Abstract
Two weeks before dying of congestive heart failure, a juvenile black rhinoceros (Diceros bicornis minor) received a single low dose of doxorubicin as part of combination chemotherapy for acute lymphoblastic leukemia. Diffuse hemosiderosis was present at necropsy in a pattern indicative of dietary iron overload, but unique iron-positive degenerative lesions were found in isolated myocardiocytes. Serum analyses revealed hyperferremia, 87% transferrin saturation, and 5- to 10-fold elevations in ferritin concentration, reflecting markedly increased tissue iron stores. Since both toxic and therapeutic effects of anthracyclines are mediated by formation of reactive free radicals via iron-catalyzed reactions, these observations suggest that iron overload may have enhanced myocardial susceptibility to cardiotoxic effects of doxorubicin. Impairments in other myocardial antioxidant defenses, such as deficiencies in catalase and glutathione S-transferase that are known to exist in rhinoceros erythrocytes, may have been underlying factors contributing to an inherent sensitivity of rhinoceros tissues to oxidant-induced injury.
Keywords
A 21-month-old southern black rhinoceros (Diceros bicornis minor) (Studbook No. 573), born in captivity at Fossil Rim Wildlife Center, was diagnosed with acute lymphoblastic leukemia on the basis of leukocytosis (>49,000/μL, >90% blasts) and cell morphology supported by cytochemistry and flow cytometry.9 Combination chemotherapy included 200 mg of the anthracycline, doxorubicin (Adriamycin, Adria Laboratories, Dublin, OH 43017) 13 days before death. This was followed by distinct clinical improvement, including resolution of palpable lymphadenopathy and leukocytosis and disappearance of blasts from peripheral blood films. Despite multiple measures, including transfusions and systemic antibiotic and antifungal therapy, the calf suddenly became lethargic and died of acute cardiorespiratory failure.
Principal findings at necropsy were those classically associated with congestive heart failure, including cardioventricular dilatation and severe acute and chronic passive congestion of lungs, liver, and spleen. No evidence of residual leukemic infiltrates was found in any organ. Numerous macrophages, heavily laden with hemosiderin, were present throughout pulmonary alveolar septa and within edematous alveolar spaces (“heart failure cells”). Moderate hemosiderosis also involved the spleen, the liver, many lymph nodes, and the duodenal villi. In most organs, hemosiderin appeared to be confined to the reticuloendothelial system, but there was an abundance of fine iron-positive granules within periportal hepatocytes accompanied by dense globular aggregates in Kupffer cells.
The ventricular myocardium had foci of edema and degenerative changes consistent with doxorubicin cardiotoxicity, including perinuclear vacuolization, disruption of microfibrils, and myocytolysis heralded by minute accumulations of mixed inflammatory cells. In addition, there were distinctive degenerative changes in isolated myocardiocytes with fine iron-positive granules in orderly arrangements along longitudinal microfibrils and transverse plates (Fig. 1). In some of these granular aggregates, von Kossa stains for calcium were also slightly positive. Aside from these microlesions, there was no evidence of cardiac parenchymal iron deposition such as that seen in hemochromatoses of humans and other species.
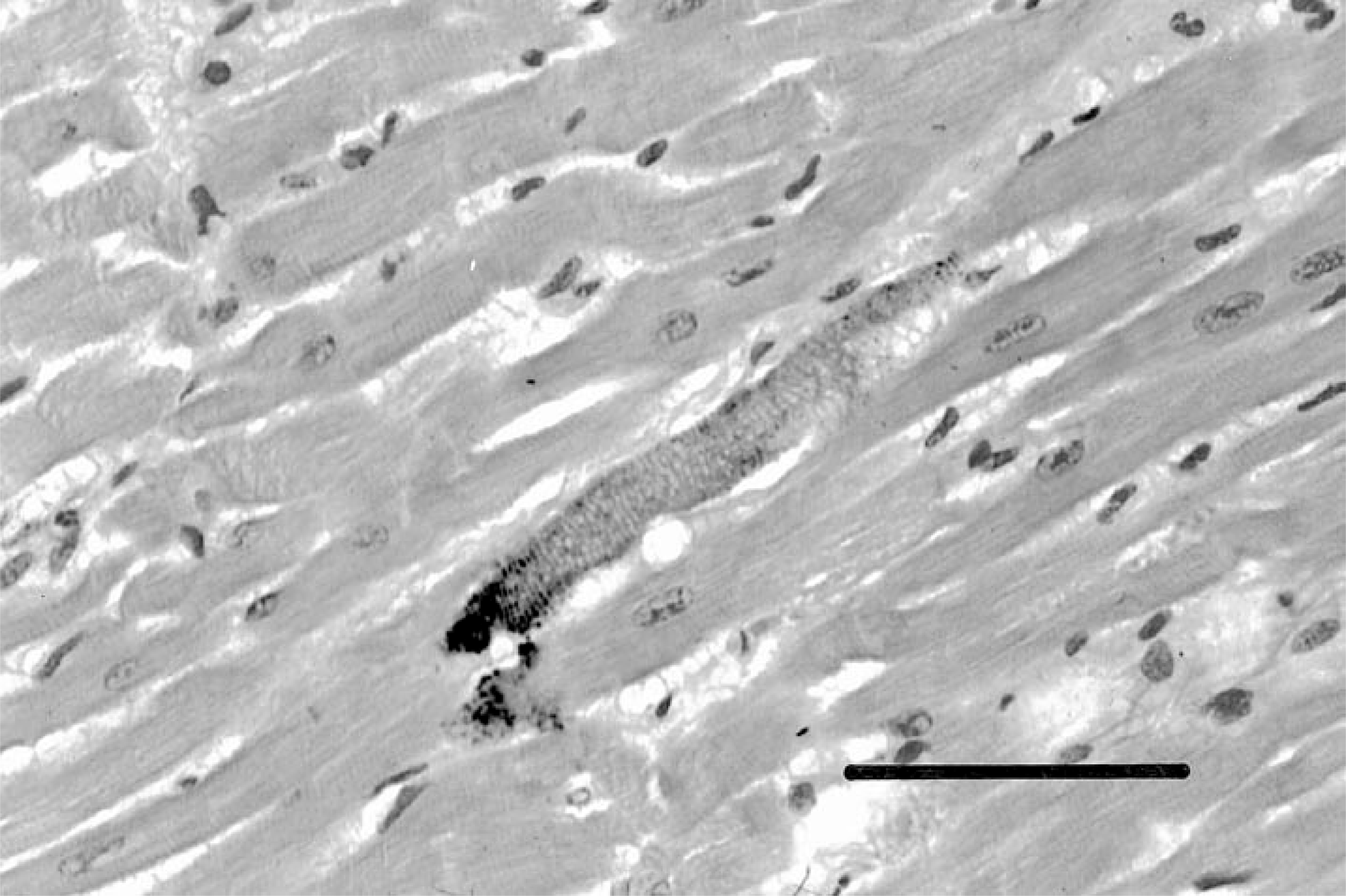
Photomicrograph of ventricular myocardium showing distinctive degenerative lesions characterized by fine iron- and calcium-positive granules deposited along myofibrils at transverse bands. Perl's Prussian blue stain for ferric iron. Bar = 100 μm.
Serum concentrations of pertinent analytes are shown in Table 1, in which values for the affected calf are compared with means from a number of different rhinoceros, equine, and human control populations. Six free-ranging D. b. minor in Malilangwe, Zimbabwe, provided the best standard for comparison among these groups. Iron analyte values for this group were comparable to those for normal equine and human controls, as well as for captive white (Ceratotherium simum) and Asian greater one-horned (Rhinoceros unicornis) rhinoceroses.
Serum analytes in affected black rhinoceros compared with those in control populations.
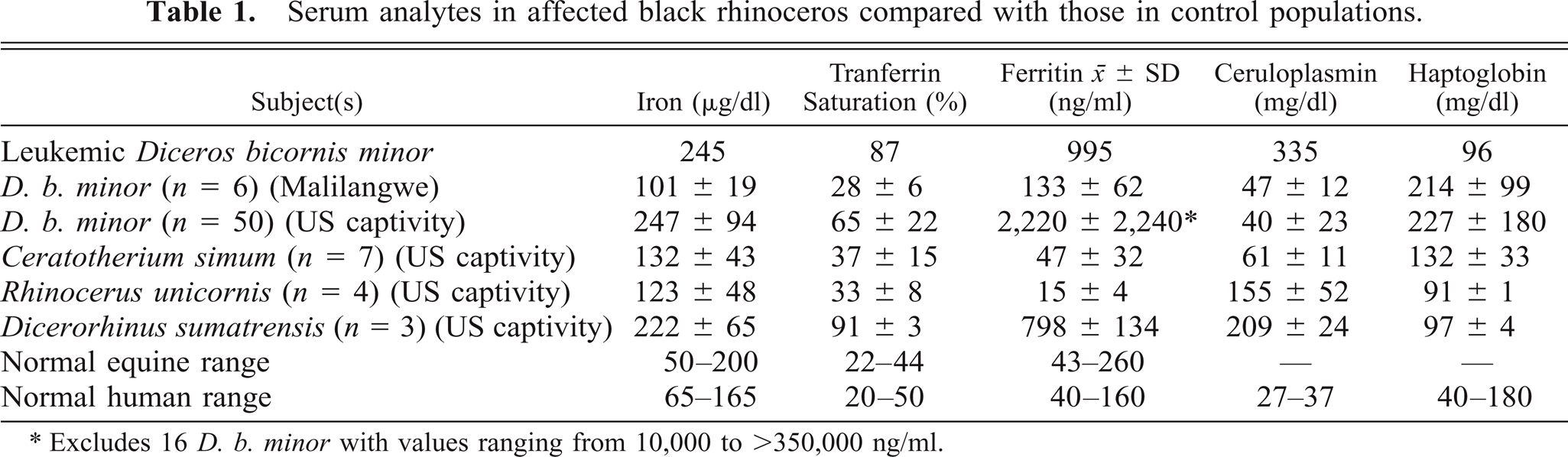
∗Excludes 16 D. b. minor with values ranging from 10,000 to >350,000 ng/ml.
In comparison, the affected calf had two- to threefold higher serum iron and transferrin saturation and almost 10-fold elevations in serum ferritin, the latter measurement being the best indicator of total body iron stores other than quantitative tissue analyses. These values were comparable in magnitude to elevated levels observed in over 50 other captive Diceros 5 and were clear indicators of significant iron overload. The other primary browser, Dicerorhinus sumatrensis, also exhibited significantly elevated iron analytes compared with white and Indian rhinoceroses, the two predominantly grazing species.
Plasma haptoglobin levels of almost 100 mg/dl ruled out active hemolysis as a source of excess iron deposition. Additional documentation of significant iron overload was provided by trace metal analyses of necropsy tissues (E. S. Dierenfeld, personal communication). Hepatic iron concentration was increased to 943 μg/g wet tissue (normal equine range = 100–300 μg/g), and liver copper was reduced to 5 μg/g (mean for 28 captive adult D. bicornis = 83 μg/g). Despite low liver copper, serum ceruloplasmin was somewhat elevated, perhaps because it also reacts as an acute-phase inflammatory response protein.
Diffuse hemosiderosis has been observed frequently in necropsies of captive black rhinoceroses. In most cases, this has been viewed as an incidental finding and interpreted as evidence of previous hemolytic episodes or subclinical hemolysis, since hemolytic anemia is among the more common problems affecting this species in captivity. In 1992, Kock et al.3 cited the absence of hemosiderosis in necropsies of six free-ranging black rhinoceroses as evidence that hemolytic anemia did not occur in the wild. They also noted that morphologic signs of excessive iron deposition began to appear at necropsy after only a few weeks in confinement, increasing thereafter in direct proportion to time in captivity. In a second seminal report, Smith et al.10 observed increased iron stores in captive black, but not white, rhinoceroses, and they provided quantitative evidence that both hepatic iron content and serum ferritin concentrations increased significantly with age or time in captivity.
In more recent necropsies, without exception, we have found both the intensity and the distribution of hemosiderin deposition to be inconsistent with solely hemolytic origins, and biochemical analyses of sera and tissues confirmed that a clinically significant, dietary-based, iron-overload syndrome currently exists in captive populations of both black and Sumatran rhinoceroses.5
Hemosiderin deposits were similar in amounts and distribution to those of other captive Diceros of comparable age, but the myocardial lesions (Fig. 1) were not those classically associated with parenchymal iron accumulation in hemochromatosis, nor did they correspond to those that we have observed in other rhinoceroses with hemosiderosis. In addition, specific changes consistent with anthracycline cardiotoxicity were present, despite the fact that the single, subtherapeutic dose of doxorubicin was equivalent to only 17% of the cumulative cardiotoxic dose in humans as calculated by allometric scaling, suggesting an inordinate sensitivity to this drug's effects.
The crucial pathophysiologic event inducing cardiomyopathy with congestive heart failure is thought to be generation of hydrogen peroxide in excess of myocardial detoxification capacity.1 Relative catalase deficiency likely contributes to this impairment, since heart muscle normally has only 2–4% of the catalase activity found in liver or kidney, and black rhinoceros erythrocytes contain only 2–5% of the catalase activity present in human red cells, significantly less than those of three other species of rhinoceroses.6–8 The inordinate sensitivity of rhinoceros erythrocytes to oxidant-induced injury may be shared by other tissues, thereby contributing to some of the multiple disorders commonly affecting this species in captivity.8
Both antineoplastic activity and toxic effects of anthracyclines depend on their ability to generate reactive free radicals by processes in which transition metals such as iron and copper play special roles.2 Hydroxyquinone nuclei of anthracyclines are capable of abstracting iron even from ferritin, forming complexes that catalyze a broad range of redox reactions. Single drug molecules can generate multiple toxic free radicals in chain reactions that continue to recycle as long as the basic reactants (superoxide and peroxides) remain available.4 As a consequence, the clinical toxicity of anthracyclines is increased in the presence of excess iron, and, conversely, reduction of free iron by chelation reduces production of hydroxyl radicals and their toxic effects.1 Cardiac function is altered by direct damage to sarcoplasmic reticulum and mitochondria by lipid peroxidation of organelle membranes, disrupting oxidative phosphorylation, on which the heart depends for ATP, and producing the histopathologic lesions characteristic of anthracycline cardiomyopathy. Myocardial function is perturbed by alteration of calcium balance, and eventually, irreversible membrane injury results in calcium influx, sequestration, and cell death.
The lesions observed in this case contained both iron and calcium in highly distinctive patterns, unlike those seen either in iron-overload syndromes or in conventional anthracycline cardiotoxicity. It seems likely that the unusual constellation of conditions in this case, namely the black rhinoceroses' known susceptibility to oxidant stress,6–8 marked deficiencies of catalase and glutathione S-transferase,6–8 and high availability of iron,5 may have combined to produce and/or enhance myocardial sensitivity to toxic effects of anthracycline. This case emphasizes the need to consider carefully the pharmacology of any drug administered to black rhinoceroses, since their inherent vulnerability to oxidants is compounded by the high iron overloads currently accumulating in captivity.5 Since uptake of excess iron is most likely a consequence of dietary departures from native browse, the same cautions apply to the other predominantly browsing species, Dicerorhinus sumatrensis, in which hemosiderosis at necropsy has also been commonly observed.5
Footnotes
Acknowledgements
We are grateful to Christine Miller, DVM, for assistance with the necropsy; to C. M. Foggin, DVM, for providing invaluable control sera from Zimbabwe; to P. S. Chavey and Dr. G. A. Andrews for assays contracted to the Cellular and Molecular Pathology Laboratory, Kansas State University College of Veterinary Medicine; to Ellen S. Dierenfeld, PhD, Wildlife Conservation Society, for results of quantitative tissue mineral analyses performed at Oregon State University; to the staff and management of Fossil Rim Wildlife Center for their support and assistance; and to I-Hsien Tsu and Misae Nakatani for technical contributions in the UCLA Hematology Research Laboratory. These studies were supported by grants from the International Rhino Foundation, the Morris Animal Foundation, and the L. B. Research and Education Foundation.
