Abstract
Degenerative myelopathy (DM) is a common, slowly progressive, debilitating disease reported in several dog breeds, including the German Shepherd Dog and Pembroke Welsh Corgi. Boxer dogs present occasionally for a thoracolumbar myelopathy for which no cause is identified on MRI or cerebrospinal fluid analysis. Despite a lack of a histologic description of DM in the Boxer in the veterinary literature, such dogs are presumed to have DM. Here we report 2 histologically confirmed cases of DM in the Boxer breed in which histologic studies disclosed marked degenerative changes in the spinal cord that were most prominent in the thoracic and cranial lumbar segments. Lesions consisted of myelin vacuolation and degeneration, myelophagocytosis, reactive astrocytosis, and ellipsoid formation most prominent in the lateral and ventral funiculi. We present a detailed histologic description of DM in the Boxer dog and compare it to DM in other purebred dogs.
Background
Degenerative myelopathy (DM) is an idiopathic neurodegenerative disorder that primarily affects the spinal cord of middle- to older aged dogs. 1,4,7 The disorder is overrepresented in the German Shepherd Dog and Pembroke Welsh Corgi and has been reported in a variety of other breeds, including the Siberian Husky and miniature Poodle. 1,3,4 Although the overall prevalence of DM in dogs has not been established, it could represent 1–5% of referral cases to neurology specialty practices. 5
Clinically, DM is a slowly progressive disorder with a high morbidity and low mortality, usually because of humane euthanasia rather than natural causes. 1–4,7 Neuroanatomic localization in dogs with DM is commonly to the third thoracic through the third lumbar (T3–L3) spinal cord segments, and the disease is typified by a progressive upper motor neuron paresis and proprioceptive ataxia in the pelvic limbs. 4 Segmental spinal reflexes typically are normal to exaggerated in the pelvic limbs, although the patellar reflex can be diminished because of lesions in the dorsal nerve root ganglia. 1,4 Late in the disease, urinary and fecal incontinence, as well as thoracic limb and brain stem dysfunction, can occur; euthanasia is a common endpoint with severe disease. 1,3
In clinical practice, the Boxer breed is overrepresented for examination and presumptive diagnosis of DM, yet a neuropathologic description of Boxer DM is lacking in the veterinary literature. 4 Herein, we characterize the disorder in 2 Boxer dogs and compare the neuropathologic findings to those described in the German Shepherd Dog and other breeds.
Clinical and Pathologic Findings
Over a 20-year period, 2 cases of DM in the Boxer breed were identified from a review of necropsies submitted to the Department of Biomedical Sciences, College of Veterinary Medicine, Cornell University; the Department of Pathobiology and Biomedical Sciences, College of Veterinary Medicine, Texas A&M University; The Animal Health Trust; and the Department of Pathology, College of Veterinary Medicine, University of Georgia. Dog No. 1 was a 9-year-old, intact male Boxer; dog No. 2 was a 10-year-old spayed female Boxer. Both dogs had a history of progressive upper motor neuron paresis and general proprioceptive ataxia in the pelvic limbs. Both dogs had markedly delayed postural reactions in the pelvic limbs and absent patellar reflexes. The remainder of the neurologic examination in both dogs was within normal limits. Neuroanatomic lesion localization was to spinal cord segments T3–L3 (with inclusion of the femoral nerve sensory ganglia because of strong suspicion of DM). No significant findings were noted on cerebrospinal fluid examination of either dog. Subsequently, both animals were humanely euthanized and a complete postmortem examination was performed except for the brain of dog No. 1, and the cerebrum and brain stem rostral to the medulla oblongata, of dog No. 2.
Tissue sections were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and mounted on glass slides. Transverse and longitudinal sections were stained with HE, and select spinal cord sections were stained with Luxol fast blue and Bielschowsky silver stain. Immunohistochemistry, with the use of a 1 : 3,000 dilution of rabbit polyclonal glial fibrillary acidic protein (GFAP) antibody (Dako, Carpinteria, CA) was performed with an Autostainer plus (Dako). Antigen retrieval was performed by microwaving for 20 minutes in 0.01 M citrate buffer. Diaminobenzidine was used as the chromagen; sections were counterstained with hematoxylin.
No gross abnormalities were detected in the spinal cord of either dog. Histopathologic examination of transverse and longitudinal sections of the cervical, thoracic, and lumbar spinal cord in both dogs revealed marked pallor of the outer two thirds to three quarters of the white matter tracts, particularly in the ventral and lateral funiculi (Figs. 1, 2). The most severe lesions were in the caudal thoracic to the mid-lumbar regions; however, in dog No. 2, lesions had extended into the caudal cervical segments. Lesions were bilateral and roughly symmetric, with marked myelin vacuolation and dilated myelin sheaths (ranging in diameter from 15 to 75 µm) that contained tight coils of degenerated myelin and pyknotic nuclear debris (Fig. 3). Some of the pyknotic cellular debris was within the cytoplasm of macrophages (myelinophages). Minimal degenerative changes were in the spinal nerve roots. Axons were conspicuously absent or fragmented in the dilated myelin sheaths; this change was most prominent in longitudinal sections (Fig. 4). In the most severely affected areas, astrocytes were more numerous and had large open-faced nuclei and an increased number of eosinophilic fibrillar cytoplasmic processes (Figs. 5, 6). In both dogs, rare spheroids were interspersed with the areas of myelin vacuolation. In dog No. 2, involvement of the sciatic nerves and the medulla oblongata was minimal. In the same dog, the cerebellum was within normal limits. In dog No. 2, a small focus of gliosis was found in the lumbar spinal cord; however, in neither dog were any changes noted in the spinal gray matter. Dog No. 2 had mild lymphoplasmacytic inflammation in the leptomeninges. Comorbid findings in dog No. 1 were severe arteriosclerosis of meningeal arterioles (age-related change) and multiple testicular tumors. Comorbid findings in dog No. 2 were chemodectoma, thyroid and adrenal adenomas, and mild, chronic pancreatitis.

Spinal cord; dog No. 1. Pallor and myelin degeneration is marked in the lateral and ventral funiculi at the level of the fifth thoracic spinal cord segment. HE.
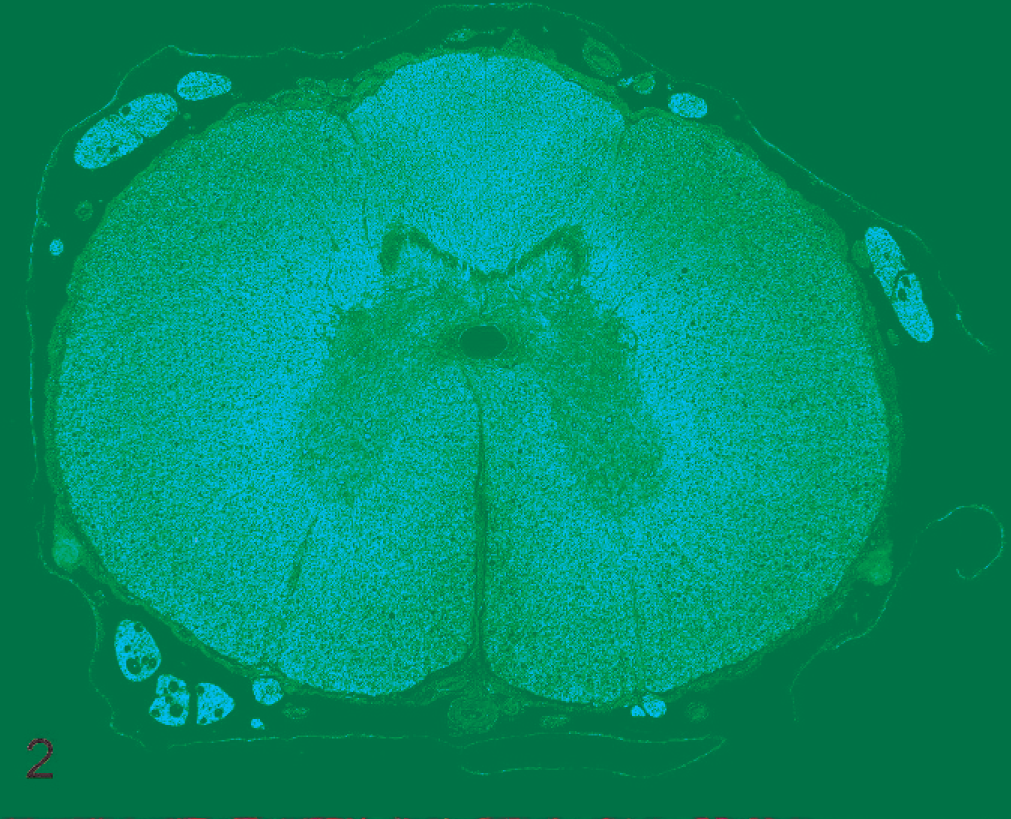
Spinal cord; dog No. 1. Myelin loss is predominantly in the lateral and ventral funiculi at the level of the eighth thoracic spinal cord segment. Luxol fast blue.

Spinal cord; dog No. 1. The dilated myelin sheaths that contain debris (arrows) are most numerous adjacent to the ventral median fissure (∗). HE.

Spinal cord; dog No. 2. Multiple axons are absent or fragmented (arrows) and surrounded by dilated myelin sheaths. Bielschowsky silver stain.
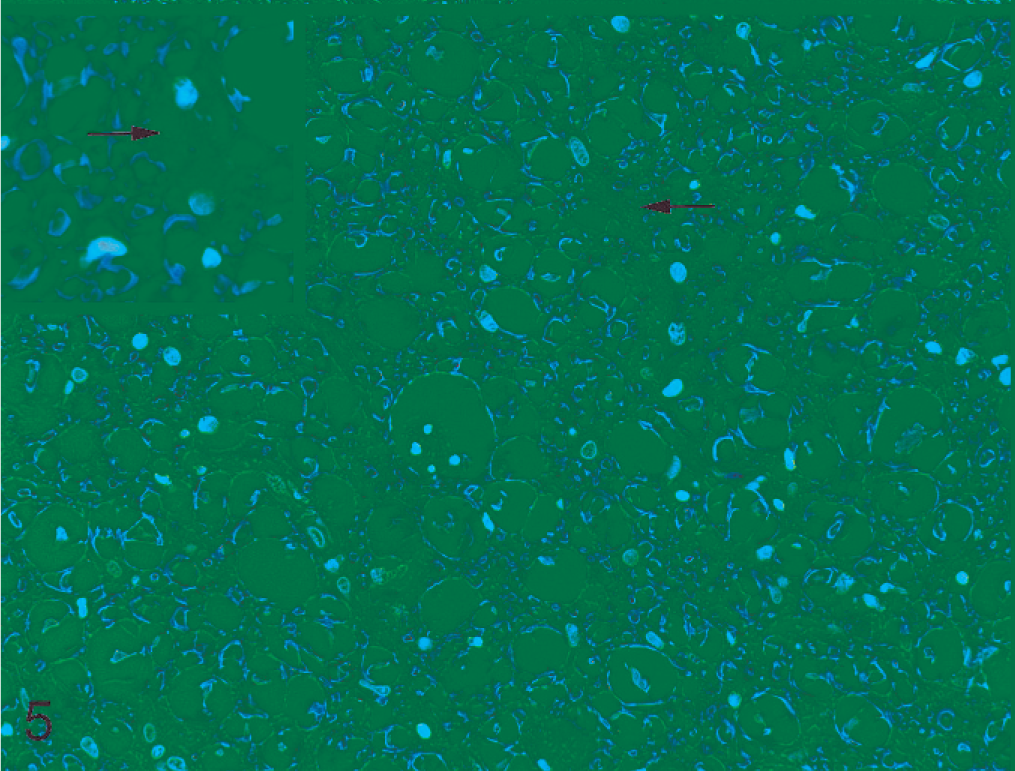
Spinal cord; dog No. 1. Plaques of astrocytic processes surrounding dilated and degenerative myelin sheaths (arrow). Inset: Higher magnification of an astrocytic plaque. HE.

Spinal cord; dog No. 2. Astrocytes are hypertrophied adjacent to the areas of vacuolation and have strongly GFAP-positive processes (arrows). Glial fibrillary acidic protein counterstained with Dako's hematoxylin.
Discussion
Degenerative diseases affecting the spinal cord of dogs are relatively common and can be secondary to focal segmental lesions (e.g., intervertebral disk rupture), inherited diseases (e.g., hereditary myelopathy in Afghan Hounds), or diseases of unknown origin (e.g., German Shepherd DM and demyelinating myelopathy of the miniature Poodle). 4 In clinical practice, DM is commonly a presumptive diagnosis in several breeds of dogs, including the German Shepherd Dog and Pembroke Welsh Corgi. 4,5 It has been the authors' (S. J. Schatzberg, S. R. Platt, M. Kent) impressions that DM also is a relatively common presumptive diagnosis for Boxer dogs, with progressive T3–L3 signs and normal magnetic resonance imaging (MRI) studies. 4 Therefore, we searched 4 pathology archives (University of Georgia, Animal Health Trust, Cornell University, and Texas A&M University) over a 20-year period but identified only 2 cases. The lack of a large cohort of cases could indicate that this is a less common disease than is presumed clinically; however, it also suggests that dogs with slowly progressive spinal cord disorders might be lost to follow-up, which also might explain the lack of any previous publication of DM in the Boxer dog.
In the 2 dogs in this report, histologic lesions were similar to those previously reported for DM in various breeds. In other breeds, the spinal cord lesions attributed to DM predominate in the mid- to caudal thoracic spinal cord segments and gradually lessen in severity cranially and caudally. All funiculi typically are affected, with the most severe lesions around the periphery of the cord and conspicuous sparing of the fasciculus proprius. 7 Lesions are bilateral, roughly symmetric, and typified by a combination of myelin vacuolation and ballooning, ellipsoid formation, myelin-laden macrophages, and a variable degree of astrocytosis. 1,2,4 Brain stem lesions, including chromatolysis, neuronophagia, and gliosis, have been identified in the red nucleus, lateral vestibular nucleus, dentate nucleus, and fastigial nucleus. 7 Unfortunately, the brain stem (other than the caudal aspect of the medulla oblongata) was not available for evaluation in either dog; therefore, the occurrence of brain stem lesions in the Boxer form of DM is unknown.
Degenerative myelopathy in the Boxer dog is distinct from an uncommon, but well-described condition of young Boxer dogs known as progressive axonopathy. 6 In this disease, numerous axonal spheroids develop in the spinal cord and brain stem nuclei in concurrence with degeneration of the lateral and ventral funiculi of the spinal cord. 6 Affected dogs manifest signs at an early age and progress rapidly, in contrast to the late onset, slowly progressive clinical course of DM. 6 The spheroids in progressive axonopathy are conspicuously absent in DM, suggesting a different pathogenesis. Indeed, the spheroids contain large accumulations of neurofilaments and therefore could reflect a form of neuraxonal dystrophy in which normal neuronal transport is inhibited. 6
The lack of inflammation in cases of DM suggests that the loss of myelin and damage to the axon is not a direct effect of an infectious agent but is rather a failure of maintenance of the continuity of the myelin sheath, of normal axon function, or both. As such, several hypotheses have been examined as to the cause of the degenerative changes in the spinal cord; however, the underlying pathophysiology remains unclear. The lesions share similarities with those found in some vitamin deficiencies; however, alterations in the levels of vitamins have not been observed consistently in affected dogs. 5,7–9 Peripheral blood lymphocytes in dogs with DM typically have a reduced response to T-cell mitogens, possibly secondary to a circulating suppressor cell, suggesting an immune-mediated mechanism. 9 Additionally, the overrepresentation of DM in certain breeds, such as German Shepherd Dogs, probably reflects a degree of genetic influence in the pathogenesis. 1,3,4,7 Ultimately, the lack of an identified cause precludes directed therapies, and there currently is no proven efficacious treatment for DM.
Older Boxer dogs might be affected by bilaterally symmetric spinal cord lesions that predominate in the thoracolumbar area. In the 2 dogs of this report, these lesions were indistinguishable from DM of the German Shepherd Dog and Pembroke Welsh Corgi. Further genetic and etiologic investigations are warranted in these breeds.
Footnotes
Acknowledgements
We thank Joy Cramer for assistance with immunohistochemistry, Kristen Toohey for assistance with the images, and Dr. Kaspar Matiasek for access to the Animal Health Trust's pathology database.
