Abstract
We report herein a case of collagenofibrotic glomerulonephropathy in a 3-year-old Shiba Inu with severe proteinuria. Histologically, renal glomeruli were enlarged with massive deposition of a homogeneous eosinophilic substance within the mesangium and capillary walls. The deposits reacted weakly with periodic acid-Schiff, stained deep blue with Masson's trichrome, and were positive by immunofluorescence for type III collagen and fibronectin. Ultrastructurally, the deposits consisted of fibrils and amorphous material in the mesangial matrix and beneath the glomerular capillary endothelium. The fibrils had transverse bands analogous to those of collagen fibrils. Electron microscopy also revealed focal detachment of podocytes and foot process effacement in glomerular tufts, which suggested that podocyte injury had contributed to the development of proteinuria in this dog. The current case resembles collagenofibrotic glomerulonephropathy (CFGN) in humans in histopathologic, immunofluorescence, and electron microscopic findings. This is the first report of CFGN in a nonhuman species with glomerular deposition of fibronectin and type III collagen.
Introduction
Collagenofibrotic glomerulonephropathy (CFGN) is characterized by massive accumulation of type III collagenous fibrils within the mesangial matrix and subendothelial area of renal glomeruli capillaries. Only a few reports have described spontaneous renal glomerular disease consistent with human CFGN in nonhuman primates, 1 dogs, 9 a cat, 10 and pigs. 13,14 Detailed features of CFGN in dogs have not been well documented, although 3 canine cases of glomerulosclerosis similar to human CFGN have been described. 9 We report the morphologic and immunofluorescent findings in a canine case comparable to CFGN with typical type III collagen deposition in renal glomeruli.
Clinical Information
A 3-year-old female Shiba Inu was presented with anorexia. Serum biochemistry abnormalities included high levels of blood urea nitrogen (BUN; 44.3 mg/dl; reference interval [RI], 10–28 mg/dl) and creatinine (2.3 mg/dl; RI, 0.5–1.5 mg/dl). Three months after the first hematologic examination, re-examination revealed increased levels of BUN (67.3 mg/dl) and creatinine (4.0 mg/dl), along with mild hypoproteinemia (total protein, 5.8 g/dl; RI, 6.0–8.0 g/dl) and hypoalbuminemia (2.1 U/liter; RI, 2.8–4.0 U/liter). Proteinuria (712 mg/dl; RI, < 100 mg/dl) and a high urine protein : creatinine ratio (7.33; RI, < 0.6), indicative of renal glomerular disorder, were observed by urinalysis. No clinical signs were observed in the parents or littermates of this dog.
Materials and Methods
A renal biopsy specimen was fixed in 10% phosphate-buffered formalin, processed routinely, and embedded in paraffin. HE stain, periodic acid–Schiff (PAS) reaction, and Masson's trichrome stain were used for histopathologic examination. For electron microscopy, paraffin-embedded biopsy tissue was dewaxed, rehydrated, postfixed in 1% osmium tetroxide, and embedded in Epon. Ultrathin sections were stained with uranyl acetate and lead citrate. The double-labeled immunofluorescence technique was applied to paraffin sections, which were dewaxed and pretreated with 0.2% pepsin. Sections were incubated with rabbit anti–type-III collagen (LSL, Tokyo, Japan) for 1 hour at room temperature. After washing with phosphate-buffered saline (PBS), sections were stained with fluorescein isothiocyanate (FITC)–conjugated goat anti-rabbit immunoglobulin (IgG; Cappel, Durham, NC). Sections were rewashed with PBS then reacted with rabbit anti–type-IV collagen (LSL) for 1 hour at room temperature. After washing in PBS, sections were reacted with tetramethylrhodamine isothiocyanate (RITC)–conjugated goat anti-rabbit IgG (GeneTex, San Antonio, TX). For immunostaining of fibronectin, paraffin sections were pretreated with 0.2% trypsin, and rabbit anti-fibronectin antibodies (Dako, Tokyo, Japan) were applied as primary antibodies. Sections were stained with FITC-conjugated goat anti-rabbit IgG. Immunofluorescence was observed with the use of an Olympus microscope equipped with epi-illumination optics and appropriate filters. Immunohistochemistry for α-smooth muscle actin (α-SMA) was carried out on the paraffin sections with mouse anti–α-SMA antibodies (clone1A4, Dako) as the primary antibody. Immunoreaction was visualized by a diaminobenzidine–hydrogen peroxide solution. Positive controls for type III collagen and fibronectin were interstitial connective tissue and, for α-SMA, smooth muscles of small arteries in the same section. Normal animal serum or IgG was used as a negative control for each primary antibody.
Results
Histologically, all glomeruli in the biopsy specimen were enlarged with accumulation of a homogeneous eosinophilic substance in mesangium and capillary walls (Fig. 1). The homogeneous substance was weakly PAS-positive (Fig. 2) and stained deep blue with Masson's trichrome (Fig. 3), indicating intraglomerular accumulation of collagen fibers. The glomerular basement membrane (GBM) was moderately thickened with subendothelial accumulation of a homogeneous substance that resulted in narrowed capillary lumens. The mesangium was expanded by the accumulation of the homogeneous substance (Fig. 2). Mild segmental mesangial cell proliferation was observed in a few glomeruli (Fig. 2). Some mesangial cells were positive for α-SMA within the segmental proliferations. Adhesions of glomerular tufts to Bowman's capsule and segmental crescents were apparent in several glomeruli. Mild infiltration by lymphocytes and plasma cells and fibrosis was found in the glomerular interstitium.

Renal cortex; dog. Marked expansion of glomeruli by homogeneous eosinophilic substance and mild interstitial lymphocytic infiltration. HE. Bar = 100 µm.

Renal glomerulus; dog. Deposits of homogeneous substance expand the mesangium and subendothelial aspect of capillary walls. Segmental mild mesangial proliferation was also observed (arrow). PAS. Bar = 50 µm.
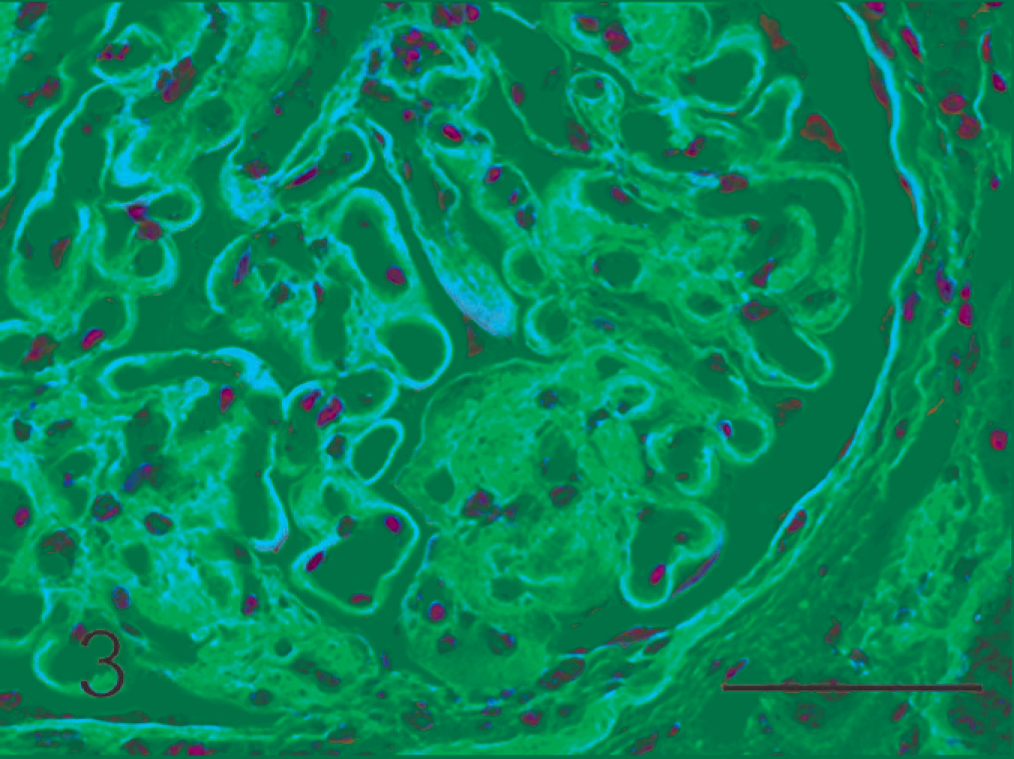
Renal glomerulus; dog. Mesangial area and capillary walls are stained deep blue. Masson's trichrome stain. Bar = 50 µm.
The deposits in glomerular capillary walls and mesangium were labeled diffusely by immunofluorescent antibody to type III collagen (Fig. 4). Double immunostaining elucidated different localizations of type IV and type III collagen in the glomerular tufts. Type IV collagen was along the epithelial side and type III along the endothelial side of the glomerular capillary wall; both had a linear appearance (Fig. 5). Fibronectin was also strongly labeled in the capillary walls and mesangium, coinciding with localization of type III collagen deposition (Fig. 6).
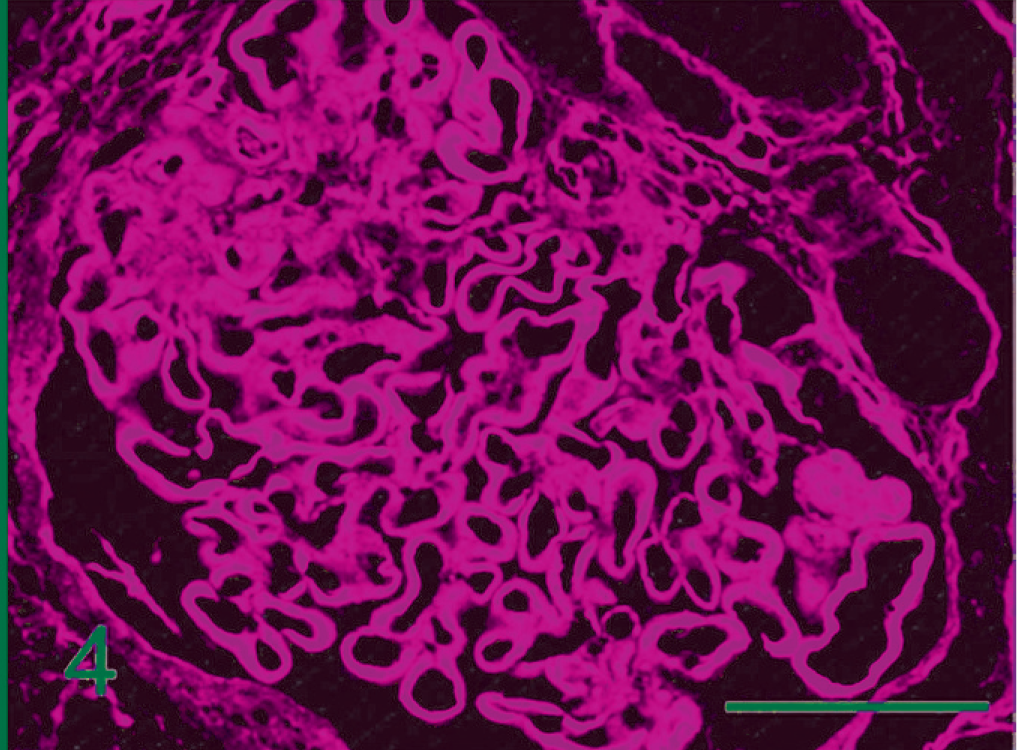
Renal glomerulus; dog. The mesangial area and capillary walls are expanded by type III collagen (red). Indirect immunofluorescence, with the use of primary antibody for type III collagen and RITC-conjugated secondary antibody. Bar = 50 µm.
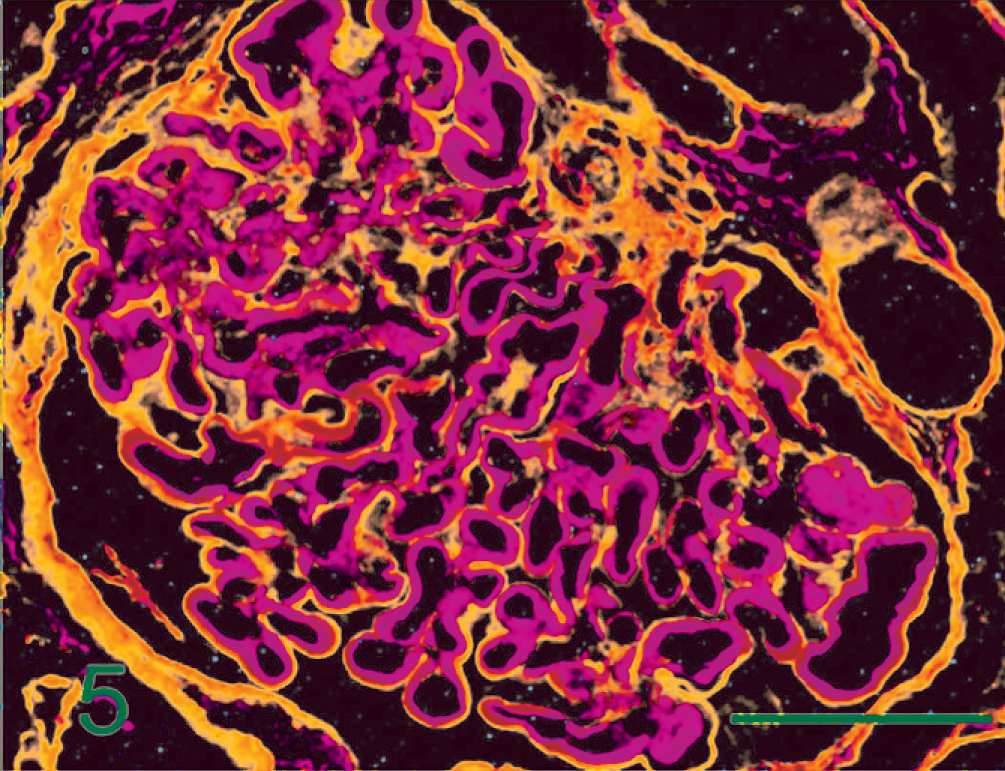
Renal glomerulus; dog. Type IV collagen (green) and type III collagen (red) in glomerular tufts is localized along the epithelial and luminal sides, respectively, with linear appearance. Indirect double immunofluorescence with primary antibodies for type III or type IV collagen and RITC- or FITC-conjugated secondary antibodies. Bar = 50 µm.
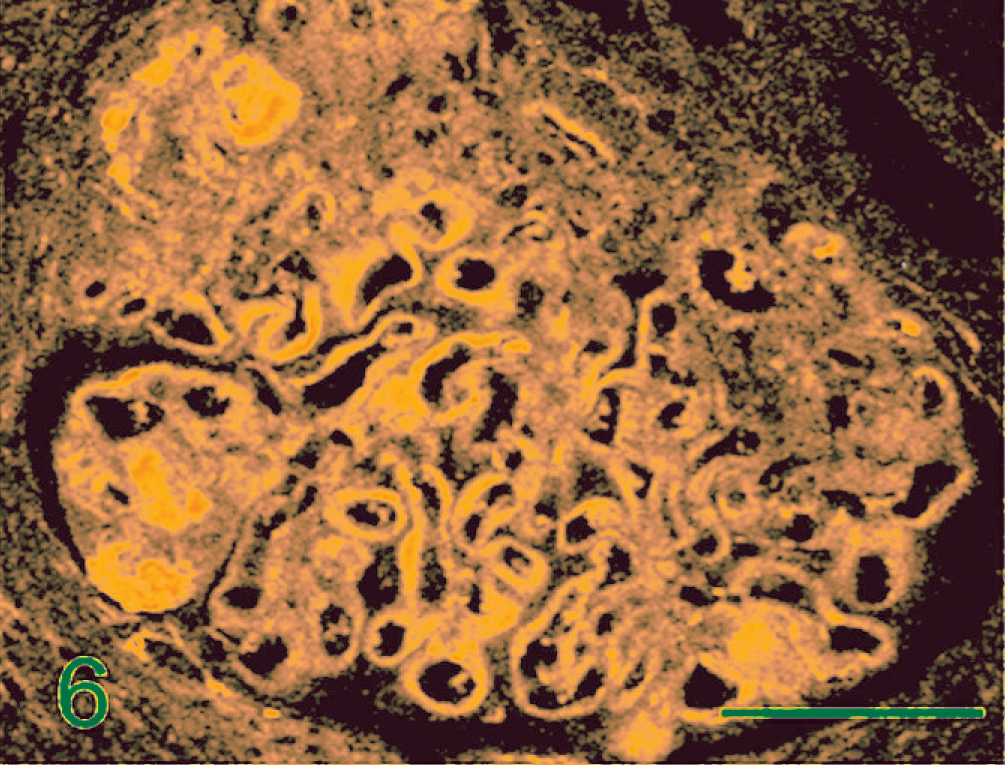
Renal glomerulus; dog. Fibronectin (green) is deposited in capillary walls and mesangial area. Indirect immunofluorescence with the use of a primary antibody for fibronectin and FITC-conjugated secondary antibody. Bar = 50 µm.
Electron microscopically, the GBM was moderately thickened, and the mesangial matrix and subendothelial space of the capillary walls were markedly expanded by the accumulation of collagenous fibrils and amorphous material (Fig. 7). The collagen fibrils had transverse bands and were thin, frayed, and spiraled or curved. Epithelial foot process effacement was observed multifocally (Fig. 8).
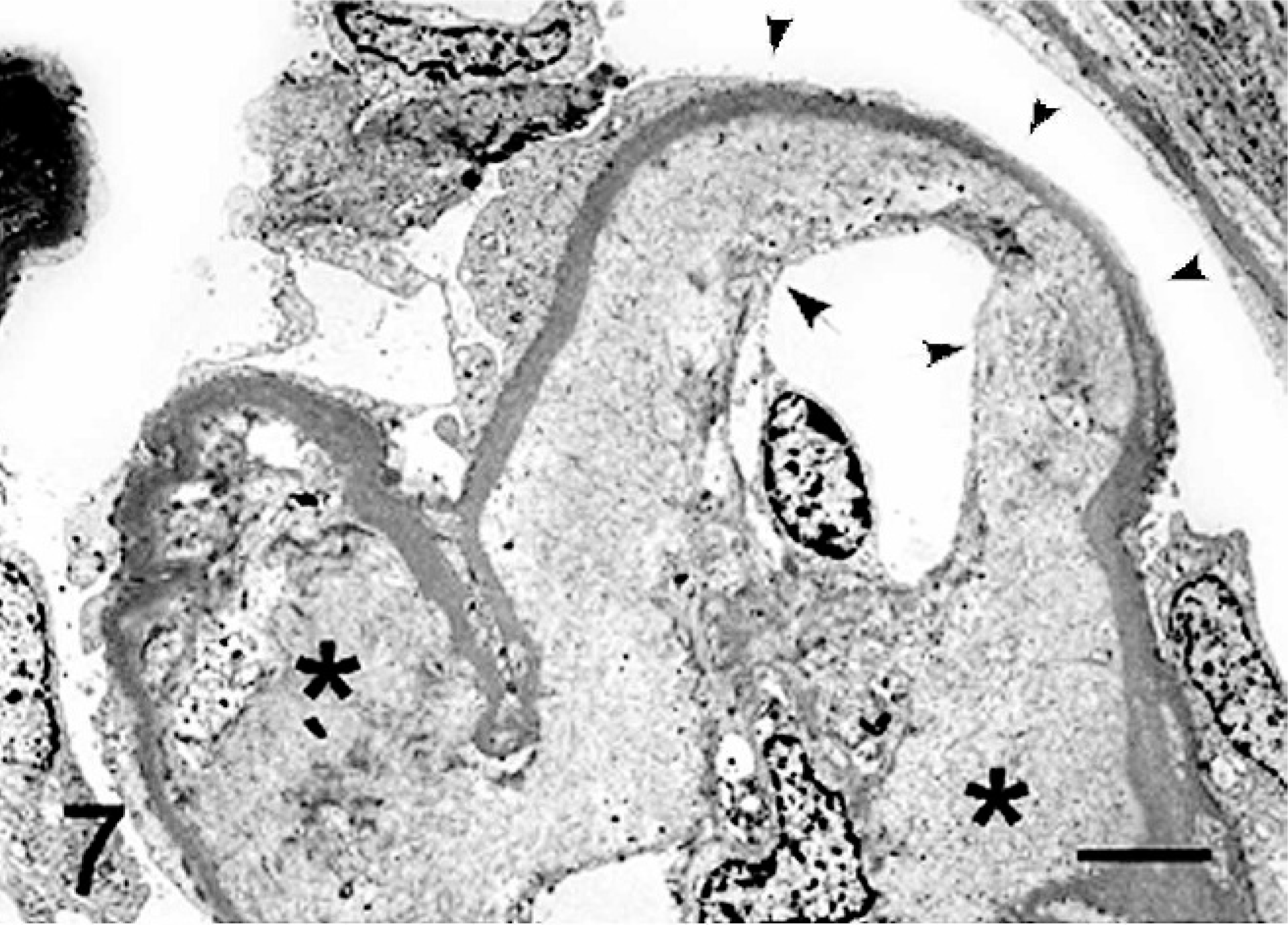
Electron micrograph. Kidney; dog. The mesangium matrix (asterisks) and subendothelial part of glomerular capillary walls (arrows) are expanded by collagen fibrils and amorphous material. Epithelial foot processes are effaced (arrowheads). Uranyl acetate and lead citrate. Bar = 5 µm.
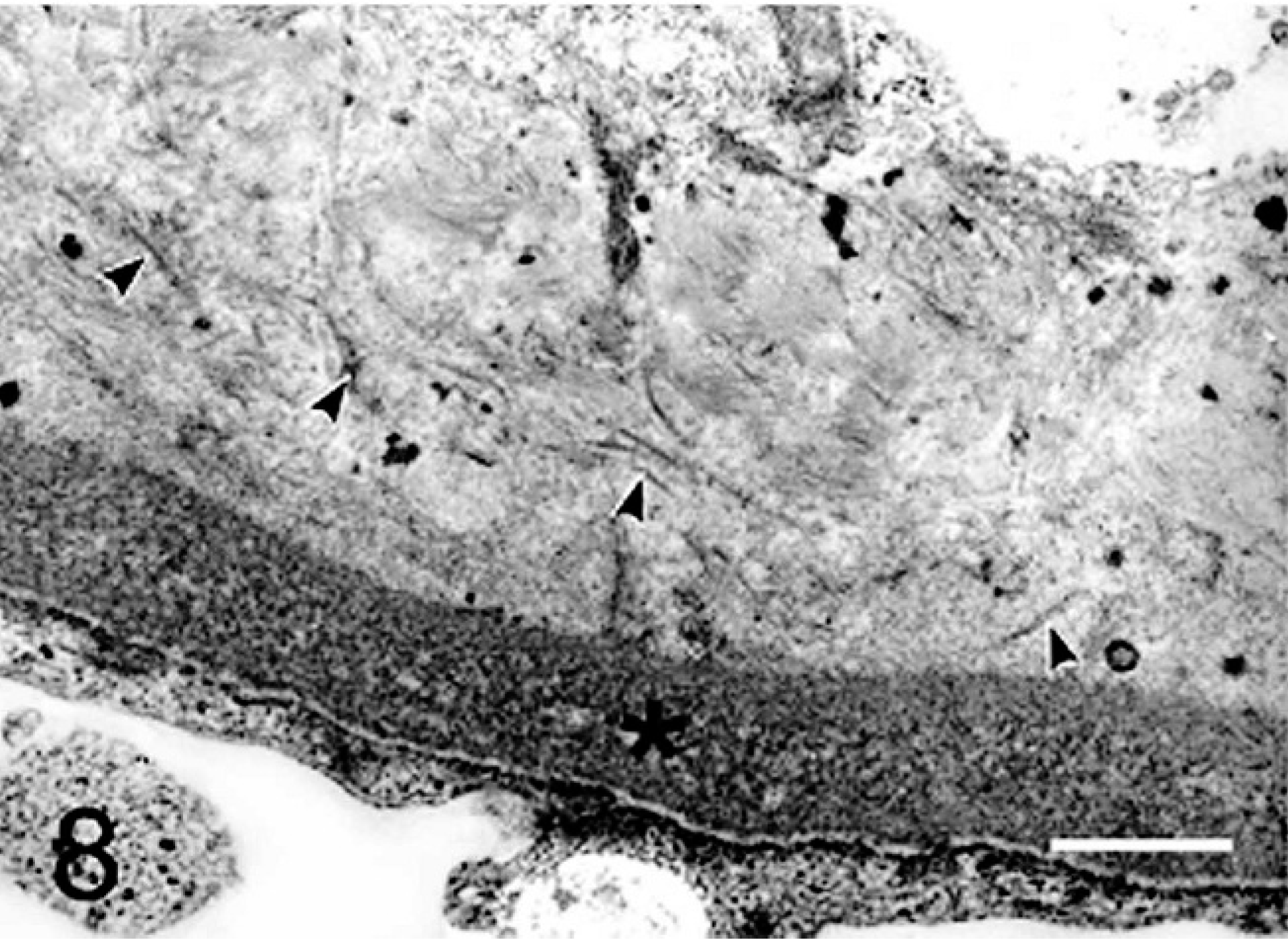
Electron micrograph. Kidney; dog. Collagen fibrils (arrowheads) and amorphous material are deposited between the glomerular capillary endothelium and the glomerular basement membrane (asterisk). Collagen fibrils are thin, frayed, and spiraled or curved. Uranyl acetate and lead citrate. Bar = 1 µm.
Discussion
All renal glomeruli of this dog were markedly expanded by deposition of type III collagen and fibronectin. Ultrastructural confirmation of collagen fibril formation in the glomeruli led to the final diagnosis of CFGN. The dog died 3 weeks after biopsy, and permission for postmortem examination was not given.
Clinical and pathologic features of CFGN in dogs have not been well documented. The glomerular accumulation of collagen fibrils with glomerulosclerosis similar to human CFGN has been reported in 3 Newfoundland dog littermates. 9 However, the presence of type III collagen in the glomeruli, which is a typical feature of CFGN in human, was not confirmed in those dogs. The current case mirrors CFGN in humans, in terms of the histopathologic, immunohistologic, and electron microscopic findings. 3,6,19 The fibrillar deposits in glomerular subendothelial and mesangial areas were identified as type III collagen by immunofluorescence. The collagen fibrils had abnormal ultrastructure, as seen in human and porcine CFGN. 13
Amorphous material was also observed in the glomerular mesangium and capillary walls in this dog by electron microscopy. Immunofluorescence findings indicated that this amorphous material contained fibronectin. The glomerular deposits of CFGN in a cynomolgus macaque contained not only collagen fibrils, but also other amorphous materials or plasmalike substances. 1 Fibronectin accumulation in glomerular capillary walls was demonstrated in 3 human cases of CFGN 5 but has not been reported. Glomerular deposition of fibronectin, the origin of which has not been determined, has not been reported previously in nonhuman cases of CFGN. Deposition of fibronectin in glomerular capillary walls was seen in hereditary glomerulopathy of Samoyed dogs, with thinning and splitting of the GBM. 17 In those dogs, fibronectin deposition was localized to the inner aspect of the GBM and was considered to be the result of increased exposure of circulating fibronectin to its binding sites on type IV collagen in damaged GBM. 17 In our case, however, fibronectin deposition was localized in the subendothelial space and the GBM was fully formed. Therefore, because fibronectin also binds to type III collagen, 15 glomerular subendothelial accumulation of fibronectin might have been causally related to the accumulation of type III collagen.
The pathogenesis of CFGN remains elusive in humans and other species. The origin of type III collagen fibrils in the glomeruli has been proposed to be either in situ formation or from extrarenal sources. 2 If the accumulated type III collagen is derived from extrarenal sources, CFGN could be a systemic disease with abnormal collagen metabolism. 2,18 In human CFGN, elevated serum levels of the N-terminal peptide of type III procollagen are a biomarker of the disease. 16 Abnormally high serum levels of type III procollagen peptide indicate excessive conversion of type III procollagen to mature type III collagen in CFGN in humans and suggest the possibility of systemic abnormalities in collagen synthesis in this disease.
Alternatively, the glomerular type III collagen deposits can be formed in situ. Type III collagen production by mesangial cells has been reported in human diabetic nephropathy, 12 and cultured rat mesangial cells produce collagen types I, III, IV, and V. 4,7 In addition, mesangial cells are activated and express α-SMA in glomeruli with type III collagen deposition. 11 In the case here, only a few mesangial cells were α-SMA positive, and only very mild mesangial proliferation was observed within a few glomeruli. These findings suggest a minor contribution of mesangial cells to type III collagen accumulation in this case.
The pathogenesis in this case was not determined because serum levels of type III procollagen peptide and other blood components including fibronectin were not evaluated. Whether podocytes or endothelial cells contributed to the production and accumulation of type III collagen in glomeruli is also unclear.
The dog in this report had severe proteinuria, a common feature of CFGN in humans. Capsular adhesions of glomerular tufts and crescent formations were observed in several glomeruli, although these findings have not been reported in most human cases. 2 Ultrastructurally, the GBM was thickened, and podocytes were detached with effaced foot processes. Podocytes regulate capillary permeability, and damage to podocytes induces massive proteinuria, 8 so podocyte injury, possibly secondary to the accumulation of collagen and fibronectin, could have contributed to the development of proteinuria in this dog.
Footnotes
Acknowledgements
We gratefully thank Ms. Yoko Kakinuma at Azabu University for her excellent technical assistance.
