Abstract
Shiga toxins (Stx) produced by Escherichia coli cause systemic vascular damage that manifests as edema disease in swine and hemolytic uremic syndrome in humans. In vitro, Stx inhibit protein synthesis and, depending on circumstances, induce necrosis, apoptosis, or both. The mechanism of in vivo Stx-mediated vascular damage is not known. The ability of Stx to cause apoptosis of vasculature in vivo was studied in pigs with edema disease that was produced by oral inoculation with Stx-producing E. coli. Arterioles of ileum and brain were evaluated by terminal dUTP nick-end labeling (TUNEL) assay for DNA fragmentation in myocytes (10 infected pigs, 5 control pigs) and by transmission electron microscopy for ultrastructural changes characteristic of apoptosis (17 infected pigs, 8 control pigs). In comparison with controls, increased numbers of TUNEL-positive arterioles were detected in 6/10 (60%) subclinically affected pigs 14–15 days after inoculation. Ultrastructurally, lesions in myocytes consisted of lysis (necrosis), with cytoplasmic debris and nuclear fragments contained between intact basement membranes. Endothelial cell changes ranged from acute swelling to necrosis and detachment from basement membrane. Subclinically affected pigs (n = 14) tended to have changes predominantly in myocytes, whereas pigs with clinical illness (n = 3) more commonly had changes in endothelial cells. The arteriolar lesions and clinical signs of edema disease are attributed to the effects of Stx on vasculature. Therefore, our findings suggest that the Stx-induced arteriolar lesions seen in this study were primarily necrotic, not apoptotic. We suspect that necrosis was the principal cause of the DNA fragmentation detected.
Keywords
Shiga toxin–producing Escherichia coli (STEC) are enteric pathogens that cause systemic vascular damage in diseases such as hemolytic uremic syndrome (HUS) in humans and edema in pigs. 2 , 11 , 14 Numerous studies suggest that the systemic manifestations in these diseases result from the vascular toxicity of Shiga toxins (Stx). 35 , 37 , 40 Verocytotoxin and Shiga-like toxin are synonyms of Stx, and the terms are used interchangeably in the literature. In various cell lines, Stx inhibit cellular protein synthesis and cause necrosis, apoptosis, or both. 12 , 15 , 18 , 32 , 33 , 35 However, the pathogenesis of systemic vascular disease in vivo is not clear. Based on the observation that Stx cause apoptosis in Vero cells, Taylor suggested that Stx-induced apoptosis might be a cause of vascular damage in HUS. 38 It is not known whether apoptosis occurs in the blood vessels of pigs with edema disease.
Stx are 70-kDa proteins composed of a single A and five B subunits. The B subunits bind to the glycolipid receptors on the cell surface, and the enzymatically active A subunit inhibits protein synthesis at the ribosomal level. 32 Stx1 and Stx2 are two types within the Stx family. These types are distinguished on the basis of their sequence and antigenic similarities with Stx produced by Shigella dysenteriae. 29 , 32 , 33 The latter has 99% amino acid sequence homology with Stx1 but only 56% homology with Stx2. 29 Antibodies against S. dysenteriae–produced Stx neutralize Stx1 but not Stx2. Stx2, and its closely related variant subtypes Stx2e, Stx2c, and Stx2d, share 84–97% amino acid sequence homology among them. 29
Stx induce apoptosis in several cell types in vitro, including epithelial, lymphoid and endothelial cells. 15 , 18 , 27 , 34 Endothelial cell sensitivity to Stx depends on the quality and quantity of surface receptors globotriaosyl ceramide (Gb3) and globotetraosyl ceramide (Gb4). 22 , 34 In general, endothelial cells derived from the microvasculature (capillaries) are more sensitive than endothelial cells derived from large vessels (umbilical vein, arteries), and the sensitivity correlates with Gb3 expression. 36 Exposure of endothelial cells to lipopolysaccharide or cytokines such as tumor necrosis factor α (TNF-α) or interleukin-1 (IL-1) increase sensitivity of some endothelial cells to the cytotoxic effects of Stx. 12 , 23 , 24 In some cases, this is due to the increased expression of receptors. 34 , 41
In susceptible swine, Stx2e-producing E. coli cause edema disease that is characterized clinically by neurologic signs (ataxia, incoordination, recumbency) or edema in various tissues. Clinical signs and gross lesions of acute disease are replicated by intravenous injection of purified Stx2e. 26 Immunization of pigs against Stx2e prior to challenge with STEC prevents development of neurologic signs and lesions. 4 , 13 , 25
A microscopic hallmark of edema disease is an arteriolar lesion commonly referred to as vascular necrosis. 2 , 16 , 19 Brain and gastrointestinal tract submucosa are common sites of the lesions. Lesions in acutely affected pigs range from fibrinoid necrosis to minimal changes characterized by necrosis of individual myocytes. Lesions in subclinically affected pigs tend to be limited to individual cell death within tunica media. 16 , 19 Histologic lesions are detected more commonly in subclinically affected pigs than in acutely affected pigs. Blood vessels with focal alterations in tunica media have some features characteristic of apoptosis, such as karyorrhectic and pyknotic changes in myocytes and, commonly, lack of inflammation. 16 , 19 , 26 , 31
The ultrastructure of Stx-induced lesions has been studied in several animal species with experimental and natural disease. Experimental STEC infection in pigs results in ultrastructural changes in the vasculature of intestinal lamina propria that is characterized by swelling of endothelial cells and necrosis of myocytes. 31 Stx induce apoptosis of enterocytes extruding into the lumen in rabbit intestinal loops. 17 Similar changes occur in rabbits orally infected with STEC, and the morphologic findings are supported by TUNEL (terminal dUTP nick-end labeling, a method for detection of the DNA fragmentation that usually occurs during apoptosis). 43 Mice orally infected with STEC develop lesions ultrastructurally characterized by degenerative and necrotic renal tubular cells. 15 , 46 DNA fragmentation occurs in numerous renal tubules and in a few glomeruli of these mice. 15 Stx1 causes lesions in the renal glomeruli and tubules of baboons. 39 Ultrastructurally, glomerular endothelial cells of the baboons are swollen, and there are fibrin thrombi in capillaries. Ultrastructural changes in tubules consist of epithelial degeneration with cell extrusion into lumen, cytoplasmic blebbing, and epithelial necrosis. In summary, ultrastructural and DNA fragmentation studies of in vivo Stx-induced lesions in animals other than swine support the notion that both necrosis and apoptosis can occur. These findings are in agreement with in vitro results.
The objective of this study was to determine if the arteriolar lesions characteristic of edema disease in swine are the result of necrosis or apoptosis. We used TUNEL staining to identify double-strand DNA breaks that often occur in apoptosis. We also examined the ultrastructural changes of affected blood vessels for the morphologic features of apoptosis such as cell shrinkage, membrane blebbing, cell membrane preservation with apoptotic body formation, nuclear condensation, and chromatin margination. 47
Materials and Methods
Inoculation and sample collection
The previously described experimental model of edema disease was used. 4 , 16 Accordingly, 3-week-old weaned crossbred pigs (Landrace, Duroc, Yorkshire, and Hampshire) were orally inoculated with a single dose of 1010 colony-forming units of STEC strain S1191 or M112 or the nonpathogenic E. coli strain 123. 4 , 16 Pigs were fed ad libitum a commercial diet containing 21% protein. Use of this well-characterized disease model ensured that vascular lesions were consistently induced in clinically and subclinically affected pigs. Strain S1191 is of O139 serotype and produces F18ab fimbriae, Shiga toxin 2e (Stx2e), and heat-stable enterotoxin B. 28 Strain M112 is a recent, previously unreferenced isolate of serotype O147 that produces F18ac fimbriae, Stx2e, and heat-stable enterotoxins A and B. Control pigs were inoculated with nonpathogenic E. coli strain 123 (serotype O43). Pigs were observed twice daily throughout the experiment for neurological disturbances (incoordination, circling, ataxia, recumbence) and subcutaneous edema indicative of edema disease. Pigs were euthanatized by intravenous administration of barbituates. Pigs challenged with S1191 or M112 that developed central nervous system signs or edema were designated as clinically affected pigs. Pigs challenged with S1191 that remained free of clinical disease during the 2 weeks after inoculation were designated as subclinically affected pigs. Samples were collected from clinically affected pigs at the time of the illness (3–6 days postinoculation [dpi]) and from subclinically affected pigs at the end of experiment 13–15 dpi. Samples from control pigs were collected 12–14 dpi.
Light microscopy: TUNEL and hematoxylin and eosin stain
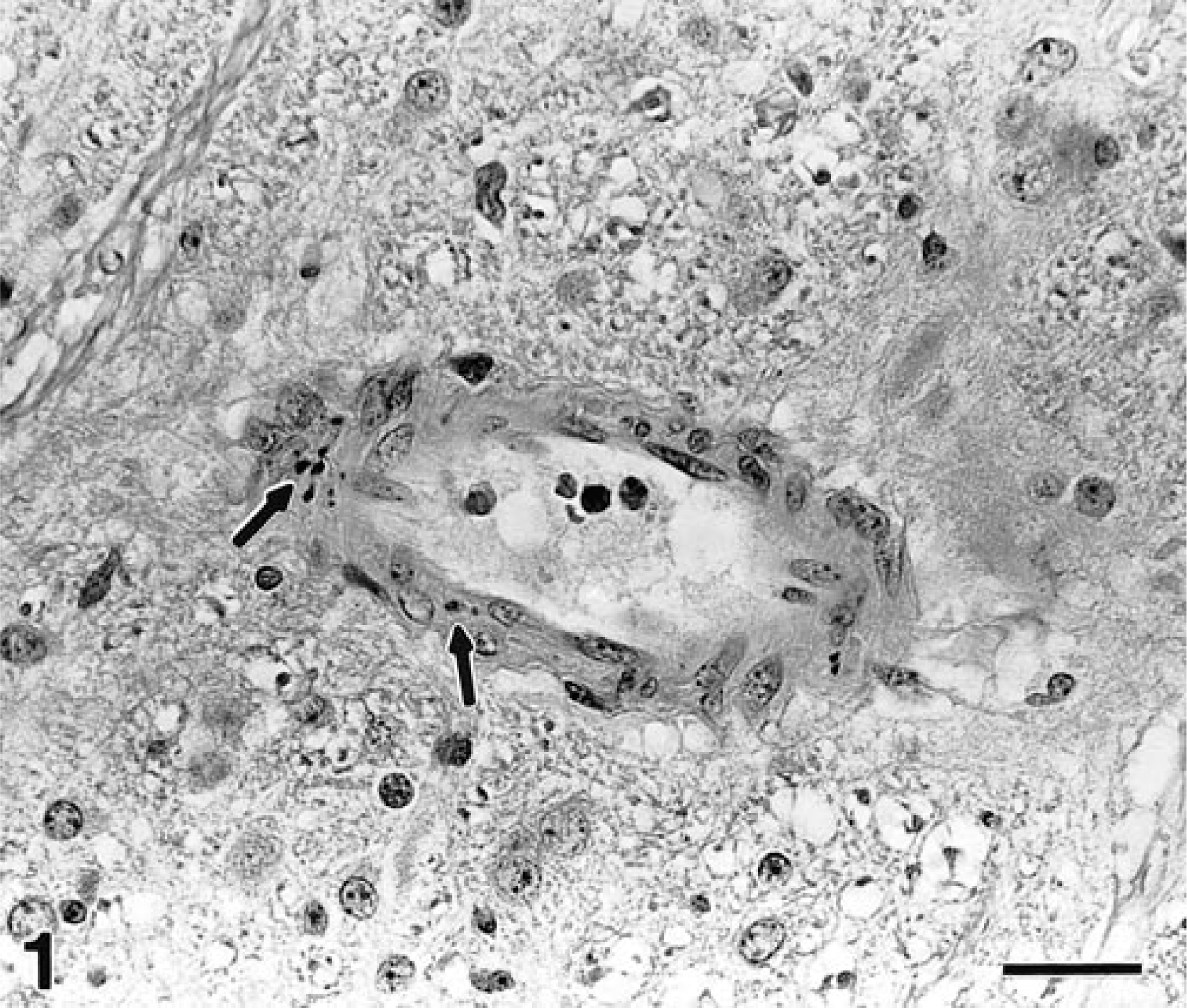
Ileum (1 m proximal to ileocecal valve) and brain stem (medulla oblongata) were collected from 10 subclinically affected pigs (Nos. 1–10, challenged with strain S1191) and 5 controls (Nos. 13–17; Table 1). Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and processed for staining by hematoxylin and eosin (HE) and TUNEL. An ApopTag kit (Oncor, Gaitherburg, MD) was used for TUNEL assay according to the manufacturer's instructions. Briefly, tissue sections were treated with proteinase K at room temperature for 12 minutes. Hydrogen peroxide was used to neutralize endogenous peroxidase. Tissue sections were subjected to an enzymatic incorporation of digoxigenin-labeled nucleotides with terminal deoxynucleotidyl transferase and then washed and incubated with anti-digoxigenin peroxidase. After washing, slides were incubated with diaminobenzidine (Boehringer Mannheim, Indianapolis, IN) and counterstained with hematoxylin.
STEC-infected and control pigs included in a study of arterioles by TUNEL.
∗ SC = STEC-infected pig that did not develop clinical signs of edema disease during 13–15 days after inoculation; clinical = STEC-infected pig that developed clinical signs of edema disease; control = control pig inoculated with nonpathogenic E. coli.
† A pig was considered to be positive if a tissue section contained two or more arteriolar profiles with pyknotic or karyorrhectic nuclei in the media.
‡ A pig was considered to be positive if the tissue section contained two or more arteriolar profiles with TUNEL staining in the media.
TUNEL-stained sections were evaluated qualitatively and quantitatively. Qualitative scoring sections were considered positive if two or more arteriolar profiles per section contained labeled myocytes. For quantitative evaluation, the number of TUNEL-positive arterioles detected was expressed as a percentage of the total number of arteriolar profiles in the section.
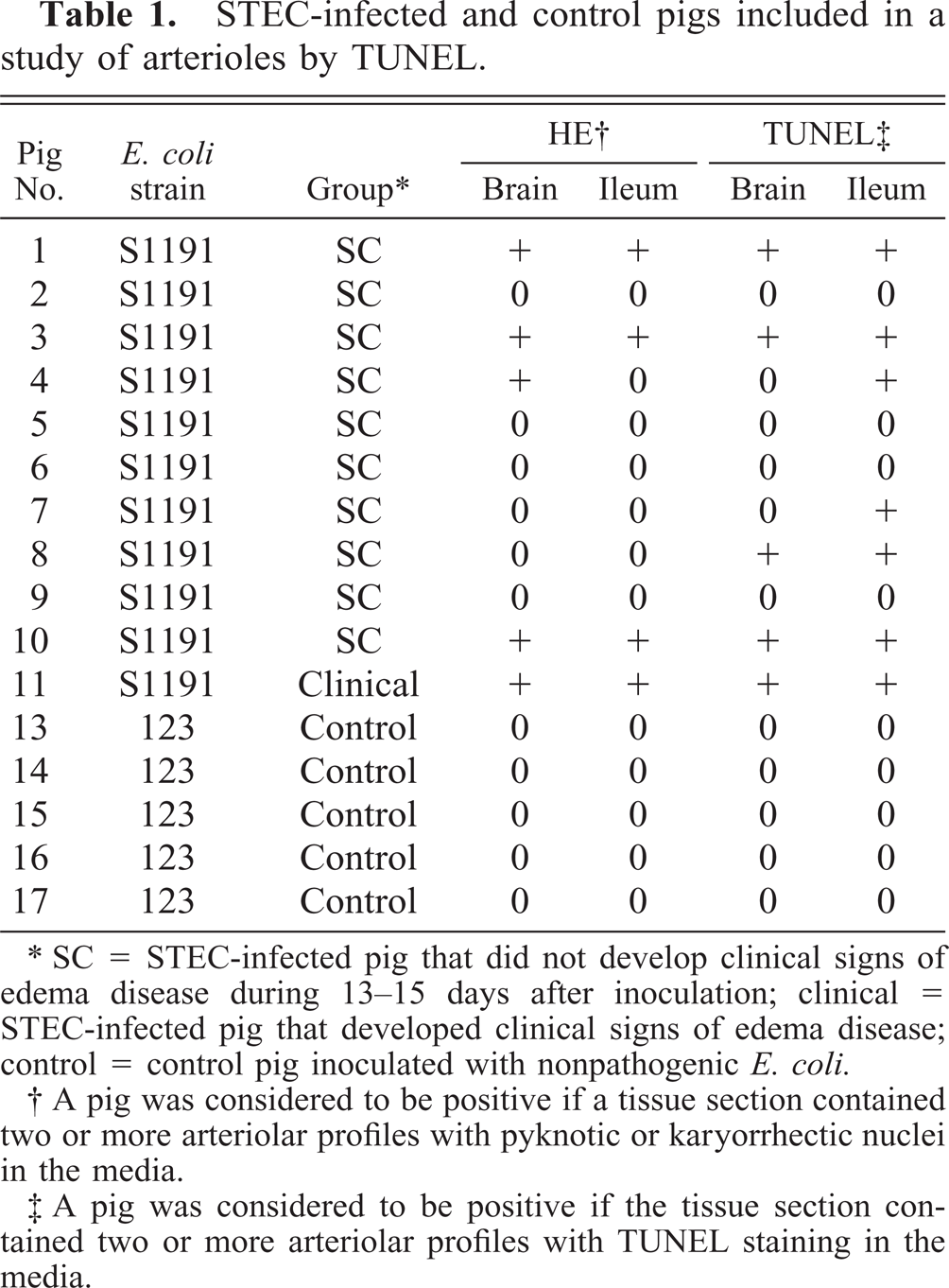
HE sections were evaluated qualitatively for the presence of arteriolar lesions characterized by karyorrhectic and pyknotic nuclear fragments in the media (Fig. 1). HE sections were considered positive if two or more arteriolar profiles per section were affected.
Brain; pig No. 12, subclinical 14 days after inoculation with STEC. Arteriole with karyorrhectic and pyknotic nuclear fragments in media (arrows). HE. Bar = 25 µm.
Transmission electron microscopy
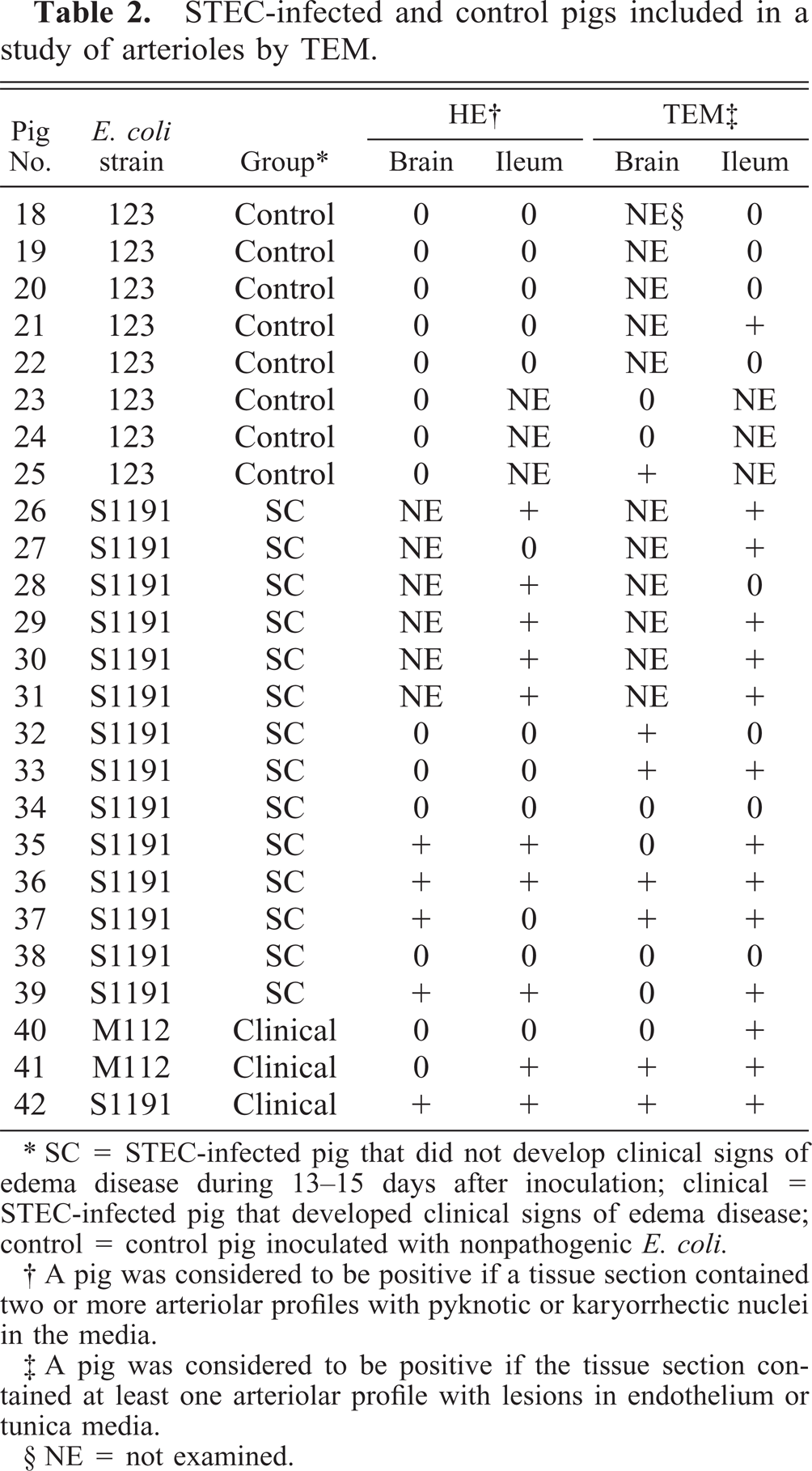
Fourteen ileal and eight brain stem samples were collected from 14 subclinically affected pigs (Nos. 26–39) challenged with S1191 in two experiments (Table 2). Both tissues were also collected from three clinically affected pigs (Nos. 40–42) challenged with S1191 (n = 1) or M112 (n = 2) in another experiment. Five ileal and three brain samples were collected from eight control pigs (Nos. 18–25) in two experiments.
STEC-infected and control pigs included in a study of arterioles by TEM.
∗ SC = STEC-infected pig that did not develop clinical signs of edema disease during 13–15 days after inoculation; clinical = STEC-infected pig that developed clinical signs of edema disease; control = control pig inoculated with nonpathogenic E. coli.
† A pig was considered to be positive if a tissue section contained two or more arteriolar profiles with pyknotic or karyorrhectic nuclei in the media.
‡ A pig was considered to be positive if the tissue section contained at least one arteriolar profile with lesions in endothelium or tunica media.
§ NE = not examined.
Animals were given intravenous sodium pentobarbital until a deep anesthetic state was attained. Ileal samples were then collected 1 m proximal to ileocecal junction and immersed immediately in 3% glutaraldehyde solution in 0.1 M Na cacodylate buffer. Animals were then euthanatized with intravenous barbituates, and samples of medulla oblongata were collected and placed in cacodylate buffered glutaraldehyde. Samples were processed further for transmission electron microscopy (TEM) by postfixation with 1% osmium tetroxide in 0.1% Na cacodylate buffer followed by dehydration in acetone series and embedding in EMbed 812 (Electron Microscopy Sciences, Fort Washington, PA). Grids were stained with 2% uranyl acetate in 50% methanol followed by Reynold's lead citrate. Ultrastructural details of at least one arteriole per sample were recorded and photographed on a Philips 410 electron microscope. On average, 3–5 arterioles in each sample were studied.
Results
TUNEL
Six of 10 subclinically affected pigs (Nos. 1, 3, 4, 7, 8, 10) had arterioles positive for TUNEL stain (Table 2). Labeling was limited to the media of arterioles. Within the myocytes, staining was confined most often to the nucleus or what appeared to be nuclear debris. An occasional cell had diffuse cytoplasmic staining (Fig. 2). Four of six TUNEL-positive pigs (Nos. 1, 3, 8, 10) had staining in brain and ileum, and two pigs (Nos. 4, 7) had TUNEL-positive arterioles only in their ileum. The proportion of arterioles with TUNEL staining (the percentage of TUNEL-positive arterioles per section) varied substantially among individual pigs. On average, the mean percentage of TUNEL-positive arterioles tended to be higher in brain than in ileum (23% and 13%, respectively). Control pig (n = 5) brain and ileal arterioles were negative for TUNEL labeling according to our scoring criteria. An occasional section from control pigs had one TUNEL-positive arteriole.
Ileum; pig No. 11, 13 days after inoculation with STEC. Arteriole with TUNEL-positive (brown) myocytes indicative of DNA fragmentation. Diffuse cytoplasmic and nuclear staining. TUNEL method, hematoxylin counterstain. Bar = 10 µm.
Four of 10 subclinically affected pigs (Nos. 1, 3, 4, 10) had arteriolar lesions identified with HE (Table 1). Three of four pigs had lesions in both tissues, whereas the remaining pig (No. 4) had arteriolar lesions only in the brain. Changes in arterioles were similar to those described previously in this experimental model and in naturally occurring edema disease. 2 , 4 , 16 , 19 They were characterized by karyorrhectic or pyknotic nuclear debris within the tunica media (Fig. 1). Occasionally, one or several myocytes had distinctly hypereosinophilic cytoplasm and an irregular, dark, shrunken (pyknotic) nucleus. Most of the tissues with arteriolar lesions identified by HE were also positively identified by TUNEL labeling. TUNEL staining was also seen in the brain stem of one pig and ileum of three pigs that tested negative for arteriolar lesions by HE in these tissues. The proportion of TUNEL-labeled arterioles was similar in tissues with and without arteriolar lesions found by HE (20% and 16%, respectively). In summary, TUNEL demonstrated increased DNA fragmentation in pigs infected with STEC. These findings could indicate either apoptosis or necrosis.
TEM
Controls
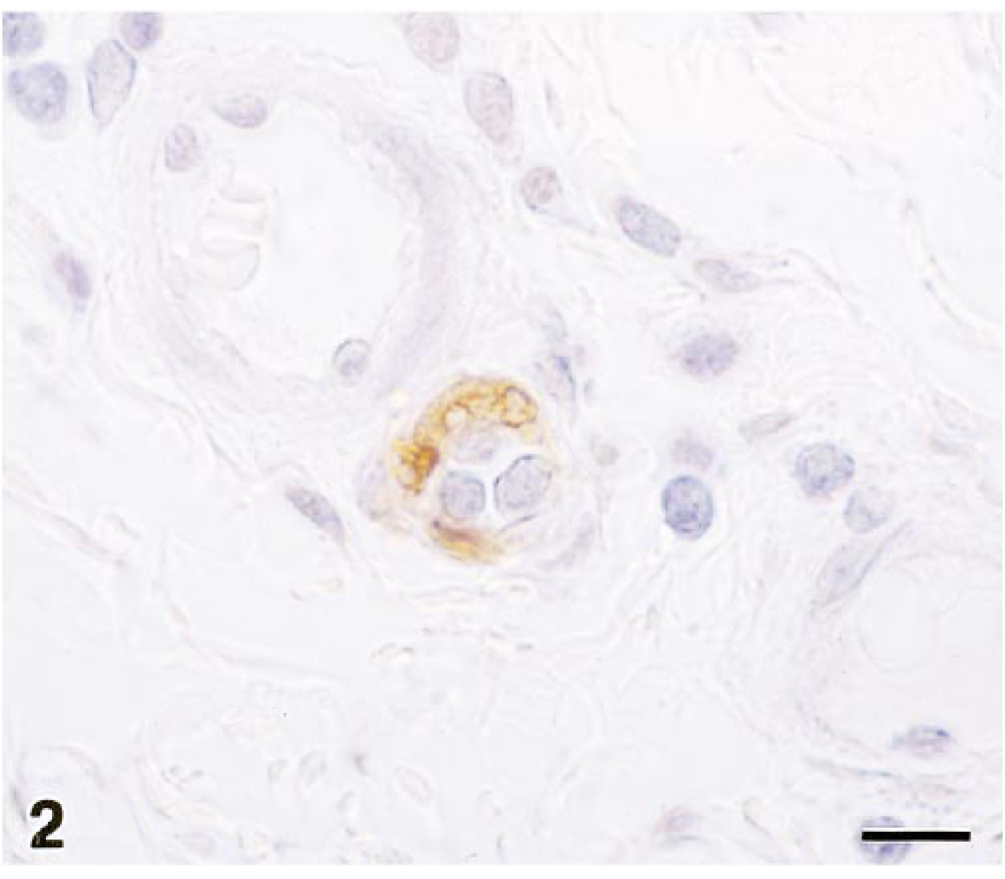
Arterioles in sections from ileum and brain stem of eight control pigs were evaluated (n = 16 arterioles in ileum; n = 11 arterioles in brain). Features of the normal arterioles included prominent marginal ridges of endothelial cells and numerous caveoli in the endothelial cells and myocytes. Arterioles of the ileum had an internal elastic membrane. Arterioles of the brain were separated from the parenchyma by Virchow-Robin space and often had granular perivascular cells in adventitia (Fig. 3). Occasionally, a few intravascular platelets and monocytes were present. One section of ileum and one section of brain (pigs nos. 21, 25) contained a blood vessel with a focal endothelial cell defect (Table 2). In one case (No. 21, ileum), it was characterized by fragmentation of several endothelial cells, denuded basement membrane, and adherent platelets. In the other case (No. 25, brain), there was a single necrotic, condensed endothelial cell devoid of organelles.
Transmission electron micrograph, brain; control pig No. 24, 12 days after inoculation with nonpathogenic E. coli. Endothelial cells of a normal arteriole have numerous marginal ridges (star) and prominent tight junctions (small arrow). Note granular perithelial cell (large arrow) and Virchow-Robin space (arrowhead). Uranyl acetate and Reynold's lead citrate. Bar = 2.3 µm.
Subclinically affected pigs
Ultrastructural changes consistent with myocyte and endothelial cell necrosis, but not apoptosis, were detected in ileal arterioles of 10/14 pigs (Nos. 26, 27, 29–31, 33, 35–37, 39) and brain arterioles of 4/8 pigs (Nos. 32, 33, 36, 37, Table 2). Three of eight pigs (Nos. 33, 36, 37) from which both tissues were collected had lesions in brain and ileum, whereas 2 pigs (Nos. 34, 38) did not have ultrastructural lesions in either tissue. Both tissues had morphologically similar changes of myocytes and endothelial cells. In the ileum, lesions of the myocytes tended to occur more commonly than lesions of endothelial cells. For example, 9/14 pigs had lesions of tunica media compared with 3/14 pigs with lesions of endothelium.
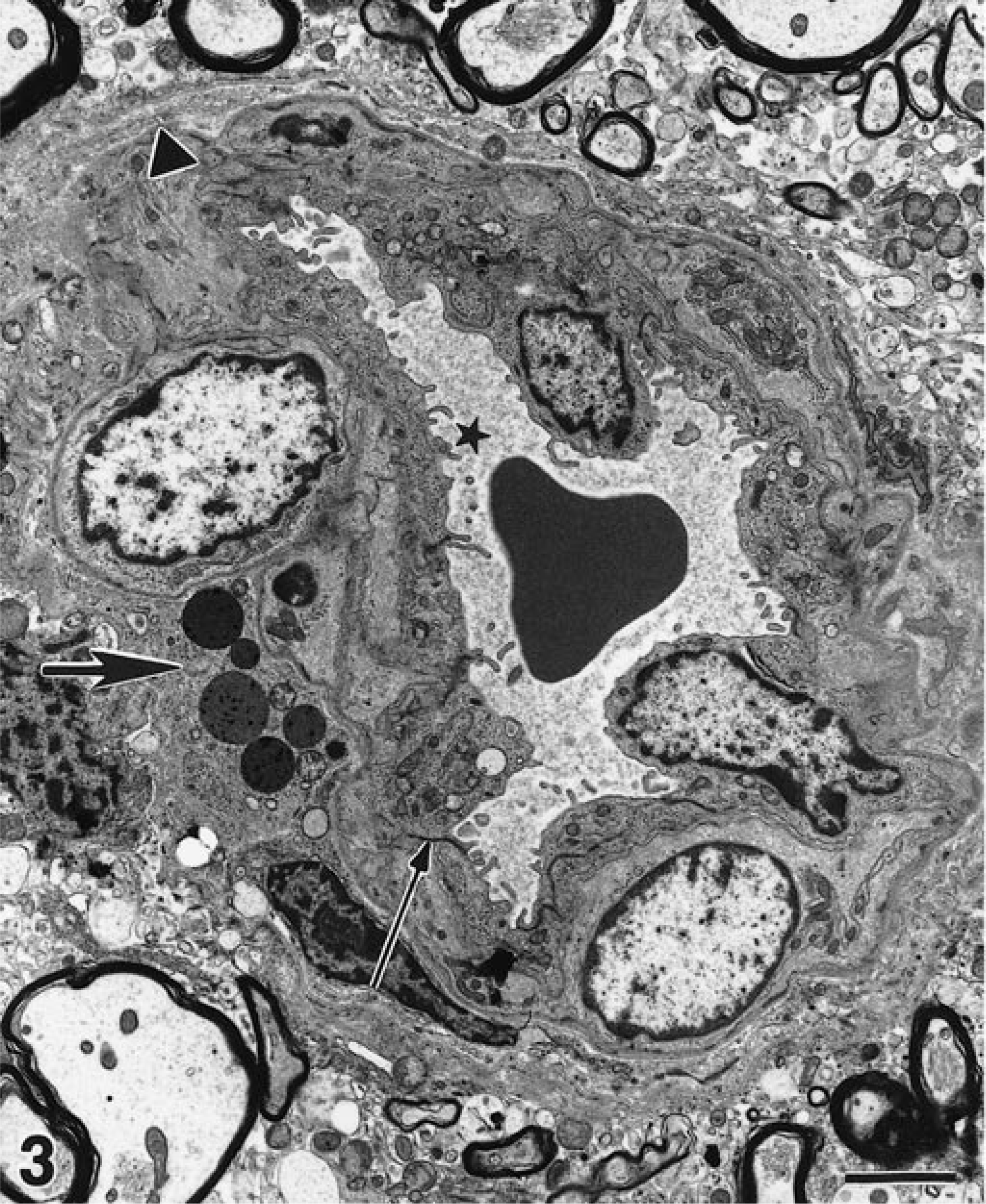
Three grades of lesions were distinguished. Focal aggregates of cellular and nuclear debris within tunica media (Fig. 4) characterized the grade 1 lesion. Cellular debris included membrane bound vacuoles and granular material indicative of lytic necrosis. The remaining myocytes were unremarkable or had limited degenerative changes, such as mild cytoplasmic pallor. There was moderate thickening of basement membranes. Some myocytes had dark, condensed cytoplasm indicative of coagulation necrosis. Grade 2 was characterized by diffuse necrosis of tunica media with cellular debris remaining between basement membranes. Grade 3 was characterized by marked expansion of the tunica media by monocytes, lymphocytes, and neutrophils and by distortion of the vascular wall by fibrin (Fig. 5). Numerous karyorrhectic and pyknotic nuclei were present within the media. An occasional cell had crescent-shaped, marginated chromatin characteristic of apoptosis (Fig. 5). However, the lack of myocyte features and the presence of mononuclear cells in the immediate vicinity indicated that such a cell was most likely a monocyte. Myocytes were replaced by a rim of cellular debris in the periphery of the vessel wall. Occasionally, erythrocytes were observed intramurally or perivascularly in the arterioles of all grades. Rarely, intravascular thrombi composed of fibrin and entrapped platelets, erythrocytes, and leukocytes were encountered. The myocyte lesion most commonly observed was grade 1 (9/14 pigs; Nos. 26, 27, 29–31, 33, 35, 36, 39). Grade 1, 2, and 3 lesions were all present in the ileum of 3/14 pigs (Nos. 26, 30, 31). We did not find apoptotic nuclear changes or apoptotic inclusion bodies in myocytes or endothelial cells of arterioles from either organ from any of the pigs.
Transmission electron micrograph; ileum; pig No. 39, subclinical 15 days after inoculation with STEC. Grade 1 arteriolar lesion with necrosis of a myocyte. Cytoplasmic debris and pyknotic nuclear fragments are contained between myocyte and basement membrane (arrows). The lumen is indicated with a star. Uranyl acetate and Reynold's lead citrate. Bar = 1.1 µm.
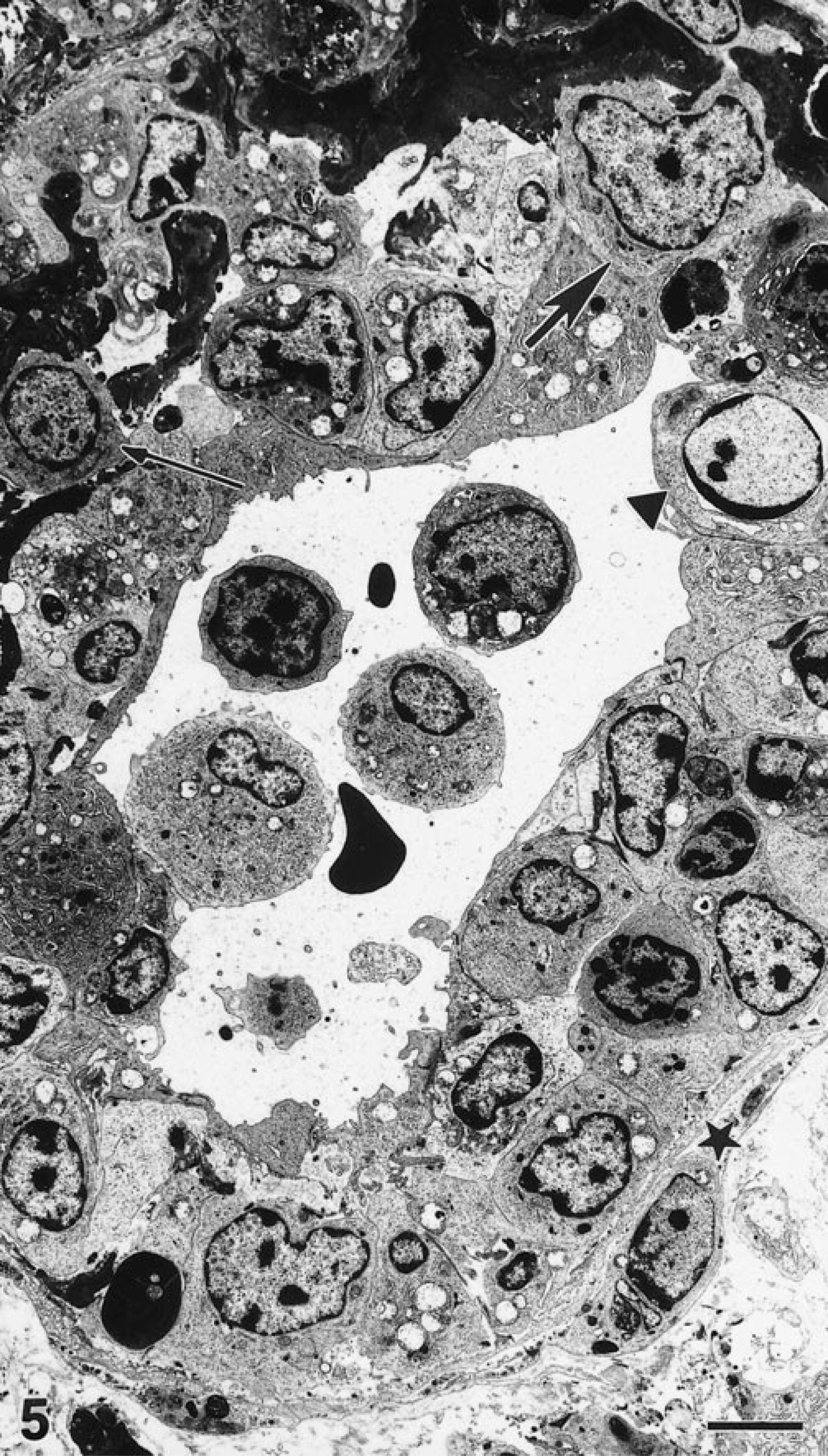
Transmission electron micrograph; ileum, pig No. 31, subclinical 14 days after inoculation with STEC. The wall of the arteriole is infiltrated by monocytes (small arrow) and macrophages (large arrow) and distorted by fibrin and edema. Some endothelial cells are necrotic. Myocytes are replaced by a rim of cellular debris (star). Note an apoptotic cell in tunica media (arrowhead). Uranyl acetate and Reynold's lead citrate. Bar = 4.1 µm.
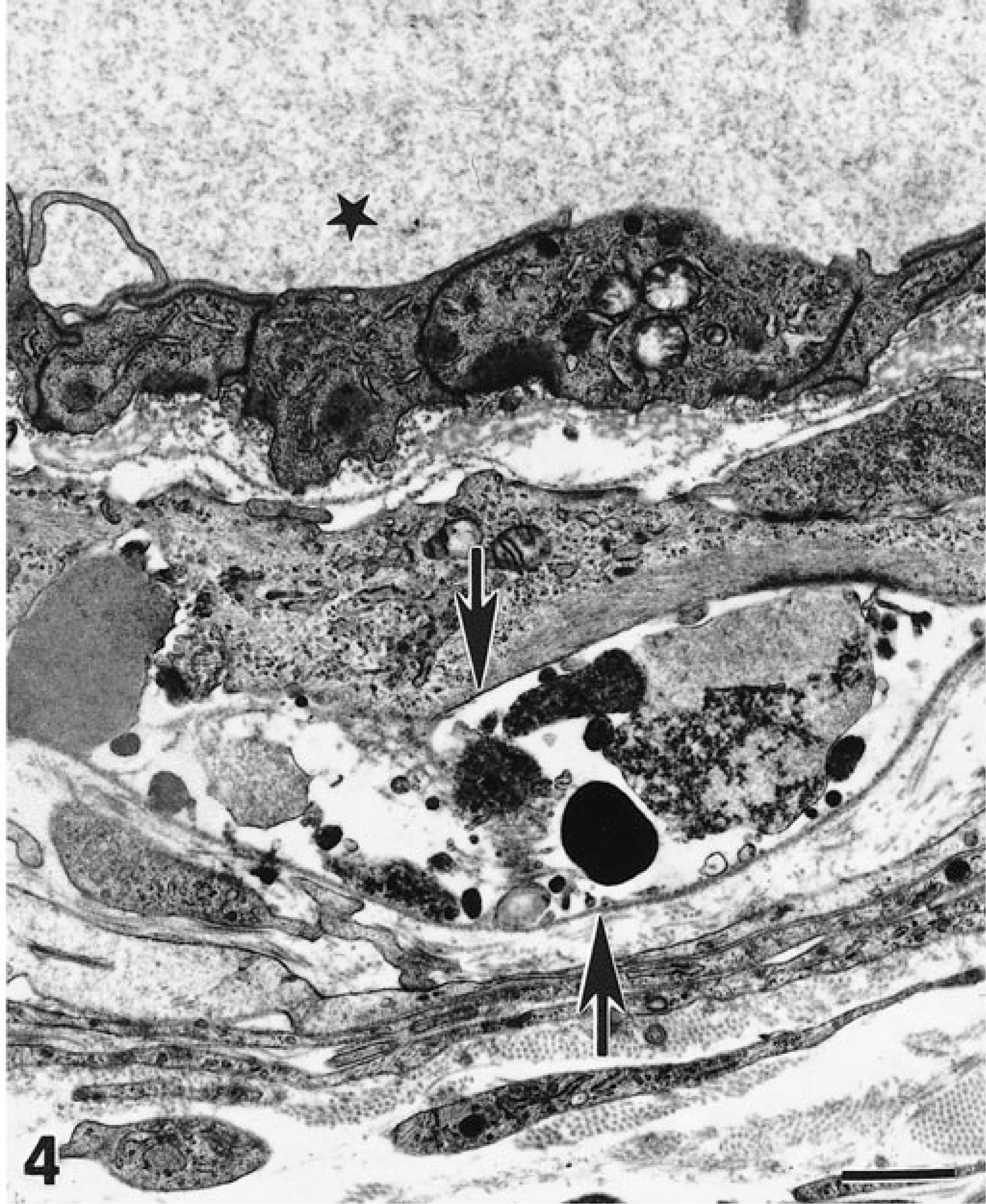
Lesions of endothelial cells were present in 6/14 subclinically affected pigs (Nos. 26, 31–33, 36, 37). Necrosis of endothelium associated with intramural vasculitis occurred in the ileum of two pigs (Nos. 26, 31). Another two pigs (Nos. 33, 36) each had a single arteriole in brain with focal endothelial lesions characterized by fragmentation of plasma membranes and condensation of cytoplasm and nucleoplasm. The myocytes directly below affected endothelial cells were unremarkable. Yet another two subclinically affected pigs (Nos. 32, 37) had advanced necrotic changes in endothelial cells of numerous arterioles in brain but not in ileum (Fig. 6). Necrotic endothelial cells were fragmented and multifocally detached from the basement membrane. Some blood vessels contained moderate amounts of intraluminal fibrillar material (fibrin) mixed with erythrocytes and reticulocytes. Platelets were present in some arterioles—adherent to intima or free in lumen.
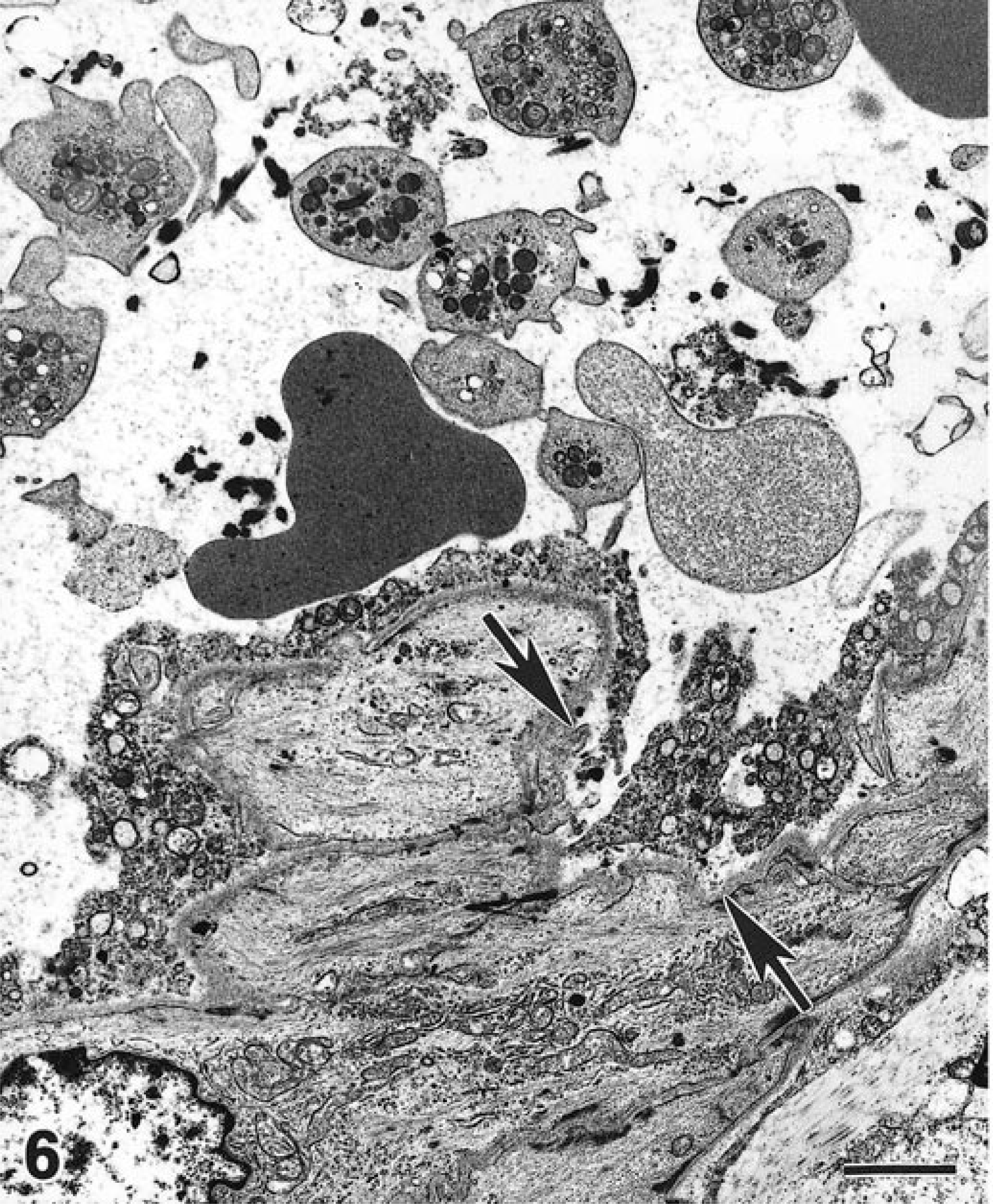
Transmission electron micrograph; brain, pig No. 32, subclinical 15 days after inoculation with STEC. The necrotic endothelial cells are fragmented and detached from the basement membrane (arrows). Platelets in the lumen are admixed with small fibrin aggregates. Uranyl acetate and Reynold's lead citrate. Bar = 1.6 µm.
Clinically affected pigs
Ultrastructural lesions were detected in the arterioles of brain and ileum from 2/3 clinically affected pigs (Nos. 41, 42, Table 2). Lesions were detected only in ileal arterioles from the remaining pig (No. 40). The severity of ultrastructural lesions and number of vessels affected varied among pigs. The lesions tended to be most frequent and severe in endothelial cells in the brain. For example, in the three pigs examined, 8/11 profiles of arterioles in brain had lesions in endothelium compared to 2/17 profiles of ileal arterioles with similar lesions. Changes in endothelial cells consisted of acute swelling (cytoplasmic rarefaction, degranulation and dilatation of rough endoplasmic reticulum, disappearance of organelles), vacuolation, and cell loss (Fig. 7). Often, these changes were accompanied by numerous platelets and few monocytes and neutrophils in the lumen or in close proximity or adherent to the intima. Occasionally, periarteriolar edema was observed in the brain. Several arterioles had focal changes within tunica media that ranged from acute swelling (Fig. 7) to necrosis of myocytes such as seen in subclinically affected pigs.
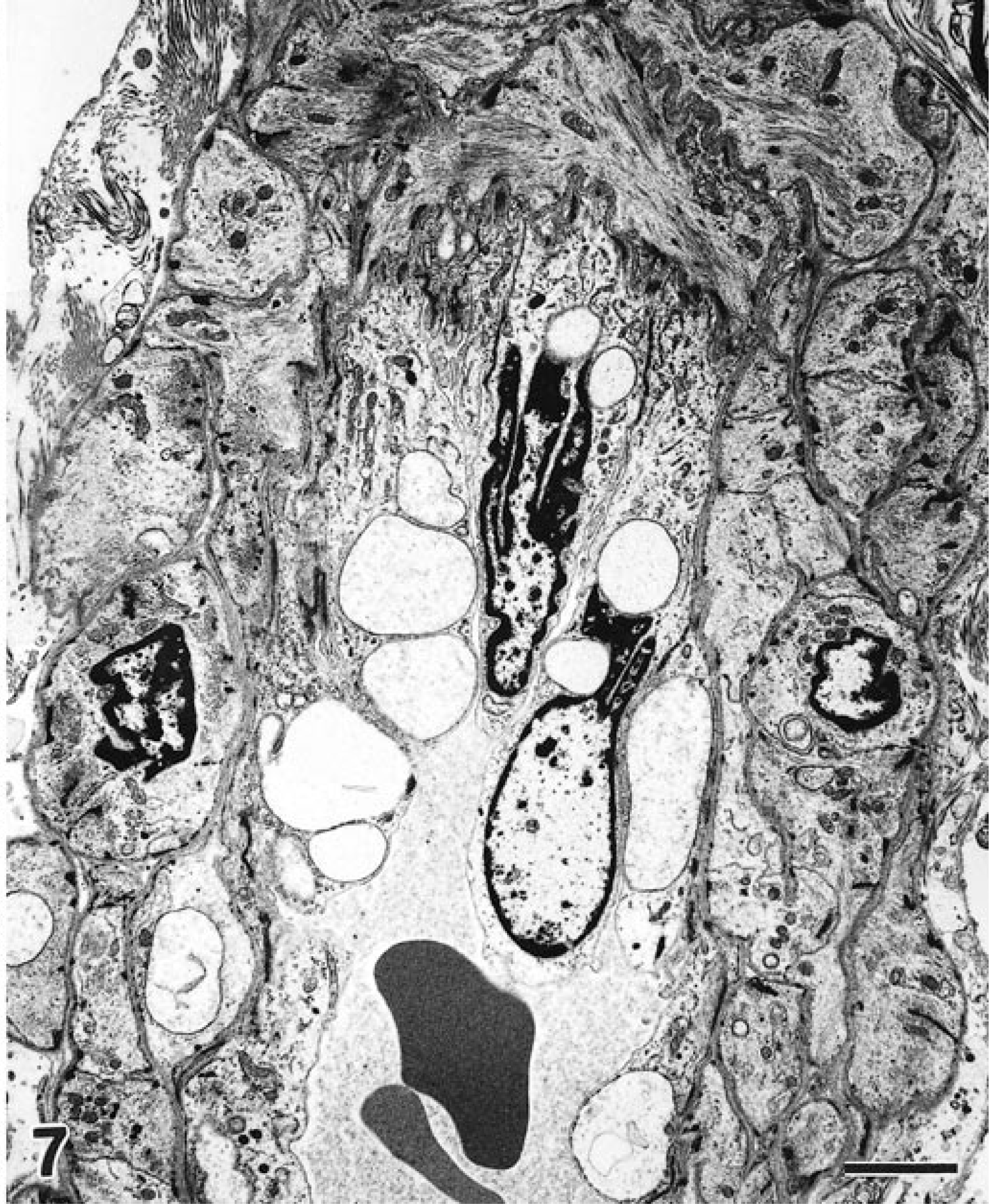
Transmission electron micrograph; brain, pig No. 41, developed ataxia 3 days after inoculation with STEC. Several endothelial cells and some myocytes are distended by clear cytoplasmic vacuoles. Uranyl acetate and Reynold's lead citrate. Bar = 2.5 µm.
Discussion
This study evaluated morphological and molecular aspects of an arteriolar lesion in STEC-infected swine. Although the TUNEL method identified DNA fragmentation suggestive of apoptosis in the arterioles of infected pigs, the ultrastructural changes were consistent with necrosis of myocytes and endothelial cells. Our inability to demonstrate ultrastructural changes characteristic of apoptosis in subclinically affected pigs led us to question whether they might be present earlier in the process or in more severe disease. Therefore, tissues from pigs with clinical disease were also examined, and ultrastructural changes characteristic of necrosis, but not apoptosis, were detected. We concluded that necrosis was the predominant change in the arterioles of both subclinically and clinically affected pigs.
Increased DNA fragmentation was detected in most of the subclinically affected animals with arteriolar lesions seen in light microscopy, indicating a direct correlation between the two parameters. DNA fragmentation was also observed in few subclinically affected pigs in the absence of light microscopic lesions, suggesting an early DNA defect. DNA fragmentation is usually a feature of apoptosis. 1 , 3 However, it was not surprising that ultrastructural changes in arterioles were more consistent with necrosis than with apoptosis. Some of the TUNEL-labeled cells had a diffuse cytoplasmic staining, indicating that DNA fragments had probably leaked out of the nucleus. This was most likely the result of increased membrane permeability, which is a feature of necrosis. Thus, the TUNEL data indirectly supported the major ultrastructural findings. TUNEL labeling in necrosis has been recognized in a number of studies. 7 , 20 , 48 Karpman et al. 15 demonstrated DNA fragmentation in necrotic renal tubular epithelial cells in STEC-infected mice. Whatever the mechanisms of the DNA fragmentation reported here are, we concluded that in this study, the predominant STEC-induced arteriolar lesion was necrosis in the endothelial cells and myocytes.
In this study, most subclinically affected pigs tended to have predominantly myocyte necrosis with limited endothelial cell necrosis. The pattern appeared to be reversed in clinical pigs that had striking endothelial cell lesions but lesser changes in myocytes. It seems probable that endothelial cell injury precedes myocyte injury and that endothelial injury is repaired quicker than myocyte injury. In vitro studies show that the cytotoxic dose and cellular response to Stx differ depending on cell type. For example, in vitro exposure to Stx inhibits protein synthesis of both renal mesangial cells (a type of myocyte) and endothelial cells. However, it causes death only in endothelial cells. 42 Therefore, it is reasonable to suggest that the myocyte response to Stx is different from the endothelial cell response.
The ultrastructural observations reported here are in agreement with those of Methiyapun et al. 31 and Drommer. 8 They described predominantly endothelial cell changes (swelling and vacuolation) in the first 2–3 days after STEC infection of swine and myocyte necrosis with intact endothelial cells 10–11 days after appearance of clinical edema disease. Moreover, endothelial cell necrosis, swelling, and regeneration have been recognized in HE sections of brain in conventional pigs with naturally occurring edema disease 19 and in gnotobiotic pigs with experimental Stx-induced disease. 9 , 10 Therefore, endothelial cell necrosis in the brain is an early event in the pathogenesis of edema disease and is probably due to the direct effect of Stx. Stx binding to arteriolar myocytes and endothelial cells of pigs has been demonstrated. 44 , 45
It is not known why the endothelial cell lesion was more common in brain than in ileum of clinically affected pigs. These findings are particularly interesting in context of the Boyd et al. 5 study of the distribution of Stx2e, Gb3, and Gb4 receptors in porcine tissue. These glycolipids were detected in several tissues but not in brain of normal pigs. Although Gb3 and Gb4 receptors have not been demonstrated in the brain, Stx2e binds to the vasculature in this tissue, and lesions occur in the brain of pigs given purified Stx2e. 5 , 26 , 44 Similarly, Stx1 damages glomerular endothelial cells in baboons, although their renal glomeruli lack Gb3. 21 , 39 These data support the notion that Stx receptor expression is increased or induced in target cells during the disease process. This may be the result of local production of tumor necrosis factor or other cytokines since increased Gb3 expression occurs in cell cultures after incubation with tumor necrosis factor. 12 , 24 , 34 , 41 Alternatively, it seems possible that tissues of pigs and baboons may contain yet unidentified, functional Stx receptors.
Lack of inflammation is a common, but inconsistent, feature of the arteriolar lesion of edema disease. The majority of infected pigs in our study had focal myocyte necrosis without signs of intramural inflammation. The reasons for the lack of inflammatory response to the necrotic cells are unknown. Immunosuppression has been reported in gnotobiotic pigs infected with STEC. 6 Furthermore, Stx1 suppresses bovine lymphocyte proliferation in vitro. 30 Gb3 receptor is also a B lymphocyte surface molecule called CD77. 27 In Burkitt's lymphoma cells, the Gb3 receptor–ligand binding results in B cell clonal deletion via apoptosis. 27 We speculate that inhibition of protein synthesis by Stx may result in decreased expression of adhesion molecules (such as selectins) or other membrane surface components necessary for recruitment of neutrophils and macrophages.
In conclusion, the predominant morphological changes in the arterioles of pigs infected with STEC were consistent with a necrotic, rather than an apoptotic, process. This confirms that the lesion of edema disease identified with HE and known for decades as vascular necrosis is indeed predominately necrotic. Increased TUNEL labeling, along with morphologic findings, indicate that DNA fragmentation may have occurred as a result of necrotic cell death.
Footnotes
Acknowledgements
This work was financed by the NIH grant AI41328. We thank Dr. N. Cornick for productive discussions and Dr. N. Cheville for advice with the interpretations of micrographs. J. Stasco and J. Olsen provided excellent assistance with electron microscopy.
