Abstract
Acute tumor lysis syndrome (ATLS) is a potentially lethal condition precipitated by the massive release of intracellular components such as nucleic acids, potassium, and phosphorus, following a rapid and widespread lysis of tumor cells. Herein, the authors describe the high incidence and characteristic histopathologic lesions of acute ATLS in p53-deficient mice used in 2 short-term carcinogenicity studies. ATLS was a frequent cause of early death in p53 +/– mice in these studies and was consistently associated with the presence of disseminated lymphoma and leukemia. Although a heavy tumor burden and leukemia were present in all affected mice, the absence of ATLS in other mice with equally severe lymphoma and leukemia indicates that these tumor burdens are necessary but insufficient to cause ATLS in mice. The hallmark histopathologic findings of ATLS in mice are the disseminated microemboli composed of nuclear and cytoplasmic debris derived from lysed tumor cells. The mechanical obstruction of capillary beds by these microemboli appeared to be the proximate cause of the early deaths of mice in these studies. Microemboli may contribute to the pathogenesis of acute renal failure and other clinical signs associated with ATLS in other species. Recognition of ATLS in laboratory animals is critical in studies intended to evaluate the efficacy and/or toxicity of anticancer treatments, where early deaths due to ATLS might otherwise be attributed to test article toxicity. Further studies on the role of microemboli in the pathogenesis of ATLS may elucidate pathogenetic mechanisms and lead to improved approaches to clinical management and treatment of this potentially lethal condition.
Acute tumor lysis syndrome (ATLS) is a potentially lethal condition that is precipitated by the massive release of intracellular components such as nucleic acids, potassium, and phosphorus, following the rapid and widespread destruction of tumor cells. The release of cellular contents and breakdown products can overwhelm normal excretory and cell-buffering mechanisms and produce an acute metabolic crisis. ATLS is most often associated with cancers characterized by rapid cell proliferation, high tumor burden, and high sensitivity to chemotherapy. ATLS has been reported with a variety of tumor types, and although most cases follow intensive induction chemotherapy, others have been associated with treatments with corticosteroids, hormones, or cytokines. 6 In humans, the massive release of cytoplasmic contents from lysing tumor cells results in the hallmark sign of hyperuricemia (uric acid is the primary toxic breakdown product of nucleic acids) as well as hyperkalemia, hyperphosphatemia with or without azotemia, and secondary hypocalcemia.
In human ATLS, hyperkalemia is often detected 24 to 48 hours before other metabolic alterations and can result in arrhythmias, syncope, or sudden death. 5 Hypocalcemia occurs at around 48 to 96 hours as a direct response to hyperphosphatemia and is usually manifested as neurologic or muscular disturbances such as tetany. 5,22 Elevated calcium and phosphate can form precipitates in the kidneys that contribute to acute renal failure. 2,22 However, the release and subsequent breakdown of large amounts of nuclear DNA can result in hyperuricemia, which contributes to acute renal failure secondary to an acute urate nephropathy and tubular necrosis. 2,22 In humans, free nucleotides are broken down into purine and pyrimidine bases. Pyrimidines are broken down and recycled as amino acids, whereas purines are converted by xanthine oxidase in the liver from hypoxanthine and xanthine to uric acid, which is excreted in the urine. 22 Significant tumor cell lysis results in increased production of uric acid, and less soluble urates may begin to deposit in the tissues. 2,5 Renal failure in human ATLS patients is attributed to the accumulation of uric acid crystals within distal renal tubule lumina, tubular epithelial cells, the medullary microcirculation, renal pelvis, and ureters, which may cause obstructive nephropathy (reviewed in Lameire et al 18 and Rampello et al 28 ). In recent years, a recombinant urate oxidase enzyme (Rasburicase) has proven effective in treating or preventing hyperuricemia and renal dysfunction in human patients at high risk for ATLS. 3
In contrast to the recognized importance of ATLS in human clinical oncology, reports of ATLS in veterinary medicine have been relatively few and sporadic. 4,17,21,24,25 Two reports have described the acute lesions of spontaneous ATLS in individual DBA/1J 19 and 129/SvEV 27 mice that had severe disseminated lymphoblastic lymphoma. The rarity of clinically significant ATLS in animals is most likely due to the fact that mammals other than primates have uricase enzymes that rapidly degrade relatively insoluble excess uric acids to allantoin. Uricase activity in most mammalian species prevents the development of hyperuricemia and the consequent nephropathy caused by deposition of uric acid crystals in the kidneys. However, it is becoming evident that other deleterious effects of ATLS can have serious repercussions in multiple species.
In this report, we describe the high incidence and characteristic histopathologic lesions of acute ATLS in p53-deficient mice being used in 2 short-term carcinogenicity studies. Heterozygous p53-deficient mice, in which one allele of the p53 gene is inactivated, were derived from C57BL/6 or CBA origins. They provide a useful model to detect genotoxic carcinogens within a shorter treatment period—6 months, as opposed to 2 years. 11,20,23 Spontaneous development of tumors is rare during these bioassays, and p53 +/– mice apparently do not respond to nongenotoxic carcinogens that can induce tumors in the liver or kidney in conventional 2-year bioassays. 30 In testing potential carcinogenic compounds, the advantages of using of p53 +/– heterozygous mutant mice instead of wild-type mice include increased sensitivity and specificity, the need for fewer animals, and a shorter duration of dosing than that of the standard bioassay.
Although the occurrence of ATLS in p53-deficient mice appears to be sporadic and unpredictable, our findings indicate that ATLS should be considered a potential cause of early deaths in carcinogenicity or toxicology studies performed using these mice. In addition, lesions similar to those that occur with ATLS in mice could be involved in the pathogenesis of this potentially lethal condition in other species, including humans.
Materials and Methods
Experimental Design
Wild-type and haploinsufficient p53-knockout mice (B6.129-Trp53tm1 N5; TSG-p53 heterozygotes) were obtained from Taconic Farms (Germantown, NY) for use in both 26-week carcinogenicity assays described in this report. Mice were 6 to 10 weeks old at the initiation of these studies. The same test compound was used in both studies described below.
Study A
In this study, the test compound was formulated in a solution and given to animals intravenously via tail vein. The purpose was to determine whether intravenous treatment with a test compound, administered 3 times per week for 26 weeks, would increase the incidence of tumors in p53 +/– mice. In sum, 398 p53 +/– and p53 +/+ (199 male and 199 female) were randomly allocated to 1 phosphate-buffered saline (PBS) control group, 1 p-cresidine (a known carcinogen) positive control group, and 5 treatment groups that received various concentrations of test article.
Study B
In this study, blood was first exposed to the test article and then washed before being given to animals by intravenous tail vein injection. The purpose was to determine whether intravenous treatment with test article–treated red blood cells, administered biweekly for 26 weeks, would increase the incidence of tumors in p53 +/– mice. In sum, 150 p53 +/– mice (75 males and 75 females) were randomly allocated to 2 control groups and 1 treatment group. The negative control group received PBS-treated red blood cells; the positive control group received the carcinogen p-cresidine; and the test group received red blood cells treated with test article.
In both studies, mice were housed in rooms maintained between 18 and 26°C and 30 and 70% relative humidity on a fixed cycle of 12 hours of light and dark (6:00
Pathology
Complete necropsies were conducted on all study animals. Mice were euthanized by carbon dioxide inhalation followed by exsanguination. All organs and tissues were examined for grossly visible lesions and then preserved in 10% neutral buffered formalin. After fixation, tissues were processed and trimmed, embedded in paraffin, sectioned to a thickness of 5 to 6 μm, and stained with HE for microscopic examination. Microscopic examinations were performed on 2 control and 2 high-dose groups for Study A and for all animals in Study B. Other tissues were collected from test article–treated mice and examined microscopically: adrenal gland, bone with marrow, brain, clitoral gland, esophagus, eye, gallbladder, harderian gland, heart with aorta, kidney, large intestine (cecum, colon, rectum), small intestine (duodenum, jejunum, ileum), larynx, liver, lung and mainstem bronchi, mammary gland (with inguinal lymph nodes), nose, oral cavity, ovary, pancreas, parathyroid gland, preputial gland, prostate gland, salivary gland, spleen, stomach (forestomach and glandular), testis (with epididymis and seminal vesicle), thymus, thyroid gland, tongue, trachea, uterus, vagina, and Zymbal gland. Complete histological examinations were not performed on positive control p-cresidine-treated controls; in these animals, microscopic examinations were restricted to p-cresidine target tissues (urinary bladder, kidney, and tissue masses) and any observed gross lesions. After routine histologic evaluation, selected sections were cut and stained with Feulgen stain or Hoechst 33342 to identify and confirm the presence of DNA or nucleic acid in the microemboli.
Results
ATLS was a frequent cause of early death in p53 +/– mice in these short-term carcinogenicity studies, and it was consistently associated with the presence of disseminated lymphoma and leukemia. In Study A, 48 early deaths out of 398 mice on study were reported. Of the 48 early deaths, 10 were from the p-cresidine-treated positive control group that died as a result of neoplasms of the urinary bladder. Of the remaining 38 early deaths, a significantly higher incidence of ATLS occurred in those mice receiving the highest dose of the test article (4.5 mg/kg); 16 out of 58 high-dose-group mice were found dead or were sacrificed early because of to a moribund condition. Disseminated lymphoma/leukemia was detected in 10 of these 16 early deaths, and ATLS was identified as the likely cause of early death in 4 of the 16—all of which were high-dose-group p53 +/– mice with lymphoma/leukemia. Lymphoma and ATLS were not present in the other 6 early-death animals in the high-dose group. In Study B, lesions consistent with ATLS were detected in 19 of the 50 PBS-treated mice. There were 12 early deaths in this negative control group, 10 of which included lesions consistent with ATLS (4 of 5 males and 6 of 7 females). ATLS was also found in a lower but significant proportion of the PBS-treated mice that survived to the end of the study (9 out of 38: 6 of 20 males and 3 of 18 females). ATLS lesions were not detected in the 8 PBS-treated mice that did not develop lymphoma; ATLS lesions were also not detected in any mice that received test article.
The most common macroscopic findings in early-death mice included enlarged lymph nodes, spleens, and livers. The affected mice typically had disseminated neoplastic disease, with marked involvement of the liver and spleen, and leukemic lymphoma.
Lymphoblastic lymphoma and leukemia were histologically apparent in all mice with lesions of ATLS. Disseminated neoplasms were composed of sheets of uniform round cells characterized by a small amount of lightly basophilic cytoplasm and a central stippled round-to-indented nucleus containing 1 to 2 indistinct nucleoli. In some areas, up to 30% of the neoplastic cells were necrotic, as characterized by pyknotic nuclei and abundant karyorrhectic debris. The spleen and multiple lymph nodes were diffusely infiltrated with neoplastic and often necrotic round cells that effaced the normal architecture. In most animals with lymphoma, neoplastic cells also infiltrated the liver (Figure 1). Neoplastic cells were also widely present within the vascular system, with numerous viable and apoptotic lymphoid cells in hepatic sinuses, pulmonary capillaries, and alveolar spaces.
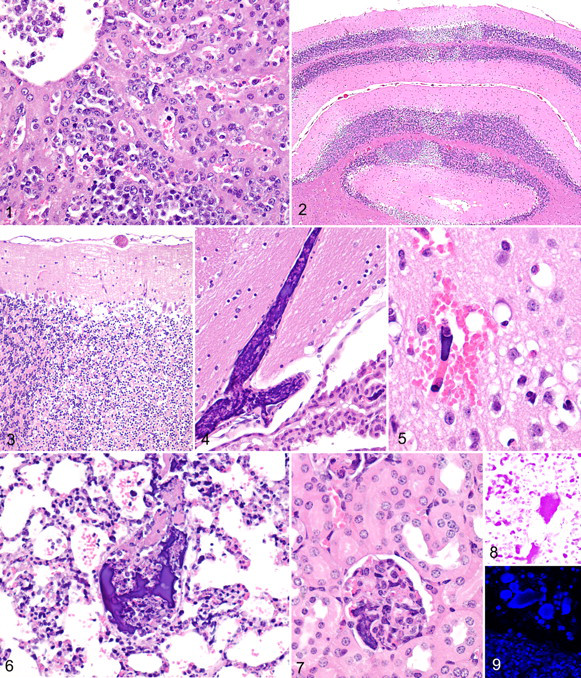
Multifocally within the brain and most clearly affecting the cerebellum were widespread areas of neuronal degeneration and necrosis due to ischemia (Figure 2). The necrotic foci in the brain were often associated with small mixed emboli composed of tumor cell–derived nuclear and cytoplasmic cell debris (Figure 3). In virtually all tissues examined, there were multifocal variably sized mixed emboli composed of deeply basophilic material variably admixed with eosinophilic material (Figure 4). In some areas of the brain, microemboli were associated with hemorrhage, neuronal necrosis, and vacuolization of the neuropil (Figure 5). The emboli were common in the lung, wherein large pulmonary vessels contained mixed emboli (Figure 6) and many alveolar capillaries were occluded by basophilic microemboli (Figure 6). The kidneys were also affected, with elongate microemboli occluding interstitial and glomerular capillaries (Figure 7). Many microemboli were composed of homogeneous, acellular, and deeply basophilic material resembling aggregated chromatin. Special stains confirmed that they contained deoxyribonucleic acids (DNA), presumably derived from the nuclear chromatin of necrotic tumor cells; the wide distribution of microemboli in the lungs was highlighted by an intense red-purple color with Feulgen stain in the lungs (Figure 8) and with bright fluorescent blue staining (Figure 9) with Hoechst 33342 dye (Molecular Probes, Eugene, OR). The Hoechst 33342 fluorescent dye is a specific stain for AT-rich regions of double-stranded DNA. 29
Discussion
The p53-deficient mouse model was initially developed because mutations in the p53 tumor suppressor gene had been associated with a range of human cancers. 15 The p53-knockout mouse was the first reported tumor suppressor knockout 9 and p53 –/– mice generally develop tumors within 2 to 9 months after birth. 7,9 On a mixed C57BL/6 and 129/Sv genetic background, up to 50% of 4-month-old and 100% of 10-month-old p53 –/– knockout mice develop CD4/CD8 double-positive T-cell lymphomas or sarcomas (which include hemangiosarcomas, osteosarcomas, fibrosarcomas, rhabdomyosarcomas, and anaplastic sarcomas). 9,16 In contrast, heterozygous p53 +/– mice do not develop tumors until later in life; they show a high incidence of nonthymic B-cell lymphomas and a variety of sarcomas. 8,14,16,26 Interestingly, the genetic background of the mice can alter the spectrum, incidence, and latency of spontaneous and induced tumors. 10,16
In experimental studies, the heterozygous p53 +/– mouse model demonstrates a high sensitivity to genotoxic events and a low spontaneous incidence of tumors until 9 to 12 months of age. 13 Because tumors are rare in normal control p53 +/+ mice during the first 6 months of life whereas tumor induction is rapid in p53 +/– mice, the heterozygous p53 +/– mouse model provides a short latency period and reduced expense for carcinogenicity testing in drug safety evaluation. 7,12,32 An additional advantage of using p53 +/– mice is that they do not produce false-positive responses in short-term carcinogenesis assays, unlike the overly sensitive 2-year mouse bioassay. 13 Twenty-six weeks has proven to be an adequate assay period for detecting most known genotoxic compounds 13 and has become the standard duration for bioassays using p53 +/– mice. 31 For these reasons, commercially available heterozygous p53 +/– mice (TSG-p53 Targeted Mutation Mice, Taconic, Hudson, NY) are increasingly used as an alternative model for short-term (26-week) carcinogenicity testing. 13
The hallmark finding in murine ATLS is the presence of widely disseminated microemboli composed of nuclear and cytoplasmic debris derived from lysed tumor cells. Previous ultrastructural studies suggest that fibrin is not a major component of these microemboli. 19 It seems likely that the characteristic microemboli we observed in mice could exist and contribute to the pathogenesis of ATLS in other species. We propose that mechanical obstruction of capillary beds by these microemboli was the proximate cause of the early deaths of mice in these studies and that microemboli may contribute to the pathogenesis of acute renal failure and other clinical signs associated with ATLS in other species. The obstruction of (1) pulmonary and cerebral capillaries by DNA microemboli and (2) pulmonary arteries by embolic cell debris probably resulted in respiratory stress and neurological signs; however, severe metabolic derangements and electrolyte abnormalities associated with the massive lysis of tumor cells may have also been lethal in some cases. Although urate crystals and renal tubular degeneration and necrosis are apparently not features of ATLS in mice, we believe that similar microemboli consisting of cell detritus could compromise renal blood flow and contribute to the pathogenesis of the acute renal failure that is characteristic of ATLS in humans. Decreased renal perfusion would result in oliguria and tissue hypoxia, which favor the formation of uric acid crystals and ischemic necrosis of tubular epithelium, while impairing regulation of serum electrolytes and clearance of toxic metabolites.
The precipitating cause of the spontaneous ATLS in these 2 studies is unknown. Lesions of ATLS in mice and other species may be underreported, although it has become clearer over time that they occur rarely and sporadically even in mice. We report here the occurrence of ATLS lesions in 2 separate studies that were completed at the same facility within a relatively short time frame. Although a heavy tumor burden and severe leukemia were present in all affected mice in this report, the absence of similar ATLS lesions in other mice with equally severe disseminated lymphoma and leukemia indicates that these tumor burdens are necessary but insufficient by themselves to result in ATLS in mice. In addition, because ATLS developed in mice that received a variety of different treatments (including vehicle controls), it appears likely that widespread lysis of tumor cells must have been precipitated by some unknown environmental factor that imposed metabolic or physiological stresses on the neoplastic cells. In humans, sporadic cases with spontaneous onset of ATLS have been described, but the syndrome is most often associated with aggressive chemotherapy of lymphoproliferative malignancies. Corticosteroids, cytokines, and hormone therapies can trigger ATLS episodes in humans. 1,5 We speculate that as-yet-undefined environmental stresses may have induced a surge of endogenous glucocorticoids that precipitated the acute tumor lysis episode and rapid death in some of these mice, but further studies will be required to confirm this hypothesis. Whether ATLS is treatment related or precipitated by an environmental factor, recognition of the syndrome in laboratory animals is critical in studies intended to evaluate the efficacy and/or toxicity of anticancer treatments, wherein early deaths due to ATLS might otherwise be attributed to test article toxicity. Further studies on the role of DNA microemboli in the pathogenesis of ATLS may elucidate pathogenetic mechanisms involved in other species and lead to improved approaches to clinical management and treatment of this potentially lethal condition.
Footnotes
Acknowledgements
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
At the time these studies were performed, the authors were employed by Charles River Laboratories, Pathology Associates International, Frederick, Maryland.
The authors declared that they received no financial support for their research and/or authorship of this article.
