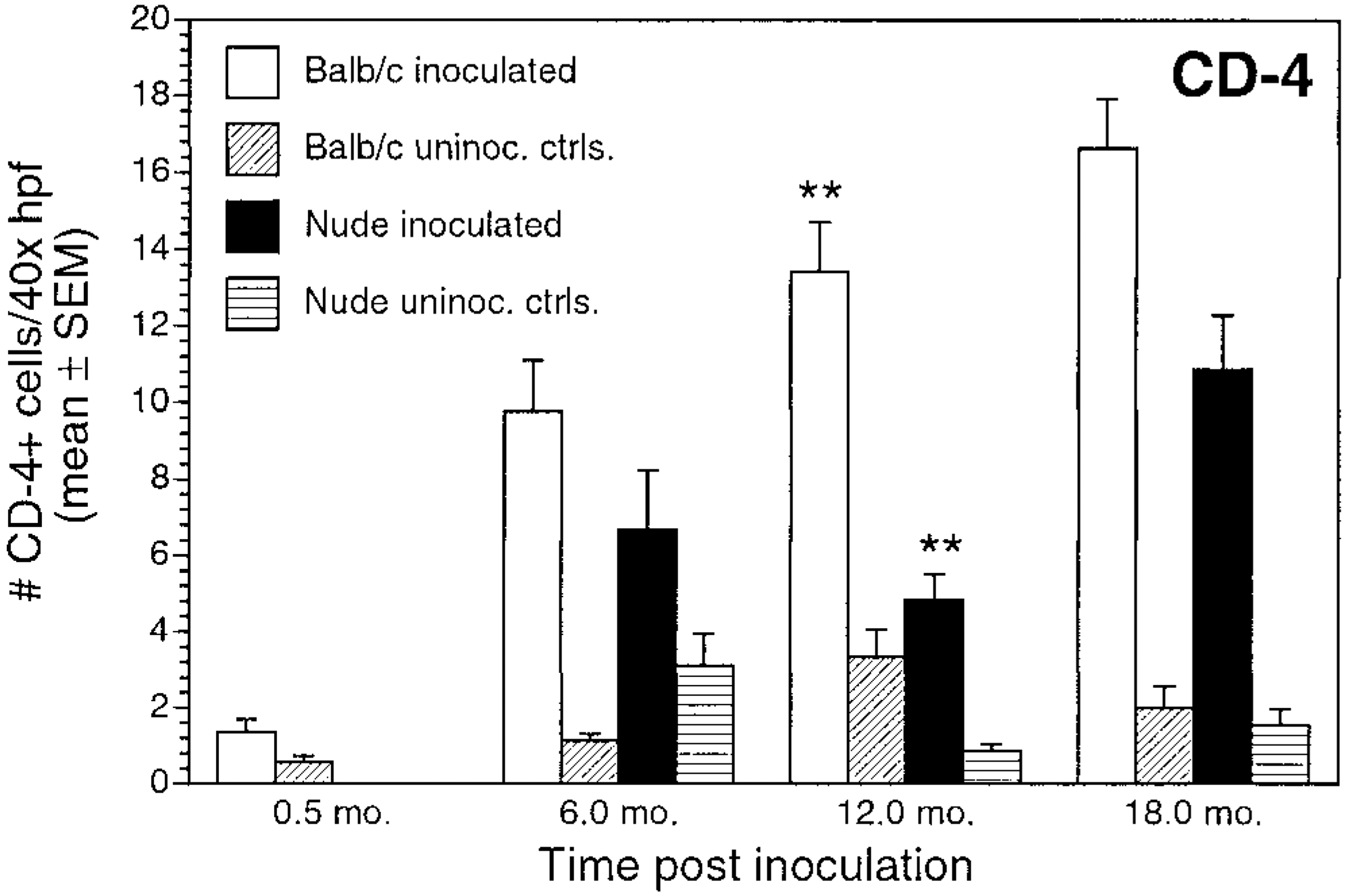
Number of CD4+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
Stomach; nude mouse, 18 months after inoculation with H.
heilmannii.
Numbers of CD8+ T cells and Mac-1+ cells did not differ between nude and BALB/c mice (Figs. 10, 11). CD8+ cells increased gradually during the study for both inoculated BALB/c and nude mice. There were substantially fewer CD8+ cells than CD4+ cells in both mouse strains, but numbers did not differ between infected BALB/c and nude mice (P > 0.05) (Figs. 9B, D, 10).
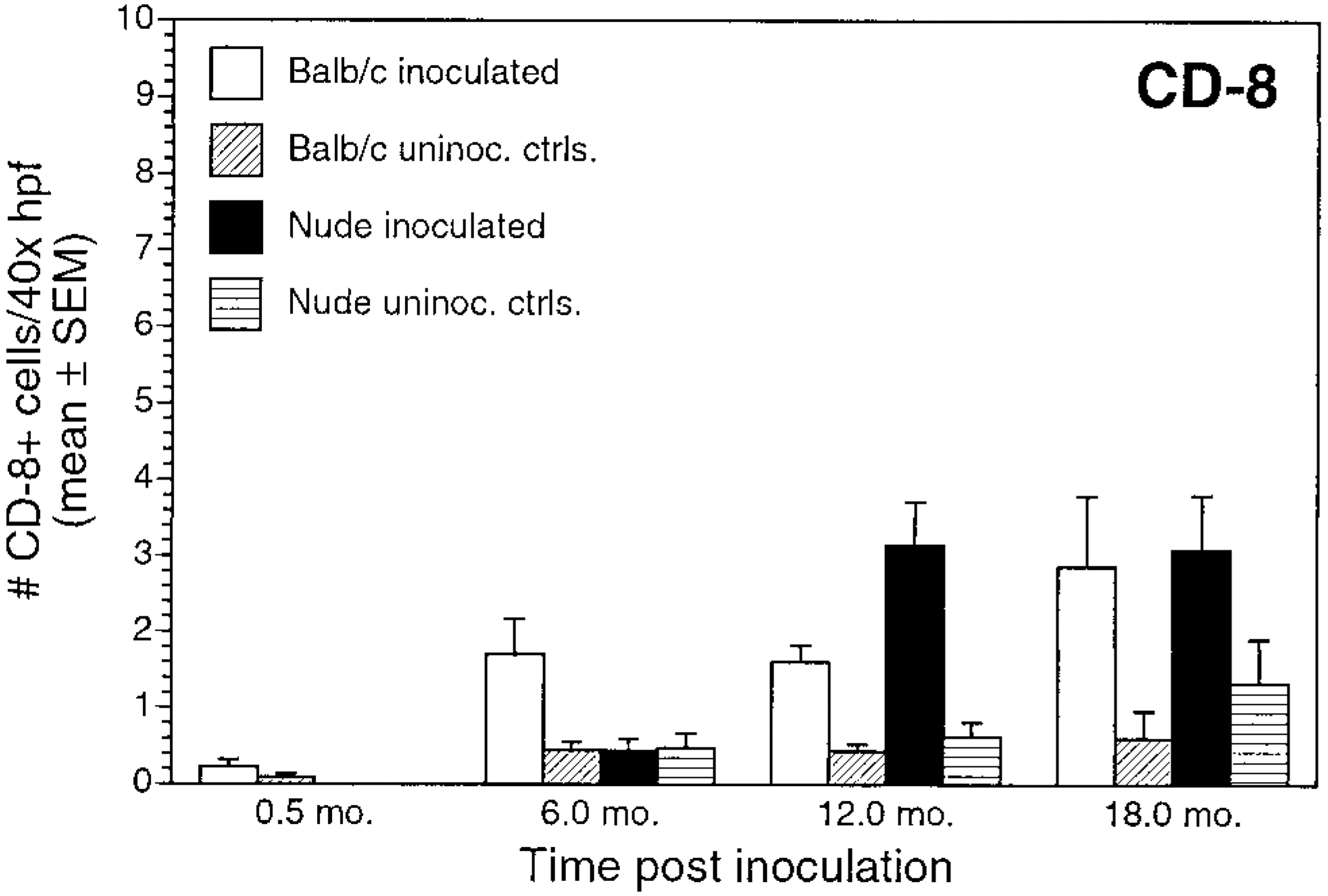
Number of CD8+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation.
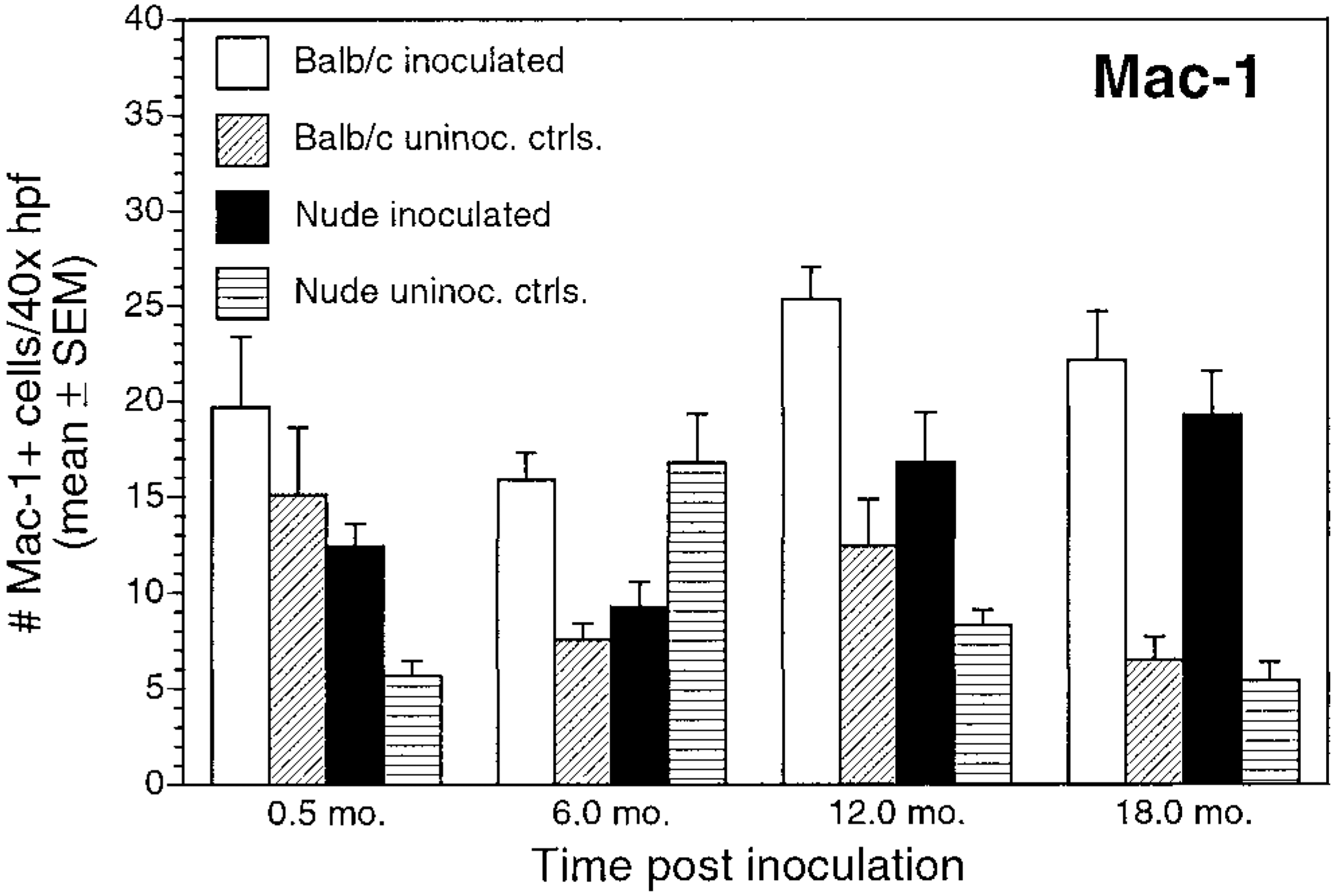
Number of Mac-1+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation.
Numbers of immunoglobulin-positive cells also differed between inoculated BALB/c and nude mice. Numbers of IgA+ cells and IgG+ cells in inoculated BALB/c mice gradually increased through 12 months PI (Figs. 12, 13). For inoculated nude mice, IgA+ cell numbers increased over time, but mean cell counts were significantly lower than those in infected BALB/c mice at all time points. Infected nude mice also had increases in number of IgG+ cells over time, but the numbers of IgG+ cells were lower than those in infected BALB/c mice and were not clearly different from controls. Mean IgG+ cell counts were significantly greater in BALB/c infected mice than in infected nude mice at 6 and 12 months PI (P < 0.05). There was a gradual increase in IgM+ cells in inoculated BALB/c and nude mice during the study (Fig. 14). IgM+ cell numbers at 12 months PI were significantly greater in BALB/c mice than in nude mice (P < 0.05).
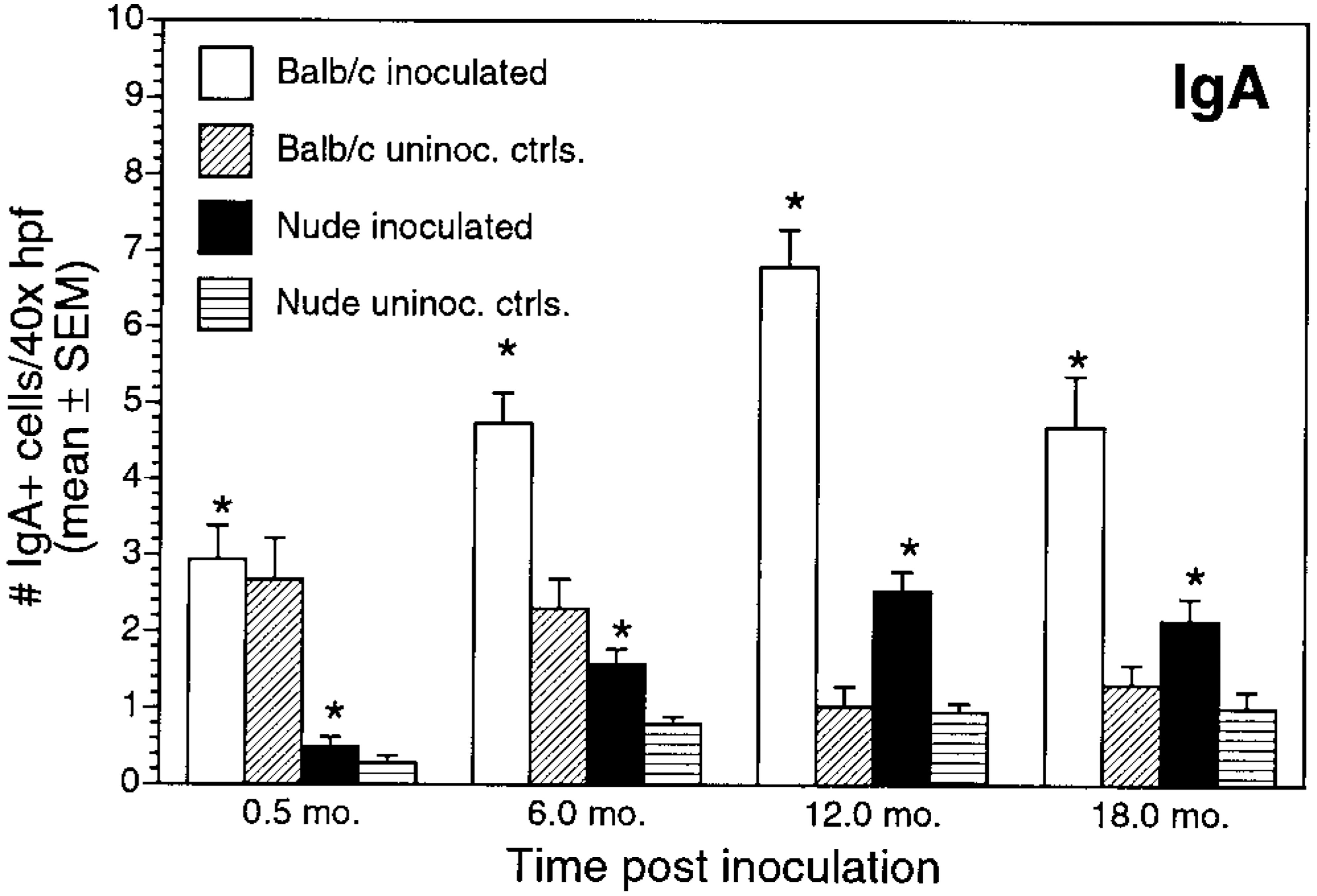
Number of IgA+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05; inoculated BALB/c versus inoculated nude mice.
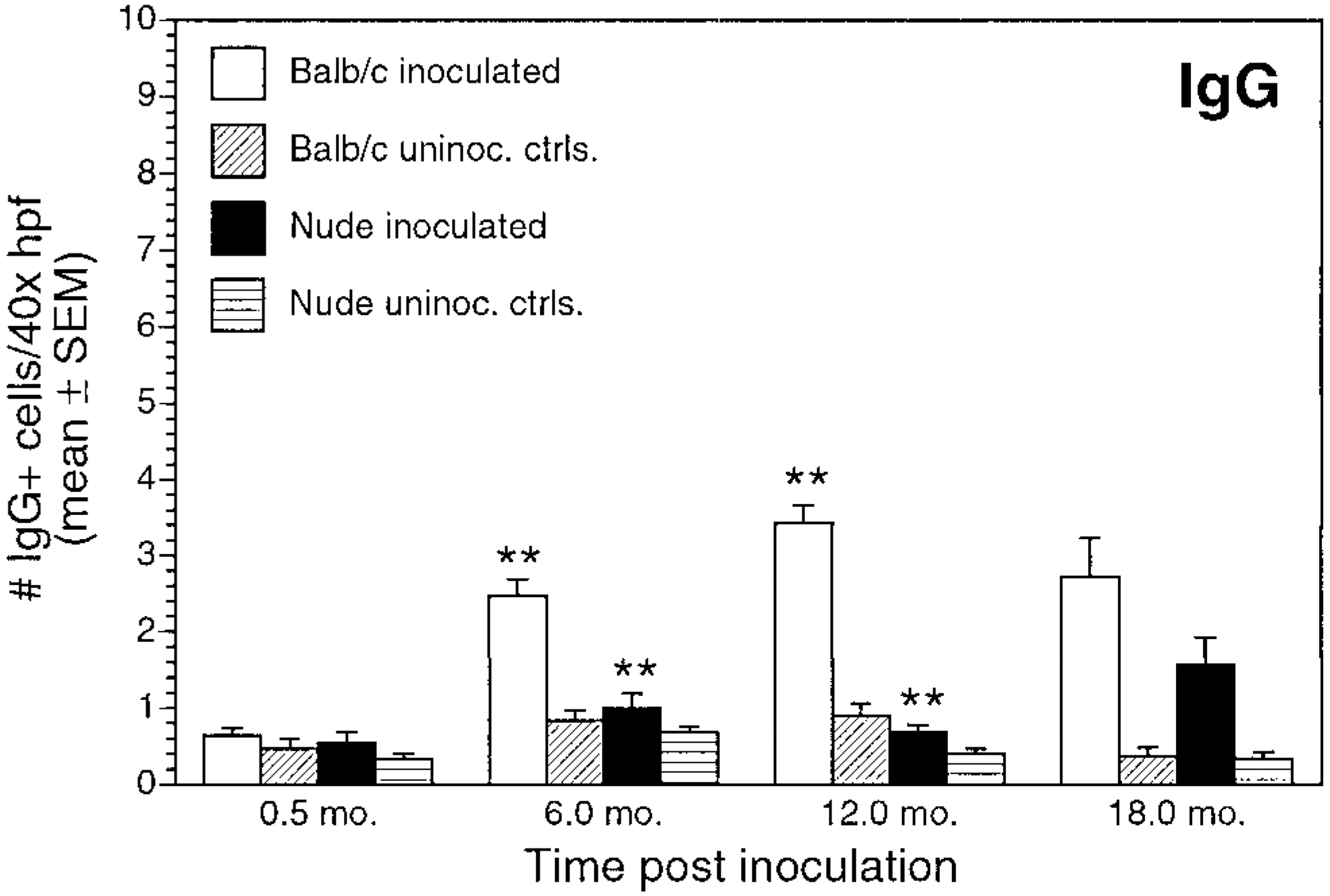
Number of IgG+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
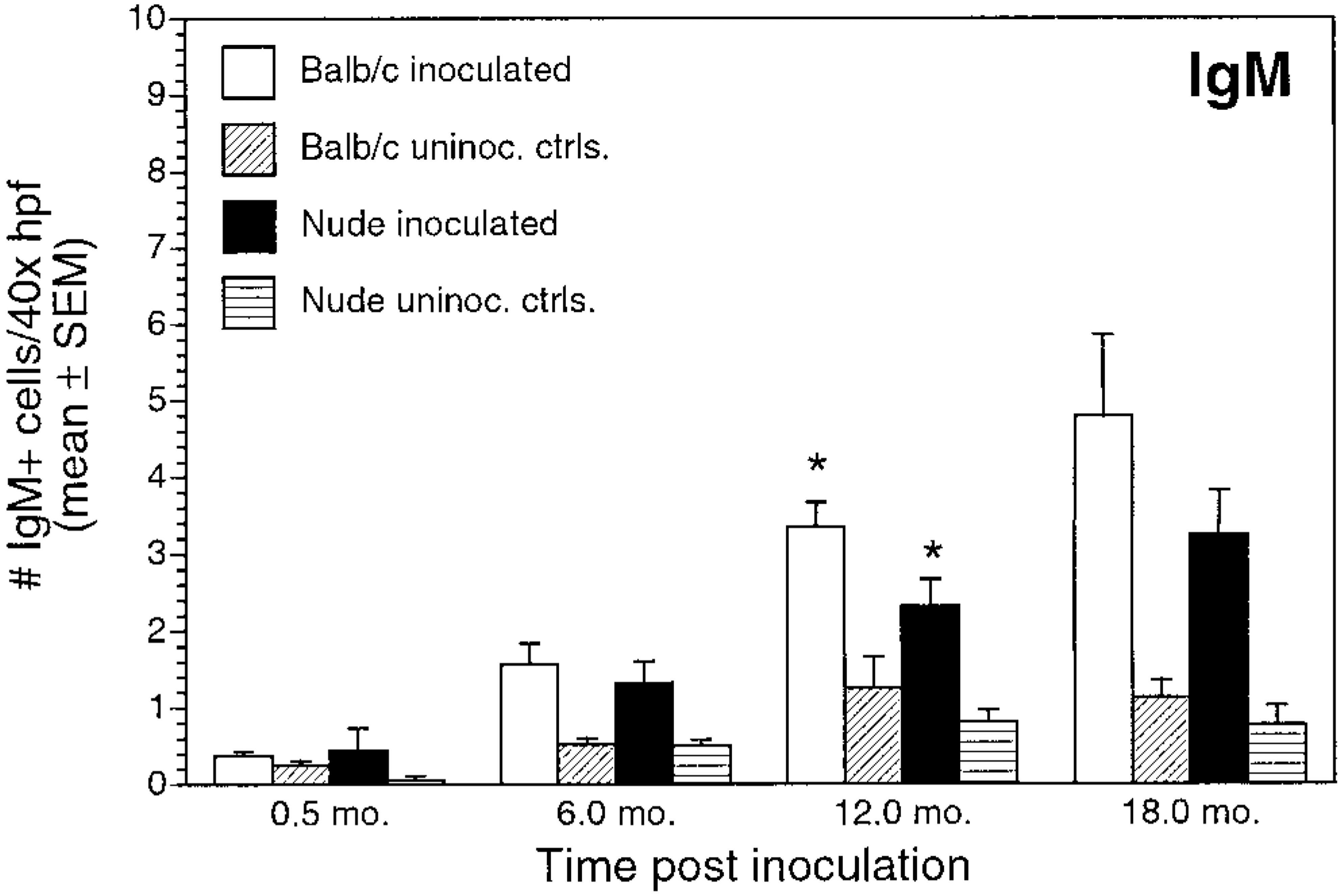
Number of IgM+ cells/40× high-power field (mean ± SEM) for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗P < 0.05, inoculated BALB/c versus inoculated nude mice.
ELISA
Control mice (BALB/c and nude) did not have circulating anti-Helicobacter IgG at any experimental time point (i.e., OD405 < 0.2). BALB/c mice had circulating anti-Helicobacter IgG at 6 and 18 months PI (i.e., OD405 > 0.5). There was a significant difference in mean OD405 values between inoculated and control BALB/c mice at 6 and 18 months PI (P < 0.05). Sera from inoculated nude mice had IgG OD levels within an intermediate zone (i.e., 0.2 < OD405 < 0.5) at 6, 12, and 18 months PI. There were no significant differences in mean OD405 values between inoculated and control nude mice at any time point (Fig. 15).
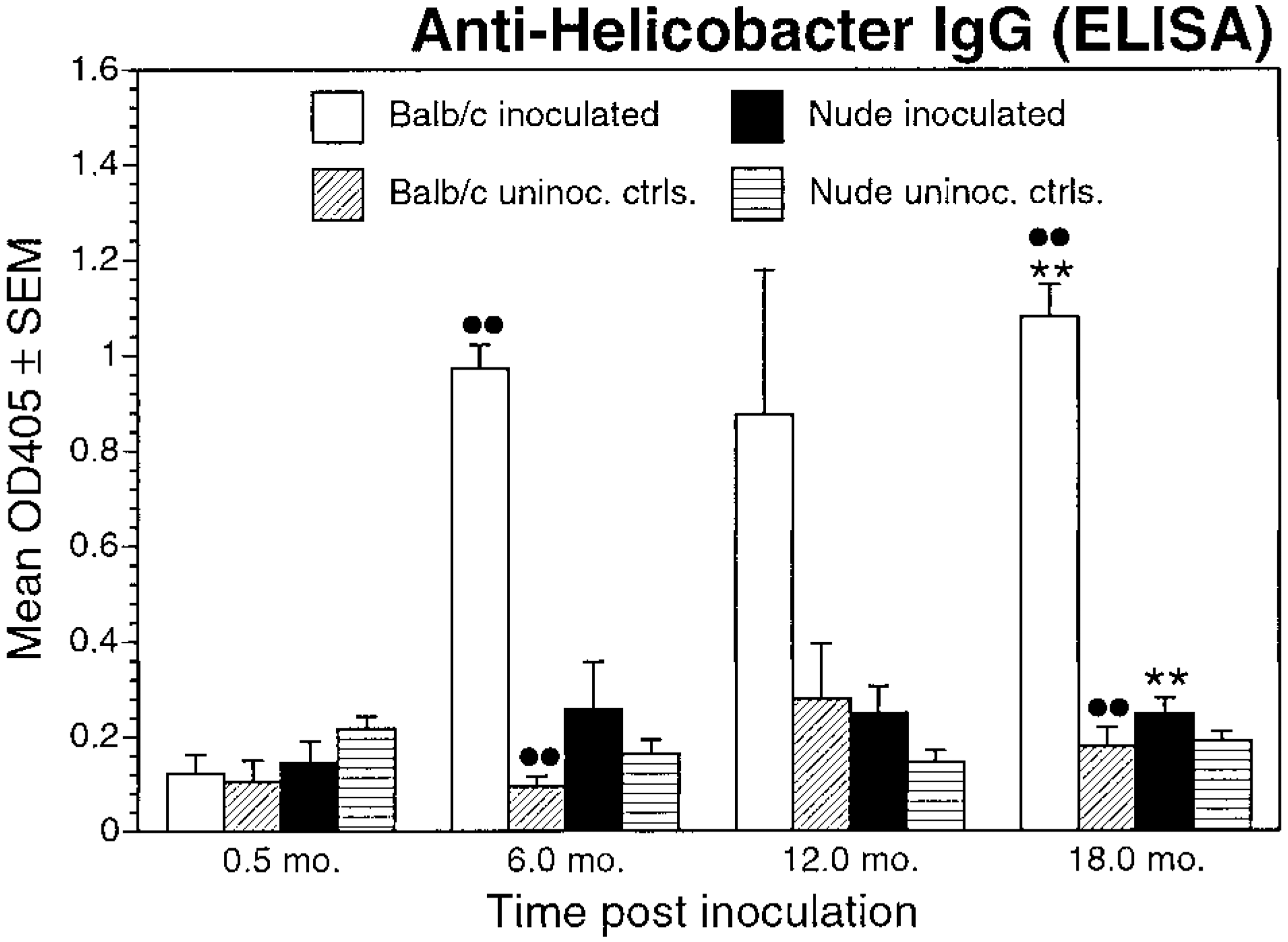
Circulating anti-Helicobacter IgG levels (as determined by ELISA, mean ± SEM) OD405, for inoculated and uninoculated control BALB/c and nude mice evaluated at 0.5, 6, 12, and 18 months postinoculation. ∗∗P < 0.05, inoculated BALB/c versus inoculated nude mice; ••P < 0.05, inoculated BALB/c versus uninoculated control BALB/c mice.
Discussion
Gastric epithelial proliferation demonstrated in this study appears to be characteristic of gastric Helicobacter infection in many species, including piglets colonized with H. pylori, 20 ferrets colonized with H. mustelae, 39 mice colonized with H. felis, 12 H. pylori, 7 or H. heilmannii, 4 5 20 and Mongolian gerbils colonized with H. pylori. 33 35 37 The results of this study confirm previous findings that nodular hyperplasia consistently occurs in BALB/c mice chronically infected with H. heilmannii. 3–5 Nodular hyperplasia is associated with focal proliferation (increased LI) of gastric epithelial cells. 5
In addition, the development of nodular hyperplasia was correlated with acquisition of T cells. Early in the course of infection (6 and 12 months PI), proliferation of gastric epithelium occurred in BALB/c mice in association with chronic gastritis of increasing severity. In young nude mice (i.e., 0.5 and 6 months PI) that were unable to produce functional T cells, 17–19, 21 29 30 34 lymphoplasmacytic gastritis and nodular hyperplasia did not occur. As nude mice aged, however, T-cell infiltrates developed, and mononuclear gastritis appeared. Concurrent with this gastric inflammatory response (12–18 months PI), mucosal nodular hyperplasia occurred in nude mice. Thus, development of lymphocytic gastritis (CD4+ cell influx) in nude mice, although delayed when compared with BALB/c mice, was correlated with induction of gastric epithelial proliferation similar to that characteristic of gastric Helicobacter infection in immunocompetent mice. The correlation of gastric inflammation, gastric lamina propria T-cell influx, and gastric mucosal proliferation/nodular hyperplasia in mice suggests that this proliferation is due, at least in part, to a T-cell-dependent host immune response to H. heilmannii infection.
BrdU LIs of nonnodular areas of the gastric mucosa of infected and control BALB/c and nude mice were not significantly different, suggesting that the development of gastric mucosal nodular hyperplasia is multifocal. The cause of the multifocal nodular hyperplasia is uncertain, but possibilities include increased colonization levels in areas of increased parietal cells (i.e., within parietal cell canaliculi), variable distribution and levels of bacterial colonization, and the presence of increased numbers of T cells within the nodules.
Because nude mice are congenitally athymic and lack normal numbers of mature T lymphocytes, they are often considered completely deficient in major histocompatibility complex–restricted cellular immunity. In this study, however, nude mice did develop gastric inflammatory infiltrates containing T lymphocytes bearing the CD4+ surface marker. Previous studies have shown that nude mice develop increased numbers of mature T cells with age and prolonged antigenic stimulation. 17–19, 21 29 30 34 Delayed T-cell maturation likely explains the delayed onset of mononuclear gastritis in chronically infected nude mice in this study. The mechanism by which T-cell maturation occurs in nude mice is not completely understood but is probably extrathymic. The results of this study confirm that athymic nude mice are capable of developing mature T cells in response to chronic immune stimulation, in this case by infection with H. heilmannii.
In this study, prolonged infection with H. heilmannii induced a chronic active gastritis in both BALB/c and nude mice. Neutrophils and Mac-1+ cells (early phase inflammatory cells) were present at all experimental time points in inoculated mice, defining this as a chronic active gastritis, as reported in previous studies. 5 12 The extent and prevalence of the neutrophilic response decreased over time in inoculated BALB/c mice in combination with an increasingly lymphoplasmacytic inflammatory response. Inoculated nude mice maintained a significant neutrophilic response over the course of the infection, whereas lymphocytes became more prevalent at later times.
Infected BALB/c mice developed a serum immunologic response to Helicobacter antigen. Inoculated BALB/c mice had an anti-Helicobacter IgG humoral response and increased immunoglobulin-containing cells in the gastric lamina propria. The immunohistochemical staining results for IgA, IgG, and IgM-positive cells, although not specific for H. heilmannii, do support a humoral response consisting of IgA, IgG, and IgM in inoculated BALB/c mice relative to uninoculated controls. The humoral response in inoculated nude mice was attenuated relative to that of BALB/c mice at all time points. This finding is consistent with the poorly developed IgA and IgG responses that have been reported in nude mice 16 17 and is most likely due to decreased cytokine stimulation of B lymphocytes early in the infection (<6 months PI).
The decrease in bacterial colonization in infected BALB/c mice over the course of infection is likely a response to changes in the gastric milieu and epithelium secondary to the chronic inflammation and nodular hyperplasia. Nude mice in this study developed lymphoplasmacytic gastritis and nodular mucosal hyperplasia later in the course of the infection, which might explain the failure of bacterial colonization to decrease in nude mice as it did in BALB/c mice.
H. heilmannii-associated gastritis and mucosal remodeling is attenuated in immunodeficient mouse strains lacking normal numbers of CD4+ T cells. Thus, CD4 immunocompetence appears to be required for the development of mucosal hypertrophy/nodular hyperplasia in experimental H. heilmannii-associated disease of mice.
Footnotes
Acknowledgements
This research was funded by National Institutes of Health grants 5-R01-CA67498, R01-A143643, and R29-DK45340. We thank S. Ringler and T. Hoepf for their excellent technical support and S. Krakowka for his critical evaluation of the manuscript.
