Abstract
The histopathologic changes induced in F344 rat kidney by oral administration of melamine for 13-week and 2-year periods in studies conducted by the National Toxicology Program, NIH,25 from 1976 to 1983 have been re-evaluated and described in detail. A constellation of tubule changes extending from papilla to cortex consistently included tubule dilatation and tubule basophilia as salient features at the subchronic time point. By 2 years, these lesions had usually resolved into fibrotic scars, in which tubule loss and collagen deposition were prominent, running from superficial cortex into the medulla. These fibrotic lesions required discrimination from chronic scars resulting from infarcts and foci of chronic progressive nephropathy (CPN). A case is presented here for interpreting the constellation of histologic changes induced in rats by melamine as representing an ascending form of nephropathy. The term retrograde nephropathy is considered to be the appropriate nomenclature for both the acute and chronic lesions. The cause for the reflux, emanating from the lower urinary tract, appeared not to be infection as an inflammatory response was not prominent. It can be speculated that melamine precipitation in the lower urinary tract created pressure effects through transient obstruction leading to the renal changes. These changes were different from those involved in a major US outbreak of renal disease and death in cats and dogs associated with triazine-contaminated pet food, in which crystalluria from insoluble melamine/cyanuric acid complexes occurred in the kidney. However, the rat findings may be relevant to melamine-associated kidney disease recently reported in infants in China.
Keywords
Introduction
Melamine, 1,3,5-triazine-2,4,6-triamine, is produced on a large scale in the United States and elsewhere. For example, China's production of melamine accounts for more than 50% of the total output in Asia and represents almost one quarter of the global output (M. L. Cunningham, NTP, NIEHS, Research Triangle Park, NC, personal communication). Melamine is used primarily for manufacturing melamine resins and melamine-based resins that have applications as starting materials for a wide variety of products, including laminates, adhesives, molding compounds, textiles, and flame retardants.25, 26 Although used mainly by the chemical industry, melamine was recently implicated in a food-borne outbreak of renal disease in domestic cats and dogs in the US.5, 6, 28 In this episode, commercial pet food was adulterated with melamine, which is high in nonprotein nitrogen, to boost the apparent protein content of the feed.
Because of its listing as a high-production volume chemical and potential for human exposure, the toxicity of melamine was studied by the National Toxicology Program (NTP), NIH, more than 25 years ago in separate single-gavage-dose, 14-day, and 3 13-week studies, as well as in a 2-year carcinogenicity bioassay in F344/N rats and B6C3F1 mice.25 In all cases (except the single-dose study), the test compound was administered in the diet. At the high doses (12,000 to 30,000 ppm) used in either the 14-day or 13-week rat studies, stone formation was observed in the urinary bladder, this effect being more severe in males than females. In male rats in particular, stone formation was associated with urothelial hyperplasia of the bladder lining. Kidney changes in male rats at 13 weeks of exposure were reported as “minimal,” but female rats at this time point were described as having “dose-related calcareous deposits in the straight segments of the proximal tubules.”25 In the 2-year carcinogenicity bioassay, male rats at the highest dose (4,500 ppm) had an increased incidence of transitional cell carcinomas of the urinary bladder, with all but 1 of the tumor-bearing rats having bladder stones as well. An association (P < .001) was therefore recorded between bladder stones and bladder tumors in male rats.25 Bladder tumors were not observed to be statistically increased in the high-dose females (9,000 ppm). The kidneys of female rats at the high dose were found on gross examination to have “pitted or roughened renal cortical surfaces,” and microscopically there was “chronic inflammation”; however, chronic inflammation was “not significant in dosed male rats.” The renal inflammation in females was considered to be distinguishable from the age-related, spontaneous condition of chronic progressive nephropathy (CPN) based on increased lymphoplasmacytic infiltrates and cortical fibrosis.25 No further histopathologic details of these renal changes were provided.
The primary intent of this re-evaluation of histopathology from the 13-week and 2-year NTP studies was to characterize the renal changes induced in rats by melamine in more detail and, based on the resulting evidence, to propose a likely pathogenesis. An additional aim was to determine if there was any similarity in the renal effects of melamine in the laboratory animal toxicity tests that would have been predictive for the renal disease encountered in cats and dogs following consumption of melamine-contaminated pet food.5, 28 Since the outbreak in domestic pets, an epidemic of infant kidney disease has emerged in China linked to melamine adulteration of milk products.27
Materials and Methods
The studies and groups examined in this histopathologic evaluation of kidney were 2 of the 3 13-week studies, and the 2-year carcinogenicity bioassay. The third 13-week study was not examined as it had included 1% ammonium chloride in the drinking water as a potential bladder stone inhibitor.25 Each of these studies had been conducted in F344/N rats and B6C3F1 mice. Only original glass histology slides were accessible for each of these studies as wet tissue and paraffin blocks were not available. The tissues had been stained with HE, and although some of the slides showed partial retraction of the mounting medium, the overall quality of the specimens was satisfactory and did not require restaining with HE.
The doses administered in the diet in these studies were as follows: first 13-week study: males and females, 0, 6,000, 9,000, 12,000, 15,000, and 18,000 ppm; second 13-week study: males and females, 0, 750, 1,500, 3,000, 6,000, 12,000 ppm; and 2-year study: males 0, 2,250, 4,500 ppm; females 0, 4,500, 9,000 ppm.
For the first 13-week study, only control (0 ppm), low-dose (6,000 ppm), and high-dose (18,000 ppm) kidneys had been sectioned and were available for histologic examination; for the second 13-week study, only control (0 ppm) and high-dose (12,000 ppm) kidneys had been prepared; for the carcinogenicity study, kidney sections for all 3 dose groups were available.
According to the NTP test protocol,25 technical grade melamine had been obtained from the American Cyanamid Company, Bound Brook, New Jersey, and was analyzed for purity by the Midwest Research Institute (MRI), Kansas City, Missouri, utilizing elemental analysis, water analysis, melting point, titration, thin-layer chromatography, high-performance liquid chromatography, and infrared and ultraviolet absorption spectroscopy. Periodic analyses by thin-layer chromatography and infrared spectroscopy indicated that no significant degradation occurred over the lifetime of the study. Test diets were prepared by first mixing a small amount of Purina Lab Chow (Ralston Purina Co., St. Louis, MO) and the required amount of melamine with a mortar and pestle, and then adding this premix to the required amount of animal meal by mixing for 10–30 minutes in a Patterson-Kelly twin-shell blender. Prepared diets containing 100,000 ppm melamine were analyzed at MRI and were found to be stable for 2 weeks at temperatures up to 45°C. For the 13-week studies, 3–4-week-old male and female F344/N rats (and B6C3F1 mice) were obtained from the Frederick Cancer Research Center (Frederick, MD), observed for 2 weeks, and then randomized by weight and assigned to test groups. For the 2-year study, 4-week-old male and female F344/N rats (and B6C3F1 mice) were obtained from Charles River Breeding Laboratories (Portage, MI), observed for 2 weeks, and assigned to individual cages according to a table of random numbers.25
In the histopathologic re-evaluation of these studies, the kidneys were examined for melamine-induced lesions but also for chronic progressive nephropathy (CPN) as this spontaneous disease entity can be a confounding influence in rat renal pathology.35 CPN was graded for severity on a scale of 4 grades: no lesions, minimal, mild, moderate, and marked, representing a modification of a previously reported schema.15 Compound-associated renal pathology was also graded on a 4-grade scale of minimal to marked severity.
Because no wet tissues or paraffin blocks were available, a single HE section that represented a duplicate slide was restained with Weigert's Van Gieson stain for confirmation of connective tissue increase.
Results
13-week studies
In the first 13-week study, for dose groups administered the high dose of 18,000 ppm of melamine, 10/10 male rats and 8/10 female rats had a constellation of lesions involving both cortex and inner medulla. At this dose, the changes appeared to be more severe in the male rats, the severity being graded as minimal to moderate in males, and minimal to mild in females. At the low dose of 6,000 ppm, only 1/9 male rats had an early form of the lesion, and none of the females was affected (Table 1). Some sections showed that the regional effects, usually observed separately in cortex and inner medulla, were continuous and part of the same overall lesion. Equivalent lesions were not observed in control (0 ppm) males or females.
Incidence of reflux nephropathy and spontaneous chronic progressive nephropathy (CPN) in melamine studies in F344 rats.
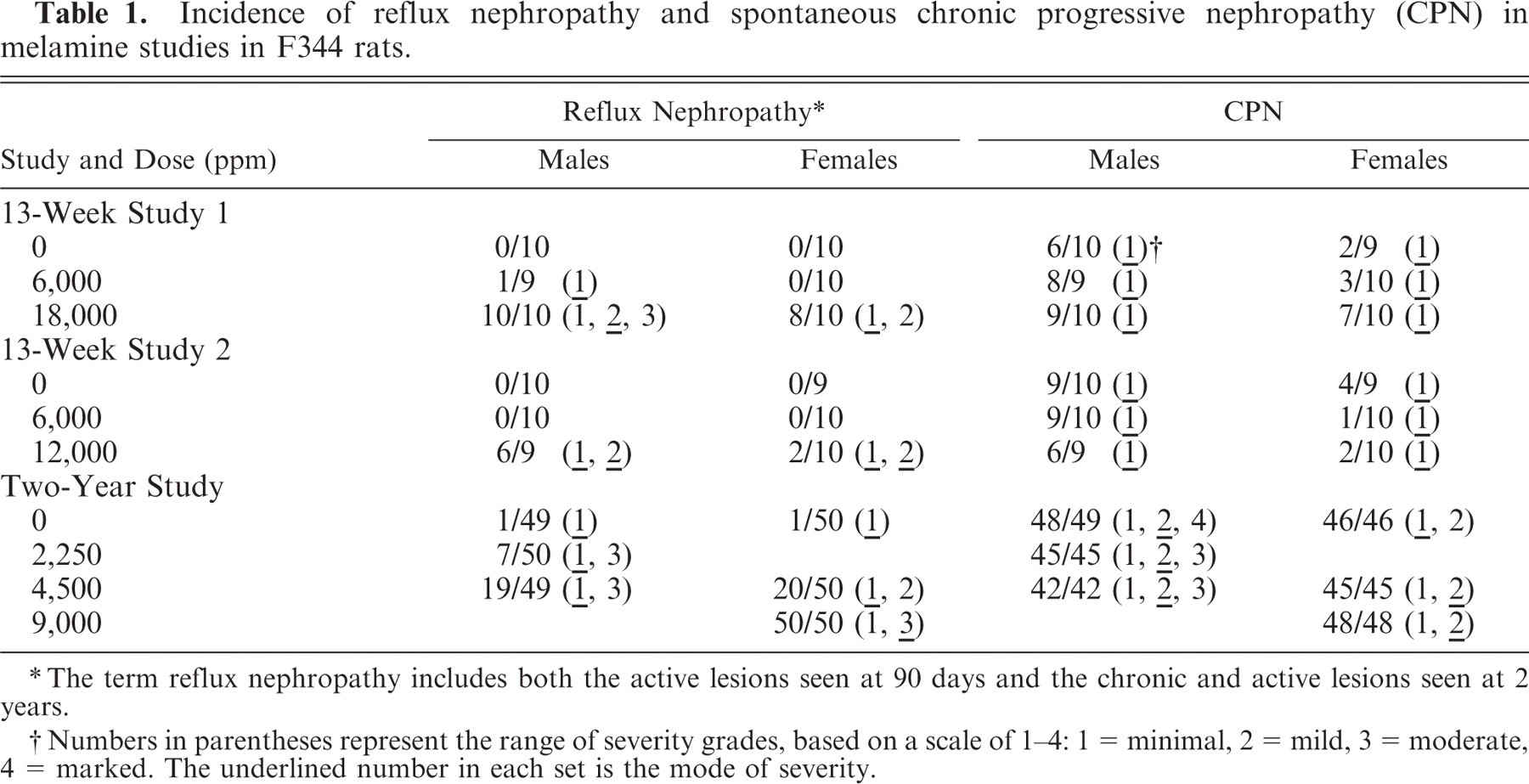
The term reflux nephropathy includes both the active lesions seen at 90 days and the chronic and active lesions seen at 2 years.
Numbers in parentheses represent the range of severity grades, based on a scale of 1–4: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked. The underlined number in each set is the mode of severity.
The cortical lesions in high-dose males and females included irregular areas of tubule basophilia involving the proximal and distal convoluted tubules of the cortex, and conspicuous tubule dilatation, which affected mainly distal tubules and collecting tubules in the cortex, extending into the outer medulla (Figs. 1–3). In contrast to the dilated distal tubules, proximal tubules were in varying stages of compression (Fig. 4). There was only a modest mononuclear cell infiltration associated with these basophilic areas, and blood vessels appeared unaffected. The cortical lesions were preferentially located in the poles of the kidney, rather than the curvature.
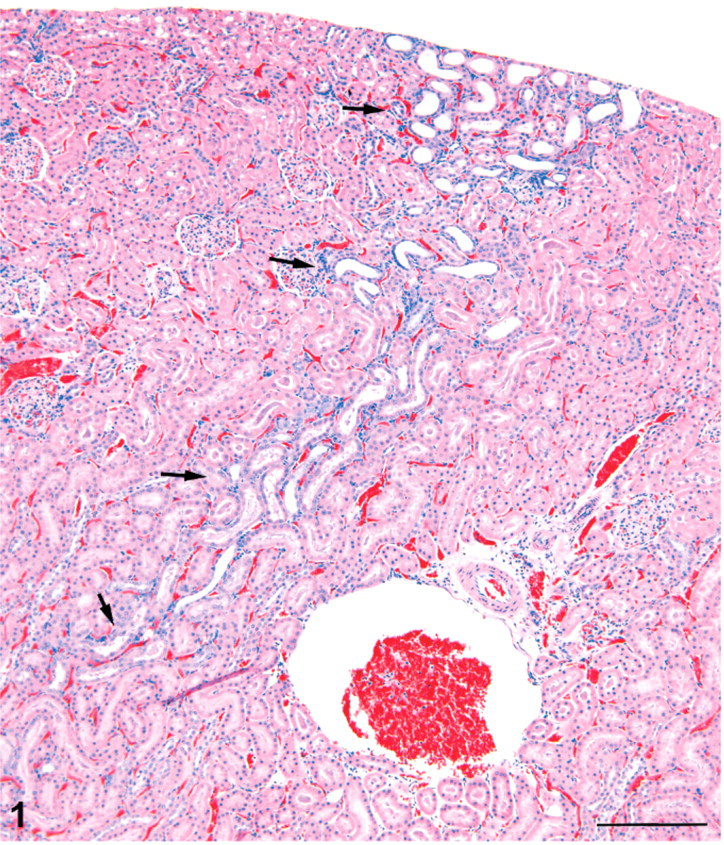
Kidney; male rat, 6,000 ppm melamine at 13 weeks. Basophilic lesion tracking axially through the cortex (arrows). Tubule dilatation is the most prominent feature. This is typical of an early stage of ascending or retrograde nephritis. HE. Bar = 200 μm.
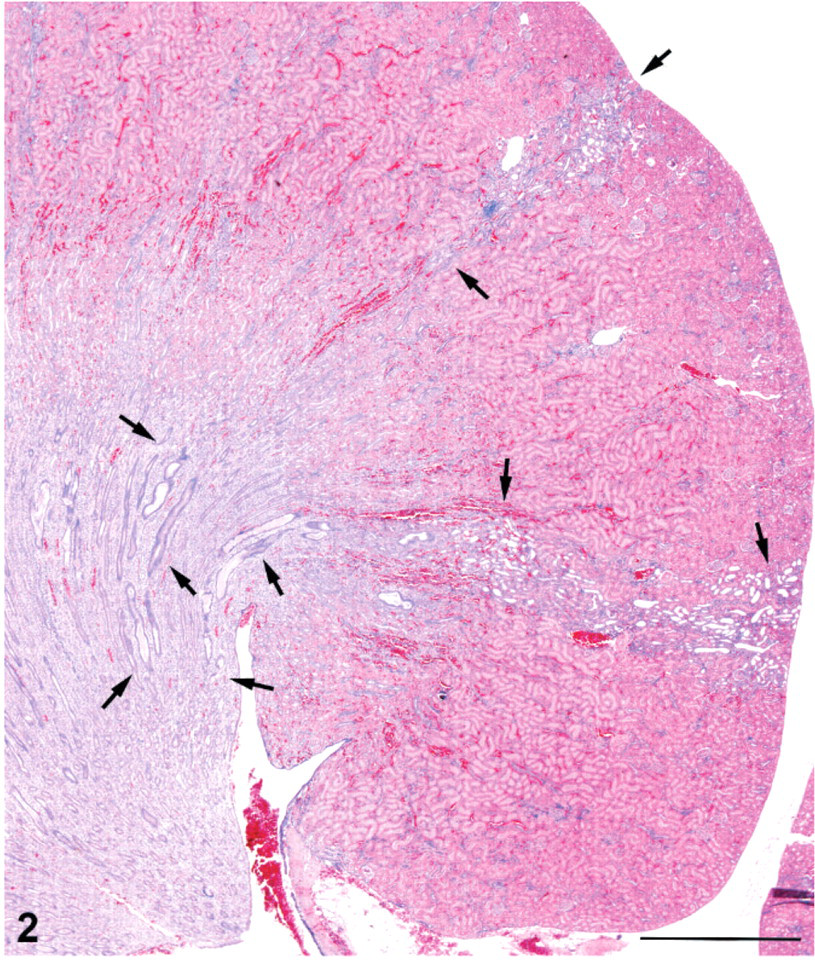
Kidney; male rat, 6,000 ppm melamine at 13 weeks. Basophilic tracts (arrows) in the cortex continue through the outer medulla and appear continuous with tracts of dilated, basophilic collecting ducts (arrows) in the inner medulla. HE. Bar = 1.0 mm.
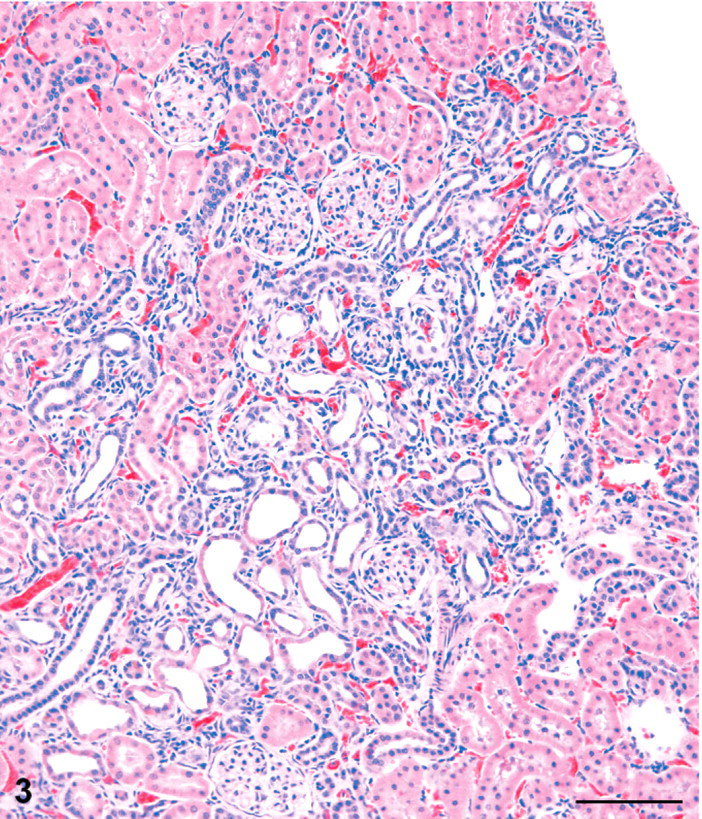
Kidney; male rat, 6,000 ppm melamine at 13 weeks. Cortical lesion is an irregular area of basophilia in which tubule dilatation is prominent. HE. Bar = 100 μm.
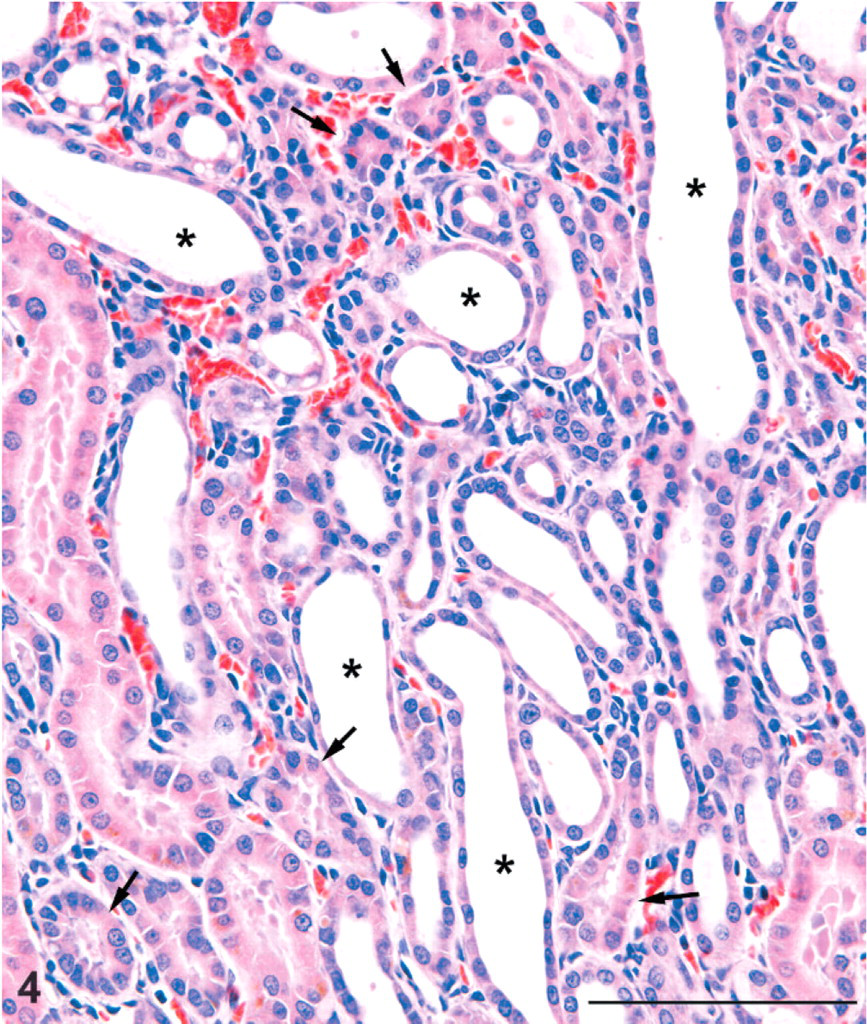
Kidney; male rat, 6,000 ppm melamine at 13 weeks. High magnification of cortical lesion showing dilatation of distal tubules (asterisks) and varying degrees of compression of proximal tubules (arrows). There is only a slight mononuclear cell reaction in the interstitium and no neutrophil infiltration. Tubule basement membrane thickening is not prominent. HE. Bar = 100 μm.
In the inner medulla, there were tracts of dilated collecting ducts, which appeared to extend from the outer medulla toward the tip of the papilla. The tracts of dilated collecting ducts in deep medulla tended to be distributed towards the lateral margins of the papilla (Fig. 5). Affected collecting ducts in the inner medulla were basophilic and characterized by a crowding of the lining epithelial cells, indicative of simple hyperplasia (Fig. 6). Some of these ducts had an occasional pyknotic cell with karyorrhexic nucleus and an increase in mitotic figures (Fig. 7). Sometimes eosinophilic debris was present in collecting duct lumens, and the affected ducts showed small foci of epithelial cell loss (Fig. 8). In only a few cases was there a minimal interstitial infiltration of neutrophil leukocytes, and occasionally these inflammatory cells were also present in collecting duct lumens. However, neutrophils were not typical of the renal alterations.
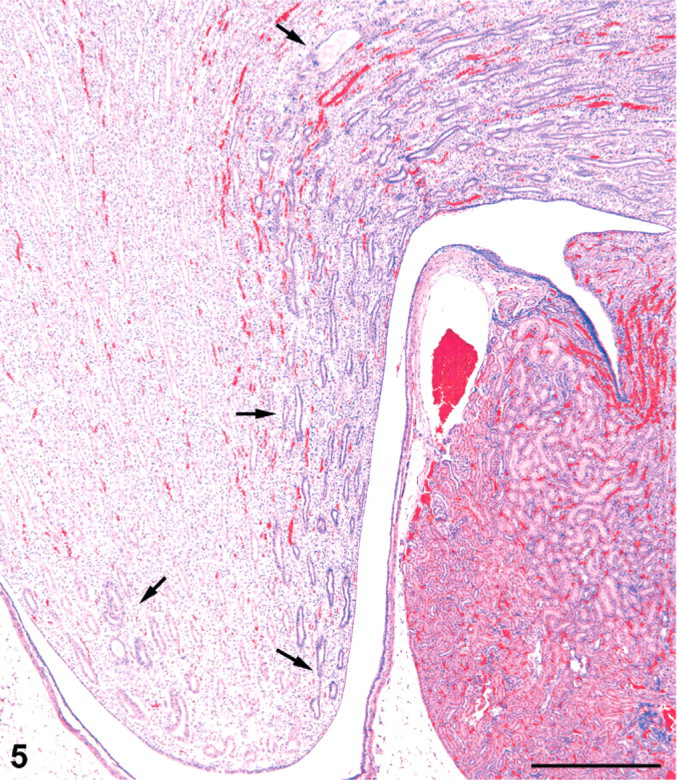
Kidney; female rat, 18,000 ppm melamine at 13 weeks. Tract of basophilic, mildly dilated collecting ducts in the inner medulla follows the margin of the papilla to the tip (arrows). A similar tract of dilated collecting ducts is located at the opposite margin of the papilla (arrow). HE. Bar = 500 μm.
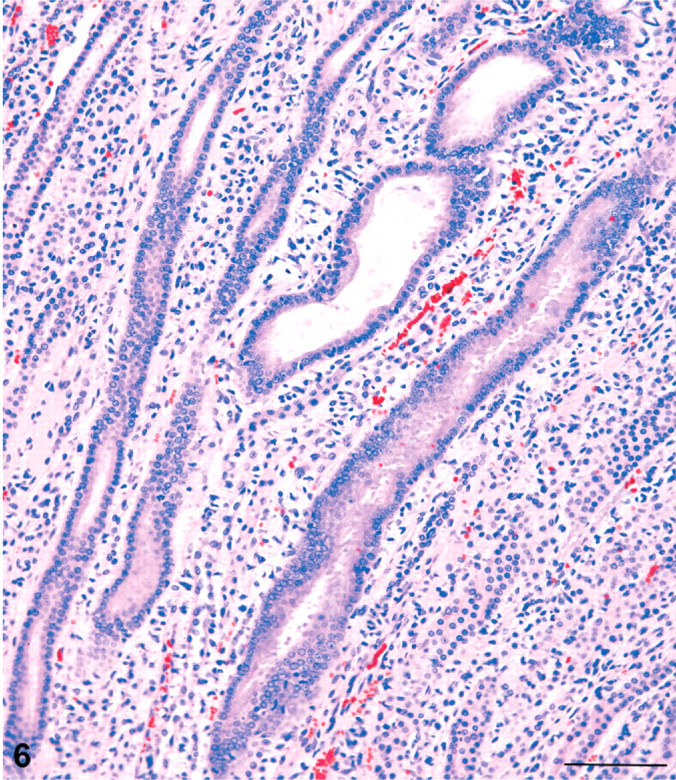
Kidney; male rat, 6,000 ppm melamine at 13 weeks. Basophilic, dilated collecting ducts in the inner medulla show marked crowding of nuclei, indicative of a hyperplastic response. HE. Bar = 100 μm.
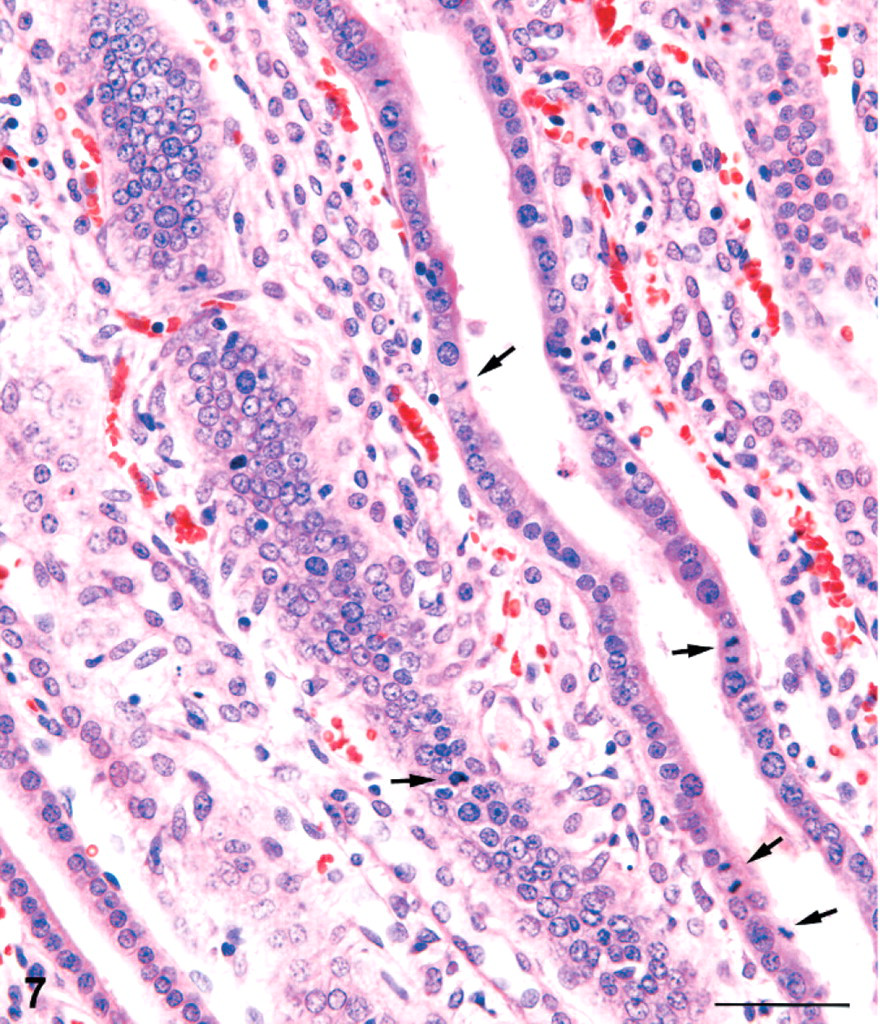
Kidney; male rat, 18,000 ppm melamine at 13 weeks. At high magnification, an increase in mitotic figures (arrows) can be observed in affected collecting ducts of the inner medulla. HE. Bar = 50 μm.
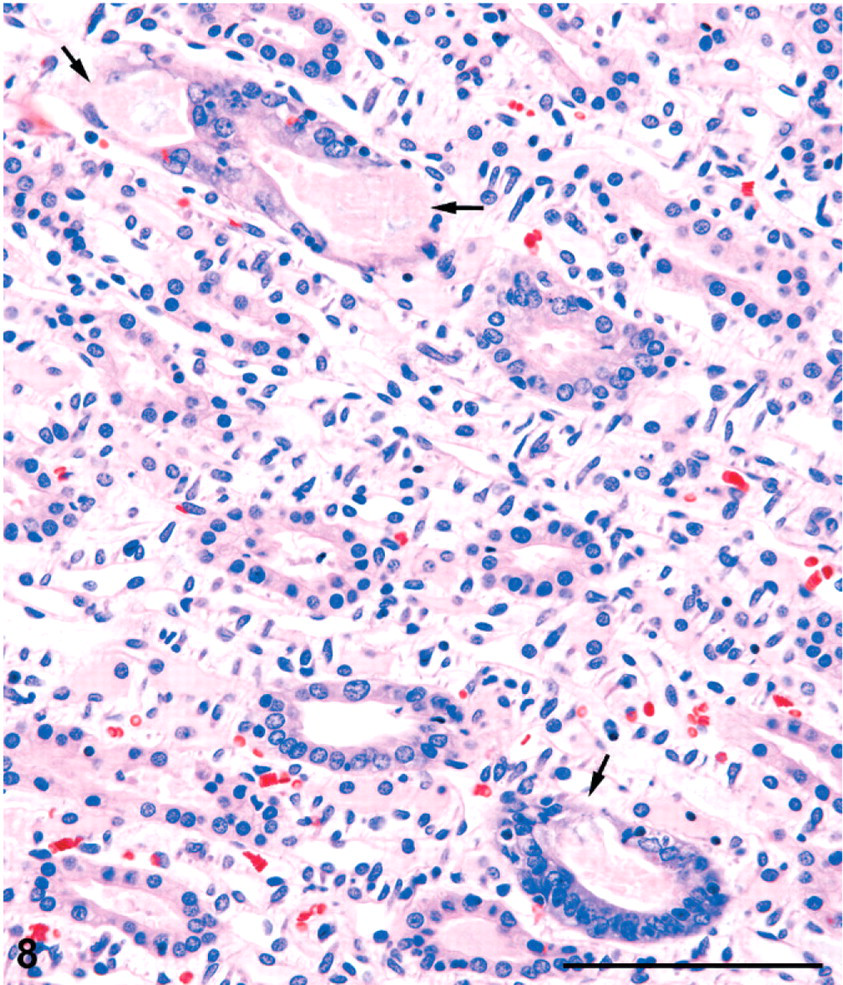
Kidney; male rat, 6,000 ppm melamine at 13 weeks. Papillary tip showing several collecting ducts with basophilic and crowded lining cells, sometimes with eosinophilic luminal contents. In 2 ducts, the lining appears not to be intact, with apparent, focal epithelial cell loss (arrows). HE. Bar = 100 μm.
In the second 13-week study where the doses administered were lower, the high dose of 12,000 ppm resulted in the same constellation of lesions as seen in the high-dose animals of the first 13-week study, in 6/9 male rats and 2/10 female rats. At the lower dose of 6,000 ppm and in the control rats (0 ppm), no similar lesions were observed in either sex (Table 1).
No birefringent crystals were present in the renal tissue in either 13-week study, suggesting an absence of melamine/cyanuric acid complex.12 However, solitary (nonbirefringent) concretions, were seen in the upper fornix of the renal pelvis in a few rats. These were small, rounded structures dissimilar to the irregular-shaped calcium-containing deposits often seen in the rat renal pelvis. Female rats in the first 13-week study had minimal to mild, dose-related tubular mineralization in the distal outer stripe of outer medulla (OSOM), an entity known to be associated with dietary mineral imbalance.21, 30 Melamine did not cause exacerbation of CPN at this time point (Table 1), with both males and females exhibiting a severity not exceeding grade 1 (minimal).
Two-year study
At the highest dose of 9,000 ppm, 50/50 female rats showed either multiple cortical scars or linear scars associated with collecting duct dilatation and hyperplasia in the inner medulla (Figs. 9, 10). In some sections, the cortical scars were continuous with prominent, fibrous tracts extending through the outer medulla and were connected to the tracts of dilated, basophilic collecting ducts in the inner medulla. Similar to the 13-week studies, affected medullary collecting ducts had crowded nuclei indicative of hyperplasia. Linear scarring (minimal) was observed in only 1/50 control female rats (Table 1).
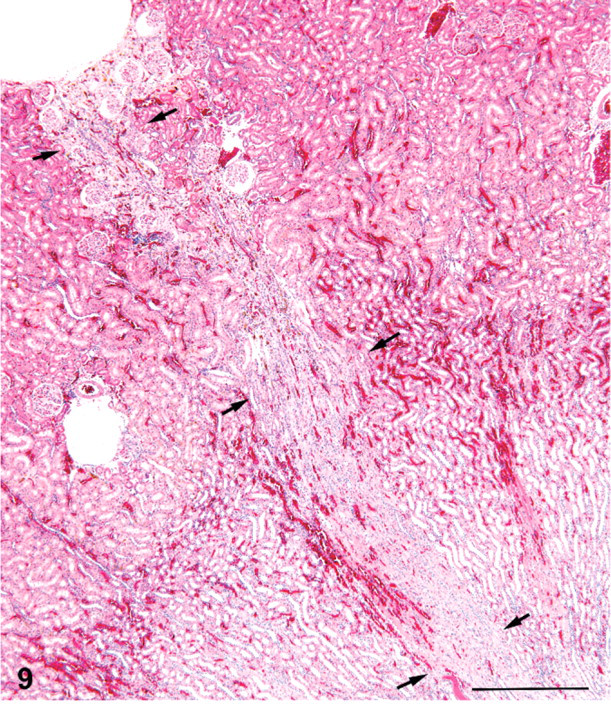
Kidney; female rat, 9,000 ppm melamine at 2 years. Fibrotic tract (scar) running from a surface depression in the outer cortex through the outer medulla and into the proximal inner medulla (arrows). HE. Bar = 500 μm.
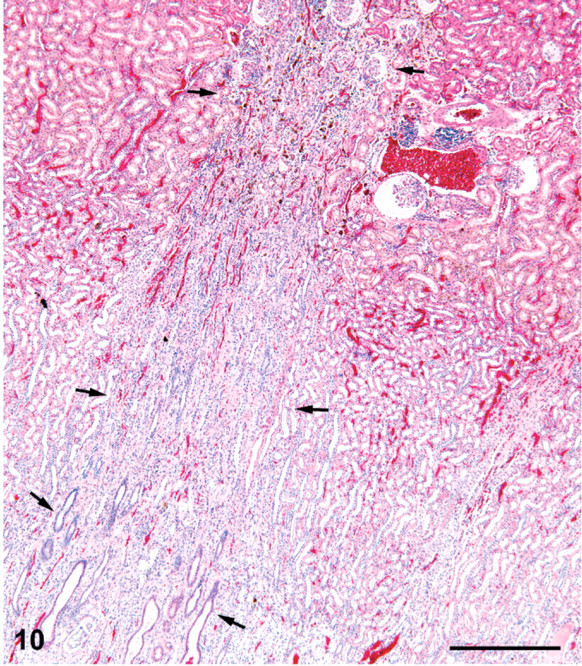
Kidney; female rat, 9,000 ppm melamine at 2 years. Fibrotic scar in cortex and outer medulla is continuous with a tract of basophilic collecting ducts in the inner medulla. HE. Bar = 500 μm.
Small cortical lesions consisted of tracts forming a dimplelike depression of the capsule. The cortical scars, however, were often quite large, causing troughlike depressions of the renal surface (Fig. 11). The linear tracts were fibrotic with a prominent loss of tubules, tubule atrophy, and crowded glomeruli in the cortex (Fig. 12). Most atrophic tubules that could be visualized contained brown pigment resembling hemosiderin deposits. Weigert's Van Gieson stain on the single section available confirmed an increase in collagen deposition and a paucity of mononuclear inflammatory cells in the scars (illustration not included). Glomeruli in these tracts were crowded because of tubule loss but showed little structural change other than slight contraction of the tuft in some cases, dilatation of some Bowman's spaces, and minimal thickening of Bowman's capsule basement membrane (Fig. 12). Dilatation of Bowman's spaces ranged from minimal to moderate in severity.
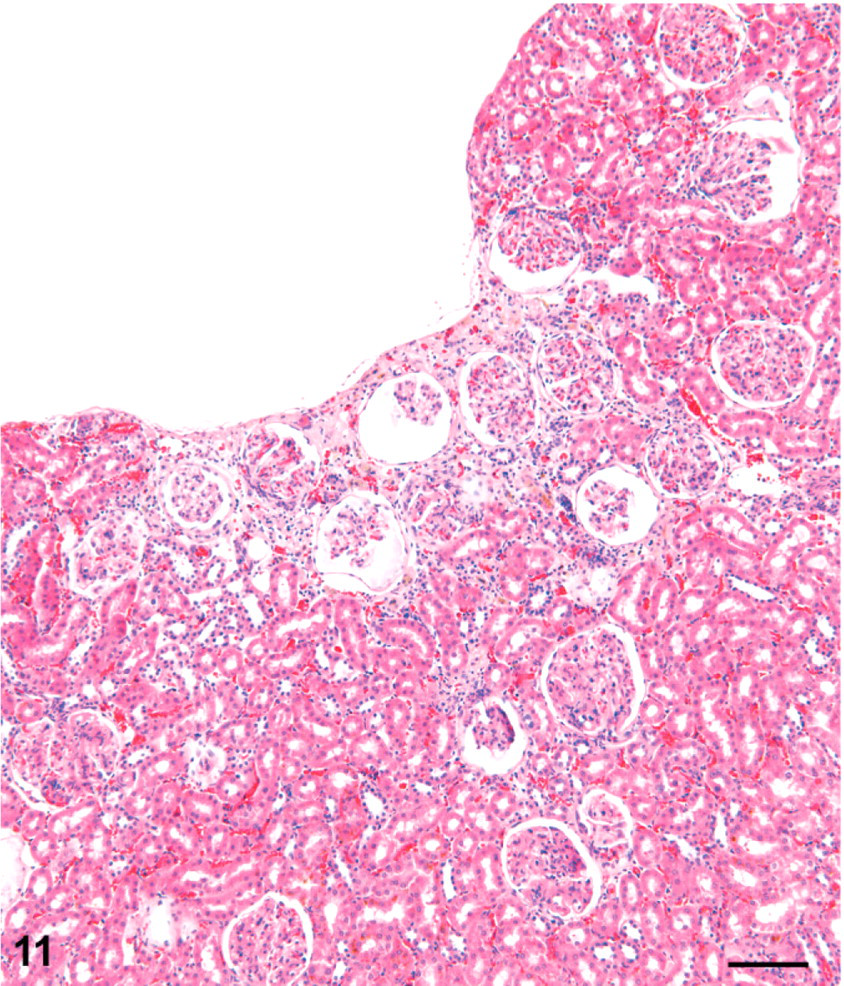
Kidney; female rat, 9,000 ppm melamine at 2 years. Outer portion of a fibrotic scar is typified by a trough-shaped depression of the kidney surface. The deeper extension of this lesion is not in the plane of section. Note the mild dilatation of Bowman's spaces in glomeruli. HE. Bar = 100 μm.
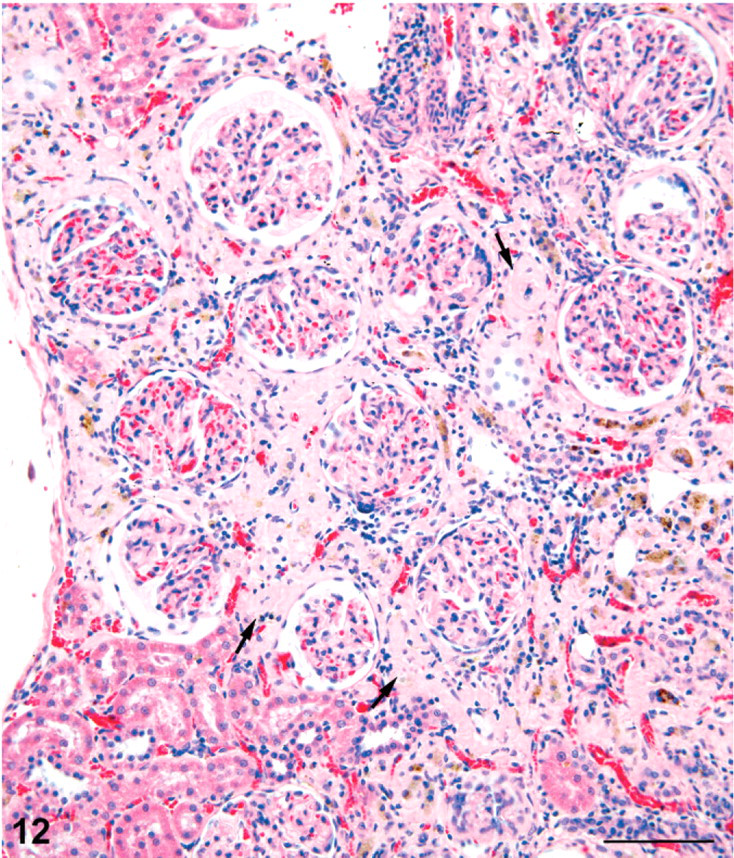
Kidney; female rat, 9,000 ppm melamine at 2 years. High magnification of cortical portion of a fibrotic scar showing prominent tubule loss, crowding of glomeruli, and brown pigment in some atrophic tubules. Apart from slight dilatation of Bowman's space and minimal thickening of Bowman's capsule basement membrane, the glomeruli are relatively unaffected. HE. Bar = 100 μm.
In male rats at the highest dose of 4,500 ppm, 19/49 showed melamine-associated renal lesions (Table 1), either cortical scars, as described above, and/or medullary fibrous tracts associated with collecting duct dilatation, or lesions more typical of the active phase of nephritis as seen in the 13-week studies (4 animals). In the control males, 1/49 rats had linear scarring (minimal severity).
CPN was present in all rats but 1, examined in the 2-year study, and melamine did not exacerbate its incidence or severity (Table 1). The mode of severity was grade 2 (mild) in all 3 groups of male rats, and grade 1 (minimal) or grade 2 (mild) in the female rat groups.
Discussion
In this re-evaluation of renal histopathology associated with dietary melamine exposure, rats were found to be affected by a constellation of changes in the 13-week studies, involving both cortex and medulla. These active lesions appeared to have resolved into fibrotic scars in the 2-year study. Occasional sections at each of these 2 time points showed that the cortical and deep medullary lesions were connected and part of the same process. Considering the incidence of these lesions and the difference in dosage between the sexes in the carcinogenicity bioassay, males were more susceptible than females.
The renal changes observed in the 13-week and 2-year studies with melamine are similar to those reported by Heptinstall16, 17 and others3, 24, 34 in experimentally induced, ascending, nonobstructive pyelonephritis in rats. In particular, the scarring observed at 2 years was very similar to the histologic lesions described in experimental nephropathy produced in both rat and swine models of ascending pyelonephritis or vesicoureteral reflux.10, 16, 17, 20, 24 The melamine-associated rat renal changes were also consistent with the features of human reflux nephropathy (chronic atrophic pyelonephritis) of early childhood.11, 19
The cortical scars observed in the 2-year study were reminiscent of chronic infarcts but differed in several ways. The larger ones caused trough-shaped depressions of the surface and connected with fibrous tracts in the outer medulla. They did not taper axially as infarcts do but tended to retain full width as they led into the deeper fibrotic tracts. As indicated above, in occasional sections, they could be seen to connect with tracts of dilated and basophilic collecting ducts in the inner medulla.
There was also a need to differentiate the melamine-associated lesions from spontaneous CPN. In comparison with the melamine-induced lesions, basophilic foci of CPN were better defined in outline, and the tubules within CPN foci exhibited conspicuously thickened basement membranes. CPN lesions were often associated with hyaline protein casts in the outer/inner medulla.
The sequence of pathologic change associated with melamine administration is interpreted as an ascending effect emanating from the urinary tract and therefore representing a retrograde or reflux type of nephropathy. In addition to the strong similarity of the pathology to experimental models of ascending pyelonephritis, the distribution of lesions is supportive. The poles of the cortex are drained by collecting ducts that successively fuse and are restricted to the margins of the papilla.16, 17, 22 The preferential involvement of the poles of the cortex, with lesions tracking to the renal pelvis via the margins of the papilla is considered to be indicative of a retrograde, ascending effect.17
Hodson and Edwards have drawn attention to the importance of intrarenal reflux in the pathogenesis of pyelonephritis18 and showed that sterile reflux can lead to renal scars in the areas of intrarenal reflux in the pig.19 In pigs and humans, intrarenal reflux has been defined as a form of pyelotubular backflow, entering the renal parenchyma via the ducts of Bellini and eventually filling both collecting ducts and nephrons.19 In rats, it has been recorded that vesicoureteral reflux is a spontaneous phenomenon, occurring under resting conditions and during bladder contraction.9, 32
There are several recognized forms of pyelonephritis: obstructive, bacterial embolic, and ascending.11, 13 Ascending pyelonephritis involving intrarenal or vesicoureteral reflux can be a result of pressure effects,4, 19 ureteral obstruction,31 or ascending bacterial infection.10, 29 However, the view is held that infection is not a prerequisite for development of reflux nephropathy.2, 19 It has been suggested also that Tamm-Horsfall protein may be involved, possibly in an autoimmune type of response.1
The precise mechanism (increased ureteral pressure, obstruction, or infection) involved in the ascending nephropathy associated with melamine exposure in rats could not be identified with certainty. The absence of PMNs in all but a few cases suggests that it was not a consequence of infection. Although no direct correlation of renal histopathology with melamine-induced stone formation in the bladder was recorded in the NTP report,25 it remains possible that melamine precipitation in the lower urinary tract created pressure effects leading to increased reflux. This is likely to have been a transient effect as active lesions were found only in the 13-week study. Spontaneous reflux in the rat may have predisposed to this course of events. Calculi formed in the urine can dissolve or become small enough to be voided, leaving no sign of their presence except for the secondary effects in the renal parenchyma.8 The apparent absence of any progressive increase in the active nephropathic change by 2 years may be related to the lower doses of chemical used in the 2-year studies as well as the tendency of rats to eat approximately the same amount of food throughout a 2-year bioassay despite their increase in body size.8 Accordingly, the concentration of chemical in the urine, and the associated damage, will also decline over time. These factors may explain why the effects were more acute in the 13-week study but had the appearance of resolution into a chronic, inactive lesion in the 2-year study. The sex difference in lesion incidence can be explained by the fact that female rats are less prone to the development of calculi than males.8
Although not reported here, we observed similar changes indicative of a retrograde nephropathy in B6C3F1 mice in the NTP 90-day study with melamine but at a much lower incidence and severity. Unlike the rat at high doses, however, no residual scarring was observed in mice exposed to melamine for 2 years. These observations suggest that the mouse is much less prone to melamine-induced renal pathophysiologic effects than the rat.
In the spring of 2007 in North America, an outbreak of renal failure in domestic cats and dogs associated with renal tubule crystalluria resulted in a major recall of pet food.6 The outbreak was traced to specific lots of food that had been formulated with wheat gluten imported from China. Analysis of the pet food and gluten demonstrated a range of simple triazine compounds, primarily melamine and cyanuric acid. Rodent tests then demonstrated that mixtures of triazines, including melamine/cyanuric acid mixture, produced melamine cyanurate crystalluria and acute toxicity in renal tubules.12 Thus, histopathology from the rodent bioassays of melamine conducted many years ago would not have been predictive for the recent domestic pet problems since cyanuric acid complexing with melamine appeared to be critical for the precipitation of crystals in the cat and dog kidney.5, 12, 28, 33
On the other hand, the kidney changes resulting from melamine exposure in rodents might be pertinent to the recent epidemic of high morbidity and low mortality from kidney injury in infants in China, linked to reported adulteration of milk products with melamine.27, 36 Powdered milk infant formulas associated with the outbreak had a melamine content of 500 ppm or greater,14 but cyanuric acid appeared not to be a contaminant in the urine of affected children.23 This outbreak was typified by the development of urinary tract stones, and premature birth and melamine-contaminated formula were the factors associated with urinary stones.14 The infant kidney may be more prone to scarring from severe intrarenal reflux because it is undergoing postnatal changes,7 particularly in premature infants.14 Thus, it is possible that the rat model is predictive for melamine-induced renal pathophysiologic effects in humans.
In summary, a review of the histopathology from the 13-week and 2-year NTP studies of melamine in F344 rats (reported in 1983) revealed cortical and medullary tubular changes with features consistent with retrograde nephropathy. Although exposure to melamine alone in these rat studies resulted in bladder calculi, the renal disease itself was relatively mild and devoid of the crystalline deposits that characterized the severe renal disease occurring in cats and dogs from contaminated pet food, which was associated with a melamine/cyanuric acid complex.
Footnotes
Acknowledgements
The preparation of this manuscript was supported by Federal funds from the National Institute of Environmental Health Sciences (NIEHS), NIH, under contract NO1-ES-95435 to Experimental Pathology Laboratories (EPL) Inc., Research Triangle Park, North Carolina. One author (GCH) is very grateful to Drs. Jerry Hardisty and Mel Hamlin of EPL, and to Dr. Ron Herbert, NIEHS, for the opportunity to work on this project. The authors acknowledge Maureen Puccini and Emily Singletary of EPL for their expert assistance with photography, and Nancy Harris (EPL) for special histologic staining of kidney tissue.
