Abstract
Eighty 3-week-old crossbred pigs were randomly assigned to six groups (13–14 pigs/group). Group 1 pigs served as uninoculated controls, group 2 pigs were inoculated intranasally (IN) with Streptococcus suis serotype 2, group 3 pigs were inoculated IN with a modified live porcine reproductive and respiratory syndrome virus (PRRSV) vaccine, group 4 pigs were inoculated IN with the same vaccine and with S. suis, group 5 pigs were inoculated IN with VR-2385 (a high-virulence strain of PRRSV), and group 6 pigs were inoculated IN with VR-2385 and S. suis. Pigs exposed to both PRRSV and S. suis were inoculated with PRRSV 7 days prior to S. suis inoculation. The pigs were 26 days old when inoculated with S. suis. Respiratory disease was significantly more severe in groups 5 and 6. Mortality rate was the highest in group 6 (87.5%). This rate was significantly higher than that observed in all other groups except group 4 (37.5%). The mortality rate in group 2, inoculated with S. suis alone, was 14.3%. No pigs from groups 1, 3, or 5 died prior to the scheduled necropsies at 10 and 28 days postinoculation with PRRSV (DPI). To study the effect of PRRSV and/or S. suis on pulmonary clearance by pulmonary intravascular macrophages, six pigs from each group were intravenously infused with 3% copper phthalocyanine tetrasulfonic acid in saline prior to necropsy at 10 DPI. Mean copper levels in the lungs of pigs in groups 2, 5, and 6 were significantly lower than those in control pigs. The mean percentage of lung tissue grossly affected by pneumonia at 10 DPI was 0%, 1%, 0%, 3%, 64%, and 62% for groups 1–6, respectively. Both gross and microscopic interstitial pneumonia lesions were significantly more severe in the VR2385-inoculated groups (5 and 6). PRRSV was isolated from bronchoalveolar lavage fluid collected at necropsy from 100% of the pigs in groups 5 and 6, 71.4% of pigs in group 4, 38.5% of pigs in group 3, and none of the pigs in groups 1 or 2. Streptococcus suis serotype 2 was cultured from the internal tissues of 7.7%, 28.6%, and 78.6% of the pigs in groups 2, 4, and 6, respectively. Streptococcus suis serotype 2 was isolated from whole blood at necropsy from 7.7%, 35.7%, and 78.6% of pigs in groups 2, 4, and 6, respectively. Significantly more pigs in group 6 had S. suis isolated from whole blood and internal tissues. In summary, both high-virulence PRRSV and S. suis decreased copper clearance, and the incidence of isolation of S. suis and PRRSV was higher in dually inoculated pigs. PRRSV-induced suppression of pulmonary intravascular macrophage function may in part explain PRRSV-associated increased susceptibility to S. suis infection.
Keywords
Since it was first recognized in the USA in 1987, 13 porcine reproductive and respiratory syndrome (PRRS) has become endemic and now is an important cause of pneumonia in 3–24-week-old pigs in swine-producing countries around the world. Several research groups have demonstrated increased severity of clinical disease in animals coinfected with PRRS virus (PRRSV) and different bacteria, including Streptococcus suis, 7 Haemophilus parasuis, 23 Salmonella choleraesuis, 33 and Mycoplasma hyopneumoniae. 27 In other experiments, a synergistic effect could not be demonstrated between PRRSV and bacterial infections. 1 , 3 , 19 Differences in experimental design, PRRSV strain, 9 S. suis strains, and host genetic factors 11 may influence the outcome of coinfection studies. For example, exposure of PRRSV-infected pigs to different S. suis strains resulted in different clinical manifestations and severity of disease, indicating that the strain of bacteria can influence the clinical disease induced in a coinfection model. 7 However, the mechanisms of the interaction between PRRSV and bacterial infections remains unclear.
The pig lung is an important site of clearance of blood-borne bacteria. 4 , 5 Pulmonary intravascular macrophages (PIMs) are thought to be very important in clearance of circulating bacteria in pigs 2 and calves. 32 We recently demonstrated detrimental effects of PRRSV on porcine PIMs both in vitro and in vivo. 28–30 PRRSV infection adversely affected uptake of copper by PIMs, and the effect differed among PRRSV strains. 28 Therefore, we hypothesized that PRRSV-induced PIM damage has a detrimental effect on removal of bacteria from the bloodstream, which results in increased incidence and severity of S. suis-associated clinical disease. The objective of this study was to further investigate the pathogenesis of PRRSV-induced increased susceptibility to S. suis-associated disease.
Materials and Methods
Experimental design
The protocol used in this study was approved by the Iowa State University Institutional Committee on Animal Care and Use. Eighty 3-week-old crossbred pigs from a herd free of PRRSV and S. suis serotype 2 were weaned at 11–12 days and moved to the research facility. The source herd was serologically monitored monthly for PRRSV and had no S. suis-associated disease or isolation of S. suis serotype 2 from pigs in the last 5 years. The pigs were randomly assigned to six groups of 13 or 14 pigs each. Pigs in groups 1 and 2 were inoculated intranasally at 19 days of age with 2 ml of sterile minimum essential medium (MEM, GIBCO Laboratories, Grand Island, NY). Pigs from groups 3 and 4 were inoculated intranasally with 2 ml of a modified live virus vaccine RespPRRS/Repro™ (NOBL Laboratories, Sioux Center, IA). Pigs from groups 5 and 6 were inoculated intranasally with 2 ml of a high-virulence PRRSV isolate, VR-2385 (Dr. P. S. Paul, Veterinary Medical Research Institute, Iowa State University, Ames, IA) on day 0 of the study. At 26 days of age (study day 7), 2 ml of S. suis serotype 2 (isolate ISU VDL 40634/94) was inoculated intranasally into uninfected pigs (group 2) or pigs that had been inoculated 7 days earlier with RespPRRS/Repro™ (group 4) or VR-2385 (group 6). RespPRRS/Repro™ vaccine is licensed by the US Department of Agriculture for intramuscular use. In this experiment, it was used in an extra-label fashion (intranasally) to represent a low-virulence PRRSV strain, as previously described. 28 The characteristics of isolate VR-2385 have been described previously. 1–11, 14 , 28 Six randomly selected pigs from each group were necropsied on day 10 of the study. The remaining pigs were euthanatized when they became moribund or on day 28 of the study (Table 1).
Experimental design of porcine reproductive and respiratory syndrome virus (PRRSV) and Streptococcus suis serotype 2 coinfection model.

∗ Six pigs from each group were randomly selected prior to inoculation and were necropsied at 10 days postinoculation (DPI). The remainder of the pigs were necropsied when exhibiting evidence of meningitis or when recumbent due to S. suis-associated lameness.
† PRRSV vaccine administered at 0 DPI, and S. suis administered at 7 DPI.
‡ No. of pigs and days postinoculation when they were found dead or euthanized due to disease.
Inocula preparation
The commercial RespPRRS/Repro™ high-passage modified-live vaccine (Lot 403B) was used as the low-virulence PRRSV strain for this experiment. PRRSV isolate VR-2385 (biologically cloned by three rounds of plaque purification in continuous cell line ATCC CRL11171) at passage 6 was used as the high-virulence isolate. The vaccine and the VR-2385 stock virus were end point titrated by observation of cytopathic effect (CPE) in serial 10-fold dilutions in 96-well microtiter plates seeded with MARC-145 cells (Dr. Kelly M. Lager, National Animal Disease Center, Ames, IA). The titers of RespPRRS/Repro™ and VR-2385 were calculated 21 as 104.99 and 104.47 TCID50/2 ml, respectively. The RespPRRS/Repro™ vaccine, the VR-2385 virus stock, and the S. suis brain homogenate stock were tested by polymerase chain reaction and were negative for the presence of type 2 porcine circovirus 15 nucleic acids.
Streptococcus suis serotype 2 isolate ISU VDL 40634/94 was originally cultured from a naturally occurring field case of meningitis in a pig submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL) in 1994. It was passed by intravenous inoculation into a 3-week-old pig that was free of PRRSV and S. suis serotype 2. The pig was euthanatized when it showed signs of central nervous system (CNS) disease. The infected brain and meninges were aseptically collected, and a 10% homogenate of the brain and meninges was prepared in Todd-Hewitt broth (Difco, Detroit, MI) with 20% glycerol using a Stomacher 80 lab blender (Seward, London, UK). Aliquots (1.5 ml) of the homogenate were stored frozen at −70 C. The homogenate served as the stock material for preparing the experimental bacterial challenge inoculum. The challenge inoculum for this study was prepared by growing an aliquot of the brain/meninges homogenate on bovine blood agar plates (BAPs) overnight at 37 C and then in Todd-Hewitt broth with 5% fetal calf serum for 7.25 hours at 37 C. The bacteria and growth medium were diluted 1:10 with Hanks' balanced salt solution and administered intranasally to the pigs as a bolus using a syringe with an 18-ga, 1.5-in. catheter sheath attached. Bacterial inoculum contained 108.30 colony-forming units (cfu)/2 ml dose. The inoculum was checked for purity by streaking onto a BAP and incubating at 37 C in 5% CO2 in air. The stock bacterium was confirmed to be S. suis based on these test results: catalase negative, Gram stain positive, Voges-Proskauer reaction negative, no growth in 6.5% NaCl, and positive for production of amylase. 6 , 12 The S. suis isolate was serotyped using antiserum supplied to the ISU-VDL by Dr. Robert Higgins (Montreal, Canada). 12 The stock bacteria were coagglutination positive for S. suis serotype 2.
Clinical evaluation
Rectal temperatures and clinical respiratory disease scores, ranging from 0 to 6 (0 = normal, 6 = severe), 9 were recorded daily for days 0–11 and every other day for days 12–28. Clinical observations including evidence of CNS disease, swollen joints, and lameness (0 = normal, 1 = mild, 2 = moderate, 3 = severe) were recorded. Clinical scoring was done in an unblinded manner by a team of two people. The number of days with fever (rectal temperature > 40 C) was determined. For respiratory, CNS, joint, and lameness evaluations, the daily scores were added and then divided by the number of days observed. For all parameters, scores were generated representing the first 10 days and days 11–28 after PRRSV inoculation. For animals that died on or before day 10 (n = 3), a score was calculated from the data collected by adding the scores and dividing by the number of observations. Scores for days 11–28 were not calculated for animals that died (n = 11) during the study because of the large number of missing daily values.
Copper particle administration and necropsy
Six pigs from each group were randomly selected prior to the beginning of the study for necropsy on day 10. Pigs were anesthesized at 10 days postinoculation (DPI), and copper was administered intravenously as previously described. 28 To assess pulmonary clearance of copper particles, pigs were euthanatized by an intravenous overdose of sodium pentobarbital (Schering-Plough Animal Health Corp., Kenilworth, NJ) 30 minutes after copper administration, and a 3- × 3- × 3-cm cube from the center of the left caudal lung lobe was collected for copper analysis.
Complete necropsies were performed on all pigs. An estimated percentage of the lung with grossly visible pneumonia was recorded for each pig based on a previously described scoring system. 9 The scoring system is based on the approximate volume that each lung lobe contributes to the entire lung: the right cranial lobe, right middle lobe, cranial part of the left cranial lobe, and the caudal part of the left cranial lobe each contribute 10% of the total lung volume, the accessory lobe contributes 5%, and the right and left caudal lobes each contribute 27.5%. Other lesions such as pleuritis were noted separately and were not included in gross lung lesion scores. Sections for histopathologic examination were taken from nasal turbinate, lung (seven sections), 9 heart, brain, lymph nodes, tonsil, thymus, liver, spleen, joints, and kidney. Immunohistochemical detection for PRRSV was performed on lungs as previously described. 8 Immunohistochemical detection for type 2 porcine circovirus antigens in formalin-fixed, paraffin-embedded blocks 24 using hyperimmune serum from rabbits was done on lungs from all pigs necropsied at 10 DPI (six pigs per group). The upper respiratory tract (nasal cavity, trachea), lungs, mandibular lymph nodes, pericardium, peritoneum, pleura, spleen, liver, brain, meninges, and joints were swabbed and cultured for S. suis serotype 2. A blood sample was collected from the cranial vena cava from each animal immediately prior to euthanasia and was cultured for S. suis.
Copper analysis
Copper analysis in homogenized lungs was performed at the ISU-VDL as previously described. 26 , 28 Samples were analyzed in triplicate.
Serology and virus isolation
Blood was collected on arrival at 11–12 days of age and from all surviving pigs on days 0, 7, 14, 21, and 28 of the experiment and at the time of necropsy. Serum antibodies to PRRSV (cutoff value of sample:positive 0.4) were detected using a PRRSV enzyme-linked immunosorbent assay (Herd Check, IDEXX Laboratories, Westbrook, MA). Bronchoalveolar lavage (BAL) was performed aseptically at necropsy with 50 ml lavage fluid consisting of MEM containing antibiotics (9 µg gentamycin/ml, 100 U/ml penicillin G, 100 µg/ml streptomycin). Lavage fluid was gently dispensed into and aspirated from the right cranial, middle, and caudal lung lobes. The BAL fluid was kept at −70 C until PRRSV isolation was attempted on a confluent monolayer of MARC-145 cells. 28 Viral CPE was confirmed to be associated with PRRSV replication by indirect immunofluorescence assay (IFA). 14 Monolayers were stained with anti-PRRSV monoclonal antibody SDOW-17 16 and fluoroisothiocyanate-conjugated anti-mouse IgG (Sigma, St. Louis, MO) and then viewed with a fluorescence microscope for evidence of specific viral antigens. If CPE was not observed within 7 days, the cultures were frozen and thawed, blindly passed two more times, and then checked for positivity by IFA.
S. suis isolation
Five milliliters of whole blood was collected in ethylenediaminetetraacetic acid tubes on days 7 (prior to S. suis inoculation), 8, and 9 DPI from pigs scheduled for necropsy on day 10 and on the day of necropsy from the remaining pigs. One hundred microliters of the whole blood was spread on BAPs, and 1 ml was put into 19 ml of Todd-Hewitt broth. Swabs of tissues obtained at necropsy were cultured on BAPs. All cultures were incubated at 37 C in 5% CO2 in air for 24–48 hours. Alpha hemolytic streptococcus-like colonies were tested for growth in 6.5% NaCl and production of amylase. 6 Representative colonies that did not grow in NaCl and were positive for production of amylase were checked by coagglutination to determine if they were serotype 2. 12
Statistical analysis
Mortality and organism isolation data were analyzed using Fisher's exact test with P < 0.05 as the level of significance for comparison. Copper levels, clinical scores, and macroscopic and microscopic lesion scores were evaluated by analysis of variance (ANOVA) using a completely randomized design with the pig as the experimental unit. If the overall ANOVA was significant (P ≤ 0.05), pairwise comparisons were performed by least significant difference.
Results
Clinical evaluation
Rectal temperatures, respiratory disease scores, CNS disease, and lameness scores are summarized in Table 2. From 1 to 10 DPI, pigs in groups 4, 5, and 6 had significantly more days with rectal temperatures of >40 C than did controls and pigs inoculated with S. suis alone. From 11 to 28 DPI, the pigs in group 5 continued to have significantly more days with elevated rectal temperatures. Group 6 was excluded from analysis because there was only one surviving pig.
Mean (±SD) rectal temperature, respiratory disease, central nervous system disease, and lameness scores for pigs inoculated with PRRSV and S. suis. Values with different superscripts are significantly different (P < 0.05) as determined by least significant difference.
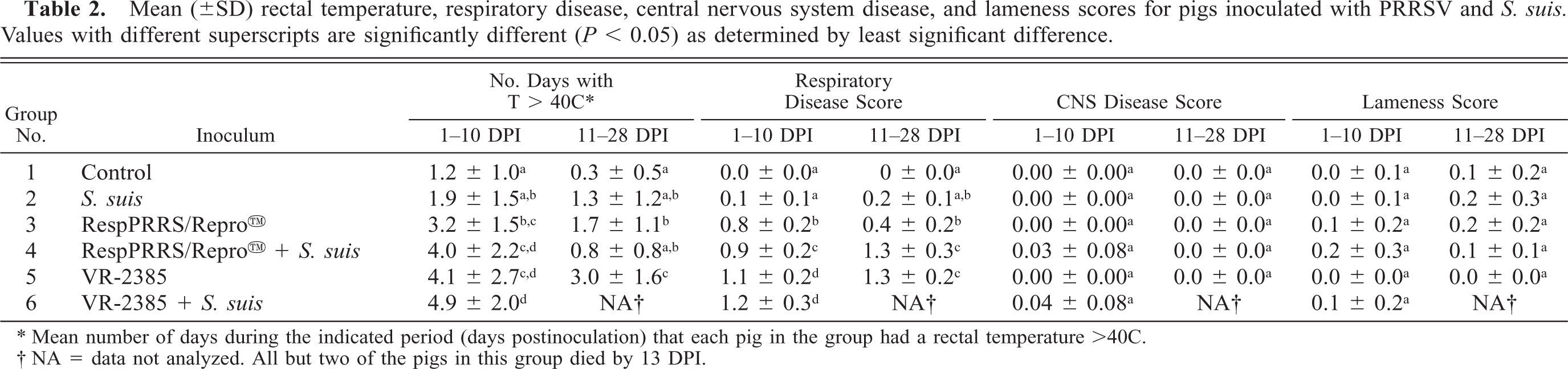
∗ Mean number of days during the indicated period (days postinoculation) that each pig in the group had a rectal temperature >40C.
† NA = data not analyzed. All but two of the pigs in this group died by 13 DPI.
From 1 to 10 DPI, pigs in groups 3–6 had significantly more severe (P < 0.05) respiratory disease than did controls or pigs inoculated with only S. suis. Pigs in groups 5 and 6 were significantly more severely affected than were pigs in all other groups. Pigs in groups 4 and 5 continued to have significantly more severe respiratory disease during the 11–28-DPI period.
Signs of CNS disease were observed in some pigs in groups 2, 4, and 6; however, when mean CNS scores were analyzed, there were no significant differences between groups. Clinical signs of CNS disease included head tilt, nystagmus, tremors, and opisthotonus. Although some degree of joint swelling and lameness was observed in all groups except group 5, there were no significant differences between the groups.
The mortality rate in group 6 (VR-2385 + S. suis) was 87.5% (Table 3). This rate was significantly higher (P < 0.05) than that observed in all other groups except group 4 (RespPRRS/Repro™ + S. suis), which had 37.5% mortality. The mortality rate in group 4 was considerably but not significantly higher than that of group 2 (14.3%), which was inoculated with S. suis alone. No pigs died in groups 1 (control), 3 (RespPRRS/Repro™), or 5 (VR-2385).
Mortality of pigs, pulmonary clearance of copper, and isolation of PRRSV from bronchoalveolar lavage (BAL) fluid and S. suis serotype 2 from internal tissues of pigs at necropsy. Values with different superscripts are significantly different (P < 0.05) as determined by Fisher's exact test for mortality, PRRSV isolation, and S. suis isolation and by least significant difference for copper level data.

∗ Number of pigs found dead or euthanatized when exhibiting CNS signs or recumbency due to lameness on or after 10 DPI (10 days after PRRSV inoculation = 3 days after S. suis inoculation). The number of pigs remaining in study group (pigs euthanatized at 10 DPI for copper analysis) are not included.
† Six pigs were evaluated per group; values are presented as a group mean ± SD.
‡ Number of pigs from which S. suis serotype 2 was recovered from internal tissues (brain, pleura, pericardium, peritoneum, liver, kidney, spleen, or joints).
Pulmonary copper clearance
At necropsy on day 10, the lungs of pigs infused with copper particles varied from light blue to dark blue, depending on the group. Control pigs had the darkest blue lungs. Blue copper particles were observed microscopically in PIMs of all pigs infused with copper. Lung copper levels of each group (n = 6) are presented in Table 3. Pigs inoculated with VR-2385 (groups 5 and 6) had significantly lower lung copper levels than did controls (group 1) and pigs inoculated with RespPRRS/Repro™ (groups 3 and 4). The pigs inoculated with only S. suis had significantly lower lung copper levels than did the control pigs. Both VR-2385 PRRSV and S. suis had an effect, but pulmonary copper levels in the combined infection were not significantly different from those in pigs inoculated with VR-2385 alone.
Serology and virus isolation
All pigs were clinically healthy and negative for antibody to PRRSV on arrival at 11–12 days of age. Control pigs (group 1) and pigs inoculated with S. suis alone (group 2) remained serologically negative to PRRSV throughout the study. All pigs inoculated with VR-2385 seroconverted to PRRSV between 7 and 10 DPI (S/P ratio ≥ 0.4) and pigs inoculated with RespPRRS/Repro™ seroconverted after 10 DPI. PRRSV serum antibodies were present in 6/7 (85.7%) and 2/6 (33.3%) of the RespPRRS-inoculated pigs (group 3 and 4, respectively) at 14 DPI and in all RespPRRS/Repro™ inoculated pigs by 21 DPI.
Isolation of PRRSV was attempted from BAL fluid at necropsy (Table 3). PRRSV was isolated from the BAL of 100% of the VR-2385-inoculated pigs (groups 5 and 6). This isolation rate was significantly higher than that in the RespPRRS/Repro™ group (38.5%) but not significantly higher than that in the RespPRRS/Repro™ + S. suis group (71.4%).
Bacteriology
The results of the internal tissue and whole blood cultures for S. suis serotype 2 are summarized in Table 3 and 4, respectively. S. suis was isolated from the internal tissues (pleura, pericardium, peritoneum, liver, kidney, spleen, brain, or joint) of significantly more pigs (78.6%) (P < 0.05) in the group coinoculated with VR-2385 and S. suis. S. suis was isolated from 28.6% of the pigs in the group inoculated with RespPRRS/Repro™ + S. suis; however, this isolation rate was not significantly different from that in the pigs inoculated with alone S. suis (7.7%). There were significantly more pigs with S. suis bacteremia in the group inoculated with VR-2385 and S. suis (78.6%) than in the pigs inoculated with RespPRRS/Repro™ and S. suis (35.7%) or the with S. suis alone (7.7%).
S. suis serotype 2 was isolated from the upper respiratory tract (nasal cavity or trachea) of 4/13 (30.8%) pigs in group 2, 6/14 (42.9%) pigs in group 4, and 8/14 (57.1%) pigs in group 6 at necropsy. Group 6 (4/14 pigs, 28.6%) was the only group where S. suis serotype 2 was isolated from the lungs.
Gross lesions
PRRSV-induced lung lesions were multifocal in distribution. The affected lung parenchyma was mottled tan and rubbery and failed to collapse. There was no evidence of pleuritis or purulent exudate in the airways. Mean gross lung lesion scores 9 were 0.3 ± 0.8 (group 3), 2.6 ± 4.5 (group 4), 63.5 ± 13.9 (group 5), and 61.9 ± 8.9 (group 6) at 10 DPI. The PRRSV-induced gross lung lesions were significantly more severe (P < 0.05) in the groups that were inoculated with VR-2385 PRRSV (groups 5 and 6). S. suis coinoculation did not significantly increase the percentage of lung with visible lesions (Table 5). Lungs from the control group and the S. suis alone group were grossly normal, except for one pig from the S. suis group that died 21 DPI. This pig had mild multifocal bronchopneumonia characterized by dark purple consolidated foci of lung parenchyma with exudate in the airways.
Streptococcus suis serotype 2 bacteremia in uninoculated pigs and in pigs inoculated with S. suis, PRRSV, and combined PRRSV and S. suis. Values with different superscripts are significantly different (P < 0.05) as determined by Fisher's exact test.

∗ At 7 DPI, S. suis serotype 2 was intranasally inoculated to pigs in groups 2, 4, and 6. Six pigs per group were randomly selected prior to inoculation, and blood samples for S. suis evaluation were obtained at 7, 8, 9, and 10 DPI. These pigs were necropsied at 10 DPI.
† Pigs were necropsied when demonstrating CNS disease or recumbency. All remaining pigs were necropsied on day 28.
Gross and microscopic lung lesions in pigs inoculated with PRRSV and S. suis. Values with different superscripts were significantly different (P < 0.05) as determined by least significant difference.
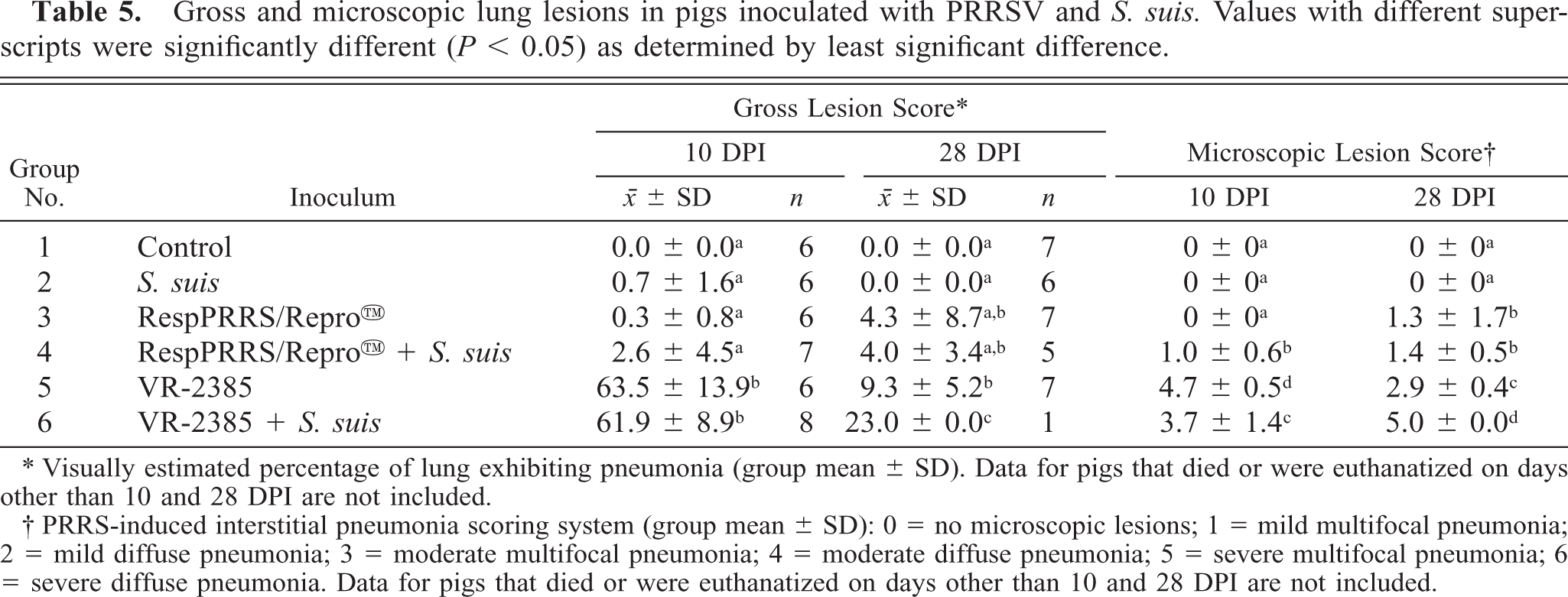
∗ Visually estimated percentage of lung exhibiting pneumonia (group mean ± SD). Data for pigs that died or were euthanatized on days other than 10 and 28 DPI are not included.
† PRRS-induced interstitial pneumonia scoring system (group mean ± SD): 0 = no microscopic lesions; 1 = mild multifocal pneumonia; 2 = mild diffuse pneumonia; 3 = moderate multifocal pneumonia; 4 = moderate diffuse pneumonia; 5 = severe multifocal pneumonia; 6 = severe diffuse pneumonia. Data for pigs that died or were euthanatized on days other than 10 and 28 DPI are not included.
Both VR-2385 and RespPRRS/Repro™ induced generalized lymph node enlargement. The enlarged lymph nodes were present by 10 DPI and persisted through 28 DPI. The lymph node changes were similar in pigs inoculated with PRRSV and pigs inoculated with both PRRSV and S. suis.
Pigs dually inoculated with VR-2385 PRRSV and S. suis (group 6) had a significantly higher (P < 0.05) incidence of pleuritis, peritonitis, and arthritis than did the pigs inoculated with S. suis alone (Table 6). Affected pigs had variable amounts of fibrin and cloudy, straw-colored fluid within the pleural, pericardial, and/or peritoneal cavities. All front and rear leg joints were examined, and arthritis was commonly present in hock, carpal, and/or stifle joints of dually inoculated pigs. Affected joint capsules were distended by serofibrinous to fibrinopurulent exudate, which often extended into the tendon sheaths and adjacent periarticular soft tissues.
Prevalence ∗ of either gross or microscopic lesions characteristic of S. suis serotype 2 infection in pigs. Values with different superscripts are significantly different (P < 0.05) as determined by Fisher's exact test.
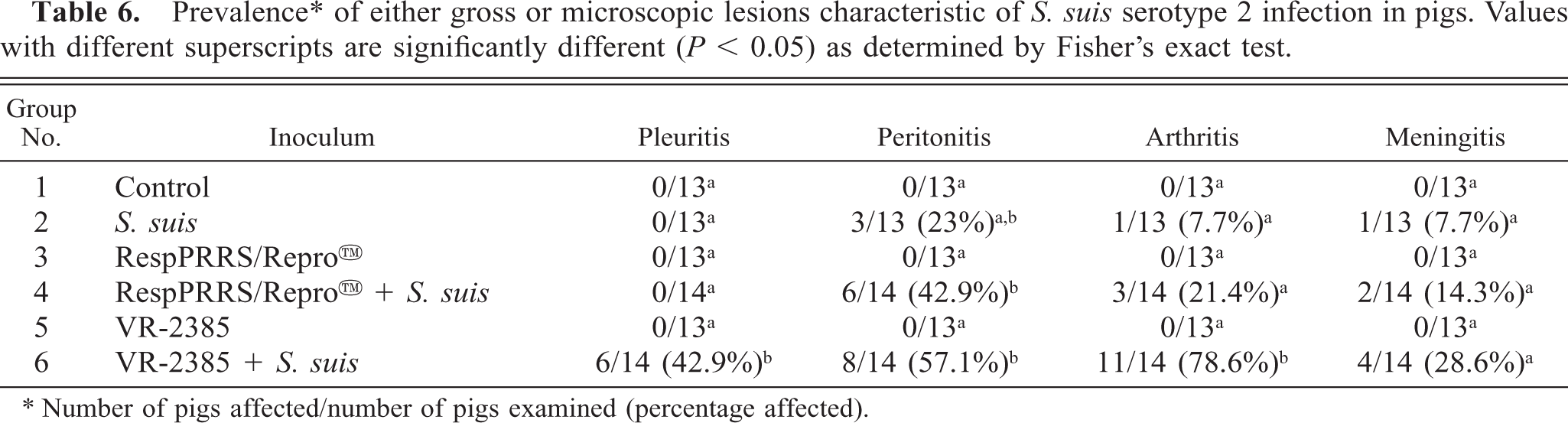
∗ Number of pigs affected/number of pigs examined (percentage affected).
Microscopic lesions
PRRSV-induced pneumonia was characterized by septal infiltration by macrophages and lymphocytes, type 2 pneumocyte hypertrophy and hyperplasia, and increased amounts of alveolar exudate consisting of macrophages, necrotic or apoptotic macrophages, neutrophils, and proteinaceous fluid. Microscopic lung lesions (Table 5) induced by VR-2385 (groups 5 and 6) were significantly more severe (P < 0.05) than those induced by RespPRRS/Repro™ (groups 3 and 4) at both 10 and 28 DPI. The RespPRRS/Repro™ vaccine induced mild interstitial pneumonia, which persisted through 28 DPI. Mild to moderate multifocal suppurative bronchopneumonia was observed in all groups inoculated with S. suis. PRRSV antigen was demonstrated by immunohistochemistry in pulmonary intravascular and alveolar macrophages in all PRRSV-inoculated groups (groups 3–6). PRRSV-positive alveolar macrophages were often found aggregated in the areas of suppurative bronchopneumonia or pleuritis. The lungs of all pigs necropsied at 10 DPI were tested by immunohistochemistry and were negative for the presence of type 2 porcine circovirus antigens.
Fibrinosuppurative peritonitis, when observed, was characterized by the presence of variable amounts of fibrin, numerous neutrophils, and fewer macrophages and lymphocytes on the surface of the liver and/or spleen. Those pigs with peritonitis also commonly had moderate to severe, diffuse suppurative hepatitis characterized by increased numbers of neutrophils in sinusoids. Articular and synovial lesions, when present, were characterized as moderate to severe, acute to subacute, fibrinosuppurative, histiocytic arthritis and synovitis. Increased numbers of neutrophils, lymphocytes, plasma cells, and macrophages were present in the articular and periarticular connective tissues.
One of 13 (7.7%) pigs inoculated singularly with S. suis, 2/14 (14.3%) pigs coinoculated with RespPRRS/Repro™ and S. suis, and 4/14 (28.6%) pigs coinoculated with VR-2385 and S. suis had moderate to severe, multifocal, fibrinosuppurative ependymitis and meningoencephalitis (Table 6). Coccoid bacterial colonies were frequently observed within the exudate of the meninges. Three of 13 (23.1%) pigs inoculated with VR-2385 had mild lymphohistiocytic encephalitis characteristic of PRRSV infection. 9 , 29
Rhinitis was observed in 5/14 (35.7%), 8/13 (61.5%), and 6/14 (42.9%) of the pigs in groups 4, 5, and 6, respectively. Rhinitis was characterized by suppurative, lymphohistiocytic, and plasmacytic inflammation in the turbinate epithelium and submucosa. Mild multifocal lymphoplasmacytic and histiocytic myocarditis was observed only in the VR-2385 PRRSV groups (23.1% and 14.3% in groups 5 and 6, respectively).
Discussion
Porcine PIMs are thought to play a major role in clearance of bacteria from the blood. 2 , 4 , 5 Clinical, pathologic, and microbiologic results from this PRRSV and S. suis serotype 2 coinfection model demonstrate a PRRSV-associated adverse effect on clearance of S. suis from the blood. The dual infection allowed for more widespread tissue dissemination of the bacteria and enhanced S. suis-associated disease. The group of pigs inoculated with the high-virulence strain of PRRSV was most severely affected by S. suis. These pigs also had the most severe decrease in pulmonary copper clearance, suggesting that PRRSV-induced damage to PIMs may be important in the pathogenesis of PRRSV-induced increased susceptibility to bacterial diseases. A similar copper clearance model was used to demonstrate that fumonisins inhibit PIMs from removing particulate matter and bacteria from the circulation and potentially predispose pigs to infectious diseases. 22
The sample used for copper analysis was collected from the same portion of the lung (3- × 3- × 3-cm cube from the center of left caudal lung lobe) regardless of whether there was pneumonia present or not. Altered blood flow to the affected tissue and associated changes in perfusion could have resulted in decreased copper levels in the sampled lung tissue. Pulmonary lesions in the VR-2385-inoculated pigs were more severe and diffuse and probably were represented in the tissue taken for copper analysis, whereas lesions were less severe and more multifocal in the RespPRRS/Repro™ group and thus an equivalent amount of affected tissue may not have been represented. The same model could be used to measure copper levels in affected lung tissue from a swine influenza virus-infected lung to determine if the effect on PIMs is PRRSV specific. Swine influenza virus replicates primarily within bronchial and bronchiolar epithelial cells and to a lesser degree in pneumocytes and alveolar macrophages 31 and probably has a less dramatic effect on PIM function than does PRRSV.
PRRSV-induced damage to PIMs may lower the threshold of S. suis necessary to induce disease. The disease induced by dual inoculation with high-virulence PRRSV and S. suis was very similar to the disease previously observed with a higher dose (109.3 versus 108.3 cfu) of the same S. suis inocula (unpublished data) administered alone to PRRSV-negative pigs. The clinical signs were similar, and S. suis was isolated from similar tissues. This finding is consistent with field observations of S. suis-associated disease on most farms where it is often of little economic importance until the herd is infected with PRRSV (unpublished data).
S. suis was administered intranasally to the pigs 7 days after intranasal inoculation with PRRSV. The culture results indicate that pigs inoculated dually with PRRSV and S. suis had a higher incidence of recovery of S. suis from the upper respiratory tract (nasal cavity or trachea) than did pigs inoculated singularly with S. suis. The only group from which S. suis was recovered from lung tissue was the group coinoculated with high-virulence PRRSV and S. suis. These results suggest that PRRSV may predispose pigs to colonization of the respiratory tract by S. suis serotype 2. PRRSV induces nonsuppurative rhinitis and metaplasia of the turbinate epithelium, 9 , 10 which may in part explain the increased colonization observed. PRRSV may damage the nasal turbinate epithelium, potentially creating a portal of entry for S. suis. 7 S. suis may enter the circulation through vessels in the submucosa of the turbinate or oropharynx or via the tonsil. If PRRSV infection allows for S. suis colonization of lung, then PRRSV-induced lysis and decreased bactericidal function of pulmonary alveolar macrophages may allow S. suis to induce septicemia. Lysis of alveolar macrophages by PRRSV may result in replacement of those macrophages by immature cells that cannot contain bacterial infections, resulting in pneumonia and septicemia. 18
Mycoplasma hyopneumoniae-induced attraction and activation of macrophages may produce an ideal environment for PRRSV to persist and subsequently may enhance the severity and duration of pneumonia in pigs coinfected with PRRSV and M. hyopneumoniae. 27 It was difficult to assess whether S. suis infection resulted in persistence of PRRSV lesions in this model because only one pig in the group coinoculated with high-virulence PRRSV and S. suis survived until the end of the study at 28 DPI. This pig did have more severe and persistent PRRSV-like interstitial pneumonia than observed in the pigs inoculated with PRRSV, alone. PRRSV was also recovered from more of the pigs (71.4%) coinoculated with RespPRRS/Repro™ and S. suis than from those inoculated with RespPRRS/Repro™ alone (38.5%). Like M. hyopneumoniae, S. suis may attract and activate macrophages in an environment conducive to PRRSV replication and persistence.
Extra-label use of the modified live PRRSV vaccine RespPRRS/Repro™ may exacerbate S. suis-induced disease in herds exposed to or carrying virulent S. suis. The highly virulent strain VR-2385 of PRRSV enhanced susceptibility to S. suis to a greater extent than did the RespPRRS/Repro™ vaccine. However, the modified live vaccine strain of PRRSV also increased susceptibility to S. suis challenge, as indicated by increased morbidity and mortality and increased recovery of S. suis from blood and internal organs. A field study comparing the incidence and severity of S. suis-induced disease in PRRSV-vaccinated and unvaccinated pigs would provide additional useful information. The RespPRRS/Repro™ vaccine was administered intranasally, although the manufacturer recommends intramuscular administration and it is licensed for intramuscular use. Some vaccines that are intended for intramuscular use can induce clinical disease when administered intranasally, 20 and it is not uncommon for practitioners to use the PRRSV vaccine intranasally. 24 There is also a precedent for a modified live vaccine (hog cholera) to increase susceptibility to bacterial infection (Pasteurella multocida). 17
We developed an experimental model that demonstrated that highly virulent stains of PRRSV predispose pigs to S. suis infection. Intranasal administration of modified live PRRSV vaccines may predispose pigs to S. suis-associated disease. This work has provided further evidence that suppression of PIM function may be a mechanism for the increased susceptibility to S. suis septicemia and disease. The PRRSV & S. suis coinfection model is straightforward and reasonably mimics what occurs in the field. This model will be very useful for testing the efficacy of various prevention and treatment regimens, such as vaccination for PRRSV, vaccination for S. suis, and antimicrobial intervention strategies.
Footnotes
Acknowledgements
We thank Drs. Eileen L. Thacker and Prem S. Paul for the use of laboratory equipment and technical advice, Theresa Young and Julie A. Kinker for technical assistance, and Jeremy D. Bruna for animal care and manuscript preparation. Funding for this project was provided by Pork Check-Off dollars from the National Pork Producers Council on behalf of the National Pork Board.
