Abstract
A firm, infiltrative mass was found in the thyroid region of an adult castrated male ferret
Keywords
The ferret (
A castrated male ferret was purchased from a laboratory animal supplier (Marshall Farms, North Rose, NY) at 10 weeks of age to serve as an educational animal in a zoological collection. It was housed singly in a room with mixed species of small mammals and birds and had been vaccinated against canine distemper (Fevac-D, United Vaccine) and rabies (Imrab 1, Rhone Merieux). The diet was dry cat food daily and a small amount of moist diet twice weekly. At approximately 31 months of age, the ferret exhibited poor appetite, moderately thin body condition, and a thin haircoat bilaterally on the chest and abdominal walls and on the tail. The diet was changed to a more palatable cat food (Max Cat Adult, Nutro Products), and a skin and haircoat conditioner (Nutraderm, SmithKline Beecham) was given. The condition resolved within several weeks.
The next abnormal sign was seen at 45 months of age, when the ferret was found in a semiconscious condition. This condition was transient and resolved within a few hours without treatment. Over the next 2 months, the ferret exhibited subtle, abnormal signs of reduced activity, diminished appetite, and some loss of luster to the haircoat. When the animal was presented for diagnostic work-up, it was afebrile and weighed 962 g, a decrease from 1,350 g 1 year earlier. Physical examination revealed a prominent soft tissue mass in the right thyroid region. The zookeeper recalled hearing short bouts of choking or coughing over the past 2 months, but because of their brevity, the symptoms were not brought to the attention of the veterinary staff.
Radiographs showed a left ventral deviation of the trachea. The fine needle aspiration was not diagnostic. Serum biochemistry data showed all values, including calcium, within normal limits, with the exception of an elevated lactate dehydrogenase, which was 1,837 IU/liter (range, 241–752 IU/liter), a marginally elevated globulin of 3.1 g/dl (range, 2–2.9 g/dl), and a fasting glucose value of 65 mg/dl (range, 107–138 mg/dl). The total T4 was 1.0 µg/dl (range, 1.01–8.29 µg/dl), but the sample was of insufficient quantity for a complete thyroid profile. 5 The tentative diagnosis of thyroid neoplasia or possible cystic thyroid was made. Surgical exploration under general anesthesia was performed 2 weeks after the initial diagnostic evaluation. Euthanasia was performed following an unsuccessful effort to excise the thyroid mass in toto.
A dense, poorly circumscribed, dark red vascular mass of 3 × 2 × 2 cm was removed from the thyroid region. It was tightly adherent to all contiguous tissues, making dissection and complete removal of the abnormal tissue impossible. The left thyroid gland appeared to be relatively normal. There was prominent and rounded enlargement (5–6 mm diameter) of both adrenal glands. The pancreas was grossly normal. Other organs and tissues had no significant gross lesions, although the brain and pituitary gland were not examined. Tissues were collected from the excised thyroid mass, from the opposite thyroid, and from the adrenal gland, heart, lung, liver, spleen, pancreas, kidney, and abdominal lymph node. Samples were placed in 10% buffered formalin for fixation and histologic preparation using standard methods for light microscopy.
The thyroid mass had histologic features characteristic of an endocrine neoplasm and more consistent with a C-cell origin than a thyroid follicular epithelium origin. The mass was comprised of irregular cords, lobules, and smaller nests of neoplastic epithelium interdigitated with a dense, arborizing fibroblastic response. At one edge, the neoplastic tissue invaded an adjacent lymph node. In the well-differentiated regions, the neoplastic cells were organized in lobules or cords, generally two to four cells broad, and were arranged in palisades of columnar cells along the thin fibrovascular septa or the abutting fibrous stroma (Fig. 1). Cells within the interior of the lobules and cords were polyhedral or spindle shaped. The cells had scant to moderate amounts of pale pink vesicular cytoplasm with indistinct borders. The nuclei were round to fusiform with indented nuclear margins and pale vesicular chromatin with small nucleoli. Mitotic figures were rare (6/10 high-power fields).
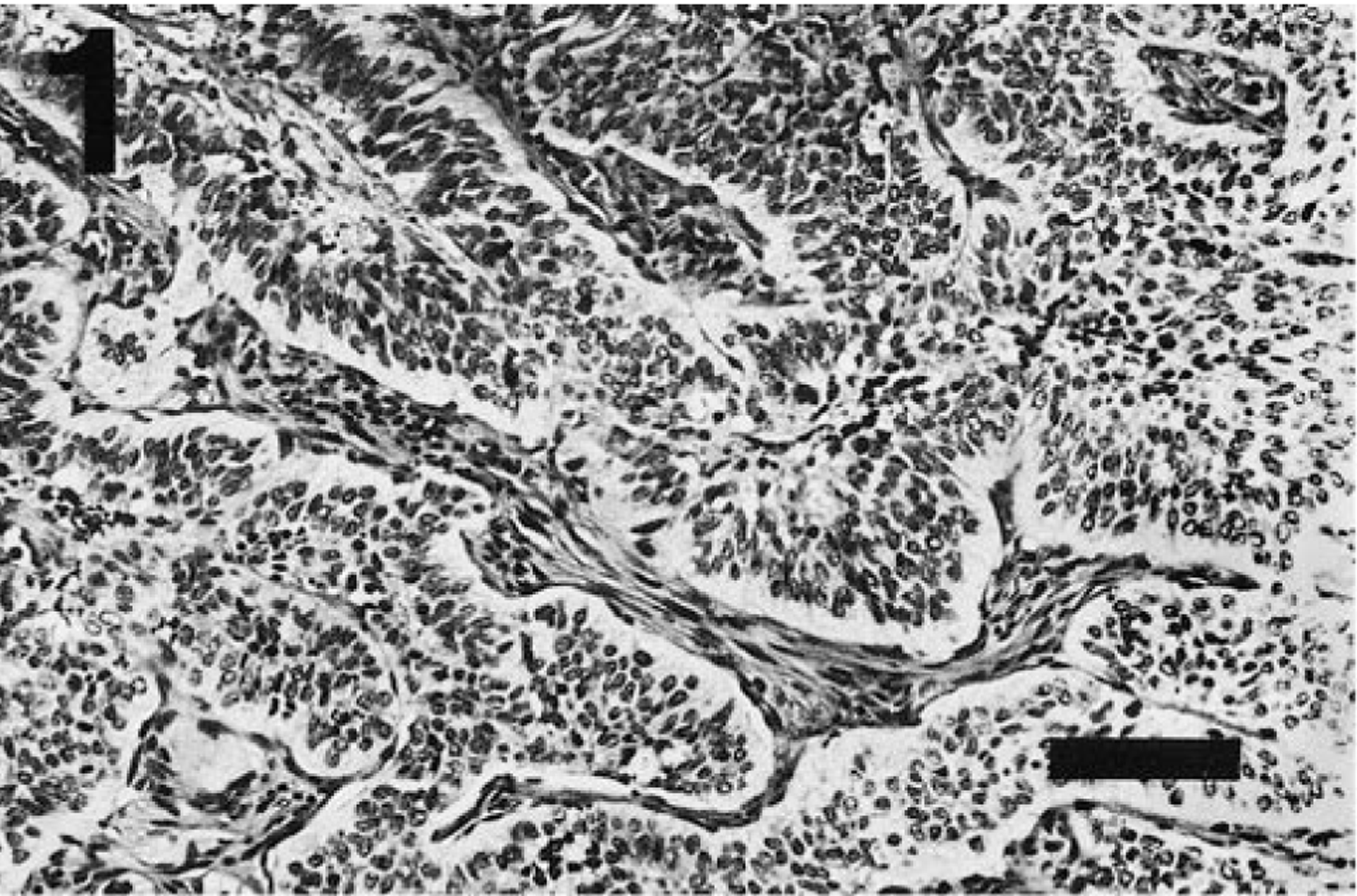
Thyroid tumor; ferret. In well-differentiated regions, the lobules and cords of neoplastic neuroendocrine cells are separated by thin septa of branching fibrovascular stroma. HE. Bar = 100 µm.
In several broad areas of the thyroid mass, the fibroblastic response was the dominant component and enclosed small nests of anaplastic cells (Fig. 2). In other areas, trabeculae of anaplastic cells also contained a lymphocytic infiltrate. Infrequent small nests of follicles lined by simple cuboidal epithelium and enclosed by an eosinophilic matrix or an intense lymphocytic infiltrate were present in one section of the mass.
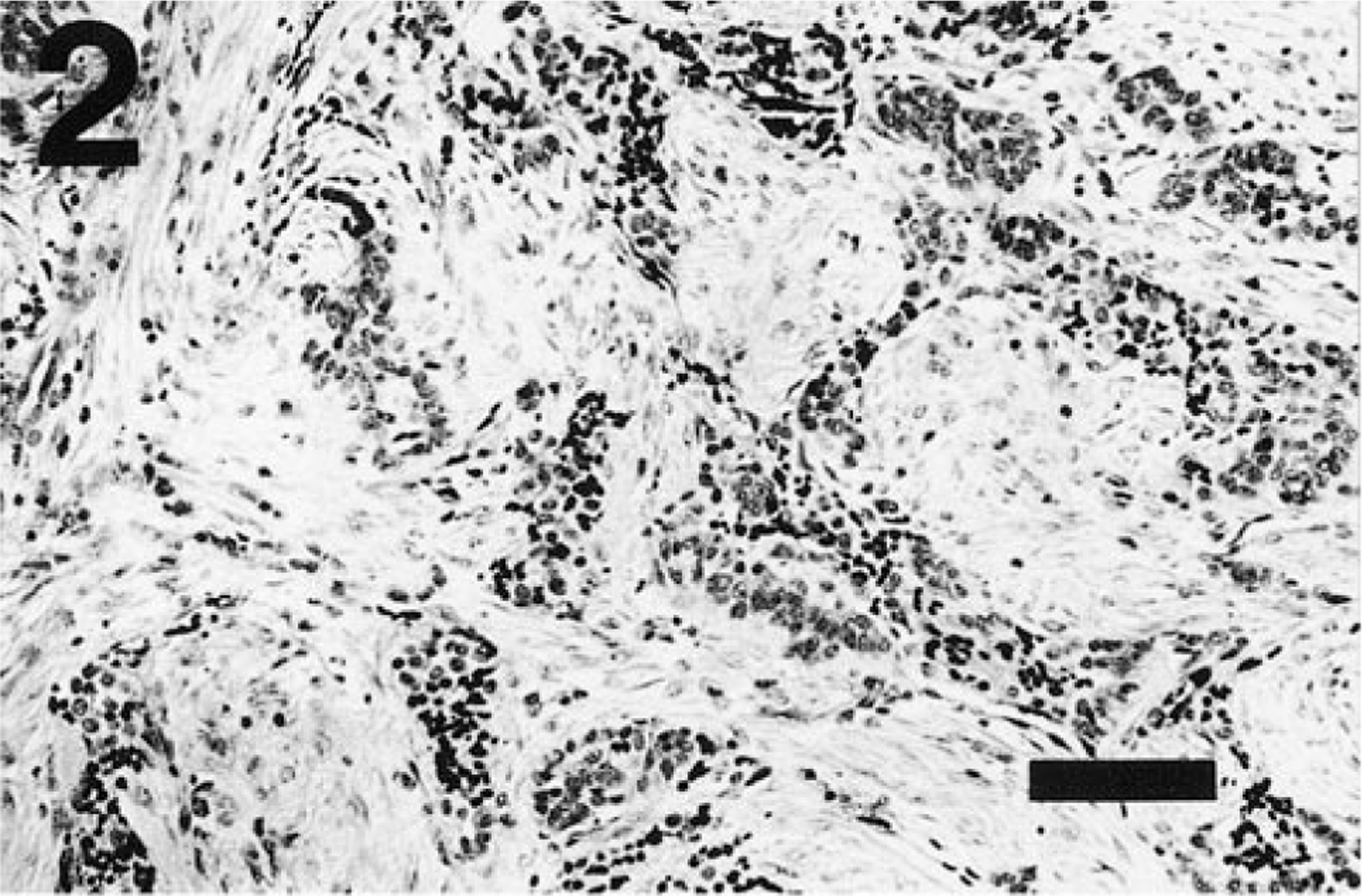
Thyroid tumor; ferret. The fibroblastic response is disproportionately high in many regions of the neoplasm. Nests of anaplastic cells are enclosed by the fibroblastic response. HE. Bar = 100 µm.
Consistent with their neuroendocrine derivation, numerous fine argyrophilic granules were present in the cytoplasm of most neoplastic cells in the thyroid tumor when stained with a modified Grimelius procedure (Fig. 3). 12 Calcitonin immunostain using human anti-calcitonin reagents was unsuccessful (C. Capen, unpublished data). Failure was likely due to either lack of cross-species reactivity of human reagents to ferret antigens or destruction of antigenic sites by prolonged formalin preservation.
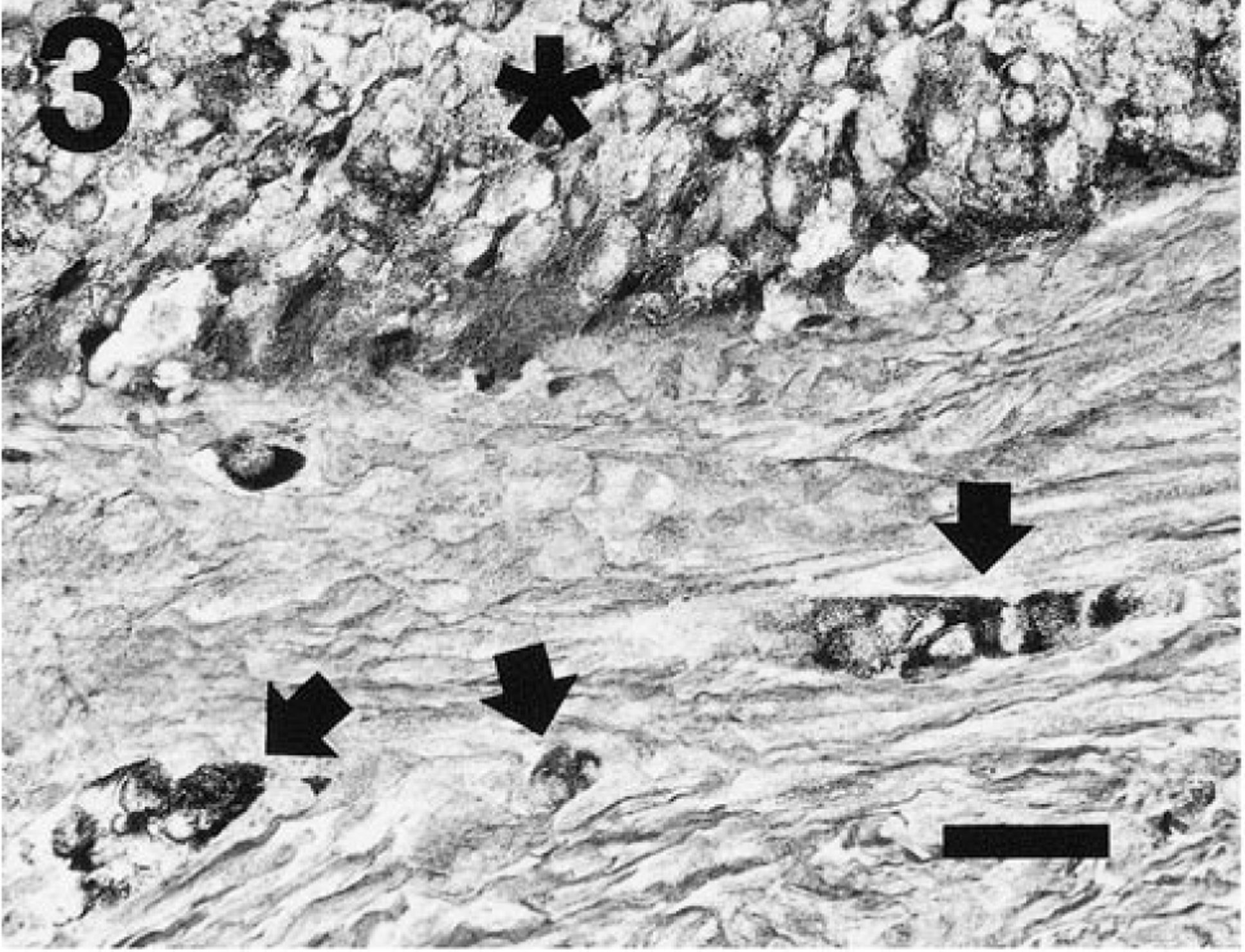
Thyroid tumor; ferret. The cords of neoplastic cells (∗) contain fine argyrophilic, cytoplasmic granules. Small aggregates of argyrophilic cells (arrows) are also isolated in the enclosing stroma. Grimelius. Bar = 30 µm.
The tumor was evaluated for the presence of amyloid by staining with Congo red and examination under polarized light. Multifocal birefringence with an apple-green hue was associated primarily with the fibrous stroma, which divided the mass and encircled neoplastic cells. This pattern of birefringence overlapped but was distinct from the intense white birefringence of the dense collagen stroma. This pattern of collagen-associated birefringence was also present in sections stained with hematoxylin and eosin.
The nonneoplastic left thyroid tissue contained an intense, diffuse interstitial infiltrate comprised chiefly of lymphocytes. The glandular parenchyma was comprised of follicles containing colloid and sloughed epithelial cells and of aggregates of C cells.
Among the tissues presented for histologic evaluation, several extrathyroid sites of endocrine neoplasia were observed. The pancreas contained a discrete, nonencapsulated tumor comprised of mildly anaplastic islet cells, which projected short distances into the adjacent exocrine tissue. The cells were organized in closely packed cords separated by limited fibrovascular stroma and had eosinophilic cytoplasm and ovoid basophilic nuclei. Two small nonencapsulated metastatic foci of endocrine lineage were also observed in the hepatic parenchyma and were comprised of closely packed cords of polyhedral cells with pale vesicular cytoplasm and round basophilic nuclei. The hepatic foci histologically resembled the pancreatic islet tumor and well-differentiated portions of the thyroid carcinoma. The origin of the metastases was not wholly established; however, the tendency of pancreatic islet tumors to infiltrate the liver suggests this tissue as the primary source.
One adrenal gland was available for histologic evaluation. This gland contained multiple neoplastic foci, which obscured the normal adrenal architecture (Fig. 4). Two large, expansile, sharply demarcated foci were comprised of closely packed nests of small polyhedral cells, which had lightly basophilic cytoplasm and round nuclei of moderate size with indented margins, marginated and finely granular chromatin, and prominent nucleoli. These cells did not stain with a modified Grimelius procedure. 12 These foci resembled adrenocortical adenomas with small cell morphology, as previously described in ferrets. 5 An adjacent, histologically distinct focus was partially displaced by the adrenocortical adenoma and was comprised of polyhedral to elongate cells arranged in sheets and smaller closely packed lobules separated by branching fibrous septa. The cells had abundant granular or finely vacuolated, densely eosinophilic cytoplasm and small, round basophilic nuclei with smudged homogeneous chromatin staining. This focus contained clusters of densely argyrophilic cells (Fig. 5) separated by cells with variable expression of fine argyrophilic cytoplasmic granules (Fig. 6), suggestive of adrenomedullary origin (i.e., pheochromocytoma).

Adrenal tumors; ferret. A dark staining sheet of small neoplastic cells (right) expands and displaces another pale staining multilobulated, neoplastic focus (pale region on left). HE. Bar = 350 µm.
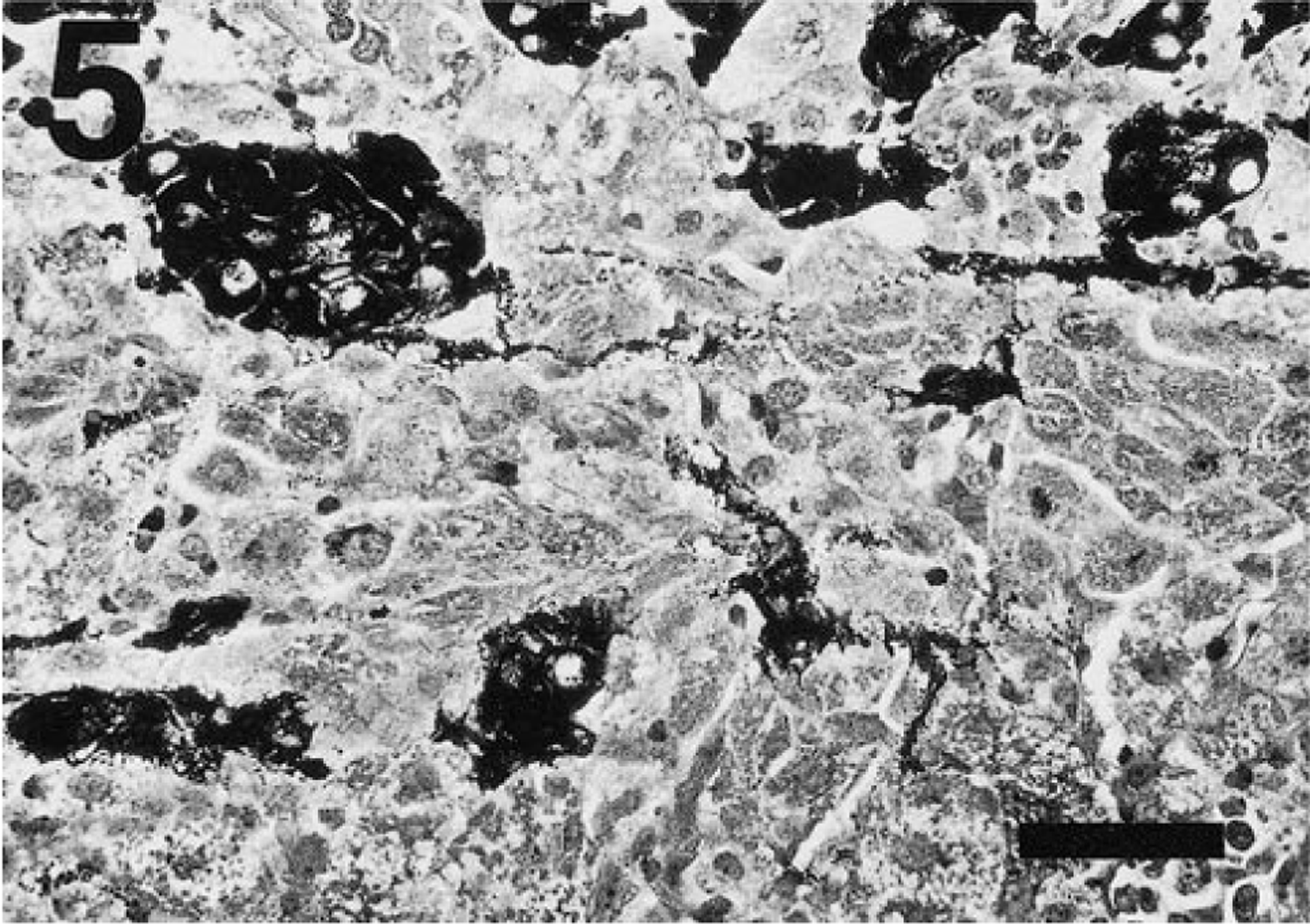
Adrenal tumor; ferret. Densely argyrophilic cells, resembling adrenal medullary cells are present in small nests among similar cells with fine granule content. Grimelius. Bar = 40 µm.
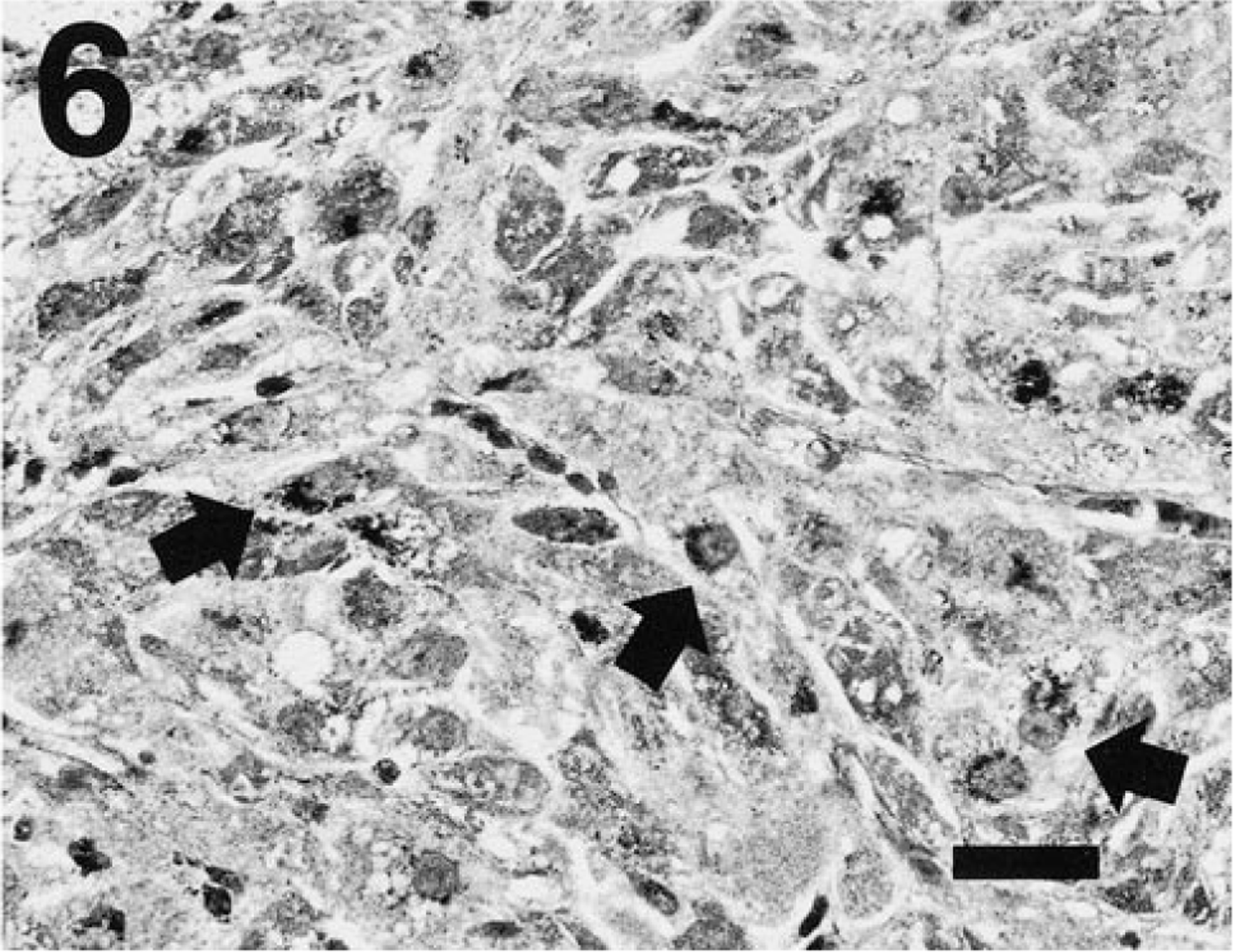
Adrenal tumor; ferret. One neoplastic focus contains sheets of polyhedral cells with variable expression of fine argyrophilic granules, in contrast to the densely stained cells in Fig. 5. Cells with notable perinuclear condensation of the argyrophilic granules (arrows) are present among other cells with more sparse granule density. Grimelius. Bar = 25 µm.
No pathologic changes were observed in other tissues examined histologically, with the exception of chronic bronchitis and bronchiolitis in the lung characterized by mononuclear cell infiltration around airways, peribronchiolar smooth muscle cell hyperplasia, and epithelial hyperplasia.
Endocrine neoplasms are the most commonly observed tumors in ferrets. In a recent review of a multi-institution database, tumors of endocrine origin were identified in 39.7% of ferrets affected with neoplasms. 8 Although endocrine tissues, such as the adrenal cortex and the pancreatic islets, are the most frequent origins of ferret neoplasms, this C-cell carcinoma, reported in ferrets for the first time, occurred in tandem with other extrathyroid endocrine neoplasms. The histologic features of the thyroid neoplasm were distinctive and consistent with a C-cell origin. The C-cell lineage was further confirmed and differentiated from thyroid follicular cells by the intense argyrophilia of tumor cells revealed with Grimelius stain. The histologic pattern of this C-cell carcinoma most closely resembled the clear cell pattern described in a survey of C-cell carcinomas in dogs. 14 A pattern of Congo red staining and birefringence centered on dense collagen stroma was also observed within the tumor, similar to that described in a majority of the canine tumors. 14 The presence of amyloid in neoplastic tissue is also a hallmark feature of medullary thyroid carcinoma in humans. 3 Care was taken to discriminate the amyloid-associated pattern of birefringence from the strong, nonspecific birefringence of the dense collagen stroma. The diagnosis of C-cell carcinoma is consistent with a lack of elevated serum thyroxine in this ferret. The nonneoplastic thyroid with the lymphocytic infiltrate suggests an immune system–mediated insult that might further reduce thyroid hormone levels. Although some dogs with C-cell carcinoma may have hypocalcemia, many dogs with this type of tumor, like the ferret in this case, have normal serum calcium levels. 2
C-cell carcinomas have been frequently observed in aged bulls and rats, in contrast to other domestic and laboratory animal species. Although early reports suggested that C-cell tumors are rare in dogs, in a recent survey of thyroid tumors in dogs, 12 of 38 (36%) were C-cell carcinomas; this is a much higher frequency of this tumor type than previously reported in dogs. 2 In bulls, pheochromocytomas are also often observed in tandem with C-cell carcinomas. 1 In a recent review of Fischer 344 rats used in long-term carcinogenicity studies, pheochromocytomas and C-cell carcinomas were independently observed in 31.9% and 14.8%, respectively, of male rats used in feeding trials. 6 In a recently published retrospective study, six of 72 horses with endocrine tumors had concurrent C-cell tumors and pheochromocytomas. 4 Although adrenocortical neoplasms are exceptionally common in ferrets, pheochromocytomas are rarely reported. 8 Consequently, the presented case is further distinguished by the concurrence of a pheochromocytoma and a C-cell carcinoma.
Although the range of clinical signs in ferrets with multiple endocrine neoplasms can be quite heterogeneous, signs due to one or more prominent lesions can predominate over others. The principal clinical features of this case, i.e., gradual weight loss, partial anorexia, reduced activity, and collapse, are probably related to the islet cell tumor and not to the C-cell carcinoma. The fasting glucose level of 65 mg/dl is highly suggestive of hypoglycemia due to excessive insulin secretion by the tumor. 7 , 9 The bilateral hair loss noted prior to the onset of signs of hypoglycemia is consistent with elevated adrenal steroids observed in ferrets with adrenal tumors. 8 Recurring and progressive hair loss are prominent features of hyperadrenocorticism in the ferret. Estrogenic steroids (e.g., 17 β-estradiol) synthesized in the adrenal tumor have been implicated in adrenal tumor–associated alopecia in ferrets.
The coincidence of medullary thyroid neoplasms and other endocrine tumors is also observed in several heritable syndromes of human disease. There are three main categories of multiple endocrine neoplasia (MEN) syndromes in humans, of which two, MEN-IIA and MEN-IIB, have a high incidence of C-cell neoplasia accompanied by pheochromocytomas.
3
The MEN-IIA and -IIB syndromes are associated with mutations of the
The third category of MEN syndrome in humans, MEN-I, is associated with a unique gene locus (
The observed similarities between the manifested neoplasms in this ferret and MEN syndromes in humans are tantalizing but incomplete. The presence or absence of other MEN-associated neuroendocrine lesions, such as pituitary tumor, parathyroid cell adenoma, or mucosal neuromas, which would support further MEN subclassification, could not be confirmed in this ferret. Likewise, the presence of a
Footnotes
Acknowledgements
We acknowledge the assistance of Dr. C. Capen in the histologic evaluation of the C-cell carcinoma. This work was supported in part by NIH grant RR07036.
