Abstract
The genus Bartonella comprises a group of gram-negative, fastidious bacteria. Because of diagnostic limitations of culture and serologic testing, polymerase chain reaction (PCR) has become a powerful tool for the detection of Bartonella spp. in blood and tissue samples. However, because many wild and domestic animals harbor Bartonella spp., transfer of Bartonella DNA during sample collection or histologic processing could result in false-positive PCR test results. In this study, we describe evidence of Bartonella DNA dissemination and transfer in the necropsy room and during the subsequent processing of formalin-fixed paraffin-embedded tissues. Bartonella DNA was amplified from different areas of the necropsy room, from the liquid paraffin in the tissue processor, and from different parts of the microtome. Unless stringent procedures are established and followed to avoid cross-contamination, the molecular detection of Bartonella spp. from tissue samples obtained at necropsy or processed in a multispecies histopathology laboratory will not be reliable.
The genus Bartonella includes at least 22 species of gram-negative, fastidious, aerobic bacilli, plus many unnamed or candidatus species that have been identified mostly by polymerase chain reaction (PCR) amplification and DNA sequencing. These intravascular bacteria appear to cause a wide spectrum of disease in animals and human beings. 4 In addition, various Bartonella spp. are efficiently transmitted by biting flies, fleas, lice, sand flies, and (potentially) ticks among a spectrum of adapted reservoir hosts. 1 Improvements in culture techniques and PCR resulted in increased detection of Bartonella spp. in association with disease. 8 However, certain species, such as Bartonella henselae in cats, Bartonella vinsonii subsp. berkhoffii in coyotes, Bartonella bovis in ruminants, and numerous Bartonella spp. in small mammals and rodents, are evolutionarily well adapted to cause persistent, nonclinical, intravascular infections in the reservoir host. 4 For example, B. bovis DNA was detected by PCR in blood of 117 of 142 beef cattle from North Carolina. 7 A similar high prevalence of B. bovis bacteremia was reported from cattle in Africa, Europe, and North America. 5,12 Thus, contamination with bovine blood is a potential source of DNA transfer to tissue samples collected in diagnostic laboratories, either at necropsy or during histologic processing. Because B. bovis is infrequently isolated from feline blood samples, 3 it would be difficult to establish whether a positive PCR test on feline tissue indicates infection with B. bovis or merely contamination with bovine blood in the necropsy or histopathology laboratory.
Because of the limitations associated with culturing these bacteria, serologic cross-reactivity among Bartonella spp., and the poor correlation between serologic test results and bacteremia, detection of Bartonella DNA by PCR has gained favor as a diagnostic test. Researchers have targeted different Bartonella genes, including the 16S ribosomal RNA (rRNA) gene, 16S–23S rRNA intergenic transcribed spacer (ITS) region, groEL gene, the citrate synthase gene (gltA), and others, for the molecular detection of Bartonella infection. 13,14 Although PCR amplification of various target genes provides a rapid diagnostic alternative for the detection of Bartonella spp., there can be limitations related to poor test sensitivity and specificity. As an example, our laboratory reported nonspecific PCR amplification of Mesorhizobium spp. by using previously published 16S–23S ITS primers designed to amplify Bartonella DNA. 14 Because Mesorhizobium DNA can be a contaminant in molecular grade water, false-positive PCR test results were documented, and the ITS primers were redesigned to increase amplification specificity. In conjunction with efforts to enhance sensitivity in the detection of Bartonella-specific DNA sequences, we recently incorporated pre-enrichment culture of aseptically obtained diagnostic samples (blood, cerebrospinal, aqueous, and joint fluids and effusions) by using a liquid insect cell culture–based medium (Bartonella alpha-Proteobacteria growth medium) before PCR testing. 8 By combining pre-enrichment culture with PCR amplification, substantial improvement in molecular diagnostic sensitivity was achieved, subculture agar plate isolates were obtained for detailed microbiologic studies, and human infections with novel Bartonella species were reported. 16 This approach is applicable to postmortem specimens, provided they are collected and transported aseptically.
When culture is unsuccessful or impractical, pathologists increasingly rely on PCR amplification to detect bacterial DNA in formalin-fixed, paraffin-embedded tissues. Archival collections of paraffin-embedded tissues provide an important and readily available resource for retrospective studies of bartonellosis and other novel or emerging bacterial infections. 10 Techniques used for DNA extraction from formalin-fixed paraffin-embedded tissue have been studied, and various PCR assays have been validated. 17 During initial efforts to define the cellular localization of Bartonella spp. in paraffin-embedded tissues, we repeatedly detected Bartonella DNA in negative control samples used during PCR testing. To investigate the source of DNA contamination, all reagents used for DNA extraction and PCR amplification were individually tested and shown to be negative for amplicon contamination. When amplicon contamination within the testing laboratory was eliminated as a source of the problem, we investigated the possibility of DNA transfer in the necropsy service area and during tissue processing and sectioning in the histopathology laboratory.
To detect DNA contamination during tissue processing, blank paraffin blocks (negative controls) and paraffin blocks known to contain Bartonella spp. (positive controls) were cut on a microtome in an alternating fashion. By using this approach, Bartonella DNA was amplified by PCR from 4 of 6 blank (negative control) paraffin blocks; however, samples obtained from the same blank paraffin blocks by using a scalpel blade were consistently PCR negative. All samples were extracted and tested in an identical manner.
After implicating the microtome as a source of DNA contamination, other potential sources of DNA transfer were investigated in our institution's necropsy room and histopathology laboratory. Five microtomes were assessed, one each from two diagnostic laboratories in our institution and 3 at a local state diagnostic laboratory. Dry, sterile cotton swabs were used to collect samples from the necropsy room (the tissue trimming board, the tissue strainer, and the necropsy table tops) at the end of a workday, after these areas had been routinely cleaned. Sterile cotton swabs were also used to collect samples from the processing area in the histopathology laboratory (the tissue trimming board, the formalin container for holding cassettes after trimming, and the water bath used for slide mounting of tissue sections). Used paraffin samples were collected directly from the tissue processor from the oldest and newest liquid paraffin–holding tank. Sampling was conducted twice, several days apart. Unused sterile swabs and new paraffin, unexposed to the processor, served as negative controls during extraction and PCR testing of the necropsy and histopathology laboratory samples.
For each of 5 different microtomes in 3 diagnostic laboratories, dry, sterile cotton swabs were individually used to obtain samples from different parts of the microtome, including the blade holder, block holder, platform, slanting platform, bench top, and used microtome blades. Samples were taken at the end of the workday, after microtomes were routinely cleaned. One new sterile swab was tested as a negative control with each set of test swabs.
DNA from the swabs was extracted by using the QIAamp DNA mini kit (Qiagen Inc., Valencia, CA) according to manufacturer's instructions. Molecular grade water was used as an additional control during DNA extraction. After extraction, DNA concentration and purity were measured with an absorbance ratio of 260 to 280 nm by using Nanodrop (Thermo Scientific, Wilmington, DE) and were stored at −20°C until used. The 16S–23 ITS primers used included 325S: 5′ CTTCAGATGATGATCC CAAGCCTTCTGGCG 3′ and 1100AS: 5′ GAACCGACGACCCCTGCTTGC AAAGCA 3′. Primers were synthesized by MWG Biotech (Highpoint, NC). PCR was performed with a final reaction volume of 25 µl, containing 12.5 µl of premix Ex Taq (perfect real time) (Takara Bio USA Inc, Madison, WI), 7 µl of molecular grade water, 0.25 µl each of forward and reverse primers (30 µM), and 10 µl of template DNA. Amplification of the ITS region was performed under the following conditions in a Mastercycler ep gradient (Eppendorf, Westbury, NY), with a single hot-start cycle at 95°C for 2 minutes, followed by 55 cycles of denaturing at 94°C for 15 seconds, annealing at 66°C for 15 seconds, and extension at 72°C for 18 seconds. Amplification was completed by an additional cycle at 72°C for 2 minutes. All amplification products were analyzed by 2% agarose gel electrophoresis, stained by ethidium bromide, and visualized under ultraviolet light. Amplicon sizes were determined by comparison with the molecular weight marker, Hyladder 1 kbp (Denville Scientific Inc., Metuchen, NJ). B. henselae DNA (0.001 pg/µl) and DNA from a healthy noninfected dog were used as positive and negative controls, respectively.
Bartonella ITS PCR amplification products were sequenced directly by gel extraction by using QIAquick gel extraction kit (Qiagen) or by cloning into the plasmid pGEM T easy vector system (Promega, Madison, WI). After Escherichia coli DH5α transformation, the recombinant white colonies were selected based on the correct insert size, and the plasmids were isolated by using QIAprep Spin Miniprep kit (Qiagen). From each transformation, at least 3 independent clones were selected for sequencing. Sequencing of each plasmid insert was done by MWG Biotech (MWG, High Point, NC). Sequence analysis and alignment with GenBank sequences was performed by using AlignX software (Vector NTI Advance 10, Invitrogen Inc., Carlsbad, CA).
Bartonella DNA was amplified by PCR from swabs taken from used microtome blades and from different parts of the microtome (Table 1). Three of the 5 microtomes sampled after daily cleaning were PCR positive for the DNA of 3 different Bartonella spp. Bartonella spp. DNA was amplified from 3 of 7 swabs taken from microtome 1, and 1 of 7 swabs from microtomes 2 and 3. DNA was also amplified from 1 of 2 used paraffin samples from the tissue processor (oldest) and from 1 of 2 swabs from the tissue trimming board in the necropsy room. All negative-control sterile swabs and all extraction and PCR controls remained negative. Amplicons from the flat platform and the block holder of microtome 1 had 97% and 99.5% sequence homology, respectively, with B. henselae (ITS strain Houston I). An amplicon, obtained from a used microtome blade, had 99.6% homology with B. vinsonii subsp. berkhoffii genotype II. This microtome was used routinely for preparation of dermatopathology slides, mainly from cat and dog tissues. The amplicons from microtomes 2 and 3 had 99.2% sequence homology to B. henselae (ITS strain San Antonio 2) and 99.8% to B. vinsonii subsp. berkhoffii genotype II, respectively. The amplicon from the necropsy tissue trimming board was 85.6% homologous to candidatus Bartonella volans; the amplicon from paraffin from the tissue processor had 99.5% homology with B. henselae (ITS strain Houston I). Cat, dog, rat, and woodchuck necropsies had been performed during the several days before obtaining samples for PCR testing.
PCR amplification results from swabs of 5 microtomes.
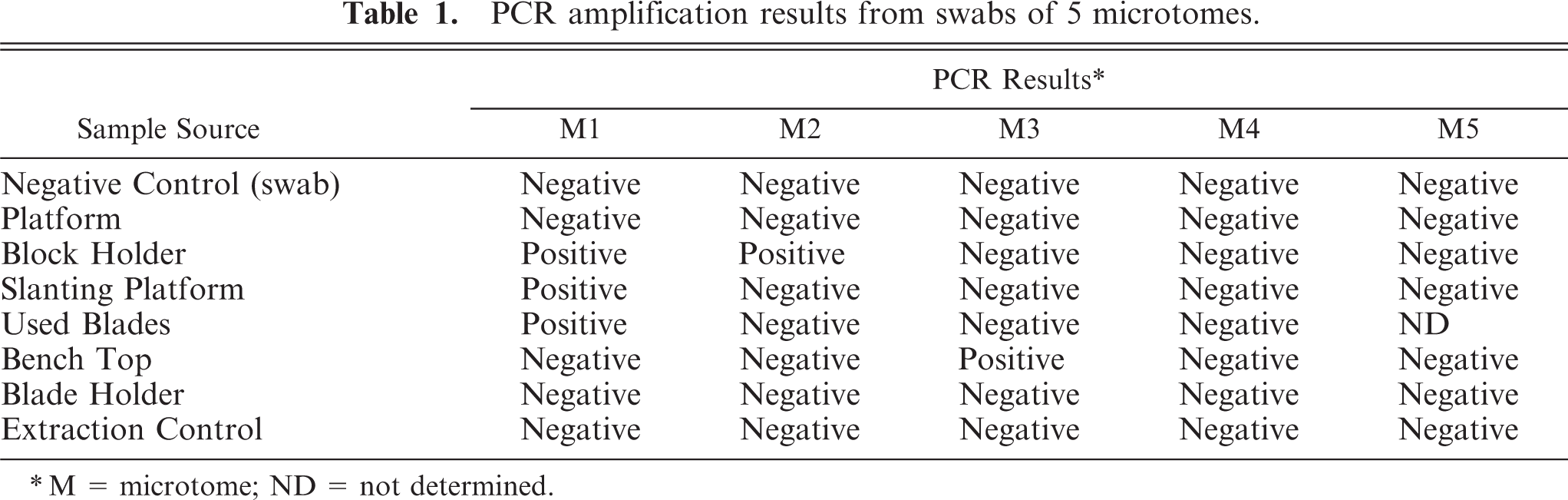
∗M = microtome; ND = not determined.
The results of this study indicated that various Bartonella spp. and strains can be amplified from necropsy rooms and histopathology processing areas. There are several possible mechanisms by which cross-contamination might occur during the collection and processing of necropsy tissues or surgical biopsy specimens. Although many Bartonella spp. have evolved in conjunction with a preferred vector and reservoir host, it is increasingly obvious that DNA of these bacteria can be found in an array of vectors and hosts. For example, B. henselae is most often found in the blood of cats and dogs but has also been isolated or amplified from blood of other animals, including dolphins, horses, porpoises, sea turtles, and whales. 9,11,15 To date, B. vinsonii subsp. berkhoffii has only been detected in dogs, coyotes, gray foxes, and human beings. 6 B. vinsonii subsp. berkhoffii genotype II is the most frequently detected genotype in canine and human samples in our laboratory. Although the most similar GenBank sequence for one amplicon was candidatus B. volans, a novel Bartonella spp. isolated from southeastern ground squirrels, (Genbank sequence EU 294521; W. K. Reeves, unpublished), the low homology suggests that this sequence represents a novel Bartonella sp. not previously sequenced in our or other research laboratories. Somewhat surprisingly, B. bovis DNA was not amplified from any microtome. This could reflect the low number of bovine tissue samples processed on the microtomes tested in this study, which was based in an urban area.
Because veterinary pathologists provide necropsy and biopsy services for a wide array of animal species, many of which can serve as reservoir hosts for Bartonella spp., transfer of DNA from these ubiquitous intravascular bacteria is highly problematic for both molecular diagnostic studies and retrospective research studies. The increasing use of PCR testing for infectious agents in postmortem specimens requires more stringent procedures for tissue collection and processing, and for cleaning of equipment and work areas in necropsy and histopathology laboratories.
In this study, Bartonella DNA was amplified from the bench top near the microtome, from used discarded microtome blades and directly from the microtome. False-positive results were previously reported when PCR was used to detect infectious agents in formalin-fixed paraffin-embedded tissues. 18 Hole-punch carryover contamination and environmental contamination of the laboratory work space and equipment have also been documented as potential sources of false-positive PCR results. 2,19
Results of this study highlight the importance of the microtome as a source of DNA contamination of paraffin-embedded tissues. Adherent fragments of paraffin or tissue from infected blocks can contaminate the next block to be processed. Thus, paraffin sections for PCR testing should be prepared according to appropriate procedures for molecular diagnostic studies, which may require more stringent cleaning of the microtome than that used for routine histology. Positive control blocks should not be processed on the same microtome or even in the same room in which diagnostic or research sample blocks are processed. Blank paraffin control blocks should be used between sample blocks to detect any DNA carryover during processing of sequential blocks. Use of a sterile scalpel blade to shave tissue from each block, instead of using a microtome for sectioning, would minimize DNA carryover; however, this approach does not preclude contamination of liquid paraffin in automated tissue processors, which was identified in this study. Prospective studies should include individual hand-embedding of tissues with new reagents. Fresh or frozen (unfixed) diagnostic samples would be favored over formalin-fixed, paraffin-embedded tissues for PCR-based testing, particularly when exposure to different tissues in automated processors cannot be avoided. This is especially critical for identifying novel associations between lesions and these fastidious bacteria. In situ tests, such as immunohistochemistry, in situ hybridization, or in situ PCR, offer additional challenges because of the need for microtome preparation of samples. Although relevant localization of agents within lesions, omission of an amplification step, and documentation of repeatable results can guard against processor- or microtome-related contamination of samples, isolated results from individual cases should be cautiously interpreted.
In conclusion, PCR is a powerful tool for the detection of DNA from Bartonella spp. and other infectious agents in blood and tissue samples from animals and human beings; however, caution should be exercised in interpreting the results. In this study, several steps in the preparation of sections from formalin-fixed, paraffin-embedded tissues were identified as potential sources of Bartonella spp. DNA carryover among specimens and among species. Standard operating procedures, like those used in molecular diagnostic laboratories, must be followed in necropsy and histology laboratories to minimize DNA cross-contamination during sample collection, histologic processing, and sectioning of paraffin blocks, when samples are intended for PCR diagnostic testing.
Footnotes
Acknowledgements
This research was supported by the State of North Carolina and in part through stipend support for Dr. Mrudula Varanat from Bayer Animal Health. We thank the histopathology laboratories for allowing us to obtain samples for PCR testing.
