Abstract
Connective tissue growth factor (CTGF) has been shown to mediate many actions of transforming growth factor-beta (TGF-β) in the fibrotic response in several diseases. We compared expression of CTGF, TGF-β, platelet-derived growth factor (PDGF), TNF-α, and interleukin-1 (IL-1) by in situ hybridization in Sprague-Dawley rats euthanized at 0, 2, 4, and 8 weeks after 5/6 nephrectomy using the rat remnant kidney model of renal failure. Collagen was evaluated by trichrome stains, immunohistochemistry, and electron microscopy. We compared expression patterns to cells undergoing metaplasia. Tubular epithelial regeneration and transdifferentiation to myofibroblasts were assessed morphologically and by proliferating cell nuclear antigen, smooth muscle actin, desmin, and vimentin immunohistochemistry. CTGF expression was minimal in controls, mild at 2 weeks and marked by 4 to 8 weeks in interstitial fibroblasts, coinciding with damage, regeneration, and fibrosis. TGF-β expression was increased in many cell types at 2 weeks, increased further by 4 weeks, then remained constant. PDGF-B messenger RNA was found in many stromal cells at 2–4 weeks, but expression decreased at 8 weeks. No significant IL-1 or TNF-α staining was detected. We conclude that CTGF and interacting factors are associated with development or progression of chronic interstitial fibrosis. Proximity of CTGF, TGF-β, and PDGF mRNA expression to regenerative epithelial cells and those transdifferentiating to myofibroblasts suggests that growth factors may modulate renal tubular epithelial differentiation.
Tubulointerstitial fibrosis is a common sequela to renal injury and is the final common pathway in several disorders leading to end-stage renal failure. Expression of a variety of fibrogenic growth factors have been demonstrated in damaged renal tissues after injury and are thought to mediate connective tissue proliferation and fibrotic damage. We asked whether the sequence of growth factors involved in tubulointerstitial fibrosis during the regenerative phases of chronic renal disease was similar to the cascade of growth factors found in other fibrotic diseases. Specifically, we wanted to determine whether evidence of connective tissue growth factor (CTGF) could be identified in areas of tubulointerstitial fibrosis in the rat remnant kidney model of chronic renal failure. We compared CTGF expression with other fibrosis-associated factors, including transforming growth factor-beta (TGF-β) and platelet-derived growth factor (PDGF), with which it interacts. Sampling points of 2, 4, and 8 weeks after renal ablation were utilized to determine in what temporal sequence expression occurred. CTGF, TGF-β, and PDGF have been demonstrated to be important mediators of fibrosis in many human and animal diseases, including chronic sclerotic diseases of renal glomeruli and interstitium. They form complex local paracrine interactions in tissues resulting in fibroblast mitogenesis, chemotaxis, and extracellular matrix production and leading to the fibrotic response characteristic of chronic scarring disease. 3 , 12 , 20 CTGF has been only recently identified as a potential mediator of damaging fibrosis in human renal disease. 19 , 27 We compared the expression of CTGF, TGF-β, PDGF, and the cytokines interleukin-1 (IL-1) and TNF-α by in situ hybridization using multiple mixed specific oligoprobes.
The rat remnant kidney model involves partial nephrectomy (complete renal ablation unilaterally and 2/3 nephrectomy on the contralateral side) and induces degenerative parenchymal changes associated with chronic renal failure. 13 , 14 , 34 By 60 days after surgery, rats are uremic and exhibit marked albuminuria, glomerulosclerosis, interstitial fibrosis, and tubular atrophy. Renal function declines rapidly after 10 weeks. 13 , 14 Lesions are largely a result of lack of functional nephrons and the progressive nature of uremic-associated tubular degeneration. 13 , 15 As tubular atrophy and interstitial fibrosis progress, there is concomitant increased tubular apoptosis and cellular proliferation. 39
In addition to these degenerative and regenerative changes, disruption of the tubular basement membrane has been shown to induce tubular epithelial-myofibroblast transdifferentiation. 26 , 29 This metaplastic transformation from epithelial to mesenchymal-like cells has been documented using intermediate filament markers. 26 , 29 It is thought to increase the number of local fibrogenitor cells and thus further hasten interstitial connective tissue expansion. Because several TGF-β and CTGF family members are involved in embryologic mesodermal determination and mesenchymal differentiation, 6 , 16 , 23 we investigated whether cells undergoing epithelial–myofibroblast transdifferentiation might be spatially and temporally associated with fibrogenic growth factor expression. Immunohistochemistry was used to compare the location of cells undergoing transdifferentiation with signals for extracellular matrix associated growth factors. Both α-smooth muscle actin and vimentin were used, as these proteins are markers of tubular transdifferentiation in this model. Desmin was included as a mesenchymal control limited to cells in the interstitial stroma. The relative amount and location of growth factor expression was also compared with evidence of tubular regeneration.
Materials and Methods
Rat partial nephrectomy
Renal tissues from 24 Sprague-Dawley rats were used in these experiments (taken from a concurrent study involving 193 rats). Subtotal nephrectomy in 3-week-old rats was performed as previously described, 7 , 25 using a two-step method. Two thirds of the left kidney was removed by excising both renal poles under metophane anesthesia. This was followed in 2 weeks by total nephrectomy of the right kidney. Renal decapsulation alone was performed in controls. Rats were euthanized at 2, 4, and 8 weeks after surgery and kidneys harvested. Six sections of nephrectomized kidneys from each time were evaluated by light microscopy, immunostaining, and electron microscopy. Controls were also examined to observe baseline expression. Tissues were placed in buffered formalin, processed routinely, and stained with hematoxylin and eosin and Masson's trichrome.
In situ hybridization
Oligoprobes were produced by the Molecular Genetics facility at the University of Georgia. Sequences encompass at least three separate sites corresponding to highly conserved regions in the growth factor and cytokine molecules. The CTGF antisense oligoprobe mixture consists of three different oligomers taken from the rat CTGF sequence and a 50-bp highly conserved sequence that also labels human and mouse CTGF. The sequences of all probes are listed in Table 1. The oligoprobes were 3′-end digoxigenin labeled using a commercial kit (Genius System, Boehringer Mannheim). Sections of renal tissues were immersed in formalin for less than 12 hours, then embedded in paraffin. Sections were cut at 6 µm, placed on Tespa-coated slides, and dried at 37 C overnight. In situ hybridization was performed routinely. 24 Briefly, slides were twice dewaxed in xylene for 5 minutes, sequentially rehydrated in graded ethanols for 2 minutes each, soaked in phosphate-buffered saline (PBS) for 5 minutes, treated with 20 µg/ml protease K at 37 C for 15 minutes, and rinsed. Slides were then postfixed in 4% paraformaldehyde for 20 minutes, rinsed in PBS twice, and dipped in 50% ethanol for 30 seconds. Specimens were acetylated in 0.2 M HCl, 0.1 M triethanolamine (pH 8), and 0.5% acetic anhydride for a total of 20 minutes to prevent nonspecific binding of the probe. They were rinsed in 0.2 M Tris (pH 7.4) with 0.1 M glycine for 10 minutes. After incubation in 2× standard saline citrate (SSC) at 70 C for 10 minutes, prehybridization was performed in buffer consisting of 50% formamide, 4× SSC, 1× Denhardt's solution, 250 µg/ml yeast transfer RNA, and 10% dextran sulfate at 37 C for 1 hour. Hybridization buffer with 1.5 pmol/slide of a digoxigenin-labeled linearized oligoprobe was then applied to sections for overnight hybridization at 40 C. Slides were washed in solutions of decreasing SSC concentration, incubated in buffer 1 (100 mM Tris-HCl/150 mM NaCl, pH 7.5) from the detection kit (Boehringer Mannheim), blocked in 2% normal sheep serum in buffer 1 and incubated with alkaline phosphatase conjugated to anti-digoxigenin mouse monoclonal antibody diluted 1:300 in buffer 1 and equilibrated in buffer 2 (100 mM Tris-HCl, 100 mM NaCl, pH 9.5). The sections were stained with 4-nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl-phosphate (NBT/BCIP) solution for up to 1 hour, air dried, and counterstained with hematoxylin. Slides from all 24 rats were run simultaneously under identical conditions for each particular probe. Conditions were optimized for each probe to minimize background staining, and multiple replicates were performed to ensure reproducibility.
Sequence of hybridization probes.

Electron microscopy
Formaldehyde-fixed kidney specimens were rinsed in phosphate buffer, postfixed in phosphate-buffered 1% osmium tetroxide, and processed routinely for examination with a Zeiss EM 900 transmission electron microscope.
Immunohistochemistry
Immunoperoxidase staining was performed routinely. Slides were pretreated with steam for 1 minute for antigen retrieval. Monoclonal antibodies to proliferating cell nuclear antigen (PCNA); Sigma), smooth muscle actin (Dako), desmin (Dako), vimentin (Dako), and type I procollagen (Sigma) were obtained commercially and used at dilutions of 1:100, 1:50, 1:50, 1:40 and 1:50, respectively. An irrelevant primary antibody (bovine viral diarrhea virus [BVD]) was used as a negative control. Secondary goat anti-mouse biotinylated antibody (1:200) incubation was followed by avidin–biotin complex reagent (Vector Laboratories), and 3,3′-diaminobenzidene (DAB) was used as a chromogen. Standarization of staining was ensured by performing the procedures on all 24 specimens simultaneously under identical conditions with multiple replicates for reproducibility and optimization for minimal background stain.
Results
Interstitial inflammatory infiltrates were observed in tissues as early as 2 weeks after surgery, with progressive fibrous connective tissue deposition in the interstitium through 8 weeks. Basal CTGF expression in control kidneys was limited to a few interstitial cells and glomerular epithelial cells. CTGF expression was mild at 2 weeks after surgery and found in a few scattered fibroblasts. By 4 weeks, abundant signal was noted in the proliferating interstitial stromal fibroblasts and myofibroblasts. Expression correlated with the degree of tubulointerstitial damage. CTGF expression remained abundant at 8 weeks, corresponding to increased amounts of interstitial fibrosis (Fig. 1). No significant basal TGF-β signal was detected in kidneys at time 0. However, TGF-β expression was abundant in tissues at 2 weeks in macrophages and lymphocytes, glomerular mesangial cells, and endothelial and tubular epithelial cells. The signal strength and distribution increased further by 4 weeks and decreased only minimally at 8 weeks (Fig. 2). PDGF-B expression was not found in the control kidneys but was observed in both glomeruli and mesangial cells in the kidneys harvested 2 weeks after surgery and in scattered fibroblasts and endothelial cells. Expression increased in the interstitium by 4 weeks in areas of interstitial expansion, but expression was sporadic in the later chronic phases of repair at 8 weeks (Fig. 3). At all three times, IL-1 expression was found in a minimal number of interstitial monocytes, and TNF-α was not detectable.
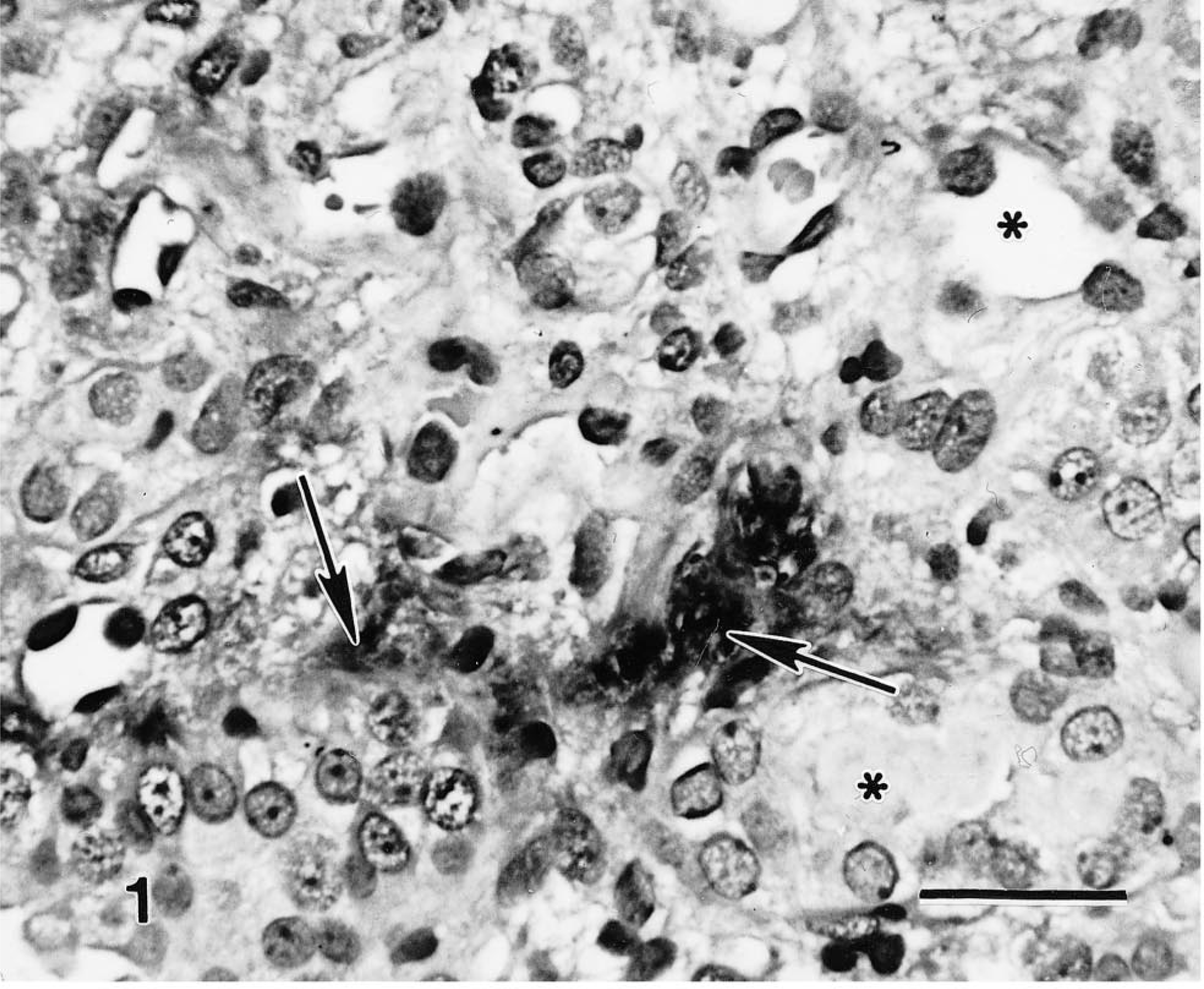
Rat kidney 8 weeks after nephrectomy. In situ hybridization with CTGF antisense oligoprobes. Marked mRNA expression is noted in myofibroblasts (arrows) from areas of interstitial fibrosis and tubular atrophy. ∗ = tubular lumen. Mayer's hematoxylin counterstain. Bar = 25 µm.
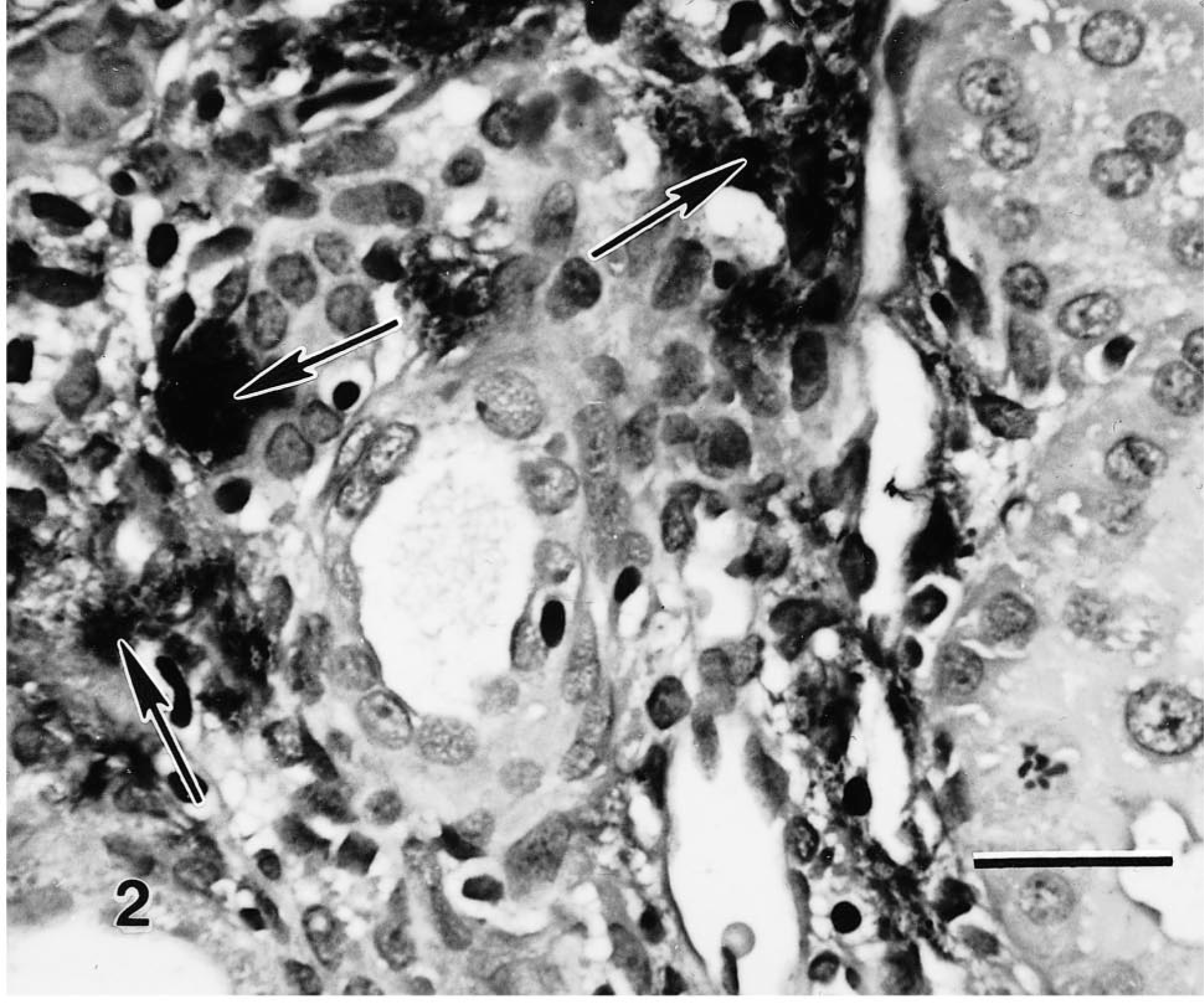
Rat kidney 8 weeks after 5/6 nephrectomy. In situ hybridization with TGF-β antisense mixed oligoprobes. Note intensity of staining surrounding ectatic tubules (arrows). Bar = 25 µm.
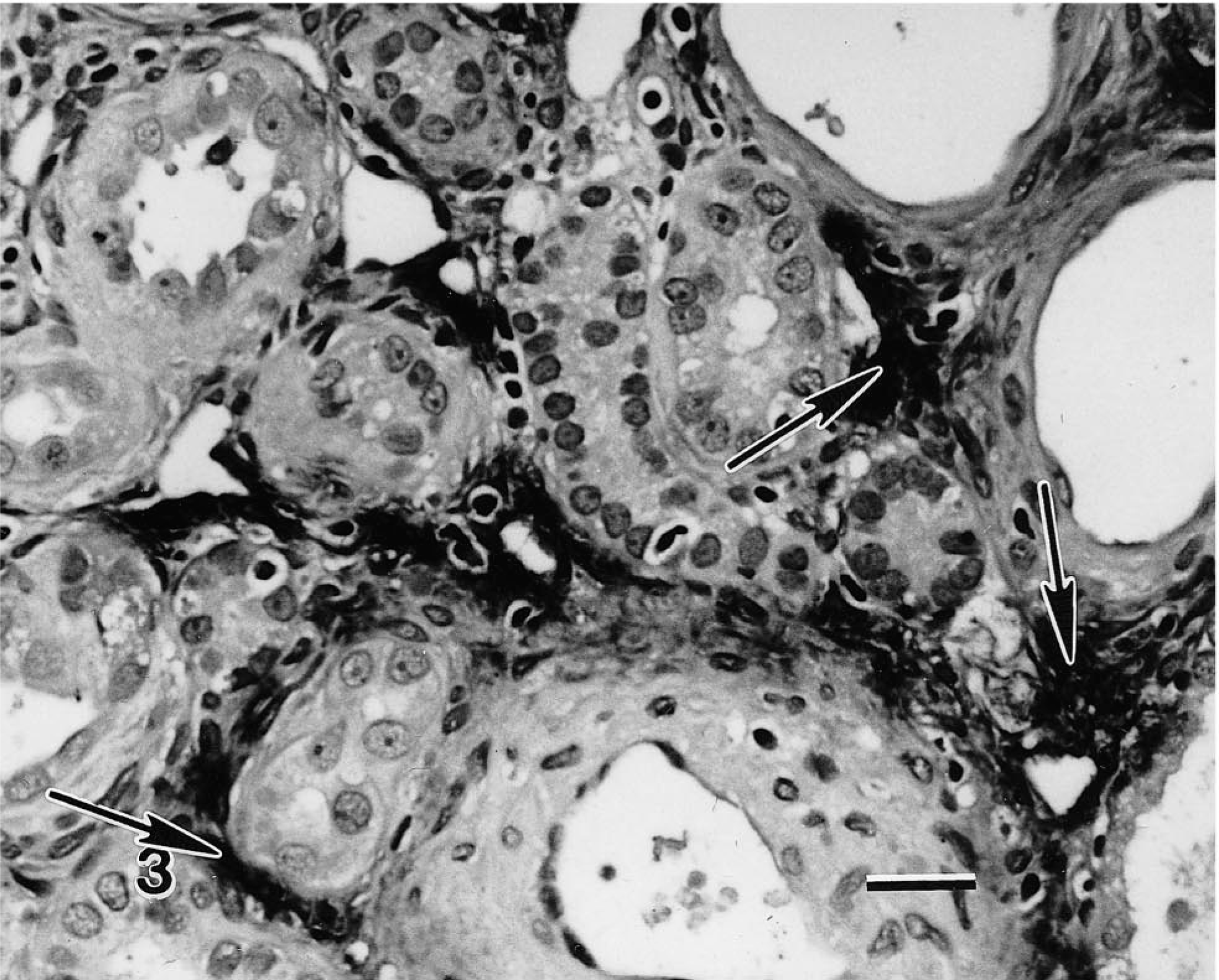
Rat kidney 4 weeks after nephrectomy. In situ hybridization with PDGF antisense oligoprobes. Endothelial cells and interstitial mesenchymal cells adjacent to ectatic tubules are stained heavily (arrows). Mayer's hematoxylin counterstain. Bar = 25 µm.
Collagen deposition was markedly increased by 4 weeks and especially at 8 weeks in the model. It was assessed by routine and trichrome staining, electron microscopy, and by immunostaining with antibodies for Collagen type I. Smooth muscle actin and vimentin immunostaining were detected in many interstitial cells characterized as myofibroblasts. In sequential sections, these same myofibroblasts were associated with both abundant CTGF and TGF-β expression. The number of cells with this myofibroblastic phenotype was minimal at 2 weeks but marked by 8 weeks. Many of the myofibroblasts stained for desmin. Tubular cells undergoing regeneration were identified morphologically by increased basophilia, increased mitoses, cellular hypertrophy, and PCNA staining. They did not stain for growth factor expression; however, many of these regenerative tubules were surrounded by marked peritubular and interstitial fibrosis, with fibrobasts or myofibroblasts that had abundant CTGF and TGF-β expression. Scattered atypical epithelial cells in tubules within damaged areas also stained positively for smooth muscle actin (Fig. 4). Vimentin staining of epithelial cells was common in cells undergoing regeneration and those in areas of damage with disrupted basement membranes (Fig. 5). The presence of epithelial cells staining positively with mesenchymal immunohistochemical markers was found at primarily at 8 weeks in areas where strong CTGF and TGF-β expression were noted.
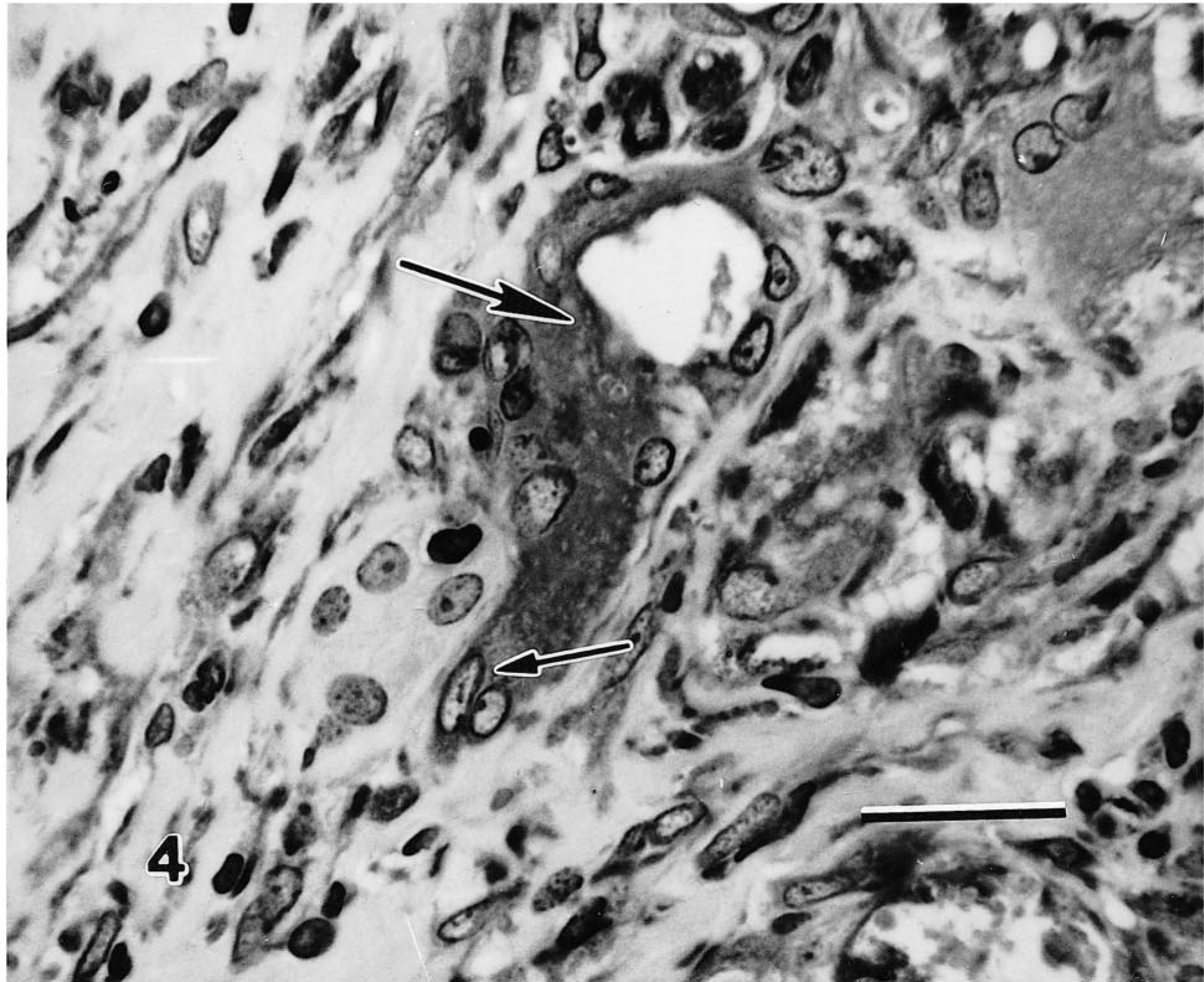
Rat kidney 8 weeks after nephrectomy. α-Smooth muscle actin immunohistochemistry. Cytoplasm from atypical, atrophic tubules (large arrow) stains positively and seems to be continuous with positively staining spindloid myofibroblastic cells in the interstitium (small arrow). This stain is a marker of transdifferentiation in renal tubular cells after injury. Avidin–biotin–peroxidase method, Mayer's hematoxylin counterstain. Bar = 25 µm.
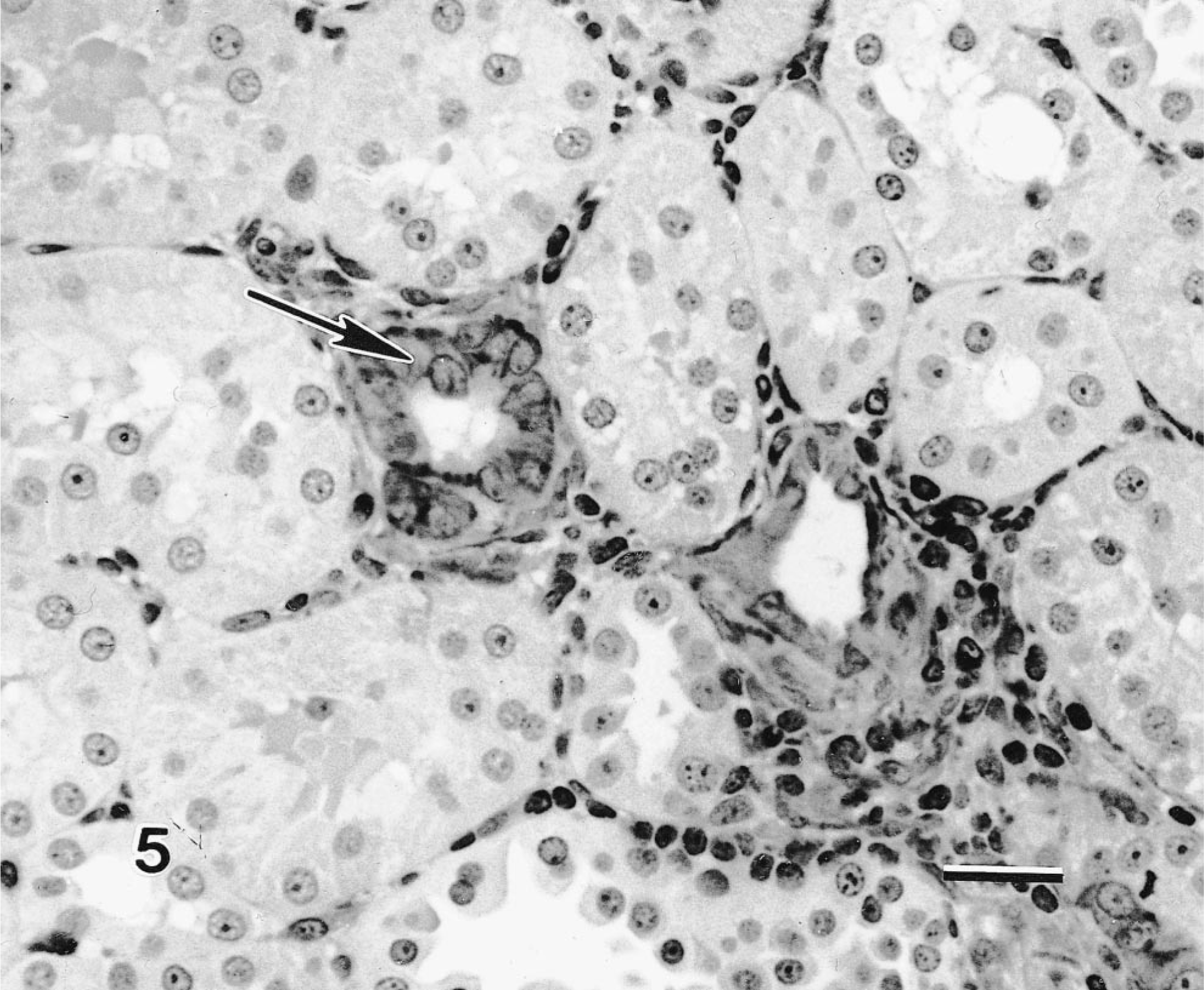
Rat kidney 4 weeks after nephrectomy. Vimentin immunohistochemistry. Note expression in both proximal tubular epithelial cells (large arrows) and interstitial fibroblasts. Avidin–biotin–peroxidase method, Mayer's hematoxylin counterstain. Vimentin is a marker of hyperplasia, mesenchymal metaplasia in renal epithelial cells or both. Bar = 25 µm.
Discussion
CTGF is a 38-kd protein secreted by fibroblasts that stimulates fibroblast proliferation and extracellular matrix production. 5 , 10–12, 24 CTGF belongs to a cysteine-rich family of peptides that function in a wide variety of biological processes, including wound repair, chondrogenesis, and embryological development. 16 , 30 , 36 Murine CTGF has been shown to have proliferative effects on a variety of mesenchymal cells and shares many of the biological effects of human CTGF. 21 , 36 CTGF expression is induced by TGF-β but not significantly by other growth factors, such as PDGF or the fibroblast growth factors. A unique, specific TGF-β response element has been identified in the CTGF promoter that directly induces transcriptional activation of the gene, 17 but expression may also be induced by corticosteroids. 8 CTGF has mitogenic action on fibroblasts 12 , 22 and can induce the extracellular matrix–associated genes type I collagen, fibronectin, and α5-integrin. 10–12 It modulates cell adhesion in fibroblasts and affects neovascularization. 4 , 35 CTGF thus has effects on connective tissue cell proliferation and extracellular matrix deposition similar to TGF-β and appears to mediate many TGF-β actions in these processes. CTGF also appears to be involved in the pathogenesis of various fibrotic disorders, including scleroderma, keloids, 18 stromal rich mammary tumors, 11 atherosclerosis, 31 hepatic cirrhosis, 16 and inflammatory bowel disease. 8 For these reasons, CTGF may be an attractive, specific target for inhibiting fibrotic pathways induced by TGF-β. 10–12
Recently, CTGF has been identified in human renal disorders. Murphy et al. 27 used suppression subtractive hybridization to find that CTGF was differentially induced when mesangial cells were exposed to high glucose levels in vitro. CTGF mRNA was also demonstrated in glomeruli of rats with streptozotocin-induced diabetic nephropathy. The addition of TGF-β to mesangial cells triggered CTGF expression. CTGF expression was suppressed by anti–TGF-β antibody and rCTGF-induced matrix expansion in kidneys. 27 In other experiments involving human renal biopsy specimens, CTGF was strongly upregulated in glomerulonephritis, immunoglobulin A nephropathy, glomerulosclerosis, and diabetic nephropathy. 19 CTGF expression was upregulated in myofibroblasts at sites of chronic tubulointerstitial damage. In contrast, CTGF mRNA expression in normal kidneys was limited to visceral epithelial cells, parietal epithelial cells, and rare interstitial cells. Results we have noted in the current study in rat kidneys parallel these earlier findings by other groups, with limited basal expression in normal renal tissue but marked upregulation of CTGF mRNA in fibroblasts within damaged, fibrotic renal interstitium. This suggests that CTGF activity in the rat remnant kidney model is intimately associated with interstitial connective tissue expansion, just as has been described in other chronic fibrotic diseases, such as hepatic cirrhosis or atherosclerosis.
Elevated TGF-β protein levels have been previously documented in glomerulosclerosis and chronic interstitial nephritis. 2 , 25 TGF-β peptides have also been demonstrated in rat remnant kidneys previously by immunohistochemistry. TGF-β was locally and temporally associated with areas of interstitial fibrosis, glomerulosclerosis, and mononuclear cell infiltration. 7 TGF-β actions enhancing fibrosis are similar to CTGF and include increased fibroblast proliferation and elevated synthesis of extracellular matrix components. We also found TGF-β mRNA expression to be associated with areas of inflammation and fibrosis, thereby correlating message expression with protein localization demonstrated in previous studies.
Several studies have also demonstrated upregulation of PDGF genes and receptors in experimental fibrotic renal diseases, particularly glomerulopathies. 20 , 28 Experimental addition of PDGF-BB has resulted in renal interstitial fibrosis. 38 PDGF-BB (but apparently not the PDGF-AA peptide homodimer) mediates renal injury by stimulation of interstitial fibroblast collagen synthesis and fibroblast proliferation, as well as mesangial proliferation in glomeruli. 28 , 38 Studies have indicated that TGF-β can stimulate the growth of various fibroblastic cell types by the induction of PDGF genes and is itself induced by PDGF. 20 , 28 PDGF facilitates the synthesis and secretion of TGF-β by proximal tubular epithelial cells in response to pathological stimuli in the kidney. 1 , 28 , 32 PDGF activity has been reported to be increased in renal disease, particularly in sclerotic glomerular diseases. 20 , 28 , 37 We demonstrated increased PDGF-B chain mRNA staining with progressive interstitial damage from 2 to 4 weeks in the rat model. However, expression seemed to paradoxically decrease in the later stages of repair at 8 weeks, even while fibrous tissue expansion increased. This could suggest that different growth factors mediate TGF-β responses at different stages of repair or in different cell types, with PDGF acting earlier and CTGF mediating later stages of fibrosis. This sequential variability in growth factor response has been noted in other fibrotic diseases. 11 , 18 , 31
IL-1 and TNF-α are important mediators of the acute phase reaction in the earliest stages of the inflammatory response and can exert a wide variety of proinflammatory influences via the stimulation of other cytokines, including other interleukins. They have direct or indirect effects on the extracellular matrix through angiogenesis, collagen secretion (IL-1), and stimulation of collagenases. We found minimal expression of either IL-1 or TNF-α during the times examined in our experiments. IL-1 was limited to a few macrophages at 2 and 4 weeks, and TNF-α staining was not observed. This is consistent with a previous report using immunohistochemistry. 34 Schiller 34 found no detectable IL-1 or TNF proteins in rat remnant kidneys, although TGF-β levels were modestly and consistently increased from 2 to 7 weeks. Our combined findings suggest these cytokines are not responsible for the progressive proliferation and expansion of extracellular matrix within the renal interstitium in the later phases of repair sampled in these experiments. It is probable that IL-1 and TNF-α are expressed in the early acute phases of repair prior to the 2-week time sampled in this study. The lack of expression of these cytokines from 2 to 8 weeks indicates that the damage associated with the rat remnant kidney model involves mechanisms associated with chronic phases of wound repair, rather than sequential or repetitive bouts of acute inflammatory cytokine cascades.
Many regenerative epithelial cells were noted in tubules, particularly in areas of severe inflammation or interstitial fibrosis. Marked tubular regeneration is an important mechanism for maintaining renal function in this model, but it has also been correlated with increased levels of apoptosis. 39 Regenerative epithelium was consistently demonstrated in areas of growth factor expression in our study based on morphology, PCNA and vimentin immunohistochemistry, and electron microscopy.
Macrophage and myofibroblast accumulation by local proliferation and chemotaxis has been associated with renal damage and loss of function in the rat remnant kidney. 40 CTGF, PDGF and TGF-β probably all interact in inducing proliferation of these cells. Both TGF-β and PDGF can induce the transformation of fibroblasts into myofibroblasts. 9 , 28 , 38 Contraction of the local extracellular matrix mediated by myofibroblasts can enhance renal interstitial scarring and enhance tubular atrophy. 29 , 31 , 40 CTGF expression is often marked in stromal myofibroblasts, 12 , 28 but whether CTGF can induce their transformation from fibroblasts is currently unknown. In addition to differentiation from fibroblasts, transdifferentiation from renal tubular epithelial cells has been described as a mechanism for myofibroblast genesis. 29 Myofibroblasts were identified in our experiments using cytoskeletal markers, including smooth muscle actin, vimentin, and desmin. Myofibroblasts were found in moderate numbers in fibrotic stroma, and they often expressed CTGF. Many of the tubular epithelial cells in the 4- and 8-week kidneys also stained positively for vimentin. Vimentin expression has been noted in damaged and regenerating renal epithelium in rat, human, and bovine kidneys previously and has been associated with changes in cell cytoskeleton, loss of cell to cell contact, and, most recently, with differentiation to myofibroblasts. 26 , 29 , 33 , 40 Some tubular epithelial cells also stained with smooth muscle actin at 4 and especially at 8 weeks. This marker is considered specific for epithelial myofibroblast transdifferentiation under these circumstances. Positively staining cells generally lacked a basal lamina and were adjacent to interstitial cells expressing CTGF and TGF-β. Because many more epithelial cells stained positively for vimentin than smooth muscle actin in our experiments, and because several other groups have noted vimentin staining in regenerative tubular epithelium, 33 it seems probable either that the switch to vimentin occurs earlier in the metaplastic event than the induction of smooth muscle actin, or that vimentin production is common to both regenerative and metaplastic cellular fates. Epithelial vimentin expression in itself is not definitive proof of transdifferentiation into mesenchymal phenotype, as vimentin can be produced during transient cell cycle movement from G0 into G1. However, the combination of smooth muscle actin and vimentin epithelial expression and the presence of many myofibroblasts adjacent to atrophic epithelial cells displaying this phenotype would argue that true metaplastic transformation is occurring. In Fig. 4, there appears to be a continuous transition between smooth muscle actin–positive epithelial cells and positively staining cells with myofibroblast morphology. The myofibroblasts appear to be arising in this and other areas directly from the degenerate epithelium. We believe this indicates that the process is one of transdifferentiation, as previous investigations have indicated. 29 The spatial and temporal association of growth factor expression and epithelial–mesenchymal transformation suggests that paracrine interactions by growth factors may potentially modulate tubular differentiation states. The proliferation of myofibroblasts from epithelium (via induction from growth factors secreted by adjacent interstitial myofibroblasts) could therefore create a potential vicious cycle of extracellular matrix expansion.
CTGF family members are known to be involved in mesenchymal differentiation. The
Sequential expression of growth factors in the rat remnant kidney model follows the paradigm of the TGF-β–CTGF pathway of fibroplasia noted in other tissues. CTGF expression plays a central role in the development of interstitial fibrosis throughout the chronic phase of renal regeneration and repair through its actions on the extracellular matrix and (myo)fibroblast recruitment. Proximity of CTGF, TGF-β, and PDGF mRNA expression to regenerative epithelial cells and those undergoing transdifferentiation to myofibroblasts suggests that paracrine interactions by interstitial growth factors may modulate regenerative or metaplastic responses in renal tubular epithelium and provide a mechanism for proliferation of myofibroblasts and amplification of fibrogenic responses.
Footnotes
Acknowledgements
We appreciate the assistance of Lisa Whittington with immunohistochemistry and in situ hybridizations and of Drs. Corrie Brown, Gary Grotendorst, and Jaroslava Halper for critical review of the manuscript.
