Abstract
A 2-year-old Basset Hound was admitted to the University of Florida Veterinary Medical Teaching Hospital with progressive spastic paraparesis. At necropsy, intradural extramedullary tumors produced areas of spinal cord swelling and softening in spinal cord segments T11–T12 and L4–L6. Histologic examination of the masses revealed sheets of polygonal blastemal cells, epithelial cells forming tubules and rosettes, and embryonal glomeruloid-like structures in the thoracic mass. Cells in the lumbar mass were less differentiated, forming rare tubules and no glomeruloid-like structures. The occurrence of two tumors in the spinal cord along with the less differentiated appearance of the lumbar tumor raises the possibility that the lumbar mass arose as a result of intraspinal metastasis. To our knowledge, this is the first report of multifocal or metastatic canine spinal nephroblastoma. In addition, the vimentin and cytokeratin immunohistochemical staining characteristics of these spinal cord nephroblastomas are described.
Spinal cord nephroblastoma is a rare neoplasm of the canine spinal cord that generally occurs in dogs less than 3 years of age.2,4,9 Clinical signs associated with spinal cord nephroblastoma are due to compression of the spinal cord by a solitary, intradural extramedullary mass, which is consistently located between the 10th thoracic (T10) and second lumbar (L2) spinal cord segments.2,3 The histologic appearance and immunohistochemical staining characteristics of these tumors suggest that spinal cord nephroblastomas originate from ectopic metanephric blastema. Metastasis (intraspinal or extraspinal) of canine spinal cord nephroblastoma or the occurrence of multiple tumors has not been previously reported to our knowledge.
A 2-year-old intact female Basset Hound was presented to the veterinary medical teaching hospital at the University of Florida for a progressive paraparesis of 3 weeks' duration. The dog had been owned since being a puppy and had been regularly vaccinated. There were no systemic health problems identified in the history. The dog's mentation was appropriate, and there were no cranial nerve deficits. The dog was weak in both pelvic limbs but was ambulatory. A moderate degree of ataxia was present, and both pelvic limbs exhibited slow postural reactions, especially on the right side. The segmental spinal reflexes were within normal limits, but there was mild reduction of the patellar reflex on the right side. The cranial thigh muscles were not atrophied, and there was no appreciable difference in their tone compared with the rest of the limb muscles. Pain was elicited on palpation of the dog's third and fourth lumbar spinal vertebrae. The anatomic diagnosis was a T3–L3 lesion with some involvement of L4–L5 on the right.
Results of hematologic and serum chemistry panels were within normal limits. Thoracic and abdominal radiographs were normal. Cerebrospinal fluid obtained from the cerebellomedullary cistern revealed elevated protein (94 mg/dl) and white blood cell count of 4/μl (normal reference range, <5/μl). No organisms or neoplastic cells were seen in the cerebrospinal fluid. A thoracolumbar myelogram was performed. The left-sided contrast column could not be detected over the body of the 12th thoracic (T12) vertebra, and mild widening of the spinal cord could be seen at this site. There was widening of the spinal cord and attenuation of all contrast columns over the body of the fourth lumbar (L4) vertebra extending caudally (Fig. 1). An intradural extramedullary lesion was suspected at T12, and an intramedullary lesion was suspected at L4. The dog was euthanatized due to a poor prognosis.
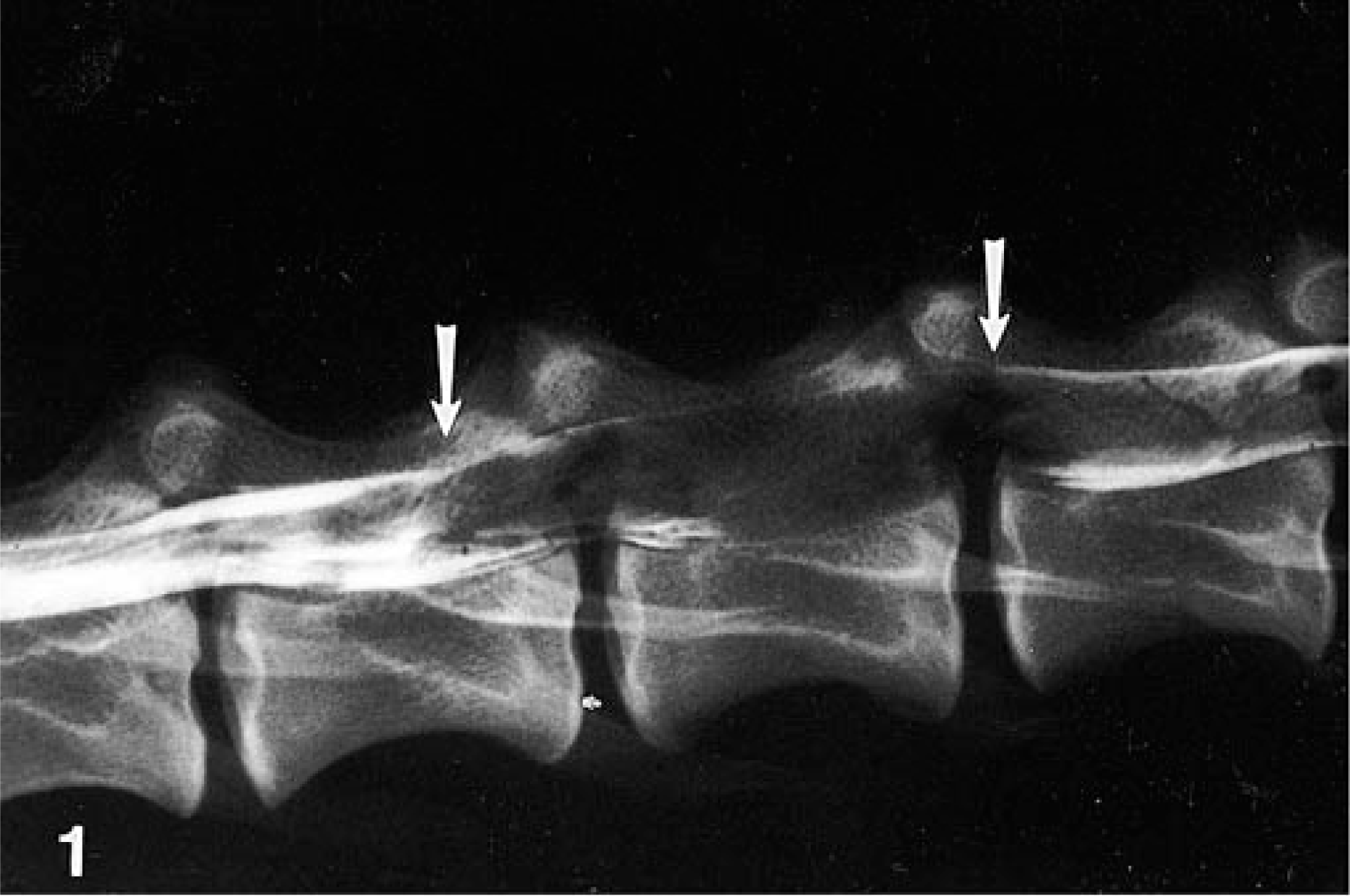
Lumbar spinal cord myelogram; dog. Arrows delineate an area of widening of the spinal cord extending from the third to the fifth lumbar vertebrae. There is complete attenuation of the contrast column over the body of the fourth lumbar vertebra.
At necropsy, gross findings were limited to the spinal cord. An intradural extramedullary mass had produced a locally extensive area of swelling and slight reddening of the spinal cord, 1.5 cm in length, in the region of the 11th and 12th thoracic spinal cord segments (T11–T12). The area of swelling was softer than the adjacent normal-appearing spinal cord. Transverse sections of T11–T12 revealed the mass to have a homogenous pale tan to clear appearance with a slightly gelatinous texture. The mass occupied approximately 30% of the cross-sectional area of the spinal cord. A second similar swelling of the spinal cord, 2.5 cm in length, was present at the fourth lumbar spinal segment (L4) extending caudally to the sixth lumbar spinal segment (L6). Cut section of the swollen cord in this region revealed a similar pale tan to clear and gelatinous extramedullary mass.
The entire spinal cord was immersed for fixation in 10% neutral buffered formalin. Sections from the right and left kidneys, liver, lung, spleen, heart, and masses present at T11–T12 and L4–L6 spinal segments were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin-eosin (HE). Formalin-fixed, paraffin-embedded sections were used for avidin-biotin peroxidase complex staining for cytokeratin, vimentin, and glial fibrillary acid protein (GFAP). The primary antibodies included a 1:200 dilution of rabbit polyclonal antibody against bovine muzzle cytokeratins 5, 6, 8, 13, and 16 (Dako Corporation, Carpinteria, CA); a 1:100 dilution of mouse monoclonal antibody against porcine vimentin (Dako); and a 1:100 dilution of rabbit polyclonal antibody against bovine GFA (Dako).
The HE-stained sections revealed a distinctive dissimilar histologic appearance of the two separate masses. The mass at T11–T12 was intradural extramedullary, unencapsulated, expansile, and composed of three distinct elements: embryonic blastemal cells, a delicate fibrous stroma, and an epithelial component forming tubules and glomeruloid-like structures (Fig. 2). The majority of the mass was composed of dense sheets of polygonal blastemal cells. The blastemal cells had variably distinct cell borders, scant eosinophilic cytoplasm, and an irregular to oval nucleus that contained finely stippled basophilic chromatin and one or more acidophilic nucleoli. In multifocal areas the sheets of blastemal cells were supported by a delicate stroma composed of spindle cells with indistinct cell borders, a scant to moderate amount of streaming eosinophilic cytoplasm, and an oval to elongate nucleus (Fig. 3). The epithelial component was characterized by cuboidal cells arranged in tubules or rosettes. These cells occasionally formed arrangements that resembled fetal renal glomeruli with rosettes of cuboidal cells surrounded by a mantle of flattened cells. Mitotic figures were common (1–3/hpf) among the population of blastemal cells throughout the mass. White matter adjacent to the neoplastic infiltrates was vacuolated, with axonal spheroids present. Neoplastic cells were observed dissecting nerve roots, separating them into bundles of fibers. Small perivascular aggregates of neoplastic cells were present within both the white and gray matter of the adjacent spinal cord.
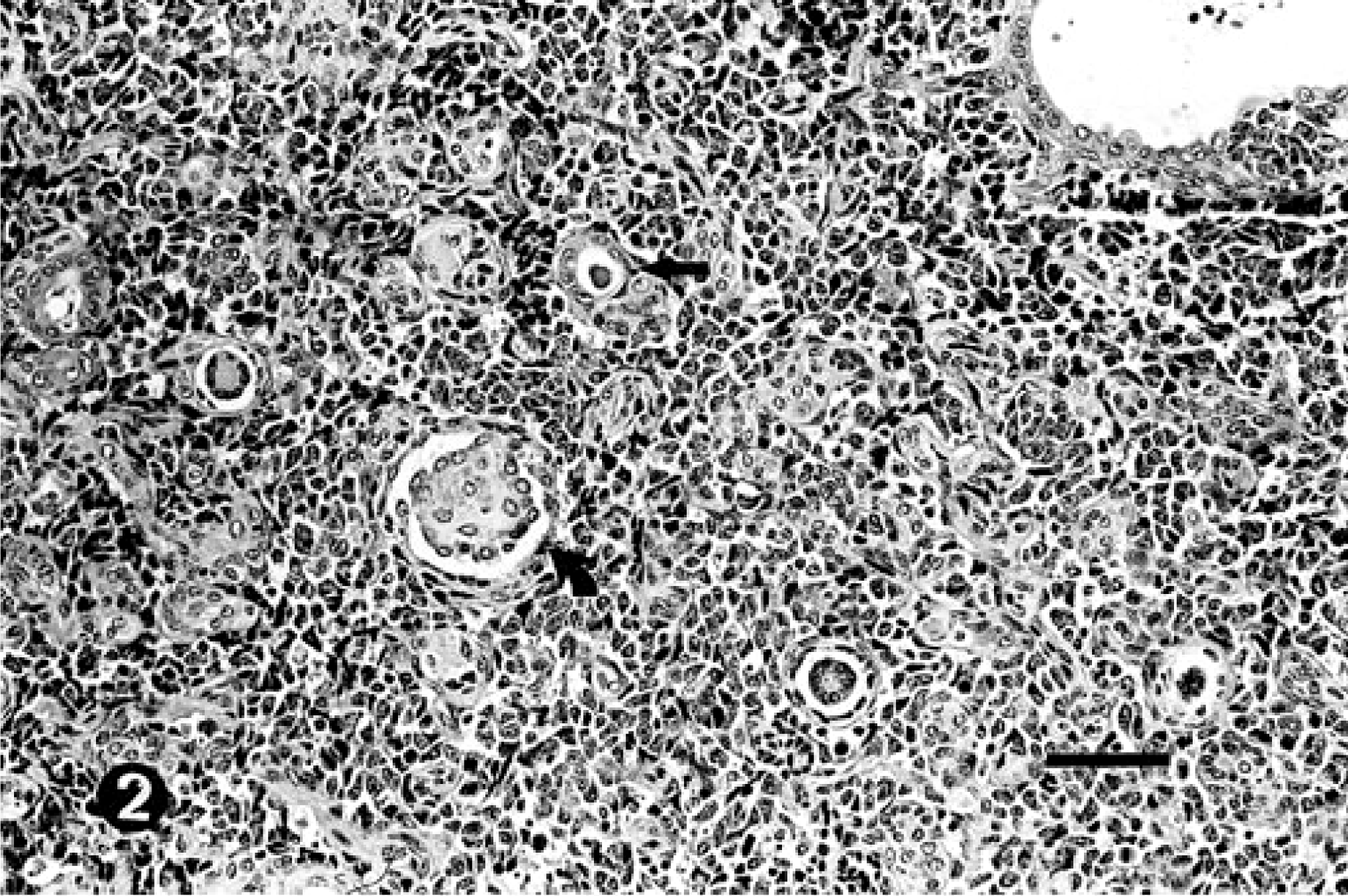
Spinal cord nephroblastoma at thoracic vertebrae T11–T12; dog. The mass is composed of three distinct elements: dense sheets of polygonal blastemal cells, a delicate fibrous supporting stroma, and an epithelioid component forming tubules (straight arrow) and structures resembling fetal glomeruli (curved arrow). HE. Bar = 60 μm.
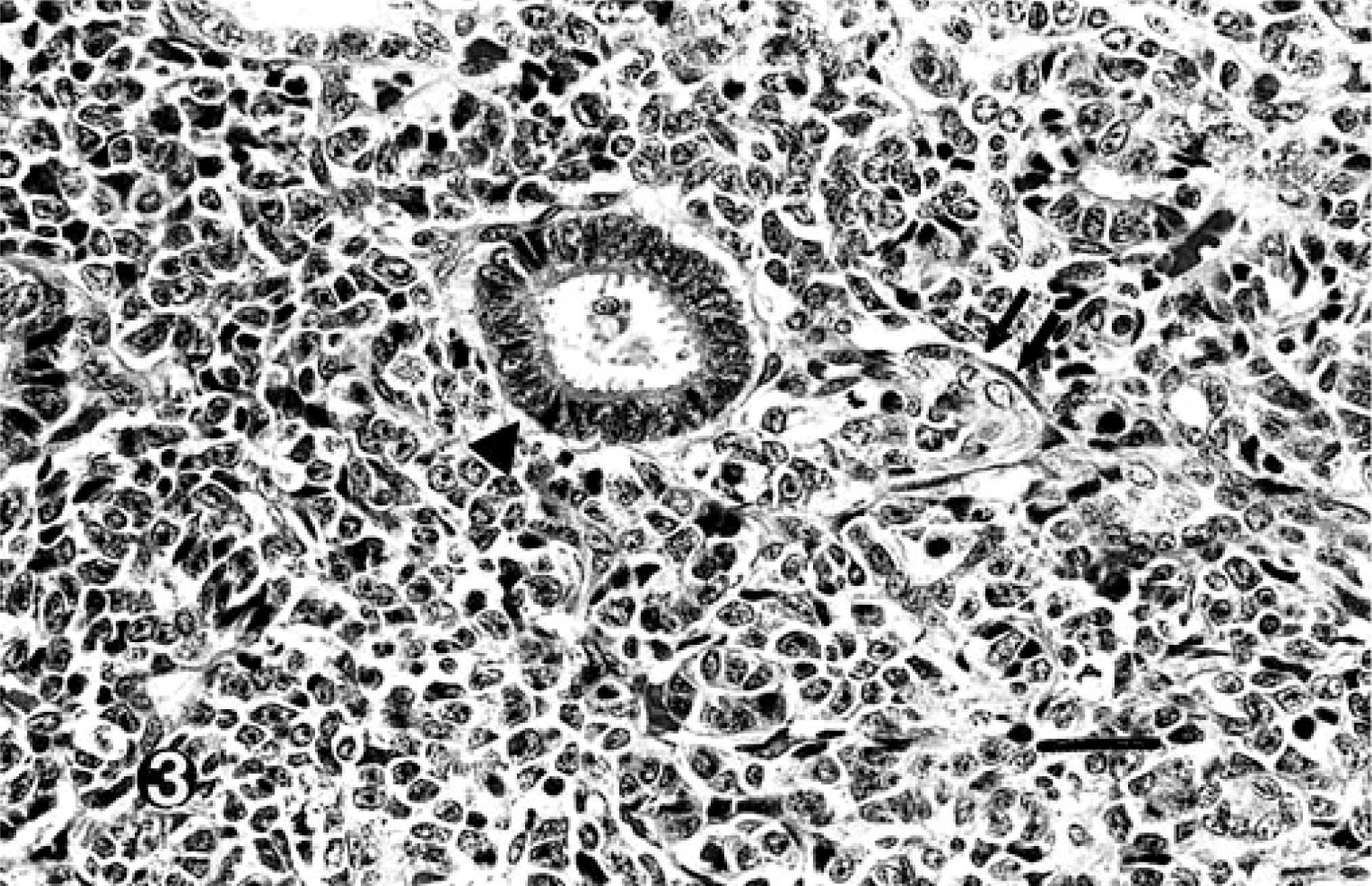
Spinal cord nephroblastoma at thoracic spinal segments T11–T12; dog. High-magnification view of polygonal blastemal cells, the supporting fibrous stroma (double arrows), and a single tubule (arrow head). HE. Bar = 30 μm.
The mass at L4–L6 was also intradural extramedullary, unencapsulated, and expansile. Unlike the mixed pattern of the neoplastic cells in the mass at T11–T12, the population of cells at this site (L4–L6) was more monomorphic, being composed almost entirely of sheets of the undifferentiated round blastemal cells supported in some areas by a delicate fibrous stroma similar to the mass at T11–T12. Only rare tubules were formed, and no glomerular component was identified at this lumbar site. Mitotic figures were more prevalent at this site (2–5/hpf), and the amount of fibrous stroma was increased. Neoplastic cells infiltrated the subarachnoid space and nerve roots and formed perivascular aggregates in the white and gray matter similar to the thoracic mass. Axonal degeneration in the spinal cord white matter adjacent to the mass was similar to the thoracic site. Neoplastic cells were not present in sections of kidney examined histologically. Based on the histologic appearance of the spinal masses, the diagnosis was T11–T12 spinal cord nephroblastoma with presumed dissemination to L4–L6. Immunohistochemical staining of the two masses revealed negative GFAP reactivity of all neoplastic cell types. Blastemal cells and the supporting fibrous stroma in both masses demonstrated positive immunoreactivity for vimentin. Epithelioid component cells forming the tubular structures in both masses demonstrated positive immunoreactivity for cytokeratin (Fig. 4).
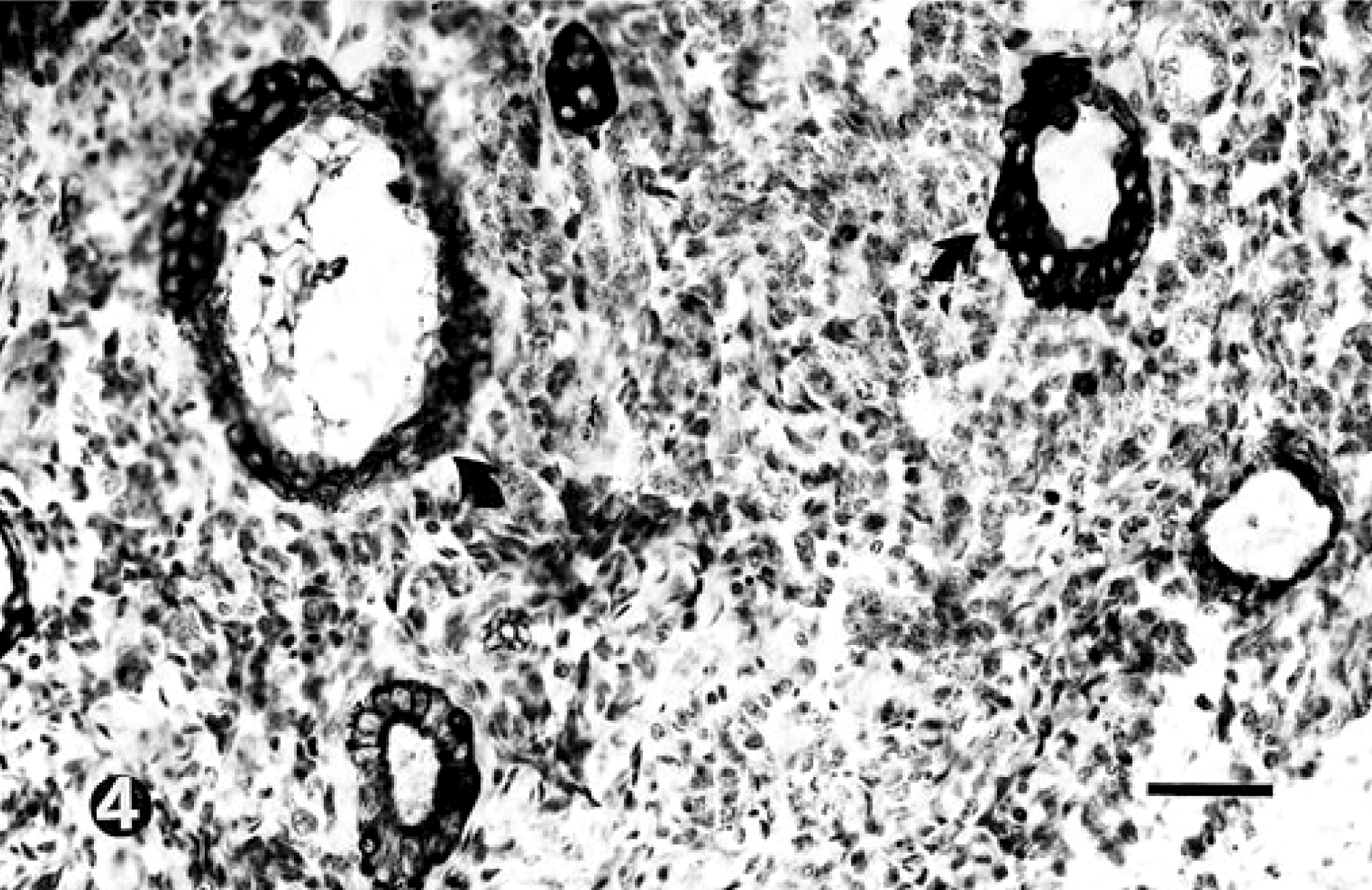
Spinal cord nephroblastoma at thoracic spinal segments T11–T12; dog. Strong positive cytokeratin immunoreactivity of epithelioid cells forming tubules (curved arrows). Avidin-biotin peroxidase staining, Mayer's hematoxylin counterstain. Bar = 30 μm.
Spinal cord nephroblastomas are thought to arise from ectopic embryonic remnants of renal tissue.3 The clinical features, histologic appearance, and immunohistochemical staining characteristics of spinal cord nephroblastomas support the ectopic renal origin. Spinal cord nephroblastomas occur in young animals and consistently occur in the region of the spinal cord adjacent to the kidney (T10–L2).2–4 Histologically, spinal cord nephroblastomas are similar to renal nephroblastomas in that they are composed of a poorly differentiated blastemal component, a mesenchymal or stromal component, and an epithelial component that may form tubules or glomeruloid structures.3,9,10 The location of the cranial mass (T11–T12) and histologic appearance of both tumors described in this case are consistent with the classic description of nephroblastoma.
The immunohistochemical staining characteristics of canine spinal cord nephroblastoma provide the strongest evidence to support the ectopic renal origin of these tumors. Baumgartner and Peixoto1 demonstrated that canine spinal nephroblastomas lack immunoreactivity for (GFAP). In contrast, canine spinal cord nephroblastoma is immunoreactive for antibody to polysialic acid, which is expressed by embryonic kidney cells and the human renal nephroblastoma.7,9 Recently a gene, the Wilm's tumor gene, has been identified in human nephroblastoma.6 The Wilms' tumor gene protein product, WT1, has been identified by immunohistochemistry in a canine spinal cord nephroblastoma, further substantiating classifying these neoplasms as renal in origin.6 Immunohistochemical staining for polysialic acid and WT1 was not performed on the tumors in this case. Canine renal nephroblastomas demonstrate distinct cytokeratin and vimentin immunoreactivity patterns that reflect the varied composition of these tumors. Blastemal and stromal components of renal nephroblastoma show positive immunoreactivity for vimentin, while epithelial components forming tubules show positive immunoreactivity for cytokeratin.8,10 The immunohistochemical staining characteristics and pattern of the tumors in this case mirror those previously reported from renal nephroblastomas.
Spinal nephroblastoma must be differentiated histologically from several other neoplasms, including primitive neuroectodermal tumors, poorly differentiated astrocytomas, and, most importantly, ependymomas. Ependymomas are tumors of the brain or spinal cord that arise from ependymal cells lining the ventricular system or central canal.5 Ependymomas form perivascular pseudorosettes and true rosettes similar to the tubular structures seen in the masses in this dog, however, cells forming rosettes in ependymomas are ciliated.5 Ciliated cells were not identified in the masses in this dog. Ependymomas classically infiltrate ventricular spaces or plug the central canal of the spinal cord. No connection of the tumor cells to the central canal was seen on any of the sections studied in these two extramedullary mass lesions.
Baumgartner and Peixoto1 used a battery of immunohistochemical stains to differentiate cord spinal nephroblastomas and ependymomas. Spinal cord nephroblastomas were consistently positive for cytokeratin immunoreactivity while ependymomas were consistently negative.1 In this dog, immunohistochemical staining of the masses revealed positive immunoreactivity for cytokeratin. The positive cytokeratin immunoreactivity of the masses in this dog also rules out the differential diagnoses of a primitive neuroectodermal tumor or poorly differentiated astrocytoma.
This case differs from all previous reports of canine spinal cord nephroblastoma in which a solitary tumor was found between T10 and L2. It is possible that both tumors present in this dog arose from two separate ectopic renal remnants, although a solitary spinal cord nephroblastoma at L4–L6 has not been recorded. Rather, we suggest that the caudal site represents a metastatic focus from the cranial mass. The cranial mass (T11–T12) had a classic histologic appearance with formation of solid blastema, epithelial tubules and rosettes, and glomeruloid-like structures. In contrast, the caudal mass (L4–L6) was almost entirely composed of a monomorphic population of less differentiated blastemal cells with a higher mitotic index. The presence of neoplastic cells infiltrating nerve roots and the perivascular location in the spinal parenchyma are unusual features for spinal cord nephroblastoma and indicate an aggressive growth pattern of the neoplasm in this dog.
Dissemination may have occurred through the subarachnoid space due to the close relationship of the intradural extramedullary mass with the subarachnoid space. A second possibility is that metastasis occurred along or within parenchymal blood vessels as perivascular accumulations of neoplastic cells were observed at both sites. Subsequent examination of sections of the spinal cord segments (L1 and L2) between the two masses revealed no neoplastic cells within the subarachnoid space, blood vessels, or lymphatic vessels.
To our knowledge, this is the first report of a multifocal or metastatic spinal nephroblastoma in a dog. Features unique to this case (compared with previously described spinal nephroblastomas) include the multifocal location with a caudal mass distant to the classic T10–L2 spinal segments, the less differentiated nature of the L4–L6 mass, the presence of neoplastic cells infiltrating and separating nerve root fibers, and the presence of perivascular accumulations of neoplastic cells within the spinal parenchyma. The unusual features of the spinal nephroblastomas in this case may be explained by an aggressive but moderately well-differentiated neoplasm at T11–T12 giving rise to a second less differentiated metastatic focus at L4–L6.
