Abstract
A 22-week-old female 129/SvEv mouse suddenly died in the context of an experiment aimed at defining the efficacy of valproic acid in a mouse model of PML/RARα-induced acute myeloid leukemia. Histologic analysis confirmed the mouse as being affected by a progressive myeloid leukemia, with infiltration of the spleen, bone marrow, liver, kidneys, and lungs. Variably sized intravascular clumps (emboli) of dense basophilic material admixed with necrotic or lytic neoplastic cells were also observed in multiple organs. A positive reaction to Feulgen and Hoechst stain confirmed the high content in chromatin of these basophilic emboli. Cleaved caspase-3 activity was demonstrated both in the leukemic infiltrates and among the intravascular necrotic or lytic neoplastic cells accompanying the basophilic emboli. A diagnosis of acute tumor lysis syndrome related to therapy-induced massive necrosis and/or apoptosis of leukemic cells with subsequent dissemination of emboli of chromatin was proposed.
Keywords
Background
Mouse models of hematopoietic tumors include spontaneously occurring lymphoid and myeloid neoplasms, with strain-specific incidence and phenotypes in a variety of inbred mice strains, as well as an ever-growing plethora of genetically engineered mouse models of lymphomas, myeloid leukemias and other hematopoietic cancers.7, 13
Acute promyelocytic leukemia (APL) is a subtype of human acute myeloid leukemia (AML), currently listed in the World Health Organization classification within the group of acute myeloid leukemias associated with recurrent cytogenetic abnormalities.21 The pathogenesis of APL resides in chromosomal translocations that involve retinoic acid receptor α (RARα) and different fusion partners.11 The molecular pathogenesis of APL has been modeled in mice with different experimental approaches, including transgenics,1, 16, 18 retroviral gene transfer of PML/RARα in murine hematopoietic progenitors,12 and “knock-in.”22 Mouse models of lymphoid and myeloid neoplasms are useful tools to explore and dissect the molecular and genetic mechanisms of hematologic malignancies in humans and may serve as a critical platform to test compounds for novel therapeutic targets.
History, Gross Findings, and Laboratory Results
The findings we describe were observed in a mouse (22-week-old female, background 129/SvEv, Harlan, Italy) in the context of a pilot experiment aimed at defining an optimal treatment schedule for subsequent studies demonstrating the efficacy of a histone deacetylase (HDAC) inhibitor (valproic acid [VPA]) in mouse models of PML/RARα and AML1/ETO induced acute myeloid leukemias.5
The mouse was part of a group (n = 4) of secondary recipients injected intravenously with spleen cells harvested from other syngeneic mice that had previously developed an acute promyelocytic-like leukemia induced with the strategy of retroviral transduction of a PML/RARα construct into murine hematopoietic progenitors.12 Five days after the spleen cells were injected, peripheral blood was collected from all mice to confirm severe leucocytosis and the presence of a significant percentage (>20%) of circulating blasts. Upon confirmation of the leukemic condition, the mice were treated intraperitoneally (IP) with VPA (400 mg/kg twice daily). One of the four treated mice died suddenly between the fourth and fifth day of therapy. A complete necropsy was performed, and, except for minimal subcutaneous, muscular, and peritoneal hemorrhages at the sites of the IP injections, no gross lesions indicating a possible cause of death were detected. Mild splenomegaly was consistent with the development and progression of myeloid leukemia. Spleen, liver, kidneys, mesenteric lymph nodes, intestine, lungs, heart, brain, sternum, and vertebrae with bone marrow were collected in 10% neutral buffered formalin, routinely paraffin embedded and sectioned, and stained with hematoxylin and eosin. Selected sections were immunostained with myeloperoxidase (Dako, Milano, Italy, N1578) for confirmation of myeloid phenotype of neoplastic cells, Von Willebrand Factor (Dako, N1505) to better delineate the lumens of blood vessels and cleaved caspase-3 (Cell Signalling, Danvers, MA, USA, Clone Asp175) to assess the apoptotic activity within the leukemic infiltrates. Fuelgen and Hoechst stains for chromatin also were performed.
The remaining 3 mice treated with VPA did not die spontaneously and were euthanatized 7 days after the beginning of therapy to assess reduction in tumor burden in comparison with untreated leukemic mice. No histologic findings consistent with acute tumor lysis syndrome (ATLS) were observed in these 3 animals.
Differential diagnoses
Sudden death without significant gross findings is nonspecific, but we considered 4 differential diagnoses based on the context of the experiment:
Repeated intraperitoneal injection might have resulted in peracute septic peritonitis with septicemia and/or endotoxic shock; however, no morphologic evidence of septic peritonitis or endotoxic shock was noticed.
Alternatively, in the context of progression of the promyelocytic leukemia, disseminated intravascular coagulation (DIC) may have ensued. Indeed, DIC is a frequent fatal complication of human APL.8 Severe impairment of hemostatic mechanisms with hemorrhagic manifestations, particularly in the lung, is a variable feature of advanced disease in the mouse model of PML/RARα-induced myeloid leukemia (Marchesi, unpublished). However, no gross or histologic evidence of hemorrhagic diathesis was detectable in the present case.
Acute VPA toxicosis was also hypothesized. Acute cerebral edematous swelling, with consequent coma and respiratory depression, as well as hyperammonemic encephalopathy, are common manifestations of acute VPA toxicity in humans.19 However, no evidence of acute cerebral edema and/or microscopic findings typical of hyperammonemic encephalopathy, such as spongiform changes of the white matter and Alzheimer type II astroglial cells were noticed.15
An additional differential diagnosis to be considered is the development of an ATLS as a consequence of massive neoplastic cell death induced by the therapy.
Microscopic findings
Histologic analysis confirmed that the mouse was affected by a progressive myeloid leukemia characterized by extensive infiltration of neoplastic myeloid cells in the spleen, bone marrow, liver, kidneys, and lungs (Fig. 1). Immunostaining for myeloperoxidase demonstrated the myeloid nature of the neoplastic cells (Fig. 2). A striking feature was represented by the occurrence in multiple organs of variably sized and shaped intravascular clumps (emboli) of dense deeply basophilic material (Fig. 3a). These basophilic emboli appeared particularly prominent in the pulmonary medium-sized vessels and capillaries (Figs. 3, 4) and in the portal-centrilobular veins of the liver (Fig. 5). The basophilic emboli were often associated with intravascular aggregates of fibrillary eosinophilic material and cellular debris interpreted as fibrin enmeshing lysed neoplastic cells (Figs. 3a). The basophilic emboli displayed positive fluorescent blue staining with Hoechst (Fig. 3b) and intense red-purple reaction with Feulgen stain (Fig. 3c), which confirmed their high content in DNA, presumably derived from the nuclear chromatin of lysed tumor cells. Cleaved caspase-3 immunostaining was plentiful among the neoplastic myeloid infiltrates (Fig. 5). Scattered cleaved caspase-3–immunopositive apoptotic bodies were also present within the intravascular aggregates of necrotic or lytic neoplastic cells frequently associated with the emboli of chromatin (Fig. 5).
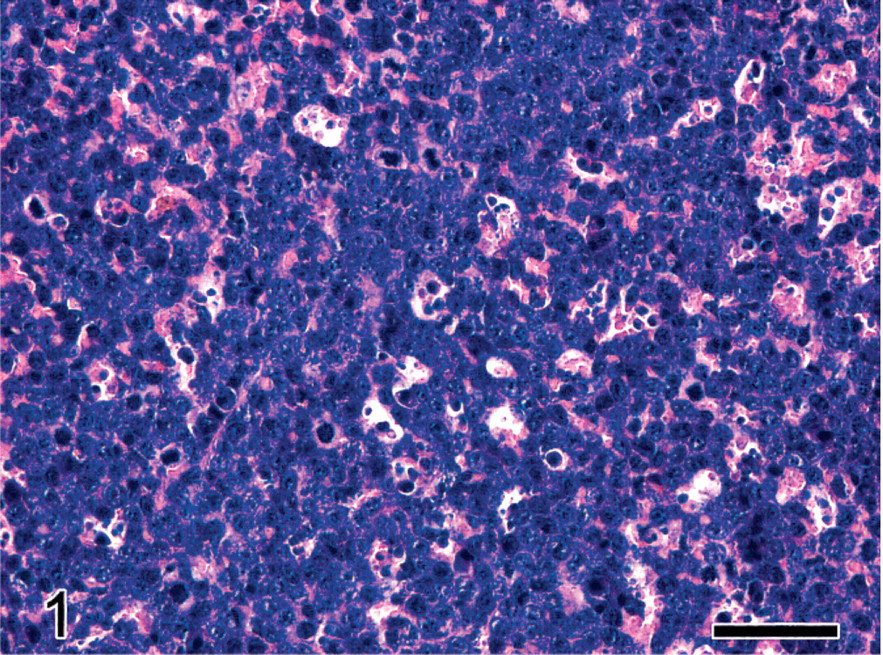
Spleen; female 129/SvEv mouse with PML/RARα-induced acute myeloid leukemia. Splenic parenchyma expanded by dense sheets of immature neoplastic myeloid cells with scattered apoptotic figures. HE. Bar = 50 μm.
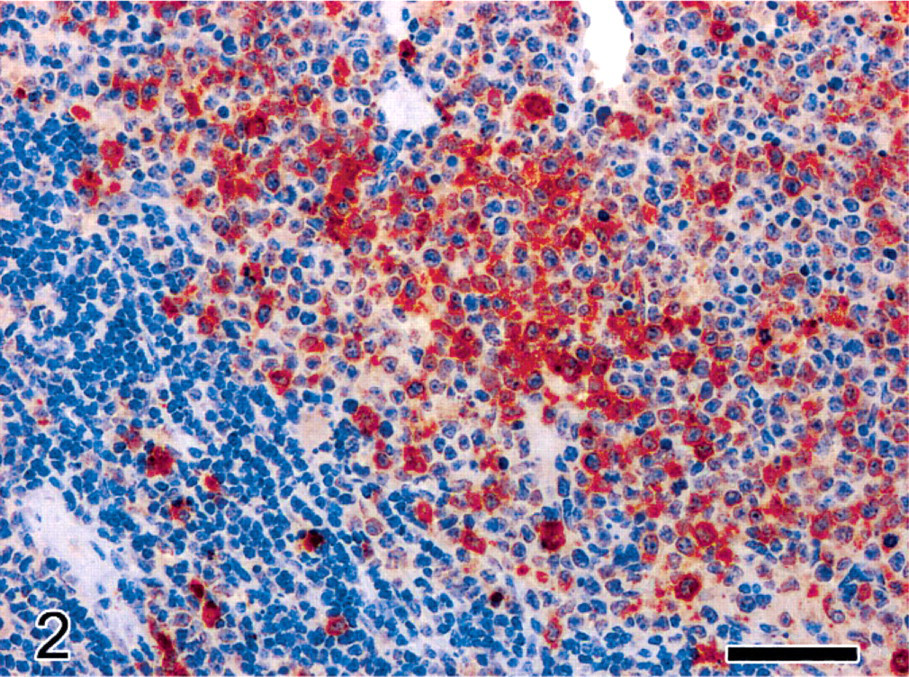
Spleen; female 129/SvEv mouse with PML/RARα-induced acute myeloid leukemia. Red pulp expanded and effaced by dense infiltrates of immature neoplastic myeloid elements, with prominent cytoplasmic myeloperoxidase immunoreactivity. Residual atrophic periarteriolar lymphoid sheath in the lower-left corner. Immunohistochemistry for myeloperoxidase, Mayer's hematoxylin counterstain. Bar = 50 μm.
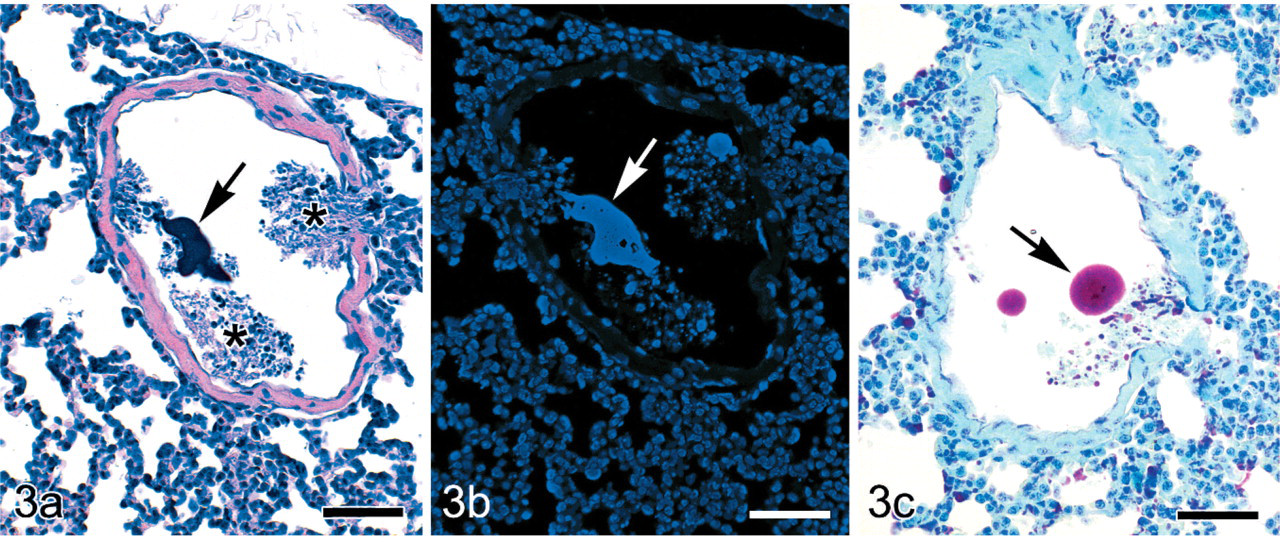
Lung; female 129/SvEv mouse with PML/RARα-induced acute myeloid leukemia. Pulmonary medium-sized vein containing a large pleomorphic clump (embolus) of dense deeply basophilic material (arrow) mixed with abundant cellular debris originating from necrosis or lysis of neoplastic myeloid cells (asterisks). HE. Bar = 50 μm.
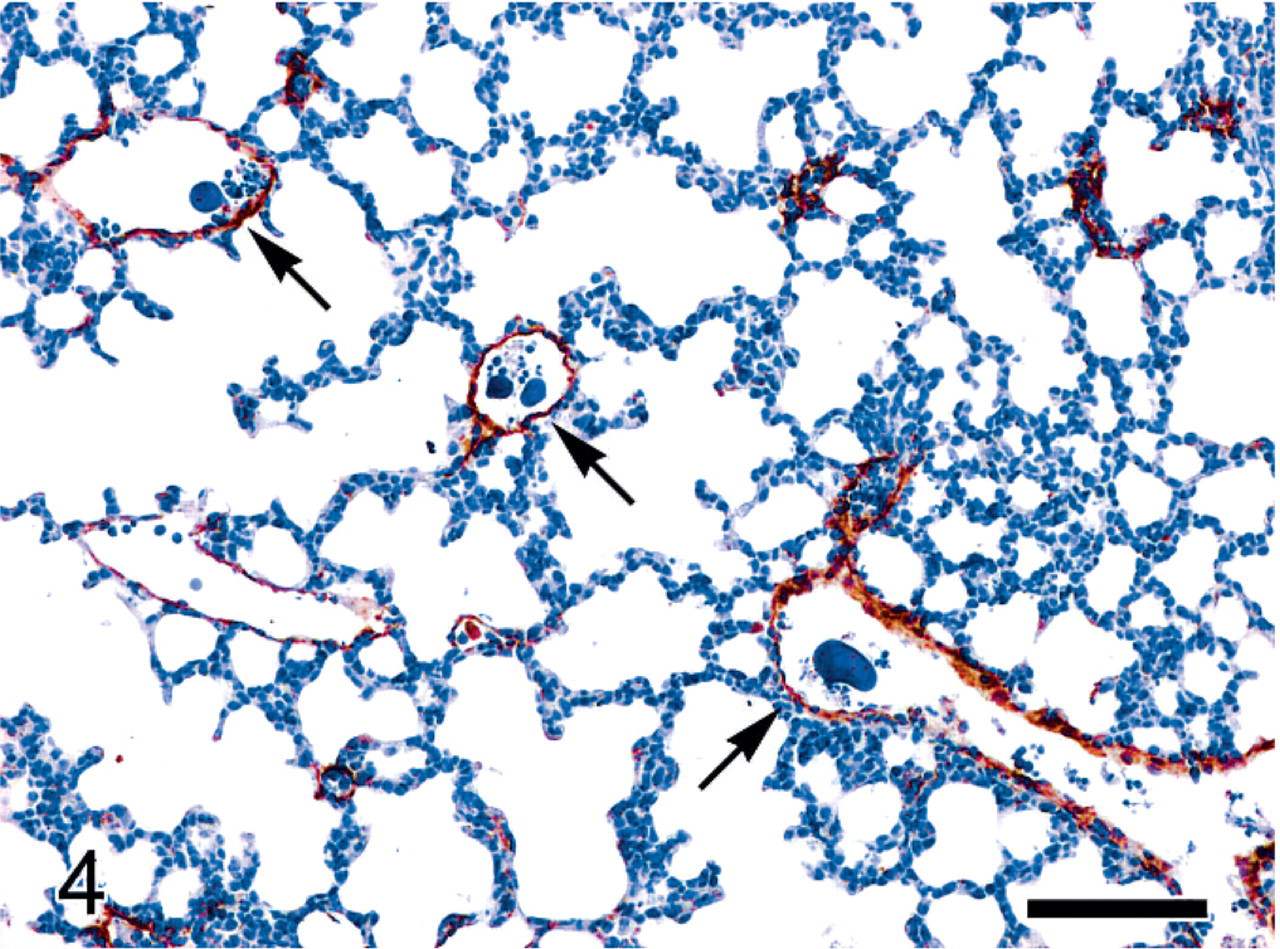
Lung; female 129/SvEv mouse with PML/RARα-induced acute myeloid leukemia. Von Willebrand factor immunostaining highlights that most of the medium-sized blood vessels in the pulmonary parenchyma contain multiple emboli of chromatin (arrows). Immunohistochemistry for von Willebrand factor, Mayer's hematoxylin counterstain. Bar = 100 μm.
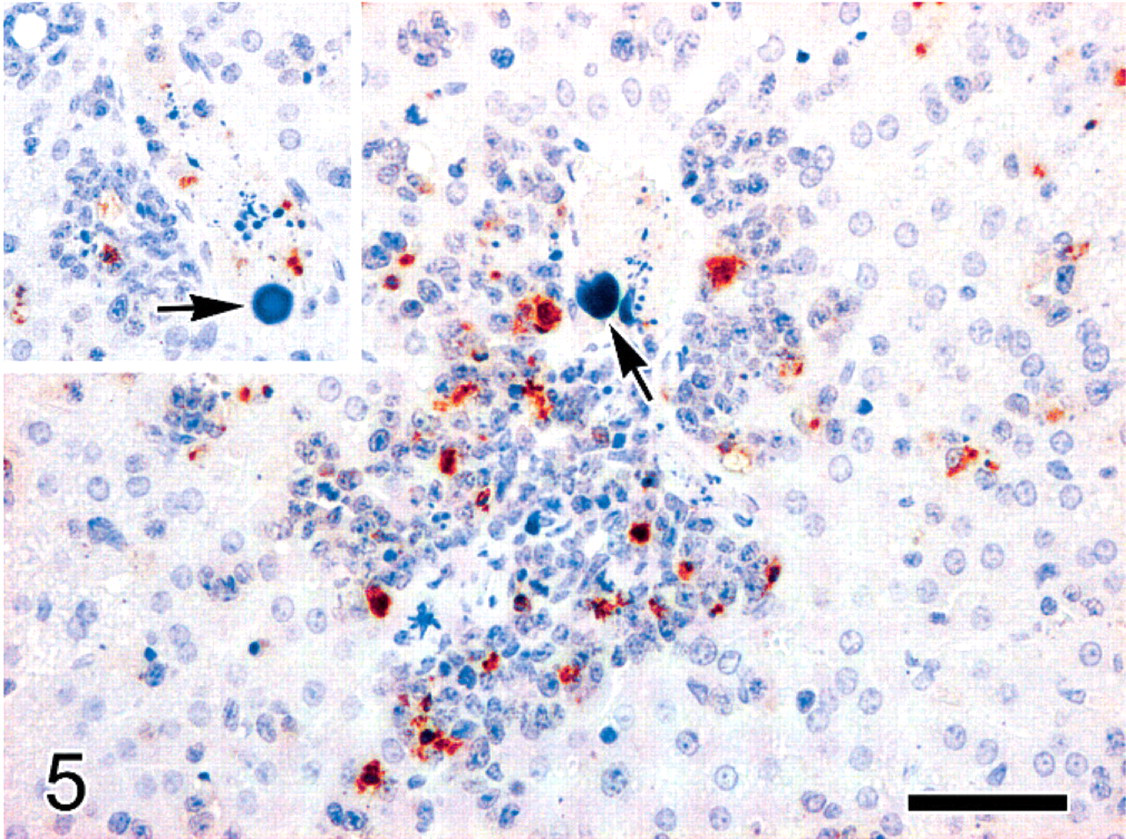
Liver; female 129/SvEv mouse with PML/RARα-induced acute myeloid leukemia. Portal-centrilobular veins containing multiple emboli of chromatin (arrows). Intense cleaved caspase-3 activity within the angiocentric infiltrates of neoplastic myeloid cells. Cleaved caspase-3-positive apoptotic bodies are also scattered among the intravascular necrotic or lytic leukemic cells (inset). Immunohistochemistry for cleaved caspase-3, Mayer's hematoxylin counterstain. Bar = 50 μm (inset, same magnification as the main figure).
Diagnosis
Based on the above findings, our diagnosis was ATLS, with massive necrosis and/or apoptosis of neoplastic cells and subsequent disseminated embolization of chromatin clumps and necrotic cell debris. This could have been induced by VPA treatment, but, if so, it is unclear why the other 3 treated mice were not affected. A clear cause of death was not possible to determine, but the extensive thrombo-embolization of chromatin clumps and necrotic debris in pulmonary vasculature could have led to acute respiratory failure, perhaps complicated by circulatory shock.
Discussion
ATLS is a serious life-threatening condition, representing a true emergency in clinical oncology. ATLS is more commonly associated with hematologic malignancies but has also been reported for solid tumors.3, 20 ATLS is characterized by serious pathophysiologic alterations, including hyperuricemia and hyperphosphatemia, caused by massive release of nucleic acids and phosphate from lytic tumor cells. These events commonly account for precipitation of uric acid and calcium phosphate in the renal tubules, with acute renal failure. Concurrent bradyarrhythmias and cardiac arrest because of potassium leakage from injured cells and hyperkaliemia may also occur.3
ATLS is more commonly reported in the context of aggressive chemotherapy (including treatment with HDAC inhibitors) or radiation therapy, but it has also been observed to occur spontaneously, both in hematologic malignancies and in solid tumors.4, 6, 20, 23 In the case we describe, the death of the mouse was attributed to ATLS and particularly to respiratory failure, because of the massive embolization of chromatin clumps and necrotic debris in pulmonary vessels and capillaries. Therapy-related ATLS has been reported to cause a similar respiratory complication in human patients with myeloid leukemia.17
A link between treatment with the HDAC inhibitors VPA and the development of ATLS is strongly suspected. Indeed, conspicuous apoptosis of neoplastic myeloid cells was demonstrated in the spleen and in other tissues. Treatment of mice bearing PML/RARα and AML1-ETO induced myeloid leukemias with the HDAC inhibitor VPA has been shown to result in increased survival due to upregulation of Fas and TRAIL apoptotic pathways in neoplastic cells with a reduction in tumor burden.5 Thus the pro-apoptotic activity of VPA in leukemic cells may underlie the pathogenesis of ATLS in this mouse.
There have been sporadic reports of ATLS in the veterinary literature.2, 9, 14 A case of spontaneous ATLS in a DBA/1J mouse with disseminated lymphoma was also reported, with a detailed description of associated histologic findings.10 The case we describe should reinforce the awareness of the possible occurrence of tumor lysis syndrome in mouse models of hematopoietic malignancies and possibly stimulate the design and conduction of experiments aimed at establishing an animal model of therapy-related ATLS that may serve as a tool for the dissection of molecular mechanisms underlying the onset and progression of this condition in the context of highly effective cancer therapy.
Footnotes
Acknowledgements
We thank Professor Pier Giuseppe Pelicci and Dr. Silvia Monestiroli of the European Institute of Oncology for providing the case and for their scientific support. We also thank Mr. Marco Brevi and Ms. Chiara Villa for the excellent technical contribution.
