Abstract
Seven of 14 newborn pups in a litter of Sprague-Dawley rats were found to have generalized detachment of the epidermis, which was thin, wrinkled, and hung in loose folds over distal extremities. Histologic and ultrastructural examination of the skin showed noninflammatory separation of the epidermis from the dermis at the lamina lucida of the basement membrane zone. Ultrastructurally, hemidesmosomes were small and had a rudimentary appearance; keratin tonofilaments in basal keratinocytes were detached from the hemidesmosomes. The skin lesions were consistent with generalized junctional epidermolysis bullosa, which has not previously been reported in the rat. In humans, generalized junctional epidermolysis bullosa is most commonly caused by autosomal recessive inheritance of defective proteins of the hemidesmosomes or anchoring filaments. The specific protein defect involved in the rat lesion was not determined because fresh frozen tissue was not available.
Keywords
Inherited epidermolysis bullosa (EB) is a family of mechanobullous diseases in which the adhesiveness of the dermoepidermal junction is compromised because of defects in junctional proteins. The defect results in subepidermal cleft formation and detachment of the epidermis that may be localized or generalized and is usually apparent immediately following birth. In humans, EB is a large heterogeneous group of diseases classified according to the level of separation in the dermoepidermal junction, the clinical features, and the mode of inheritance of the disease. 17 The location of the junctional separation is identified by transmission electron microscopy and immunoepitope mapping. 1 The junctional form of EB (JEB) is characterized by separation within the basement membrane zone, most commonly at the lamina lucida, by both lethal and nonlethal clinical presentations, and by an autosomal recessive pattern of inheritance. 1 In veterinary medicine, sporadic cases of JEB have been reported in dogs, 4 , 13 , 16 cats, 9 , 14 cattle, 18 and horses. 6 , 8 , 10 Mouse models of JEB have been found in genetically manipulated strains deficient in the integrins α6, 7 β4, 12 , 19 or α3 2 and in a spontaneously occurring mutant mouse strain with a defective laminin-5 gene. 11 This case report is the first to document the spontaneous occurrence of JEB in a rat.
JEB was observed during a study designed to evaluate the potential reproductive and developmental effects of inhalation exposure to hydrogen sulfide in Sprague-Dawley rats. 3 The dam and sire of the affected pups had been exposed to 30 ppm hydrogen sulfide for 6 hours/day during a 2-week prebreeding, 2-week mating, and 3-week gestational period. Newborn pups from 1 of 37 litters of Sprague-Dawley rats (Charles River, Raleigh, NC) involved in the study were observed to have an epidermis that was thin, wrinkled, and easily detached and that hung in loose folds over the distal extremities, especially the forelimbs (Fig. 1). Seven of 14 pups in the litter and both genders were affected. Shortly after birth, the dam cannibalized most of the affected pups, which were weak and moribund. One pup was immersion fixed in neutral buffered formalin. Neither the dam nor the sire of the affected litter had any grossly apparent skin lesions or other abnormalities when necropsied at the termination of their hydrogen sulfide exposures. The surviving pups of the litter also appeared to be grossly unaffected when necropsied at approximately 23, 61, and 63 days of age. No other animals in the study had similar skin abnormalities. Some affected pups also had additional congenital abnormalities, including shortened limbs and brachygnathia.
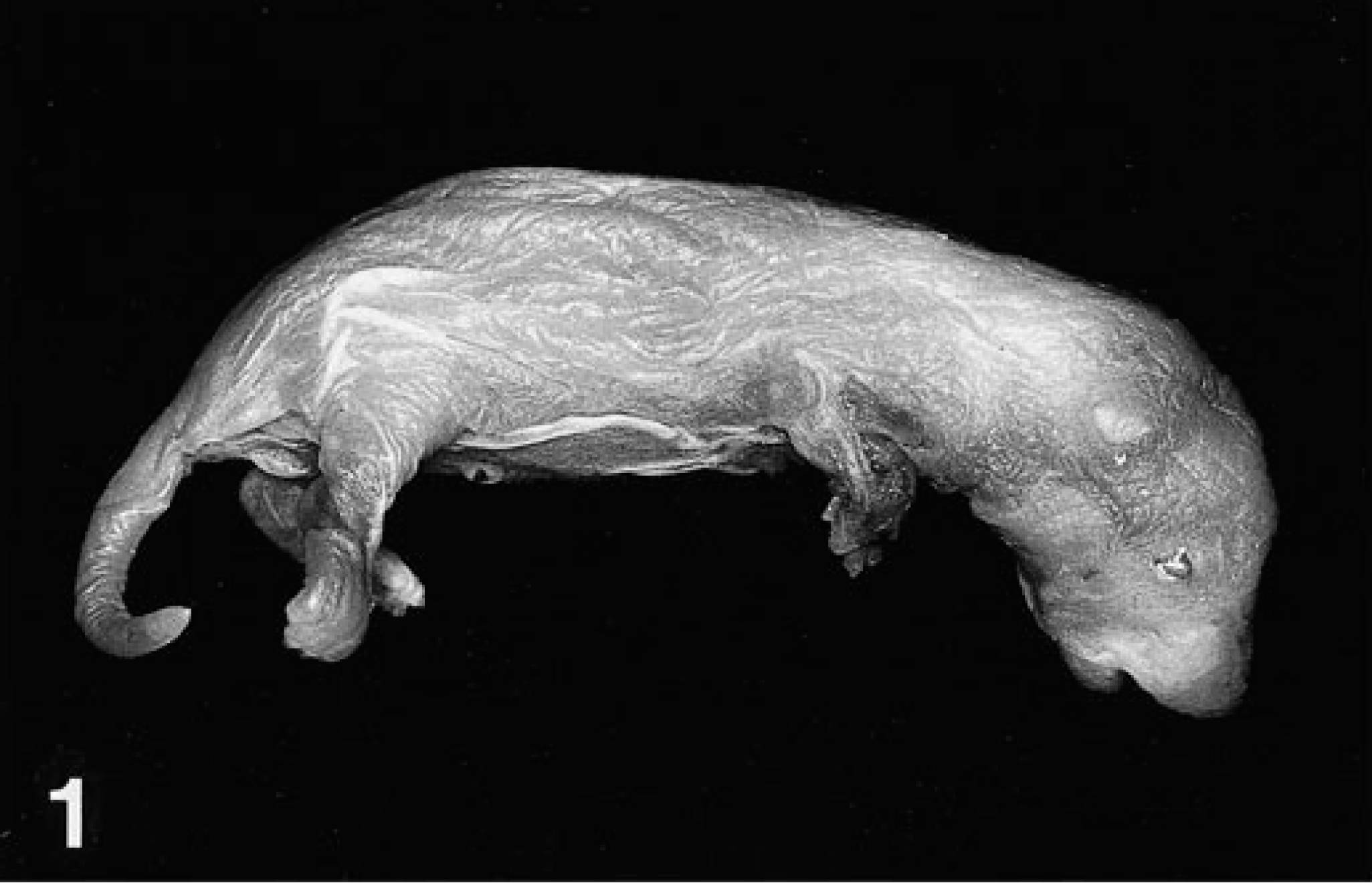
Newborn Sprague-Dawley rat. Generalized junctional epidermolysis bullosa is indicated by a thin, wrinkled, loosely adherent epidermis that hangs in loose folds over distal extremities.
Full-thickness sections of skin from the formalin-fixed pup were taken from five sites (neck, forelimb, cranial flank, caudal flank, and ventral abdomen), embedded in paraffin, routinely processed, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Histologic examination of the skin from all sites showed localized to regionally extensive separation or loss of the epidermis from the dermis at the basement membrane zone without associated hemorrhage or inflammation (Fig. 2, inset). A periodic acid–Schiff reaction failed to localize the basement membrane to either the floor or roof of the subepidermal cleft. Formalin-fixed skin was routinely processed for transmission electron microscopy as previously described. 15 Ultrastructural examination of subepidermal clefts demonstrated separation at the lamina lucida, with the floor of the cleft consisting of the lamina densa (Fig. 3, inset). Ultrastructurally, several components of the dermoepidermal adhesion complex appeared to be malformed. Hemidesmosomes were small and had an amorphous, rudimentary structure. The keratin tonofilaments in the cytoplasm of the basal keratinocytes were not attached to the hemidesmosomes. In contrast, anchoring fibrils appeared similar, in numbers and morphology, to those of an unaffected control pup (Figs. 4, 5).
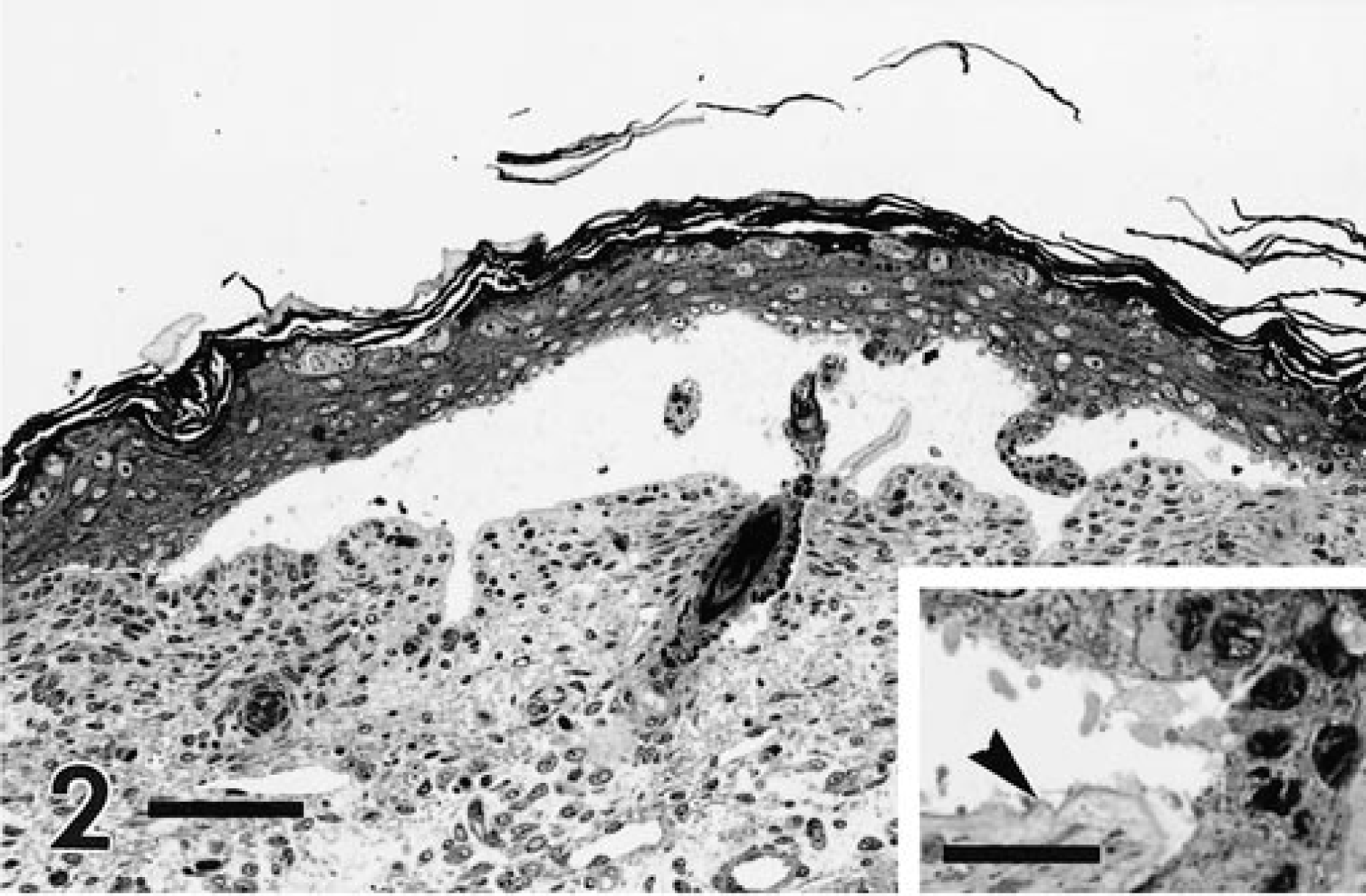
Skin; newborn Sprague-Dawley rat with junctional epidermolysis bullosa. The follicular and surface epithelia of the epidermis are detached from the dermis, forming a subepidermal cleft. HE. Bar = 75 µm. Inset: The basement membrane appears to be located at the base of the cleft (arrowhead). HE. Bar = 30 µm.
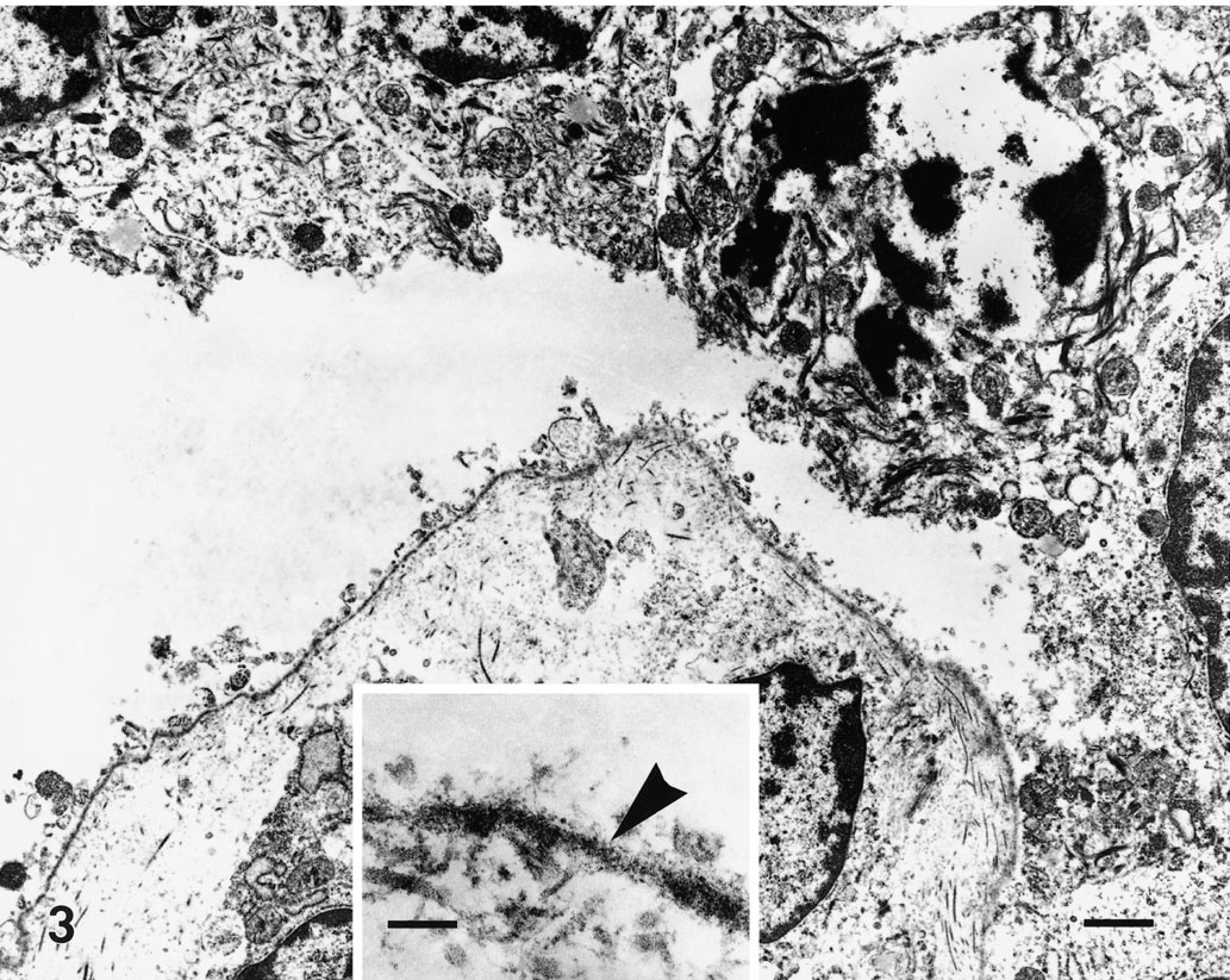
Transmission electron micrograph. Skin; newborn Sprague-Dawley rat with junctional epidermolysis bullosa. Margin of a subepidermal cleft showing the separation of the dermoepidermal junction within the lamina lucida. HE. Bar = 2 µm. Inset: The lamina densa lines the floor of the cleft (arrowhead). HE. Bar = 350 nm.
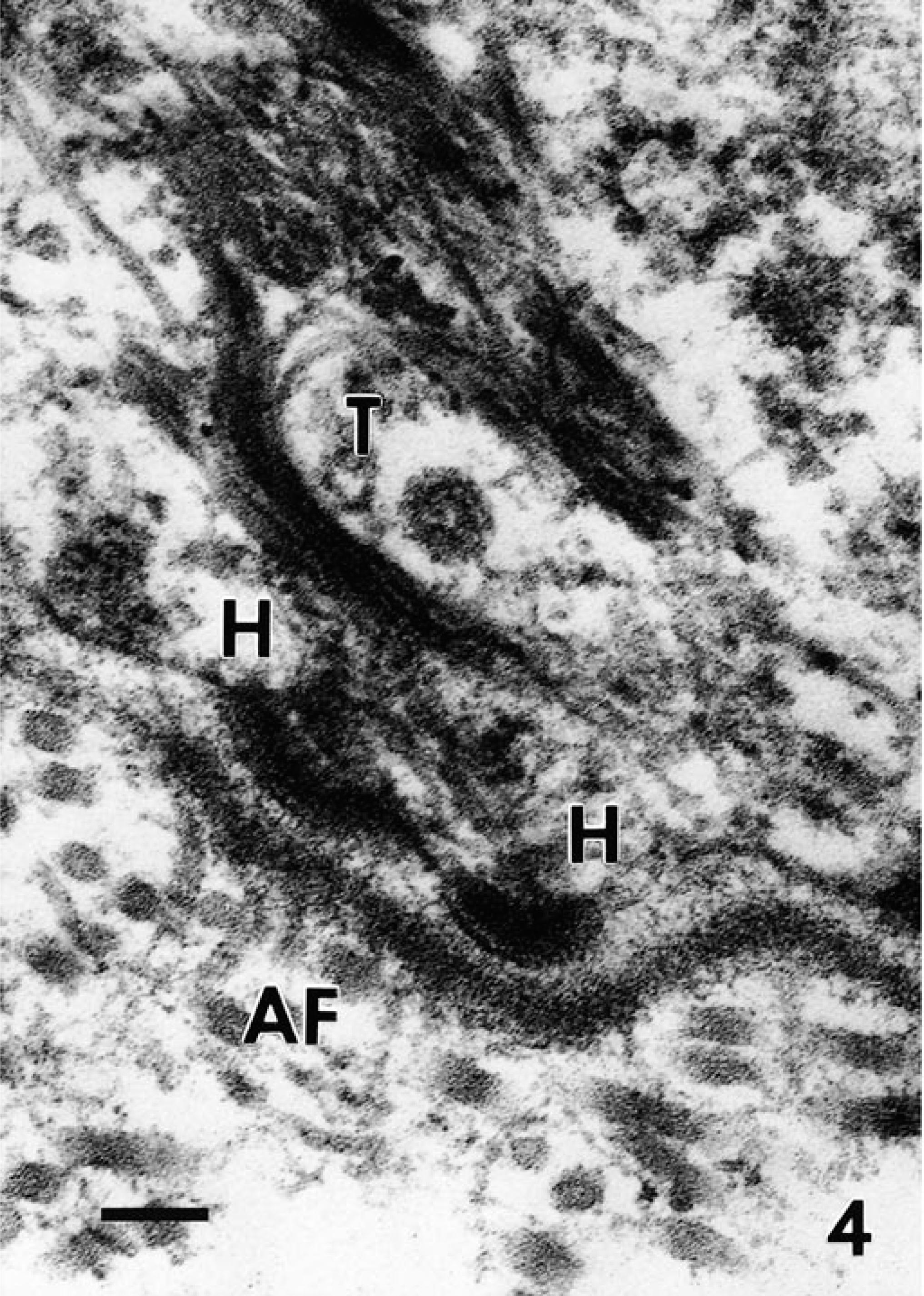
Transmission electron micrographs. Skin; newborn Sprague-Dawley rats. The cutaneous basement membrane zone separating the cytoplasm of a basal keratinocyte (top) from a dermis (bottom) from a control pup (
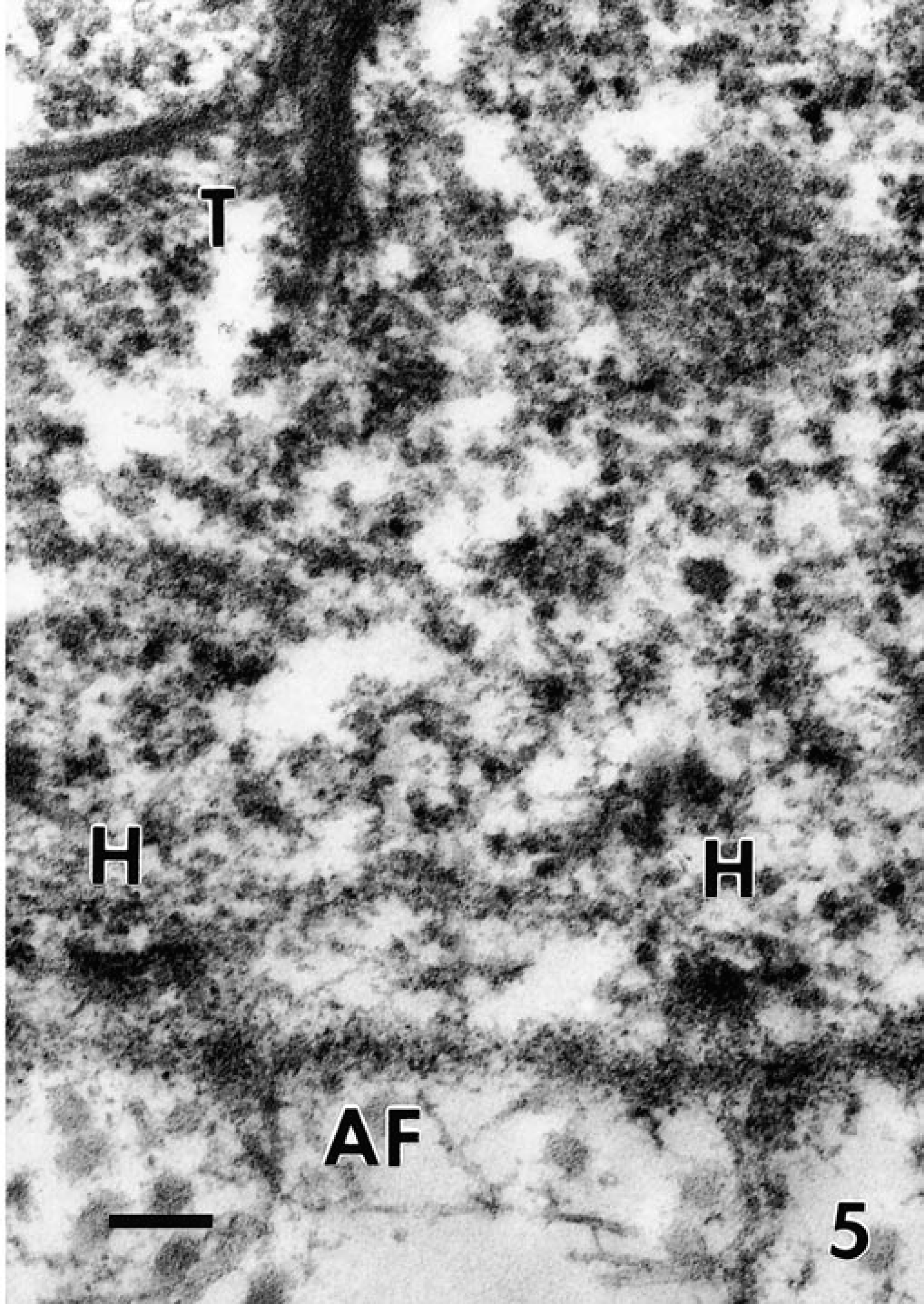
In the affected pup, the hemidesmosomes (H) are small and have a rudimentary appearance; the tonofilaments (T) in the cytoplasm of the basal keratinocyte are detached from the hemidesmosomes. The anchoring fibrils (AF) in the superficial dermis appear similar to those of the normal control pup. Bar = 300 nm.
These gross, histologic, and ultrastructural findings confirm a diagnosis of congenital generalized JEB. The severity, lethality, and generalized distribution of JEB in the Sprague-Dawley rat are most analogous to the Herlitz variant of JEB in humans. The Herlitz variant is the most frequently recognized form of JEB in humans and presents as generalized blistering of skin and mucous membranes, leading to death in early childhood. 5 The protein defects identified in cases of Herlitz JEB are found in the three polypeptide chains of laminin 5, a major component of anchoring filaments that bridge the lamina lucida. JEB with pyloric atresia is one other lethal form of EB in humans with skin lesions potentially analogous to those present in the case of JEB in the rat. In this form, the defective protein is either chain of the α6β4 integrin, a transmembrane glycoprotein that mediates adhesion between the hemidesmosome and the lamina lucida of the basement membrane. 17 Because fresh frozen tissue was not available, immunoepitope mapping of the skin could not be used to ascertain which proteins may have been defective in the affected rat pups.
All clinical variants of JEB in humans have an autosomal recessive mode of inheritance. 1 In the Sprague-Dawley rat, a similar autosomal recessive pattern of inheritance is strongly suspected because of the rare sporadic occurrence of the disease, the normal gross appearance of the parents, and the 50% incidence in the affected litter. Sprague-Dawley rats are an outbred strain; however, the male and female rats used for the breeding experiments in our study may have been littermates, and a fortuitous inbreeding may have occurred, resulting in homozygosity of a recessive trait and phenotypic expression of JEB. It appears unlikely that the hydrogen sulfide exposure contributed to the pathogenesis of JEB in the rat for several reasons. Only one litter in a midrange exposure group was affected out of 28 total litters exposed to hydrogen sulfide, congenital EB has not been reported as a response to toxin exposure, and there is no conclusive evidence that hydrogen sulfide is mutagenic. This is the first case report of the spontaneous occurrence of JEB in a rat.
Footnotes
Acknowledgements
We thank Ms. M. Struve, Ms. K. Miller, Mr. R. A. James, and Ms. E. Gross for their excellent scientific contributions and Dr. B. Kuyper for her editorial assistance. This project was funded in part by the American Petroleum Institute.
