Abstract
There are anecdotal reports of iron storage disease in captive kori bustards (Ardeotis kori), but detailed descriptions of this disease have not been reported. The goals of this retrospective, multi-institutional study were to (1) describe microscopic findings associated with iron accumulation in postmortem tissues of kori bustards and (2) use an adapted grading scale to score iron accumulation and associated hepatic lesions. Tissue sections from 19 adult captive kori bustards (age range 3–28 years; 12 males and 7 females) were evaluated histologically with hematoxylin and eosin, Masson’s trichrome, and Prussian blue stains, and scored for iron accumulation. Hemochromatosis was diagnosed in cases with iron storage (in hepatocytes and/or Kupffer cells) and concurrent parenchymal damage (defined as having both necrosis and fibrosis). Hemosiderosis was diagnosed in animals with evidence of iron storage without necrosis or fibrosis. Ten of the 19 cases (age range 8–27 years; 7 males and 3 females) were diagnosed with hemochromatosis, including 6 with mild disease, 3 with moderate disease, and 1 with severe disease. Histologic evidence of iron accumulation was also identified in kidney, intestinal tract, adrenal gland, and spleen, but there were no associations between severity of iron accumulation in the liver and accumulation in other organs.
Hemochromatosis is defined as increased iron deposition in tissues leading to organ toxicosis. Hemosiderosis is excessive iron deposition in tissues without pathologic sequelae. 1,6 Hemochromatosis, also known as iron storage disease (ISD), is well-documented in a number of animal species, 2,4,7,8,10 –13 including various species of wild and captive birds. 16 Fruit or nectar-eating avian species such as birds of paradise, toucans, mynahs, starlings, lorikeets, and rarely other psittacines may be particularly prone to developing ISD. 8,10,17,18 Affected birds often show no clinical signs until the disease is advanced. Iron storage disease is most often diagnosed at postmortem examination without known clinical correlates. When present, clinical signs in birds include coelomic distension, ascites, labored breathing, and lethargy. Hemochromatosis may cause death in birds due to hepatic failure, which is relatively common in some species such as toucans and lorikeets. 15
Bustards are in the family Otididae. As one of the largest and heaviest extant flying birds, kori bustards (Ardeotis kori) are popular in zoos in North America and Europe. Their natural habitats are the dry savannahs and grasslands of Southern and Eastern Africa. They are omnivorous birds, but consume more animal items than other bustards. 5,9 In the wild, their diet includes small mammals, lizards, snakes, seeds, and berries. In a retrospective study of captive kori bustards, lameness, gastrointestinal parasitism, and wing trauma were the most common clinical conditions, and general trauma was the most common cause of mortality. 5 Iron storage disease has been anecdotally reported in kori bustards, but ISD has not be formally described in this species. 5 The aim of this study was to characterize the histologic findings associated with iron accumulation in captive kori bustards.
Materials and Methods
Study Population and Immunohistochemistry
The pathology archives from 2 institutions (Northwest ZooPath, Monroe, Washington, and National Zoological Park, Washington, DC) were searched for kori bustards that had diagnoses of “hemosiderosis,” “hemochromatosis,” or “iron storage.” Hematoxylin and eosin (HE)-stained sections from formalin-fixed paraffin embedded (FFPE) tissue blocks were reviewed from all cases with the aforementioned diagnoses. Cases were included in the study that had pigment within hepatocytes confirmed as iron with Prussian blue (PB) stain (Ventana Medical Systems). Additional 5-μm-thick sections from FFPE blocks containing liver were stained with Masson’s trichrome (MT; Dako Artisan) to screen for fibrosis. Hepatic sections stained with HE, PB, and MT were scored using a modified grading scheme for iron accumulation in hepatocytes and Kupffer cells, fibrosis, and hepatocellular necrosis. 4 Further details on the modified grading scheme is given below. Complete blood count and blood chemistry values were collated when available.
Grading Scheme Parameters
Iron accumulation was evaluated using PB and based on the distribution of hepatocellular iron within 10 high-power fields (HPFs; 2.37 mm2). Iron accumulation in hepatocytes was grade 0 if no stainable iron was in hepatocytes, grade 1 if iron was limited to periportal hepatocytes, grade 2 if iron was in periportal and midzonal hepatocytes (Suppl. Fig. S1), and grade 3 if iron was in hepatocytes in periportal, midzonal, and centrilobular hepatocytes (Suppl. Fig. S2). Iron accumulation in Kupffer cells was grade 0 if no stainable iron was in Kupffer cells, grade 1 if occasional Kupffer cells contained iron (<5 cells per 10 HPFs; Suppl. Fig. S3), grade 2 if moderate numbers of Kupffer cells contained iron (5–10 cells per 10 HPFs; Suppl. Figs. S4a and S4b), and grade 3 if there was iron deposition in large numbers of Kupffer cells (>10 cells per 10 HPFs or cell aggregation/granuloma formation; Suppl. Figs. S5a and S5b).
Hepatocellular necrosis was evaluated based on the number of necrotic hepatocytes within 10 HPFs. Grade 0 indicated no necrotic hepatocytes in 10 HPFs; grade 1 indicated few necrotic hepatocytes (<5 necrotic cells per 10 HPFs; Suppl. Fig. S6); grade 2 indicated moderate numbers of necrotic hepatocytes (5–10 necrotic cells per 10 HPFs); and grade 3 indicated large numbers of necrotic hepatocytes and coalescing foci of necrosis (>10 necrotic cells per 10 HPFs; Suppl. Fig. S7). As in other iron deposition studies, the histologic sections of liver varied in size, so regions with the most necrosis were used to count 10 consecutive HPF. 7
Fibrosis was scored as grade 0 if there was no identifiable increase in collagen within portal regions (Suppl. Fig. S8); grade 1 if increased collagen deposition expanded portal regions (Suppl. Fig. S9); grade 2 if increased collagen deposition extended to midzonal regions (Suppl. Fig. S10); and grade 3 if bridging fibrosis (connecting portal regions) was present (Suppl. Fig. S11).
Based on the aforementioned grading scheme, hepatic sections were classified as having hemosiderosis (iron accumulation with no necrosis or fibrosis) or hemochromatosis (iron accumulation with either necrosis or fibrosis). Hemochromatosis was scored as the cumulative sum of all values (iron accumulation in hepatocytes and Kupffer cells, necrosis, and fibrosis). Mild was defined as a score of 8 or less, moderate as a score of 9 and 10, and severe as a score of 11 and 12.
Additionally, HE sections of available tissues were evaluated for each animal, and histologic lesions were recorded for extrahepatic tissues.
Results
Study Population
Nineteen kori bustards in human care that were confirmed histologically to have increased hepatic iron using Prussian blue stain were included in this study. Causes of death or euthanasia included chronic musculoskeletal lesions (such as recurrent lameness or osteoarthritis) (n = 9), sepsis (n = 4), trauma (n = 4), and unknown (n = 2). The cases were received between October 1993 and November 2019 by 2 institutions. Animal ages ranged from 3 to 28 years and included 12 males and 7 females.
Histologic Findings
Of the 19 cases with hepatic iron accumulation, 10 (age range 8–27 years; 7 males and 3 females) were diagnosed with hemochromatosis, including 6 with mild disease (ages 8–26 years; average age 17 years; 4 males and 2 females), 3 with moderate disease (age range 9–27 years; average age 18 years; 3 males), and 1 with severe disease (age 24 years; female). All cases of hemochromatosis had iron accumulation in hepatocytes and Kupffer cells. The grade was ≥2 in all cases of hemochromatosis (Figs. 1–4). Mild extramedullary hematopoiesis was occasionally present, but was considered a background finding. Twelve cases had hepatocellular necrosis (age range 6–27; average age 17 years; 8 males and 4 females), which was mild in 9 cases (age range 6–27; average age 17 years; 6 males and 3 females) and severe in 3 cases (age range 8–24; average age 17 years; 2 males and 1 female; Fig. 5). In the MT-stained sections, 10 cases had fibrosis (age range 8–27; average age 18 years; 7 males and 3 females) which was mild in 7 cases (age range 8–26; average age 17 years; 5 males and 2 females), moderate in 1 case (age 27 years; male), and severe in 2 cases (ages 9 and 24; average age 17 years; 1 male and 1 female; Fig. 6 and Table 1).
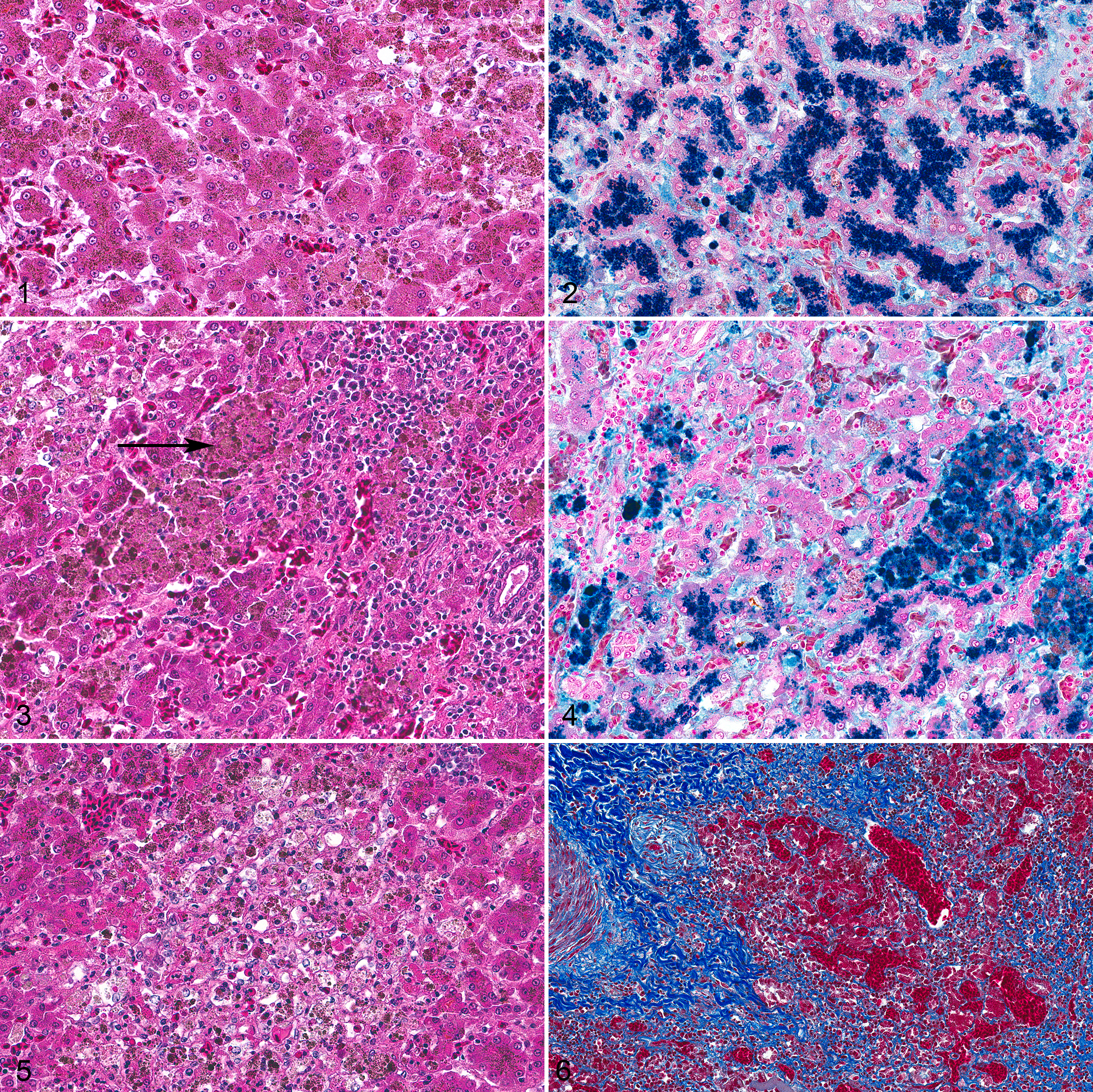
Liver, bustard, case 4.
Signalment and pathologic findings in 19 kori bustards with hepatic iron accumulation. The data show histologic grades for iron deposition, fibrosis, and necrosis. 4
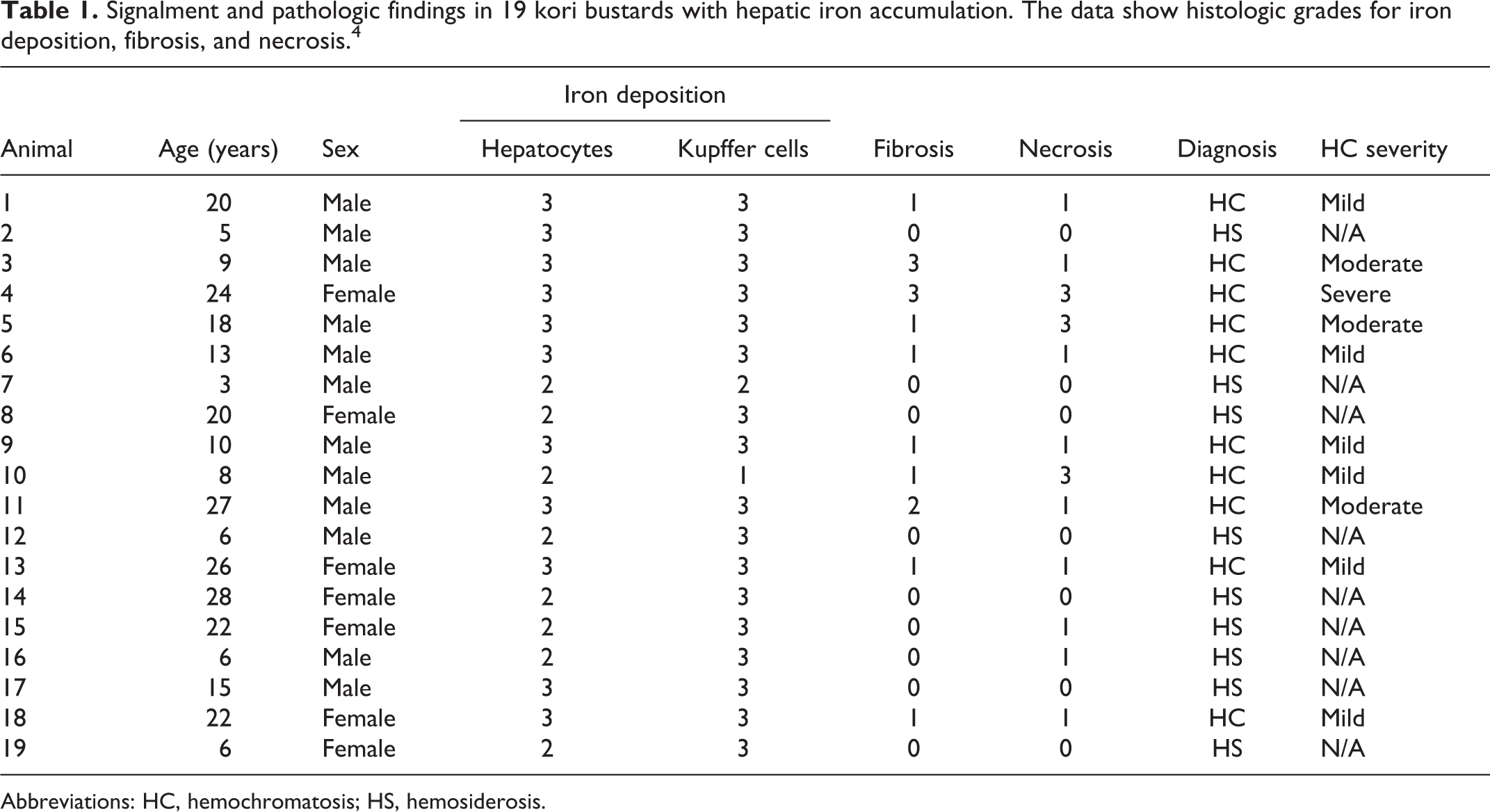
Abbreviations: HC, hemochromatosis; HS, hemosiderosis.
A variety of complete blood count and serum chemistry values were available for 13/19 cases (Suppl. Table S1). Multiple different labs were used to generate these values. 3,14 There was no correlation between the severity of hemochromatosis and changes in liver-associated values (aspartate transaminase, cholesterol, albumin, or total protein). Bile acids were not evaluated in any cases.
All 19 cases had evidence of renal iron, including brown granular intracytoplasmic pigment in tubular epithelium, predominately in the proximal renal tubules. Nine of 19 cases with intestine available had evidence of enteric iron within macrophages in the lamina propria of the small intestine (age range 3–28; average age 15 years; 6 males and 3 females) but not within enterocytes. All 12 cases with available adrenal glands had evidence of iron within interrenal cells (age range 6–28; average age 19 years; 6 males and 6 females). Eleven of 17 cases with spleen available had evidence of iron in the red pulp and less frequently white pulp macrophages with phagocytized brown granular material (age range 3–28; average age 17 years; 6 males and 5 females; Suppl. Table S2).
Discussion
This study identified 9 cases of hemosiderosis and 10 cases of hemochromatosis in kori bustards in human care. The diet of captive bustards in US zoological institutions varies, but generally includes a commercial meat-based diet with regular supplementation with fresh fruits and vegetables. 5 Kori bustards in human care are speculated to be at risk for developing hemosiderosis because they may lack nutritional components present in their natural diets. 9 For instance, wild bustards may ingest low levels of iron or there may be compounds which chelate iron in their natural diets, like tannins or phytates. Bustards inherently may have an increased ability to absorb available dietary iron, and are thus susceptible to iron overload from diets prepared in human care.
In the present study, increased iron deposition in hepatocytes and Kupffer cells (determined by Prussian blue stain) did not correlate with increasing evidence of hepatic damage including hepatic necrosis, fibrosis, or changes in blood chemistry hepatic values. The packed cell volume and/or red blood cell count was low in a few of the cases indicating anemia, but this did not correlate with more severe histologic lesions or more hepatic damage. Therefore, it was assumed that severity of disease does not lead to disruption of hematopoiesis. These findings should be interpreted conservatively, as quantitative hepatic iron levels were not performed, and iron deposition may have been more severe or less severe in portions of the livers that were not available for histologic evaluation. Additional limitations involved the use of a grading scheme in which there is the potential for variability between pathologists. The use of other factors like evidence of hepatic degeneration or swelling to indicate hepatic disease was considered but it was determined that hepatocellular necrosis was the most reproducible for all pathologists involved in the grading. Unfortunately, serum chemistry values for hepatocellular damage (eg, hepatocellular leakage enzymes: alanine transaminase, aspartate transaminase, sorbitol dehydrogenase, glutamate dehydrogenase, lactate dehydrogenase) were rarely available in these cases and correlations with iron accumulation were not possible.
Little is known about hepatic iron (trace element) levels in kori bustards. The only literature regarding iron processing in this species is a comparison of circulating iron, total iron binding capacity, and percent transferrin saturation by Marrow et al, 9 who suggested that kori bustards in human care may have a slightly higher saturation of transferrin than their wild counterparts. However, these findings were not statistically significant. Additionally, Marrow et al recommended a combination of hepatic iron levels and histopathologic evaluation to evaluate iron storage disease in this species. Quantitative iron analysis was not done in this study.
Many of the cases in this study had suspected iron deposition in the spleen, renal tubular epithelium, adrenal cortical cells, and intestinal tract based on evaluation of HE-stained sections. In these tissues, there were no histologic lesions associated with the presumed iron accumulation and there was no association between the amount of hepatic iron storage and storage in these additional organs.
The most common causes of death or euthanasia in this population of bustards were sequelae to traumatic injury such as recurrent lameness, chronic musculoskeletal lesions, or sepsis. These findings correlate with the study by Hanselmann et al 5 who found that trauma is an important cause of morbidity and mortality in captive kori bustards and that hemosiderosis or hemochromatosis are common incidental findings but rarely appear to lead to the death of the animal.
Conclusions
This study employs a novel grading system to qualitatively evaluate hepatic changes in kori bustards with evidence of hepatic iron accumulation. The major finding was that increased iron deposition did not correlate with increasing evidence of hepatic damage including hepatic necrosis, fibrosis, or changes in blood chemistry values. In future studies, if this grading scheme is used, the addition of quantitative measurements would be beneficial to assess for correlations between hemochromatosis scores, hepatic damage, and hepatic iron levels.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211035386 - Hepatic lesions associated with iron accumulation in captive kori bustards (Ardeotis kori)
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211035386 for Hepatic lesions associated with iron accumulation in captive kori bustards (Ardeotis kori) by Sarah K. Cudd, Michael M. Garner, Andrew N. Cartoceti and Elise E. B. LaDouceur in Veterinary Pathology
Footnotes
Authors’ Note
The views expressed in this article are those of the authors and do not reflect the official policy of the Department of the Army/Navy/Air Force, Department of Defense, or US government.
Acknowledgements
The authors thank the following institutions and individuals for contributing case material to this study, including Northwest ZooPath and the Animal Care Sciences and Wildlife Health Sciences staff at the Smithsonian National Zoological Park. We thank the histology staff at the Joint Pathology Center and Walter Reed National Military Medical Center for preparation of slides, IHC and special stains, especially W. McNeil, S. Tamer, S. McNair, K. Gathers, A. Brown, and A. Cherilus. We are also grateful to Leroy Brown of Histology Consulting Service for Slide preparation, and Cathy Minogue of Northwest Zoopath for data retrieval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
