Abstract
Rat respiratory virus (RRV) is the working name for a novel respiratory pathogen of laboratory rats in North America, Europe, and Asia. Although the agent has not been definitively identified, evidence supports a viral etiology. Because no serologic or molecular assays for RRV are available, diagnosis depends on histopathologic evaluation of the lung. We introduced 104 Wistar Han rats, free of known pathogens and of RRV-associated lesions, into a rat production colony positive for RRV-type lesions, but free of other histologic, serologic, or microbiologic evidence of infectious disease. Lungs of 8 of the naïve rats were examined grossly and microscopically each week, weeks 0–13. Irregular gray-white lesions suggestive of interstitial pneumonia were grossly evident from weeks 6 through 13. Primary histopathologic evaluation of all lungs by one pathologist found multifocal, lymphohistiocytic interstitial pneumonia or prominent perivascular lymphoid cuffing from weeks 5 through 13. Based on results of the initial evaluation, diagnostic criteria for RRV infection (i.e., changes seen only after exposure to the RRV-positive colony) were tentatively selected and used by 2 other pathologists to classify each lung as RRV positive, RRV equivocal, or RRV negative. The secondary evaluation found 95% concordance in RRV diagnosis between pathologists, and correlated well with the initial evaluation, thus confirming the consistency of the criteria. These data show that RRV-naïve rats introduced into an RRV-endemic colony develop equivocal microscopic lesions of RRV by 5 weeks of exposure, and positive diagnostic lesions by 7 weeks. Interstitial pneumonia becomes grossly evident after 6 weeks of exposure.
In 1997, Elwell et al. 3 reported distinctive inflammatory lesions in the lungs of F-344 rats examined over several years in prechronic toxicity studies performed at various sites in the United States for the National Toxicology Program of the National Institute of Environmental Health Sciences. The lesions consisted of “a prominent increase in perivascular lymphocytes throughout the lung and a variable increase in the amount of peribronchiolar lymphoid tissue,” as well as interstitial pneumonia with macrophages, neutrophils, and lymphocytes. The same year, Gilbert et al. 4 reported similar lesions in Sprague-Dawley rats in a cyclosporine liposome inhalation study; the pulmonary lesions were a confounding factor in results interpretation. Pulmonary lesions morphologically consistent with those reported by Elwell et al. were subsequently reported by Slaoui et al. 11 in 1998, in Wistar rats in Belgium. In both the Elwell and Slaoui reports, lesions diminished with time, and disappeared before the conclusion of the 2-year studies. At this point, the condition was appropriately considered as a “disease” as it constituted a distinctive morbid process with a characteristic train of lesions.
Early investigations at the Research Animal Diagnostic and Investigative Laboratory at the University of Missouri detected a cytopathic agent in mammalian cell cultures inoculated with tissue homogenates of lungs from Sprague-Dawley rats with similar lesions. This and the other findings presented in an abstract in 1997 8 and in the American Committee on Laboratory Animal Diseases (ACLAD) newsletter that year, 9 led to the tentative conclusion that the lesions were caused by a novel virus of rats. As current evidence supports a viral cause, the term rat respiratory virus (RRV) has been suggested as a useful working term to describe this entity, although it has not been definitively established that the etiologic agent is, in fact, a virus.
Because the causative agent has not been identified and sequenced, or reproducibly grown in vitro, there is no available serologic or molecular assay. Thus, diagnosis of an RRV infection depends on histopathologic detection of characteristic pulmonary lesions. 2
Pneumonia caused by RRV appears widely distributed in North America and Europe, and has also been detected in Asia (data not shown). At the Charles River Research Animal Diagnostic Services, histologic lesions characteristic of RRV infection are observed in approximately 6% of rats submitted for routine health surveillance from sources outside of Charles River barrier production rooms. 7 Thus, not only is RRV infection more common than infection due to the viruses typically screened by serology, but unlike many of the currently prevalent viruses that only rarely cause lesions in any tissue, RRV infection often results in morphologic damage, specifically to the lung. Because detection depends on histopathologic diagnosis, publication of diagnostic criteria in the peer-reviewed literature is important. This not only assists in disease detection and management, but also helps provide a framework for future discussion of RRV research and consequences.
To refine the histologic criteria for diagnosis, estimate the time from exposure until lesion development, and better define the time course of RRV lesions, we introduced naïve rats into an endemically infected colony, then followed pulmonary histopathology during the subsequent 13 weeks.
Materials and Methods
Animals
Naïve test rats
Wistar Han rats (Crl:WI(Han)), 4 weeks old, 52 of each sex, were obtained from the Charles River IGS Foundation Colony (Charles River, Wilmington, MA). This small colony was cesarian-rederived, then maintained in isolators. Routine monitoring of sentinels found no evidence of antibodies to Sendai virus, pneumonia virus of mice, sialodacryoadenitis virus (rat coronavirus), Kilham rat virus, Toolan's H-1 virus, rat parvovirus, reovirus, rat theilovirus (monitored using GD-VII antigen), lymphocytic choriomeningitis virus, hantavirus, rat adenovirus (monitored using mouse adenovirus antigen), Encephalitozoon cuniculi, cilia-associated respiratory bacillus, and Mycoplasma pulmonis. In addition, routine cultures onto appropriate media were negative for Salmonella spp., Streptobacillus moniliformis, Corynebacterium kutscheri, Helicobacter spp. (monitored by polymerase chain reaction [PCR]), and other pathogenic bacteria. Examination of the pelage and gastrointestinal tract found no parasites. In addition, all isolators in the foundation colony were historically negative for histologic lesions characteristic of RRV, and as of present, no rat in any of the isolators has ever been found to be infected with RRV. Therefore, the isolator colony was considered to be RRV negative.
RRV positive colony
A RRV-positive rat colony was among a group of rat colonies included with a business acquisition. This colony was negative, before and after the duration of this study, for all of the pathogens listed previously for the naïve test rats. The RRV status was determined through routine health monitoring, which included histologic evaluation of lung for characteristic RRV lesions. Lung lesions consistent with those previously described in the literature as associated with presumed RRV infection, and often observed at our diagnostic laboratory, were confirmed in the colony animals during multiple evaluations, by multiple pathologists, throughout the year before beginning this experiment. Of the lungs, 39% from that colony during the previous year were considered to have lesions consistent with RRV infection (data not shown).
Exposure, housing, and husbandry
The naïve test rats were introduced into the RRV-positive colony. The exterior surfaces of shipping crates were thoroughly sprayed with Clidox (1:5:1) in a sealed, pass-through room. After 20 minutes of crate disinfection, the internal boxes were introduced into the positive barrier room. Animals were segregated by sex and grouped four per cage in wire-top solid-bottom cages located on the bottom racks at various locations throughout the RRV-positive barrier room.
Bedding in the test rat cages comprised a 50:50 mix of clean and soiled hardwood bedding from colony animals. The soiled bedding was collected at routine bedding changes of cages of rats at least 8 weeks of age, in the positive colony. Food (Purina 5008) and water were provided ad libitum.
All animals were housed and cared for in accordance with the Guide for the Care and Use of Laboratory Animals. All procedures conducted in the research reported in this article were reviewed and approved by the Charles River Institutional Animal Care and Use Committee. Charles River is fully accredited by Association for the Accreditation of Laboratory Animal Care International.
Necropsy and histology
At week 0 (week of introduction) and every week for the next 13 weeks, four male and four female test rats were submitted to the Charles River Research Animal Diagnostic Services, Health Monitoring Laboratory for necropsy, except that no necropsies were conducted for week 3, and only 4 rats were examined week 13 (four rats died when their automatic watering system malfunctioned). Animals were euthanized by CO2 asphyxiation. Blood was collected, and serum was frozen for viral serology. A necropsy was performed, with emphasis on careful examination of the lung. Lungs were harvested and divided. The left lung was gently inflated with 10% neutral-buffered formalin (NBF), and the right lung frozen for future investigations. After fixation for a minimum of 24 hours, tissues were trimmed and routinely processed for paraffin embedding. Paraffin-embedded sections of lung, one section through the length of each left lobe, were cut at 5 µm onto glass slides and stained with hematoxylin and eosin.
Evaluation
Hematoxylin and eosin-stained lung sections were examined by a board-certified (American College of Veterinary Pathologists, ACVP) veterinary pathologist (C.B.C.) using a light microscope. All morphologic findings were recorded and assigned a subjective severity score of 1 (minimal) to 5 (severe). Because it was not known which early changes might signal RRV infection, or the full spectrum of changes accompanying infection, the initial evaluation graded all alterations. These morphologic criteria are listed in Table 1 and included perivascular hemorrhage, perivascular leukocytic infiltrates (eosinophils, lymphocytes, macrophages—each scored separately), lymphocytic cuffing of pulmonary vessels, focal increases in intra-alveolar macrophages, multifocal interstitial inflammation, and increases in bronchus-associated lymphoid tissue (BALT).
Histologic criteria included in initial evaluation of lung sections by first pathologist.
Subsequent to the initial evaluation, two additional ACVP board-certified pathologists with extensive experience in rodent diagnostic pathology (T.M.A. and M.A.S.) microscopically examined the same hematoxylin and eosin-stained sections. Using the changes identified in the initial evaluation as most strongly correlated with RRV infection, they classified each lung with as either “positive” (presence of characteristic RRV lesions), “equivocal” (presence of lesions which may be characteristic of RRV lesions), or “negative” (no characteristic RRV lesions).
Results
Viral serology
Banked frozen serum from two rats from each time point (0, 1, 2, 4, 5, 6, 7, 8, 9, 10, 11, 12, and 13 weeks after introduction into the room) was processed for antibody serology (multiplexed fluorometric immunoassay) for a panel of rat pathogens, including Sendai virus, pneumonia virus of mice, rat coronavirus, rat virus, H-1 virus, rat parvovirus, rat minute virus, reovirus, and Mycoplasma pulmonis. In addition, routine testing of both the isolator-based colony serving as the source of the naïve rats, and the RRV-positive colony remained negative by monthly testing for all pathogens listed in the Materials and Methods section.
Gross examination
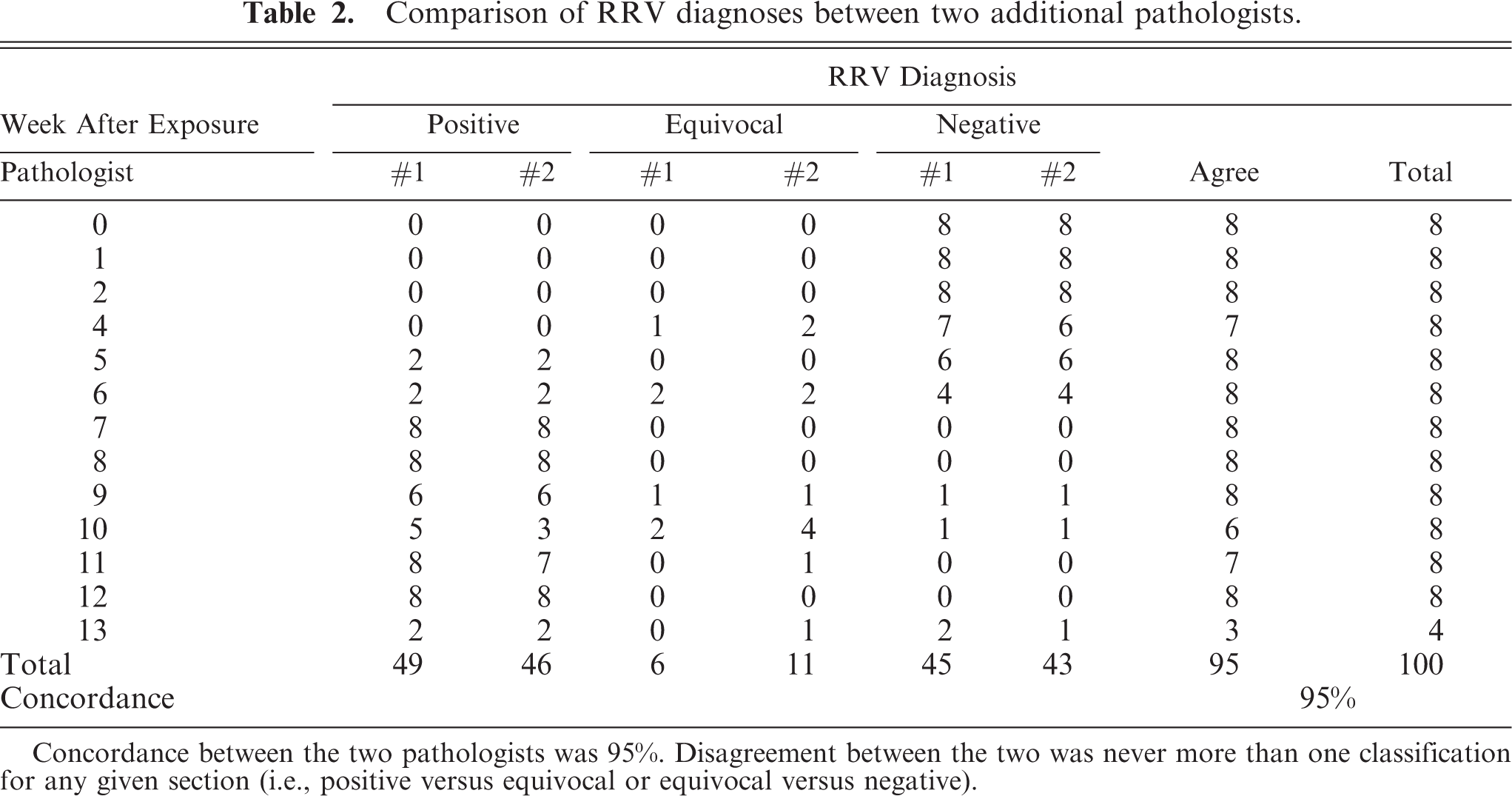
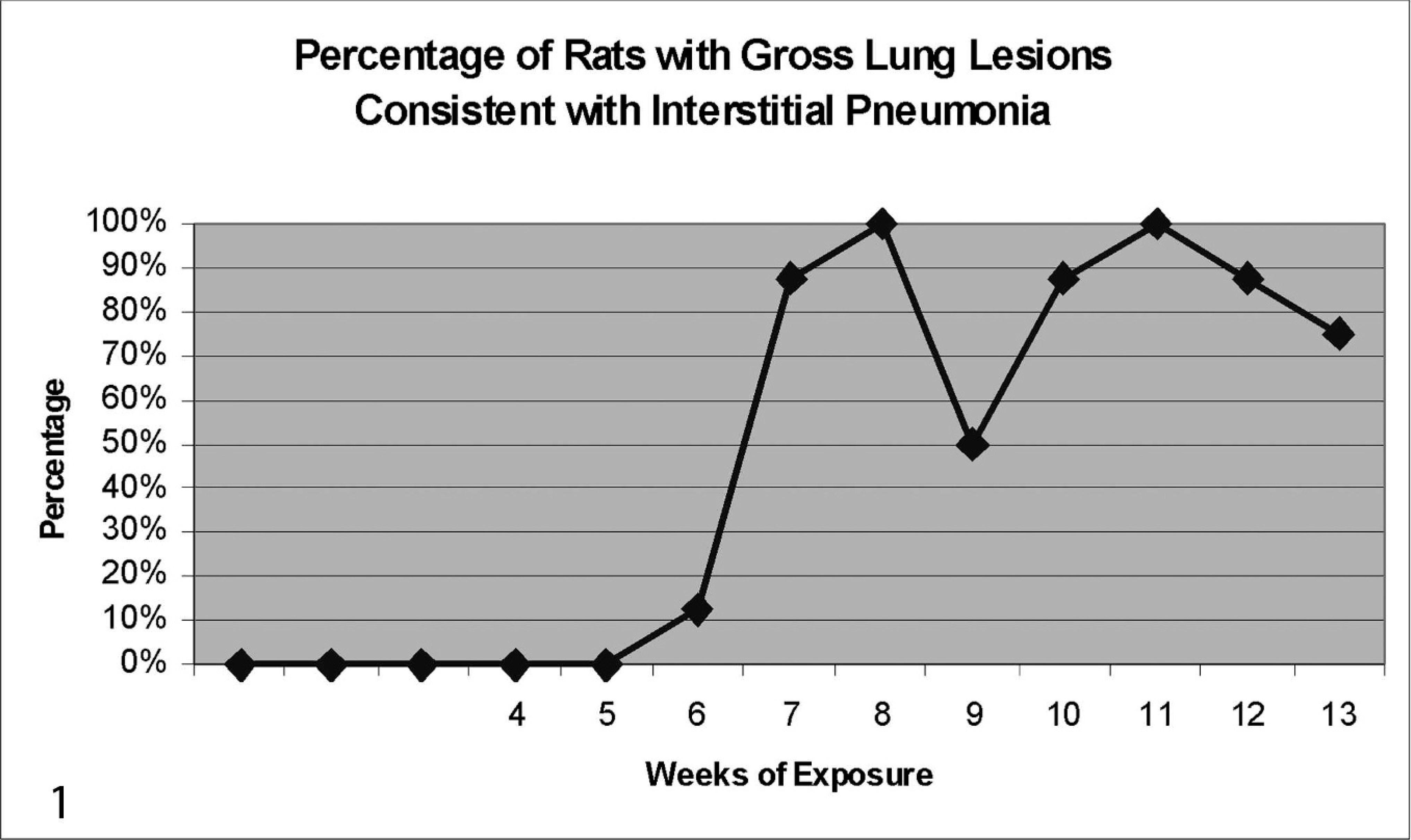
No gross pulmonary lesions were noted in any animals during weeks 0, 1, 2, 4, and 5 (note: necropsies were not conducted during week 3). Gross pulmonary lesions were first noted at week 6 after exposure in 1 of 8 animals. By week 7, 7 of 8 animals had gross pulmonary lesions. Gross pulmonary lesions were subsequently noted at each necropsy time point throughout the remainder of the 13-week study (Fig. 1). All gross pulmonary lesions were described as gray or white, 1- to 2-mm patchy areas or as gray/red mottling, suggestive of interstitial pneumonia (Fig. 2). No lesions were noted in any other tissue.
Percentage of the 8 rats sampled each week that had gross lung lesions consistent with interstitial pneumonia. Only 4 rats were evaluated at week 13.
Lung; rat 7 weeks after exposure. Numerous pale areas (indicated by black arrow) are visible across the lung and are consistent with interstitial pneumonia.
Microscopic
Initial evaluation
No changes were noted in animals in the week 0 rats, shipped to the necropsy facility directly after receipt. On weeks 1, 2, and 4 a minimal increase in perivascular eosinophils was observed, preceding a slight increase in perivascular lymphocytes. Slight perivascular hemorrhage was also noted at this time. By exposure week 5, perivascular eosinophils had peaked; henceforth, they declined. Week 5 also saw a substantial increase in perivascular lymphocytes and macrophages, both of which remained elevated for the remainder of the 13 week exposure period. By week 7, 4 histologic features had greater severity than any of the others: perivascular macrophages, perivascular lymphocyte infiltration, lymphoid cuff formation (Figs. 3, 4), and multifocal interstitial inflammation, which was always lymphohistiocytic (Fig. 5). The perivascular infiltrates of lymphocytes and macrophages and the interstitial pneumonia peaked at a mean severity of 2 (mild) or greater and remained elevated above all other features for the remainder of the study, except that interstitial inflammation declined to baseline (0) in the 4 animals examined at exposure week 13. Hemorrhage (i.e., perivascular erythrocytes) was observed in some sections at all time points except week 0. Essentially no increases in BALT or alveolar histiocytosis were observed. No changes were found in airways. The blood vessels most often involved by perivascular infiltrates were interstitial vessels, away from bronchi and bronchioles. Results of the initial microscopic evaluation are summarized in Fig. 6.

Lung; rat 7 weeks after exposure with multifocal lymphohistiocytic interstitial pneumonia. Lymphocytes and macrophages form cuffs around blood vessels, distend the alveolar septa, and fill alveoli. Alveolar septal thickening is prominent subjacent to the pleura at the upper left. HE.

Lung; rat 7 weeks after exposure. A thick band (cuff) of lymphocytes and macrophages encircles a pulmonary vein adjacent to an area of thickened alveolar septa. HE.

Lung; rat 7 weeks after exposure. This is a higher magnification of the field shown in Figure 4. Lymphohistiocytic interstitial pneumonia thickens alveolar septa, fills alveoli, and forms a thick cuff around a small pulmonary vein. HE.
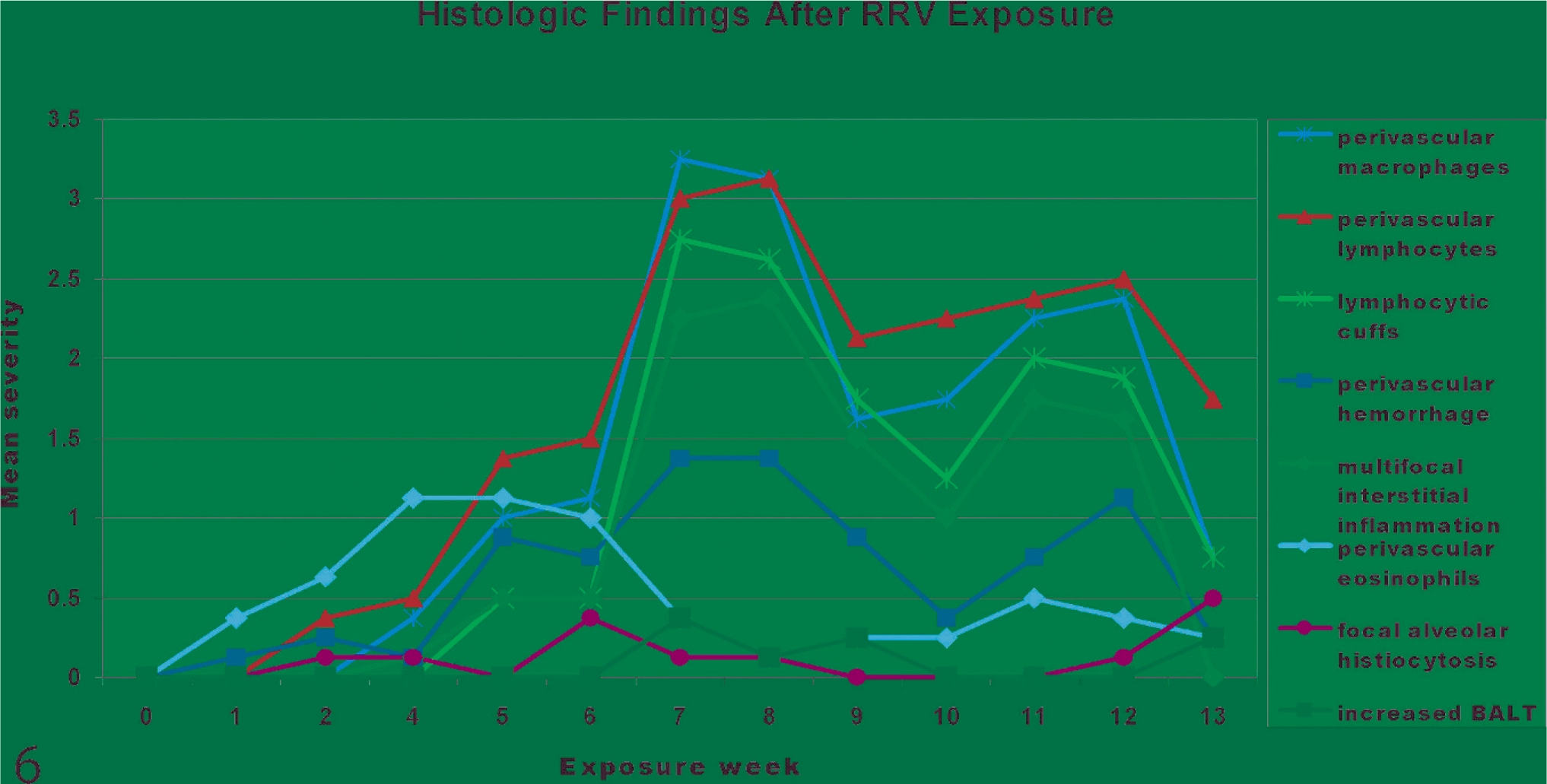
Histologic findings on rat lungs, by week, after exposure to a RRV-positive colony. All changes of potential interest were recorded regardless of correlation with RRV infection. The changes that peaked at weeks 7 and 8 (i.e., perivascular infiltrates of macrophages and lymphocytes, including perivascular lymphocytes organized into dense cuffs, as well as multifocal interstitial pneumonia) were selected as useful criteria for histologic diagnosis of RRV.
Secondary evaluation
Two additional pathologists evaluated all lung sections using the criteria found to best correlate with overall lesion severity and that were believed to be least likely to give false-positive diagnoses. Criteria used included interstitial inflammation, mixed perivascular infiltrates of lymphocytes and macrophages, and perivascular lymphoid cuffs. Using these criteria, no microscopic changes characteristic of RRV infection were noted at weeks 0, 1, and 2. Microscopic pulmonary lesions suggestive of RRV (graded as equivocal) were noted beginning at week 4 after exposure and continuing throughout the duration of the study. From weeks 7 through 12 after exposure, most animals were classified as “positive.” Results of the comparison are presented in Table 2. Of 100 lungs evaluated, the two pathologists agreed on the classification in 95, and disagreed on 5, a 95% concordance.
Comparison of RRV diagnoses between two additional pathologists.

Concordance between the two pathologists was 95%. Disagreement between the two was never more than one classification for any given section (i.e., positive versus equivocal or equivocal versus negative).
Discussion
Although the etiologic agent referred to as RRV remains unidentified, the pulmonary inflammation has distinctive features and a predictable course. Thus, we agree with previous investigators that it is useful, both diagnostically and from a colony health management perspective, to regard it as a disease. Our results confirm and extend previous reports of the transmissibility of the condition, the lesions associated with it, and provide the first peer-reviewed description of the time course of the visible manifestations of RRV infection.
We show that naïve rats exposed to a RRV-positive breeding colony will develop lesions similar to those in the colony, and consistent with RRV infection. By itself, this study by design does not provide conclusive evidence of the infectivity of RRV, as it does not exclude the possibility of an environmental factor causing the interstitial pneumonia in the RRV-positive room. The infectivity of RRV has previously been supported by other reports, 2,8,9 and our findings are consistent with those studies. Subsequent to the experiment, several times we have set up groups of naïve rats in isolators, then introduced rats from a RRV-positive colony, and found lesions appearing in the previously naïve rats (data not shown), thus further supporting the infectious nature of this disease. Gross lesions appear in 6 weeks and the characteristic microscopic lesions necessary for a positive diagnosis appear slightly earlier, at 5 weeks. RRV-associated lesions reach maximum severity 8–12 weeks after exposure. The characteristic microscopic lesions of RRV are lymphohistiocytic interstitial pneumonia or dense perivascular cuffs of lymphocytes and macrophages around interstitial blood vessels, and we propose that these lesions be used as diagnostic criteria (i.e., the presence of either of these changes is sufficient to make a positive diagnosis), as outlined in Table 3. We wish to emphasize that a single lymphoid cuff is considered nonspecific.
Suggested criteria for the diagnosis of RRV infection based on pulmonary histopathology.

∗The criteria for an equivocal diagnosis are intentionally broad to maximize sensitivity, despite the necessity of resampling populations to further investigate equivocal results.
The results of this study also provide some clarification of previous lesion descriptions. The initial report by Elwell et al. 3 included neutrophils within alveoli and increased BALT as noteworthy components of the inflammation. Subsequent investigators have not mentioned the neutrophils as a significant component, 1,8,11 nor were they noted in the lungs in this study. Although the remainder of the lesions described by Elwell et al. are consistent with RRV infection, it is likely that they had a secondary bacterial infection concomitant with RRV. Transmission electron microscopy on those lungs demonstrated bacteria. We also did not observe an appreciable increase in BALT, the explanation for which is uncertain. The lack of increased BALT in our study may be the result of differences in the strain or infective agent, the shorter 13-week duration of our study, or that perhaps the increased BALT previously reported was because of stimuli other than RRV. Regardless of the explanation, increased BALT is a nonspecific finding, is not a constant feature of RRV infection, and should not be included in the criteria for diagnosis of RRV infection.
The lesions described in the present study, although generally consistent with previous descriptions, are distinct from most other respiratory infections of rats. 6 The lesions of RRV infection are nonsuppurative, which distinguishes them from known bacterial and mycoplasmal infections of laboratory rats. The lesions of RRV infection are not bronchocentric or bronchiolocentric, and no changes have been found in airways. This distinguishes RRV infection from infections as a result of Sendai virus, rat coronavirus, and polyomavirus. The only known virus of rats that produces nonsuppurative interstitial pneumonia and perivasculitis is pneumonia virus of mice (PVM). Although these lesions can resemble RRV, PVM is rare in contemporary laboratory rats 7 and the PVM-related lesions described previously were produced by experimental infection, whereas natural infection is considered asymptomatic. 5 It should also be noted that rats with RRV-associated lesions, and the cohorts of these rats are serologically negative to all of these other agents. Thus, the distinctive histologic features are sufficient for a positive diagnosis, although it is prudent to include serology, microbiologic culture, and perhaps other ancillary tests to exclude alternative diagnoses before taking actions that might disrupt research studies or breeding colonies.
The long time course of RRV infection and the necessity to use histopathology for detection have important implications in formulating rat health surveillance programs. Once exposed, a given sentinel rat is most likely to have diagnostic RRV lesions 8–12 weeks after the initial exposure. This period is longer than that required for other types of serologic and molecular testing in rats; seroconversion generally occurs to most agents within 2–3 weeks, 10 preceded by shedding and tissue infection, which can be detected by PCR. Thus, sentinel exposure periods for rats should be extended to be between 6 and 13 weeks, with 8–12 weeks being an optimal exposure duration. Although RRV was readily transmitted in our exposure experiment, the exposure load of RRV in most research facilities may be less and transmission to sentinels may vary in some situations from our study. In particular, the duration of RRV infectivity in soiled bedding is unknown. Thus, sentinel programs relying on soiled bedding transfer should seek to use bedding as fresh as possible and as much volume of it as possible. At least 2 factors will make RRV surveillance difficult on incoming rats into a research facility. First, the long lag time until development of lesions could require an extended quarantine period, especially if sentinels are monitored instead of the principal animals. Second, the necessity for euthanasia of rats to collect the appropriate diagnostic sample (lung) means that either sentinels will need to be used despite the extended quarantine period or that additional animals will need to be procured so that some may be used for RRV screening. However, if the additional rats for RRV surveillance are procured from an endemically infected colony, the prevalence of lesions may be less than that in naïve rats. 9 Additional considerations in using sentinel animals exposed by soiled bedding are that not all exposed rats will develop diagnostic lesions, that the concentration of the putative infectious agent in soiled bedding is unknown, and that its resistance to inactivation in aging soiled bedding are unknown. Thus, the efficacy of soiled bedding exposure of naïve sentinel animals is unknown.
RRV may be the most important infection of contemporary laboratory rats for 2 reasons. First, as noted previously, our diagnostic laboratory classifies approximately 6% of rats being submitted for health monitoring from external clients as positive for RRV infection, a prevalence several times higher than the second most prevalent rat pathogen, rat parvovirus. 7 In addition, unlike infection with rat parvovirus, which does not produce lesions, most, if not all, naïve rats infected with RRV will develop lesions. However, despite being the most common (putative) virus of laboratory rats, and the only common pathogen to produce gross or microscopic lesions, RRV is still a relatively unknown, poorly understood infectious agent in laboratory animal medicine and pathology.
Many crucial aspects of the infectious agent currently referred to as RRV remain to be clarified. Key unknowns include the taxonomy of the infectious agent, the pathogenesis including the spectrum of tissues and cell types infected, and why the pulmonary lesions develop so slowly, host resistance, the possibility of viral persistence, duration and routes of shedding, and whether other species are infected with or without production of lesions. Many of these questions may yield to investigation once the putative virus can be reproducibly cultivated in vitro. Importantly, isolation and characterization of the infecting agent is likely to allow development of serologic and molecular methods of detection. These will not only eventually supplant histopathology as the primary diagnostic methodology, but will permit more precise epidemiologic surveys, as well as facilitating further laboratory investigation of RRV.
Footnotes
Acknowledgements
We thank the Necropsy, Histology, and Serology laboratories at Charles River Research Animal Diagnostic Services for their excellent technical assistance, and Steve Jennings for his careful assistance in the trafficking and archiving of specimens.
