Abstract
Fourteen cases of feline leukemia virus (FeLV)-associated enteritis were immunohistologically examined for the expression of FeLV proteins gp70, p27, and p15E in the jejunum, mesenteric lymph nodes, spleen, and bone marrow. Results were compared with those of FeLV-infected cats without intestinal alterations. Other viral infections and specific bacterial, fungal, and parasitic infections were excluded by standard microbiologic methods, histopathology, immunohistology, and in situ hybridization. In FeLV-associated enteritis, FeLV gp70 and p15E were strongly expressed in intestinal crypt epithelial cells. In contrast, FeLV-positive cats without intestinal alterations showed only faint staining for gp70 and p15E and comparatively strong p27 expression in these cells. Findings suggest a direct relation between FeLV infection and alterations in intestinal crypt epithelial cells that may be attributed to the envelope proteins gp70 and p15E and/or their precursor protein. Distinct similarities to the intestinal changes in the experimentally induced FeLV-feline AIDS syndrome are obvious, suggesting that naturally occurring feline AIDS variants may be responsible for FeLV-associated enteritis.
Persistent feline leukemia virus (FeLV) infection is associated with a wide spectrum of nonneoplastic diseases. 29 – 31 FeLV-associated enteritis (FAE) is one of the conditions observed in cats persistently infected with FeLV. The definition of FAE is based on light microscopic alterations of the small intestine comparable to those observed in cats with feline panleukopenia and the absence of panmyelophthisis in bone marrow, in connection with the demonstration of FeLV infection and the failure to demonstrate parvovirus in intestine and bone marrow. 12 , 13 , 28 In FAE, intestinal epithelial changes are often milder than in feline panleukopenia. Epithelial regeneration is only rarely seen. Bone marrow is either unaltered or hypercellular. Furthermore, cats with FAE are significantly older than animals with feline panleukopenia. 28 The purpose of this study was to demonstrate the major FeLV proteins gp70, p27, and p15E in FAE to elucidate their potential pathogenic role in the disease and to compare the findings in FAE with those in FeLV-positive cats without intestinal alterations.
Materials and Methods
Animals and tissue processing
The study was retrospectively performed on 28 cats that had been routinely necropsied at the Departments of Veterinary Pathology of both Leipzig and Giessen universities between 1984 and 1997. Fourteen animals were immunohistologically positive for FeLV antigen and exhibited histopathologic alterations consistent with those observed in FAE (group I, Table 1). 28 For comparative examinations, immunohistologically FeLV-positive cats without alterations of the small intestine were examined (n = 14, group II, Table 2). Immunohistologic detection of FeLV infection had been performed on spleen, bone marrow, mesenteric lymph nodes, and small intestine as previously described. 10 Samples from jejunum, spleen, and bone marrow were tested from all animals. Mesenteric lymph node samples from 10 cats of group I and 13 cats of group II also were tested. Cats immunohistologically negative for FeLV that had unaltered small intestine and that had died or were euthanatized because of alterations not affecting the intestine served as negative control group (n = 17). All samples were fixed for 12–18 hours in 10% nonbuffered formalin or methanol, respectively, and embedded in paraffin. Sections 5 µm thick were stained with hematoxylin and eosin (HE) and Giemsa. Specimens from the small intestine were further stained with a modified Gram's stain and with the Warthin-Starry stain. 15 , 22
Cats with FeLV-associated enteritis (group I).
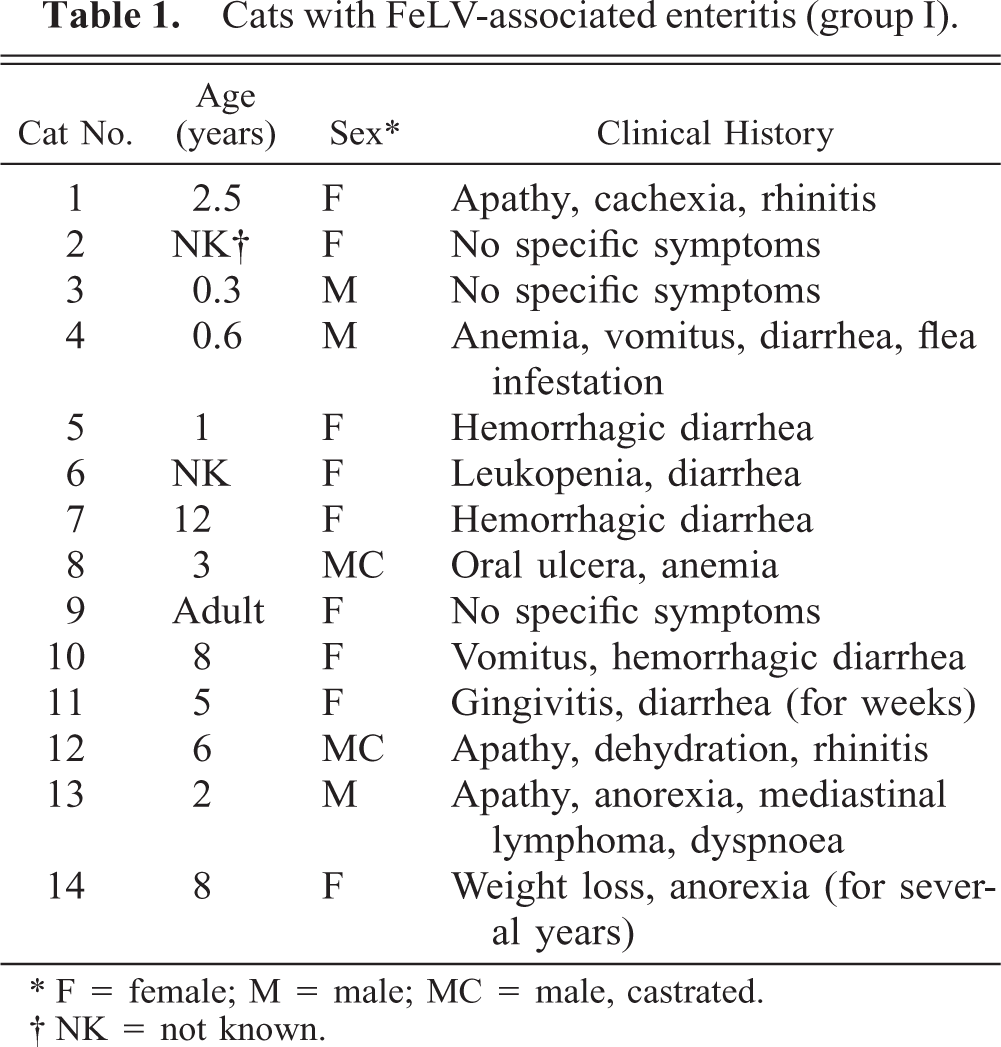
∗ F = female; M = male; MC = male, castrated.
† NK = not known.
Immunohistologically FeLV-positive cats without intestinal alterations (group II).
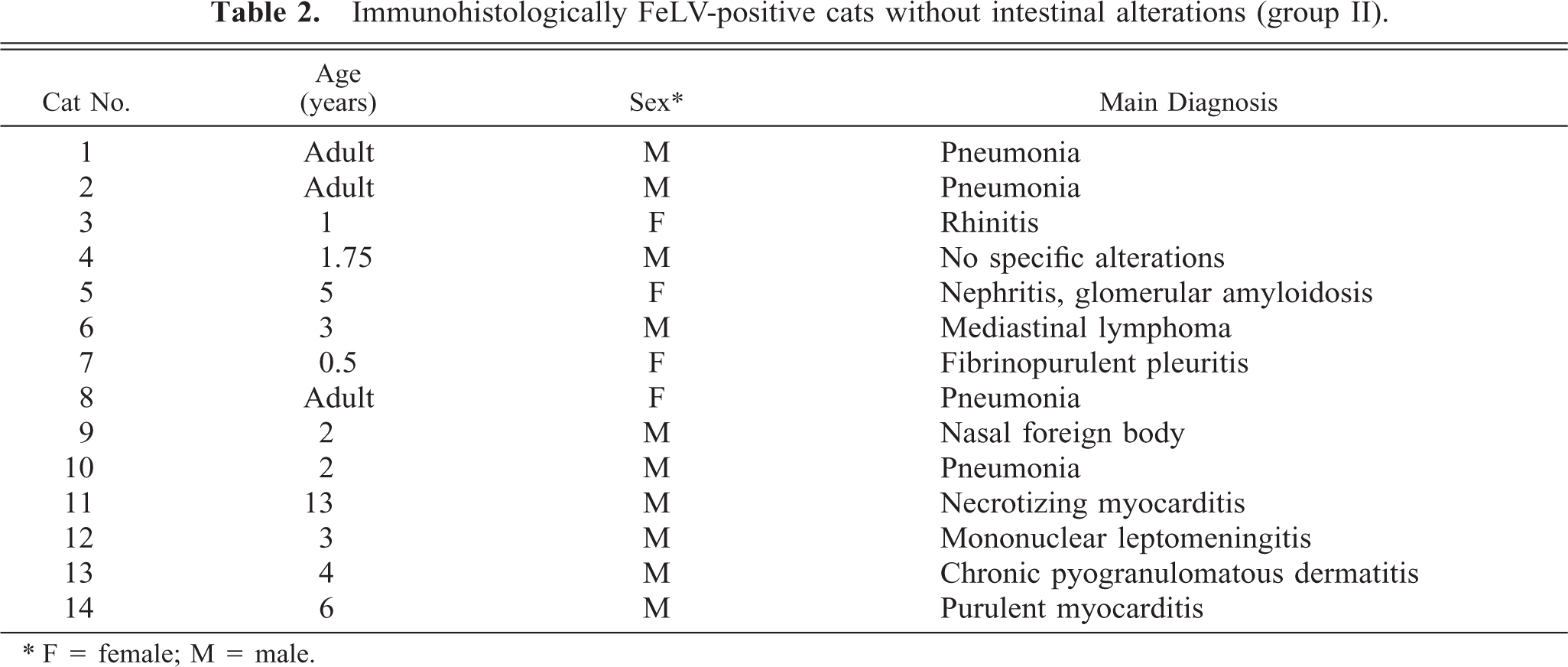
∗ F = female; M = male.
Scoring system for histopathologic alterations
Histopathologic alterations of the small intestine were evaluated according to the degree of change observed in the intestinal crypts and villous tips that was distinguishable from post mortem alterations. The changes were graded on the number of degenerating epithelial cells and the degree of depletion of crypts, dilation of residual crypts, and villous atrophy.
Transmission electron microscopy
Feces or ingesta from cats of group I were examined ultrastructurally for viral particles by applying the negative staining technique. 1 , 37 For transmission electron microscopy, formalin-fixed jejunal and bone marrow tissue of one cat (No. I-14) was available. Specimens were washed with 0.166 M cacodylate buffer and embedded as previously described. 14
Immunohistology
For immunohistology, the indirect peroxidase and the peroxidase–antiperoxidase method were performed on both formalin- and methanol-fixed, paraffin-embedded tissues as previously described. 8–10, 32 The antibodies applied are listed in Table 3. Controls were established by incubation of sections with normal rabbit and rat serum and an irrelevant monoclonal antibody directed against chicken lymphocytes, respectively. 3 Formalin-fixed, paraffin-embedded intestinal tissues from cats with panleukopenia and granulomatous necrotizing lesions of feline infectious peritonitis (FIP) served as positive controls for parvovirus (PV) and feline coronavirus (FCoV) antigen. 8–10, 34 , 38 The antibody against FCoV had been tested by immunohistology for demonstration of FCoV antigen both in FIP lesions and in intestinal FCoV infections. 8 , 9 The antibody against canine PV (CPV) had been tested successfully by western blot with CPV, by capture enzyme-linked immunosorbent assay with polyclonal anti-CPV antibodies as capture, and by immunohistology for cross-reaction with feline PV.
Mouse monoclonal antibodies and catalog numbers. ∗
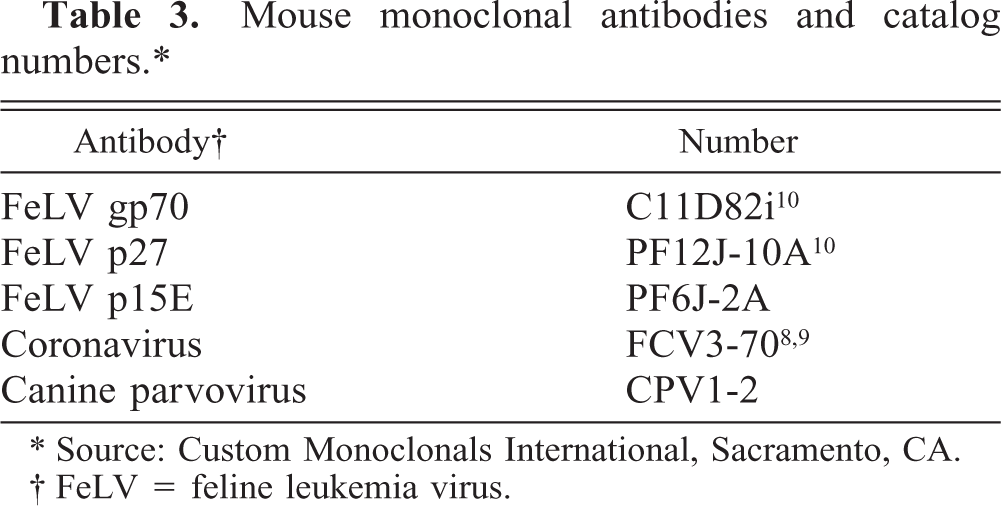
∗ Source: Custom Monoclonals International, Sacramento, CA.
† FeLV = feline leukemia virus.
Scoring system for immunohistology
The expression of FeLV antigens was estimated according to the staining intensity (faint, moderate, strong, very strong) within individual cells and according to the estimated number of cells exhibiting a positive immunohistologic reaction (single, few, some, many positive cells). Because the staining intensity and the number of positive cells increased simultaneously, the data points represent the mean estimated values based on the number of positive cells and the staining intensity: − = no staining; (+) = single/faintly positive cells; + = few/moderately positive cells; ++ = some/strongly positive cells; +++ = many/very strongly positive cells.
Gel electrophoresis, western blotting, immunoblotting
The proteins of washed and vacuum-dried cell suspensions of the permanently FeLV-infected lymphoblastoid cell line FL74 were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) on 12% minigels under nonreducing and reducing conditions. 11 , 36 Prestained SDS-PAGE marker proteins (Bio-Rad Laboratories, Richmond, CA) were used to determine the molecular weight. Proteins were transferred to an Immobilon®-P-membrane (Millipore, Eschborn, Germany) by western blotting. After blocking with 5% milk powder, membranes were incubated at room temperature for 2 hour with mouse anti-FeLV gp70 (1:2,000), mouse anti-FeLV p27 (1:1,500), or mouse anti-FeLV p15E (1:2,000), followed by a 1 hour-incubation with a 1:2,000 dilution of alkaline phosphatase-conjugated goat anti-mouse IgG (H&L; Dianova GmbH, Hamburg, Germany) as secondary antibody. All reagents were diluted in phosphate-buffered saline (pH 7.4) with 0.05% Tween®-20 (Merck, Darmstadt, Germany). Membranes were developed in a solution of 5-bromo-4-chloro-3-indolyl-phosphate (50 mg/ml 70% dimethyl formamide; Boehringer Mannheim Biochemica, Mannheim, Germany) and 4-nitroblue tetrazolium chloride (75 mg/ml dimethyl formamide; Boehringer Mannheim Biochemica).
In situ hybridization for demonstration of the PV genome
In situ hybridization for the demonstration of the PV genome was performed on formalin-fixed specimens of small intestine as previously described. 38 The probe (pNCPV-2) derived from a pBR322 plasmid containing the entire VP1- and VP2-coding sequences of the canine PV genome, which was split by the Hae III restriction enzyme and labeled with biotin-16-dUTP by random-primed labeling (Boehringer Mannheim Biochemica). 27 , 38 Feline and canine intestinal and canine myocardial specimens, immunohistologically positive for PV antigen and exhibiting positive hybridization signals for PV genome served as positive controls. Controls were represented by specimens incubated with the biotinylated control probe pBR322.
Microbiologic examinations
Routine bacteriologic and virologic examinations were performed on single cats (Nos. I-5, I-7).
Results
Case histories
All animals were domestic shorthair cats. Cats with FAE (group I) ranged from 4 months to 12 years of age, with an average age of 4.5 years and a median age of 3 years. In six cats, a clinical history of diarrhea was reported. Another four cats had shown general symptoms like weight loss and anemia. One cat exhibited a thymic lymphoma, and the remaining three cats were euthanatized because of a diagnosis of FeLV infection without specific symptoms (Table 1). FeLV-positive cats without intestinal alterations (group II) ranged from 6 months to 13 years of age, with an average age of 3.6 years and a median age of 3 years. Cats had died with or were euthanatized because of various clinical symptoms but none affecting the alimentary tract (Table 2). No macroscopic or histologic intestinal alterations were recorded.
Immunoblotting
The monoclonal antibodies against FeLV gp70 and FeLV p27 showed the strongest bands at the expected molecular masses (approximately 70 kd and 27 kd) both under nonreducing and reducing conditions. A band at 15 kd with anti-FeLV p15E was seen only under reducing conditions (Fig. 1).
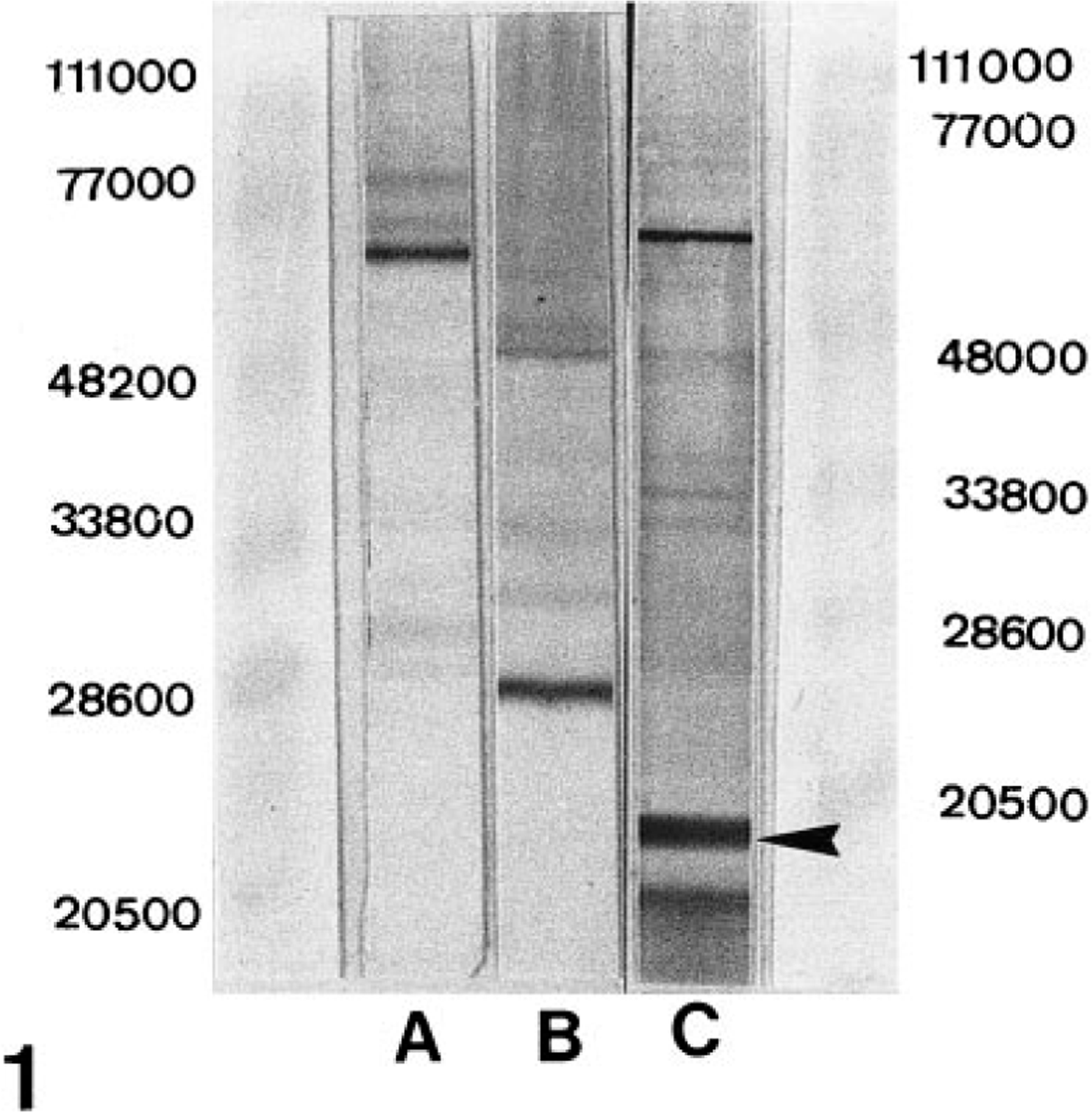
Immunoblot of formalin-fixed permanently FeLV-infected feline lymphoblastoid cells (FL74). 37 Strong bands with anti-FeLV gp70 at approximately 70 kd (A) and with anti-FeLV p27 at approximately 27 kd (B) under non-reducing conditions. Appropriate band with anti-FeLV p15E (C; arrowhead) under reducing conditions.
Histopathologic findings, immunohistologic demonstration of virus antigens, and in situ hybridization for the PV genome in jejunum
Special stains did not reveal any signs of specific bacterial, mycotic, or endoparasitic intestinal infections in any cat. FeLV antigen expression was generally observed in crypt epithelial cells and infiltrating mononuclear cells in the mucosa. In positive controls, PV antigen was expressed by intestinal crypt epithelial cells, and coronavirus (CoV) antigen was mainly expressed by villous tip epithelial cells. In situ hybridization signals for PV genome were observed in intact nuclei of crypt epithelial cells. Controls for antigens, PV genome and nonspecific staining did not show any reaction.
FAE (group I)
Histologic alterations were comparable to those described for FAE and feline PV enteritis. 12 , 13 , 28 They were represented by degeneration of crypt epithelial cells, depletion of crypts, dilation of residual crypts, and villous atrophy. In the majority of cats (8/14; 57%), changes were moderate (Fig. 2a). In the remaining cats, the alterations were graded as marked in three cats (21%) and as severe in three cats (21%; Fig. 2b). In addition to minimal changes of crypt epithelial cells, cat No. I-4 exhibited markedly shortened villi and degeneration and sloughing of single or few villous-tip epithelial cells. In most of the cats (9/14; 64%), the mucosa was markedly infiltrated by mainly mononuclear cells. In four cats (29%) infiltration was moderate, and in one cat (7%) it was massive (Fig. 2a, b). Gut-associated lymphoid tissue was present in sections from six cats. This tissue exhibited moderate follicular hyperplasia in four cats but showed normal follicular activity in the remaining two cats.
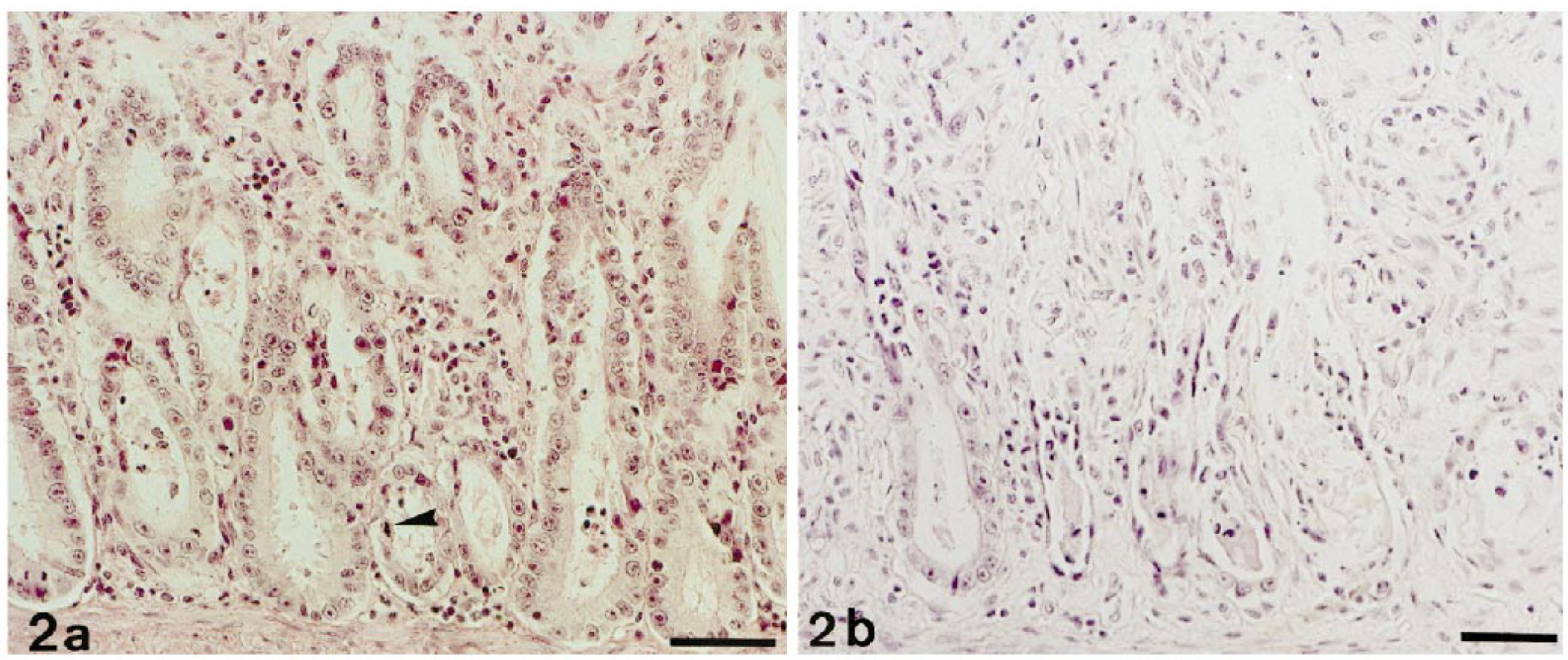
Jejunum; cat. FeLV-associated enteritis. HE. Bar = 50 µm.
In most cats (10/14; 71%), FeLV gp70 was the viral protein that was most strongly expressed by crypt epithelial cells. In all except one cat, it was at least moderately expressed (Figs. 3a, 5). Staining for p15E showed a comparable pattern but was generally less intense (Figs. 3b, 5). In contrast, p27 was most often faintly to moderately expressed (11/14; 79%) (Fig. 3c). Two cats did not show any p27 expression in epithelial cells (Fig. 5).
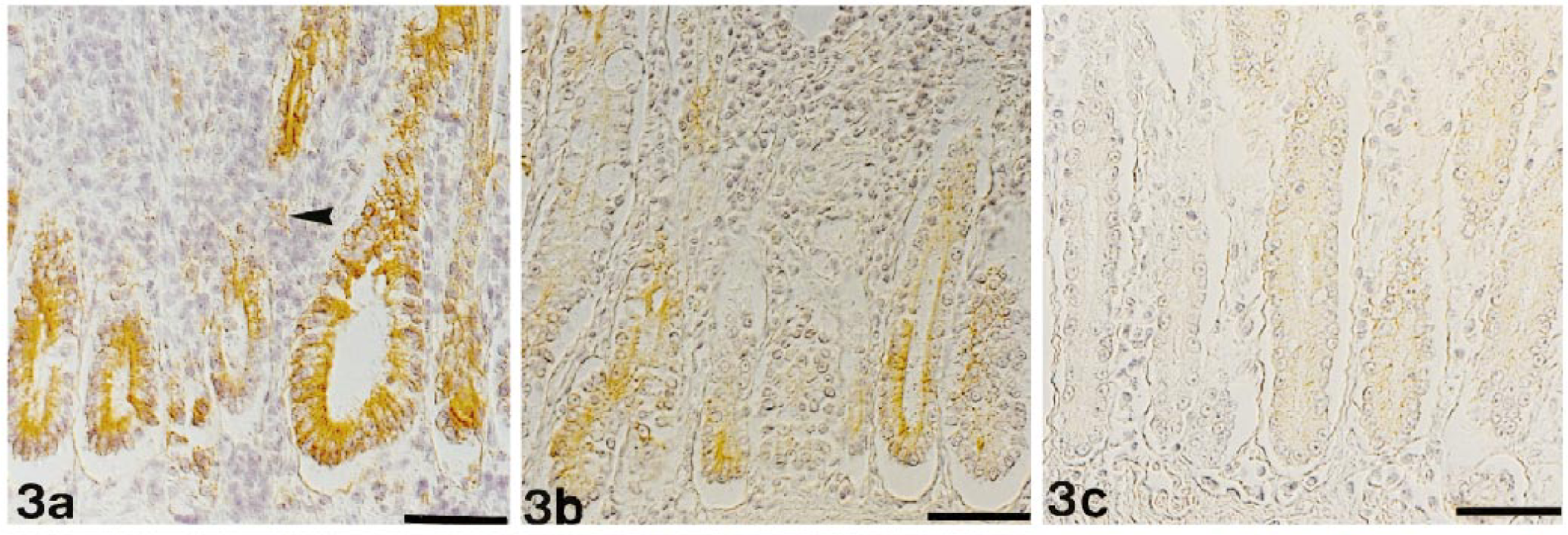
Jejunum; cat No. I-8. FeLV-associated enteritis. Expression of FeLV antigens in epithelial cells. Indirect peroxidase method, Papanicolaou's counterstain. Bar = 50 µm. Fig. 3a. Very strong cytoplasmic staining of many crypt epithelial cells for gp70. Single infiltrating cells in the mucosa stain moderately positive (arrowhead). Fig. 3b. Moderate to strong cytoplasmic staining of many crypt epithelial cells for p15E. Fig. 3c. Moderate cytoplasmic staining of many crypt epithelial cells for p27.
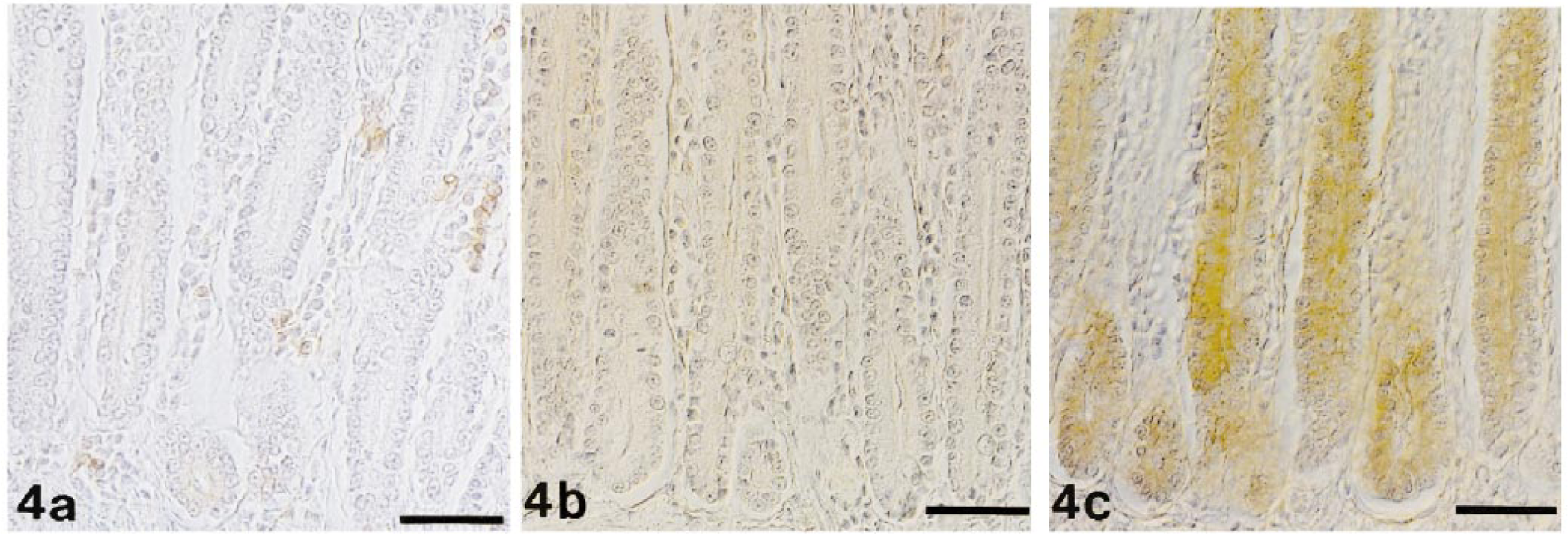
Jejunum; cat No. II-2. FeLV-positive cat without intestinal alterations. Indirect peroxidase method, Papanicolaou's counterstain. Bar = 50 µm. Fig. 4a. Faint to moderate cytoplasmic staining of numerous crypt epithelial cells for gp70. Numerous infiltrating cells in the mucosa stain moderately positive. Fig. 4b. Crypt epithelial cells and infiltrating cells in the mucosa are p15E negative. Fig. 4c. Strong positive cytoplasmic staining of many crypt epithelial cells for p27.
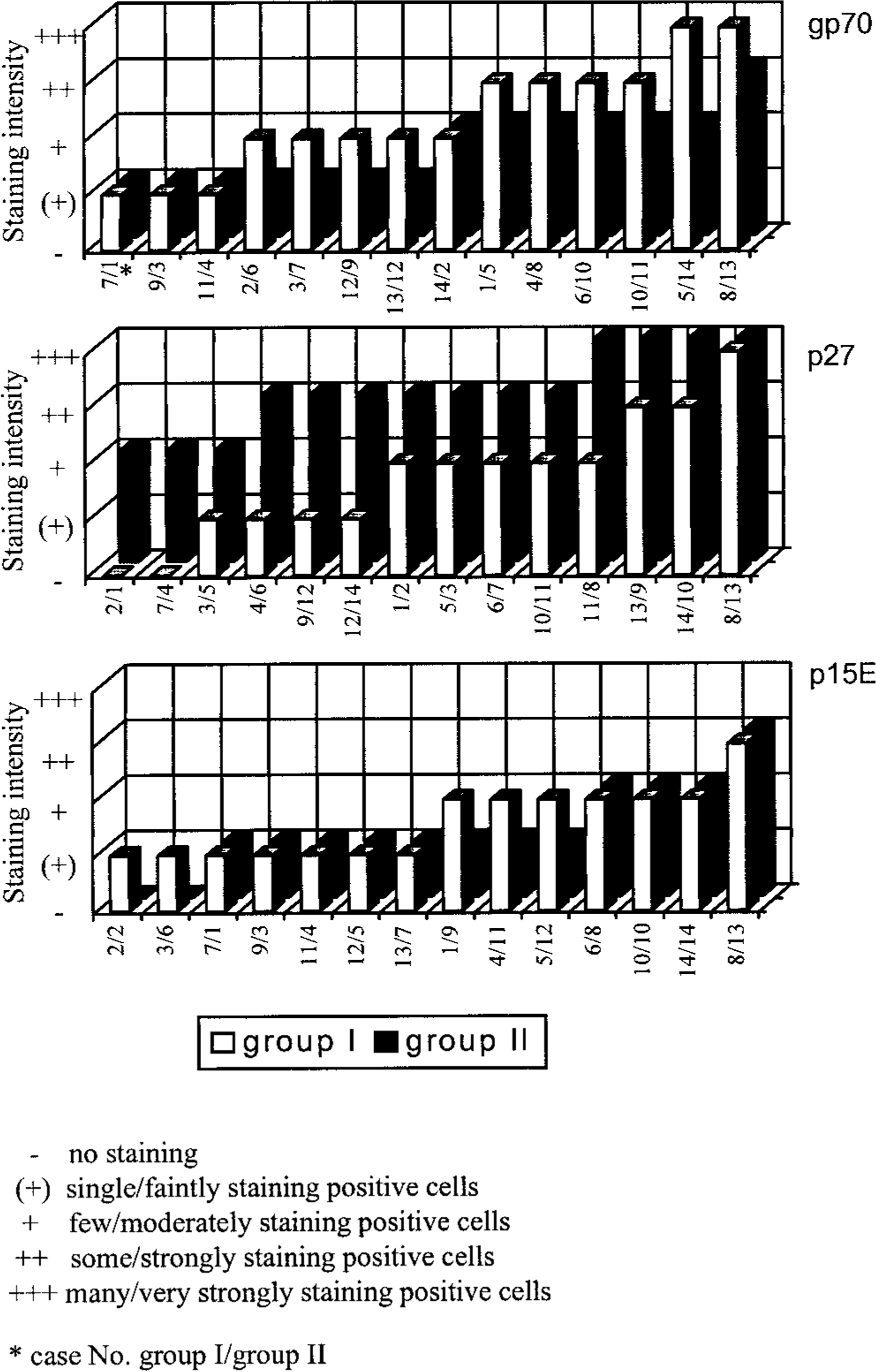
Expression of FeLV proteins gp70, p27, and p15E in crypt epithelial cells. In cat jejunum. Each bar represents the mean estimated value for one animal. Values are arranged in order of increasing magnitude.
Infiltrating cells in the intestinal mucosa were usually moderately to strongly gp70 positive and showed a faint to strong staining for p15E but did not express p27 (Fig. 3a–c). CoV antigen expression was seen in single villous tip epithelial cells detached from the villous tip in cat No. I-4.
PV antigen and genome were not detected in any cat.
Cats without intestinal alterations (group II)
Intestinal epithelial cells did not show any alterations. In most cats (12/14; 86%), the mucosa showed minimal to moderate infiltration by mononuclear cells. Gut-associated lymphoid tissue was present in sections from two cats and showed normal follicular activity.
FeLV antigen expression was observed in crypt epithelial cells of all cats. In most cats (11/14; 79%), staining for p27 was most intense; it was generally moderate to strong (Fig. 4c). In contrast, gp70 expression was faint to moderate (Fig. 4a) and was completely lacking in two cats. In three cats (21%), staining for p27 and gp70 was equally strong. The p15E protein was usually expressed the least intensely (Fig. 4b) and was completely lacking in two cats. Infiltrating cells, however, stained generally intensely positive for gp70 (Fig. 4a). They variably expressed p15E but did not stain for p27 (Fig. 4b, c).
CoV and PV antigen and PV genome were not detected in any cat.
Histopathologic findings and expression of FeLV antigens in spleen, mesenteric lymph nodes, and bone marrow
Spleen
In the FAE cats (group I), B-cell zones exhibited normal activity to moderate hyperplasia. In FeLV-positive cats without intestinal alterations (group II), B-cell activity ranged from moderate depletion to moderate hyperplasia. The red pulp seemed unaltered in all cats of both groups. In several cats (Nos. I-7, I-13, II-10, II-13), moderate extramedullary hematopoiesis was indicated by the presence of few megacaryocytes.
Staining for FeLV antigens was found in mononuclear cells (lymphoblasts, lymphocytes, macrophages) and dendritic cells of both the red and white pulp. It was generally strongest in FAE cats (group I) where a variable number of all cell types expressed gp70 and, to a lesser extent, p27 and p15E. In cat Nos. I-8 and I-14, megacaryocytes in the red pulp stained positively for gp70 and p15E. No cat of group II expressed all antigens in spleen. In cat No. II-14, the spleen was completely FeLV negative.
Mesenteric lymph nodes
In the FAE group (I), most cats (7/10; 70%) showed normal lymphoid follicles. Mild to moderate follicular hyperplasia was observed in the other three cats. In FeLV-positive cats without intestinal alterations (group II), variable follicular activity, ranging from moderate depletion to mild hyperplasia, was observed.
Staining for FeLV antigens was generally less intense in the lymph nodes than in the spleen. In group I, staining was often restricted to single weekly gp70- and p15E-positive follicular dendritic cells. In group II, all FeLV antigens were expressed in many cells in all except two cats, in which the mesenteric lymph nodes were FeLV negative.
Bone marrow
Bone marrow activity was variable. In the majority of cats (group I: n = 11, 79%; group II: n = 10, 71%), it was moderate or marked and was rated as physiological with respect to the cats' ages. In the FAE group (I), four cats with marked bone marrow activity were ≥2 years of age. In group II, four cats with marked and 2 cats with high bone marrow activity were adults.
In the FAE group (I), staining for gp70 was generally observed. It was expressed most intensely, whereas p27 and p15E were not detected in four cats. In group II, bone marrow was negative in one cat. In the remaining cats, gp70 was usually expressed by megacaryocytes and cells of the lymphoid and myeloid lineage, and expression of p27 and/or p15E was occasionally absent (n = 6).
Histologic features of jejunum, spleen, mesenteric lymph nodes, and bone marrow in FeLV-negative cats with unaltered small intestine
Intestinal epithelial cells did not show any alterations. The mucosa was generally minimally to moderately infiltrated by mononuclear cells. FeLV antigens, CoV antigen, and PV antigen and genome were not detected in any cat. The B-cell zones in the spleen and lymphoid follicles in mesenteric lymph nodes showed normal activity. Bone marrow activity ranged from marked to very high and was physiological for the cats' ages.
Transmission electron microscopy
Viral particles were not demonstrated in feces or ingesta of any cat with FAE. In cat No. I-14, few immature and mature type C viral particles were seen in bone marrow and jejunal crypts.
Microbiology
A high number of Escherichia coli bacteria were isolated from the small intestine in cat Nos. I-5, I-7, and I-11 and from the mesenteric lymph nodes in cat Nos. I-7 and I-11. Routine virologic examination of homogenized lung, liver, kidney, and spleen tissue by tissue culture was negative for cytopathic viruses.
Discussion
We investigated the morphologic and immunohistologic characteristics of FAE with regard to the differential expression of FeLV antigens. Exclusion of other infectious agents as the cause for intestinal lesions in cats with FAE by histopathologic, immunohistologic, electron microscopic, and molecular biologic methods has confirmed that FAE is an unique entity. It definitely differs from panleukopenia-like syndrome, PV enteritis, and CoV enteritis; other rare enteric viral infections or specific bacterial, protozoan, or fungal pathogens can also be excluded. 5 , 6 , 9 , 12 , 13 , 16 , 23 , 28 , 33 , 38
FeLV antigens were immunohistologically demonstrated using monoclonal antibodies against the major FeLV proteins, gp70, p27, and p15E, which were examined for their specificity by immunoblots. The results indicated that anti-gp70 binds strongly to gp70, even when the disulfide bonds to the transmembrane protein p15E are still intact. Anti-gp70 also binds to the gp85 envelope (env) precursor protein. 24 In contrast, the lack of a distinct band with anti-p15E under nonreducing conditions indicates that p15E is only bound when not associated with gp70. 2
FAE is dominated by the degeneration of intestinal epithelial cells. This feature is also characteristic of the feline acquired immune deficiency syndrome (FAIDS), which can be observed after experimental infection with FeLV–FAIDS variants, leading to enterocolitis with necrosis of crypt epithelial cells and villous atrophy. 7 In FAE, C-type viral particles were ultrastructurally demonstrated in intestinal crypts. The presence of these particles does not prove viral infection but does indicate viral replication, which was also anticipated in FAIDS because of strong p27 immunofluorescence in crypt epithelial cells and hyperplastic lymphoid follicles. 2 Expression of p27 without any evidence of cell degeneration also was observed in other than intestinal epithelial cells (e.g., mucosal epithelium of esophagus and airways) both in FeLV-positive cats with variable alterations, including FAE, and in cats with FAIDS. 7 , 35 The presence of p27 in unaltered (epithelial) tissues and intestinal crypt epithelial cells of FeLV-positive cats without FAE rules out a direct relation between the FeLV core protein p27 and cell degeneration. 7 , 35
In contrast, the strong expression of both FeLV env proteins, gp70 and p15E, in intestinal crypt epithelial cells in cats with FAE indicates that a potential excess of env proteins is related to cell alterations. In FAIDS, structural changes of gp70 and differences in glycosylation have been proposed as the cause of a potential disorderly assembly of viral particles with subsequent presence of excess soluble gp70. 24 , 25 Inhibition or alteration of cellular glycoprotein processing due to viral env precursor protein accumulation in the rough endoplasmic reticulum and Golgi apparatus are considered potential mediators or direct causes of cytopathogenicity. Alternatively, interference of the altered epitope with a vital cell receptor has been proposed. 4 , 25
FAIDS is accompanied by the appearance of unintegrated, variant forms of viral DNA in bone marrow, intestinal epithelial cells, and nonseverely depleted lymphoid tissues. 7 , 17 FeLV–FAIDS variant forms proved replication defective but transcription competent. 20 A chimera, created from prototype clones of the common form and the variant form, showed distinct tropism for intestinal germinal epithelial cells and induced a rapidly fatal enteropathy; unintegrated DNA and high amounts of viral p27 were demonstrated in altered crypt epithelial cells. 18 , 20 , 21
The striking similarities between intestinal alterations in FAE and FAIDS suggest that naturally occurring FeLV–FAIDS variants are responsible for changes in FAE. Because the studies on FAIDS were performed with specific-pathogen-free cats, specific enteropathogenic bacteria, viruses, and parasites could be excluded. Therefore, a primary correlation between selective virus amplification in certain target tissues, such as intestinal epithelial cells, and the cytopathic damage to these cells had been suggested. 7 , 18 , 19 This hypothesis was in part clarified by a recent study on replication kinetics and cell tropism of FAIDS variants, which indicated a unique cytopathogenicity rather than a unique cytotropism. 26
However, because this study was performed retrospectively on routinely necropsied cats with natural FeLV infection and because diagnosis of FAE is only possible after histopathologic and immunohistologic examination, direct proof of a potential cytopathic effect of the FeLV env proteins and/or their precursor protein is not possible.
Footnotes
Acknowledgements
We are grateful to Dr. A. Waldvogel (Department of Veterinary Pathology, University of Bern, Bern, Switzerland) for providing the canine parvovirus DNA probe and to Prof. Dr. A. Pospischil (Department of Veterinary Pathology, University of Zürich, Zürich, Switzerland) for providing the facilities for the parvovirus in situ hybridization. Furthermore, we thank Mrs. Roseline Weidenmann for excellent technical assistance and Dr. S. Menger for help with the ultrastructural examination. The work was supported by the Deutsche Forschungsgemeinschaft (RE 532/5-1). AK and JK were supported by the “Hochschulerneuerungsprogramm” of the State of Saxony and the Bundesministerium für Forschung und Technologie.
