Abstract
A 10-year-old intact male Labrador Retriever dog was euthanized because of rapid deterioration after suffering from mild chronic cough, hematuria, acute blindness, ataxia, and lethargy. Clinical examination revealed blepharospasm and hyphema, with clear discharge from the right eye; a firm mass in an enlarged right testicle; a mass in the irregularly enlarged prostate; and nodules in the left kidney, liver, and spleen detected by abdominal sonography. Cytologic evaluation of fine needle aspirates from the prostate, testis, and kidney comprised large, clustered or individualized, anaplastic cells that lacked convincing tissue differentiation. Necropsy examination revealed an irregularly enlarged prostate with dark tan to red zones and multiple, discrete, beige to dark red nodules that ranged from 0.5 to 6 cm in diameter in the lung, liver, left kidney, right testis, colon wall, stomach wall, and brain. On histologic examination, discrete nests of anaplastic carcinoma-like tumor cells were found in sections of all affected organs. Results of immunohistochemical examination revealed widespread expression of von Willebrand factor and the absence of cytokeratin in neoplastic cells. The diagnosis was metastatic epithelioid hemangiosarcoma, primary site unknown.
History, Gross Findings, and Laboratory Results
A 10-year-old intact male Labrador Retriever dog was referred to the Veterinary Teaching Hospital (VTH) at Oregon State University (OSU) for evaluation of a thoracic mass. The dog had a 1-month history of mild coughing, intermittent hematuria, acute right ocular pain, and lethargy. A physical examination revealed blepharospasm and hyphema, with clear fluid discharge from the right eye; a firm mass 1 cm in diameter within an enlarged right testis; and an irregularly enlarged prostate. An abdominal ultrasonographic examination confirmed an irregularly enlarged prostate with a 5.9 × 3.8-cm mass, and nodules in the liver, spleen, and left kidney that measured from 1.2 to 4 cm in diameter. A blood count and biochemistry profile were within normal limits. Urine was brown and turbid and contained many red blood cells (RBC) (>100 cells/hpf) and white blood cells (20–30/hpf), with normal pH level pH 7, and 1.035 specific gravity. Cytologic examination of fine needle aspirates was performed on samples obtained by ultrasound guidance from the prostatic mass, prostatic parenchyma, left kidney mass, and the mass in the right testis. The sample from the prostatic mass contained numerous clustered or individualized neoplastic cells, with a background of RBCs (Fig. 1), scattered leukocytes, and necrotic cellular debris (Fig. 1). Individual neoplastic cells were large and typically contained 1–3 round nuclei with multiple prominent, bizarre nucleoli. They were surrounded by a variable amount of eosinophilic cytoplasm with relatively distinct borders. Most cells contained small, discrete cytoplasmic vacuoles, with a few cells containing 1–2 large vacuoles. Marked anisocytosis and anisokaryosis were present, and mitotic figures were rare. In some fields, cohesion was identified, but this was not a consistent finding. Similar aggregates of neoplastic cells were present in the samples from the right testis and the left kidney. The sample from the solid portion of the prostate was consistent with benign prostatic hyperplasia, and the sample from the spleen was suggestive of nodular lymphoid hyperplasia. Palliative treatment that consisted of an appetite stimulant and pain medication was provided. The dog was euthanized 1 week after presentation at the VTH because of rapid deterioration associated with development of anorexia, acute bilateral blindness, and generalized ataxia, and was submitted for necropsy to the Veterinary Diagnostic Laboratory at OSU.
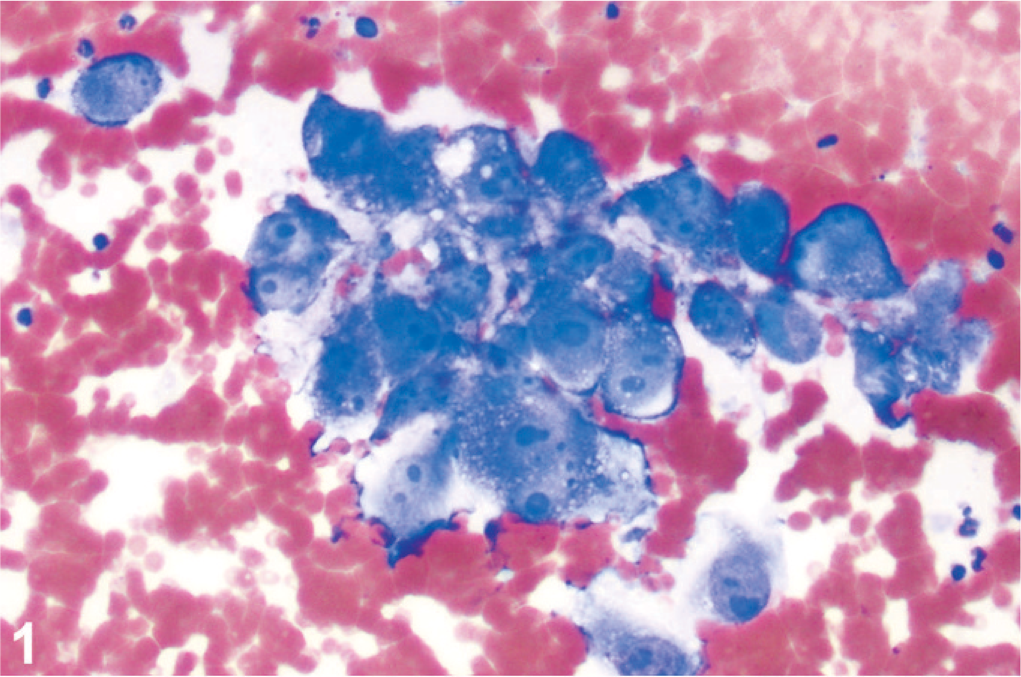
Anaplastic cells in the aspirate from the prostatic mass occasionally show cohesion. Wright's Giemsa stain.
At gross necropsy, the 25-kg Labrador Retriever dog was in moderate body condition and good postmortem condition. Muscle mass was reduced and adipose stores minimal. A mottled beige to red soft mass that measured 4.5 × 5 × 6 cm was located in the left caudal lung lobe. Multiple similar nodules that ranged in size from 0.3 to 1 cm in diameter were scattered throughout all lung lobes. There were a few red-black soft masses up to 0.5 cm in diameter that were located at the mesenteric insertion of the colon and in the stomach wall. Half of the left kidney was replaced by a mottled beige-gray to red, slightly firm, 4 × 4 × 6-cm mass, and multiple liver lobes had similar masses up to 4.5 cm in diameter (Fig. 2). The prostate gland was irregularly enlarged, some areas were spongy and beige, other areas were soft and mottled red-beige (Fig. 2). The muscular layer of the urinary bladder wall was thickened to 8 mm. The right testis had a solitary, soft, mottled black-beige mass 1 cm in diameter, and there was hyphema of the right eye. The brain had multiple, randomly distributed, small, discrete, hemorrhagic foci that varied from 0.1 to 0.3 cm. The spleen had a small number of roughly spherical, beige-red, subcapsular, soft nodules up to 1 cm in diameter.
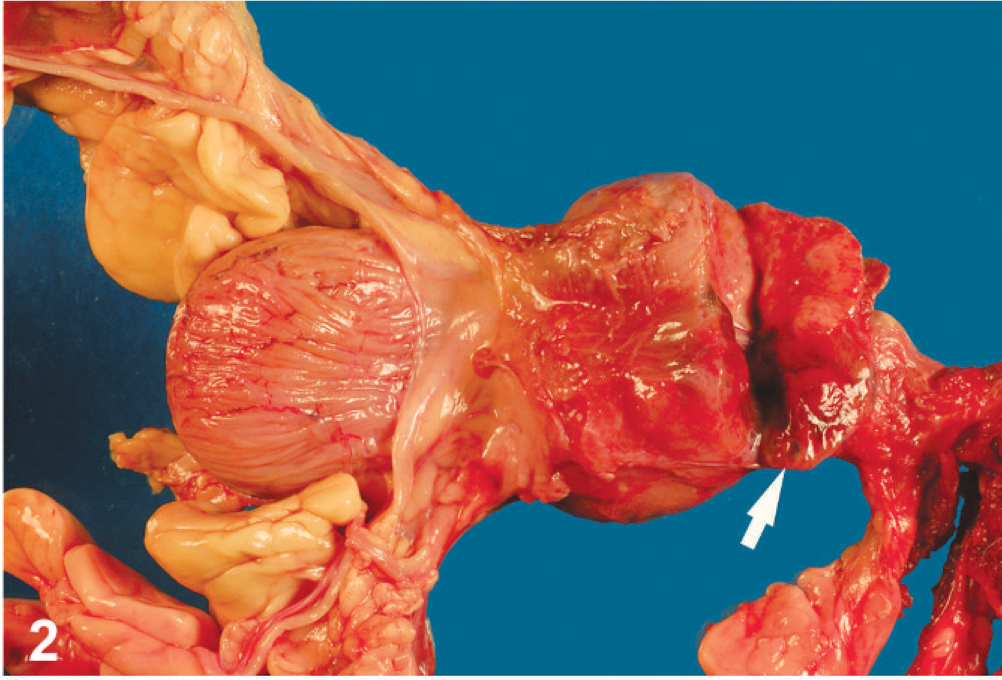
Prostate; dog. The prostate is severely enlarged by a mottled beige brown mass (arrow).
Differential Diagnoses
Based on clinical findings of an enlarged prostate with a mass and masses in other organs, and the presence of anaplastic neoplastic cell with cohesion on cytology in the aspirate taken from the prostatic mass, a metastatic anaplastic carcinoma, possibly of prostatic origin, was the primary differential diagnosis. Cytologic features did not, however, completely rule out the possibility of a poorly differentiated sarcoma. The gross necropsy appearance was more suggestive of metastatic hemangiosarcoma (HSA) based on the dark red color of the disseminated lesions, although a primary site was not identified. Immunocytochemistry was not done and could have provided valuable information about the phenotype of the neoplastic cells.
Microscopic Findings
Routine HE stained sections of masses from the lung, spleen, prostate, kidney, liver, colon, and testis, and sections taken from the brain, and right eye were examined by light microscopy. The prostate had solid clusters of anaplastic cells infiltrating the prostatic interstitium and vasculature and replacing prostatic parenchyma. Tumor cells were large, polygonal to round to ovoid, had large, vesicular nuclei, with moderate anisokaryosis and multiple nucleoli (Fig. 3). Mitotic figures were common and often bizarre (Fig. 3). The surrounding prostatic tissue had prostatic epithelial hyperplasia and mild prostatitis. Solid aggregates of large anaplastic cells penetrating vascular walls were present within multiple pulmonary arterioles, whereas other masses effaced alveoli. Mild hemorrhage was also present. Anaplastic cells formed solid, multilobular masses in the kidney, liver, colonic wall, and testis that were supported by delicate fibrous but highly vascular stroma and associated with massive hemorrhage in the liver, colon, and testis, and large areas of necrosis in the kidney. The right eye contained appreciable amounts of degenerate erythrocytes and fibrin in both chambers. The iris was focally expanded by a multilobular, solid mass composed of anaplastic cells morphologically identical to those seen in other organs that was associated with severe hemorrhage. The lesions in the brain consisted of solid nests of anaplastic neoplastic cells that occluded arterioles and were associated with extensive perivascular hemorrhage. The nodules in the spleen presented, on histologic examination, as nodular lymphoid hyperplasia.
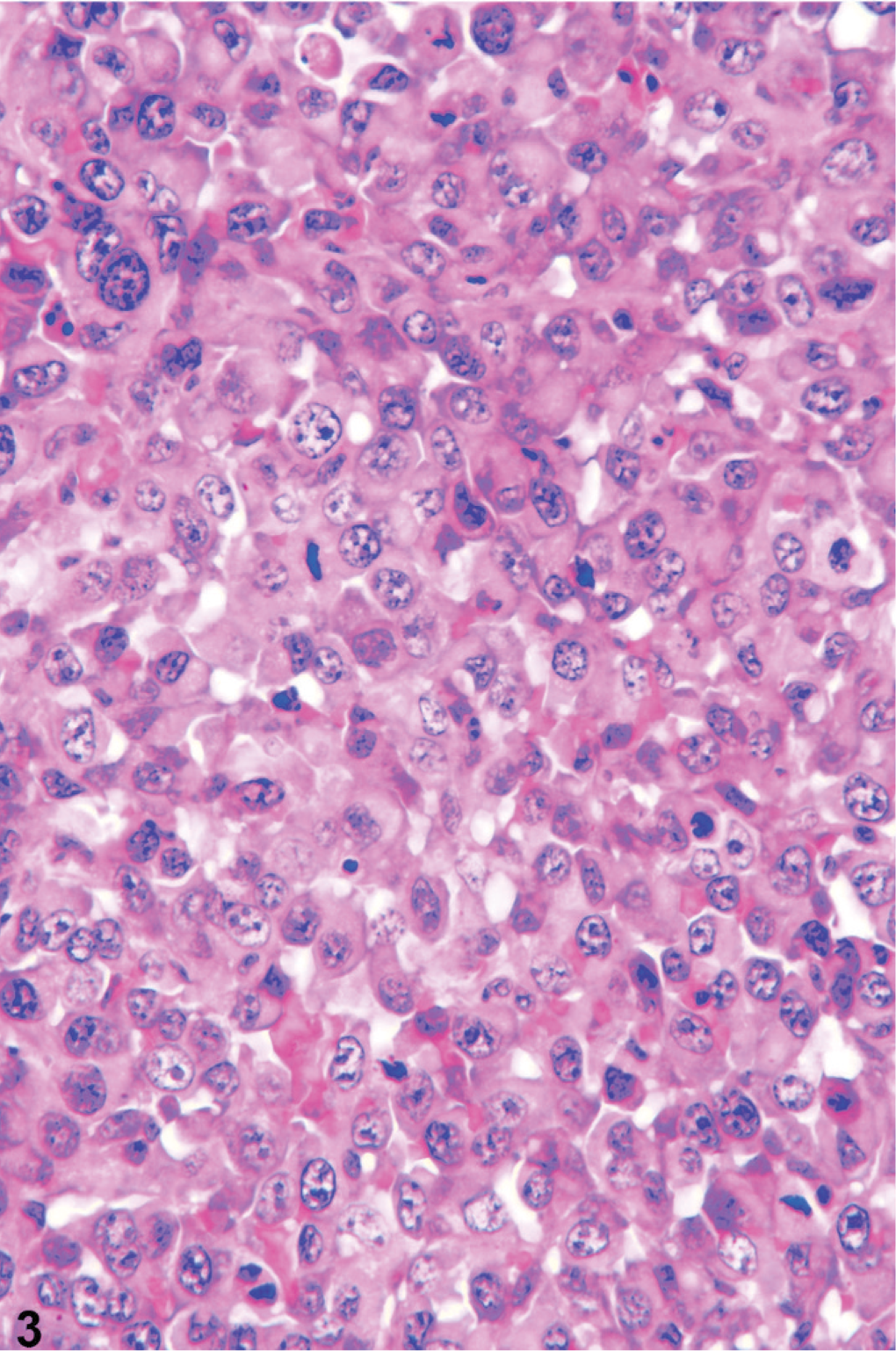
Prostate; dog. Epithelioid tumor cells are arranged in solid lobules, are large, and have pleomorphic, vesicular nuclei with marked anisokaryosis and frequent, often bizarre, mitotic figures. HE.
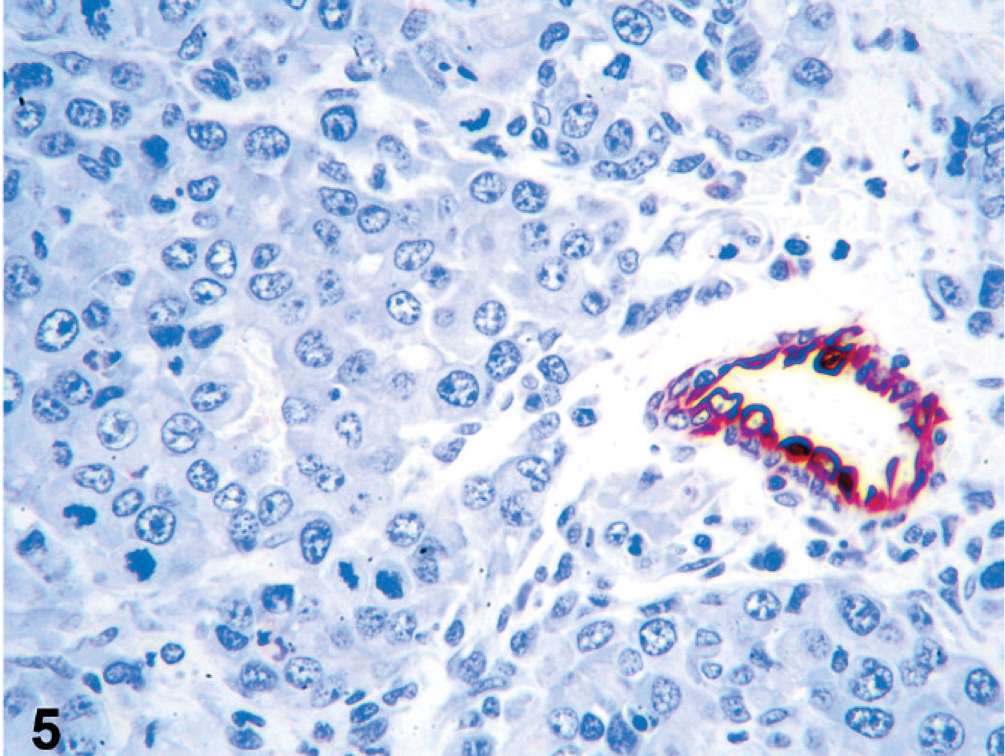
The histopathologic presentation as anaplastic epithelioid neoplasm supported the clinical and clinicopathologic diagnoses but was not consistent with the gross presentation of the neoplasm. Therefore, further characterization of the neoplastic cells by immunohistochemistry was pursued. Immunostaining was performed on an autostainer (Dako Autostainer Universal Staining System) with Nova red chromogen (Vector Laboratories, Burlingame, CA) and Mayer's hematoxylin counterstain (Sigma, St. Louis, MO). Microscopic sections were high temperature antigen retrieved with BD Retrieval A solution (Dako, Carpinteria, CA). Rabbit anti-human von Willebrand factor (vWF aka factor VIII-related antigen; Dako, diluted 1 : 400) and wide spectrum cytokeratin (WSS; Dako, diluted 1 : 500) antiserum and monoclonal mouse anti-human cytokeratin antibody (AE1/AE3; Dako, diluted 1 : 200) were used. Normal canine skin served as a positive control, and serial sections of neoplastic tissue were incubated with nonimmune serum to serve as negative controls. Neoplastic cells in all lesions from all examined locations had faint-to-strong cytoplasmic staining for vWF (aka factor VIII-related antigen, Fig. 4). Neoplastic cells in serial sections of the same tissues did not express cytokeratin, whereas adjacent resident epithelium was strongly positive (Fig. 5).
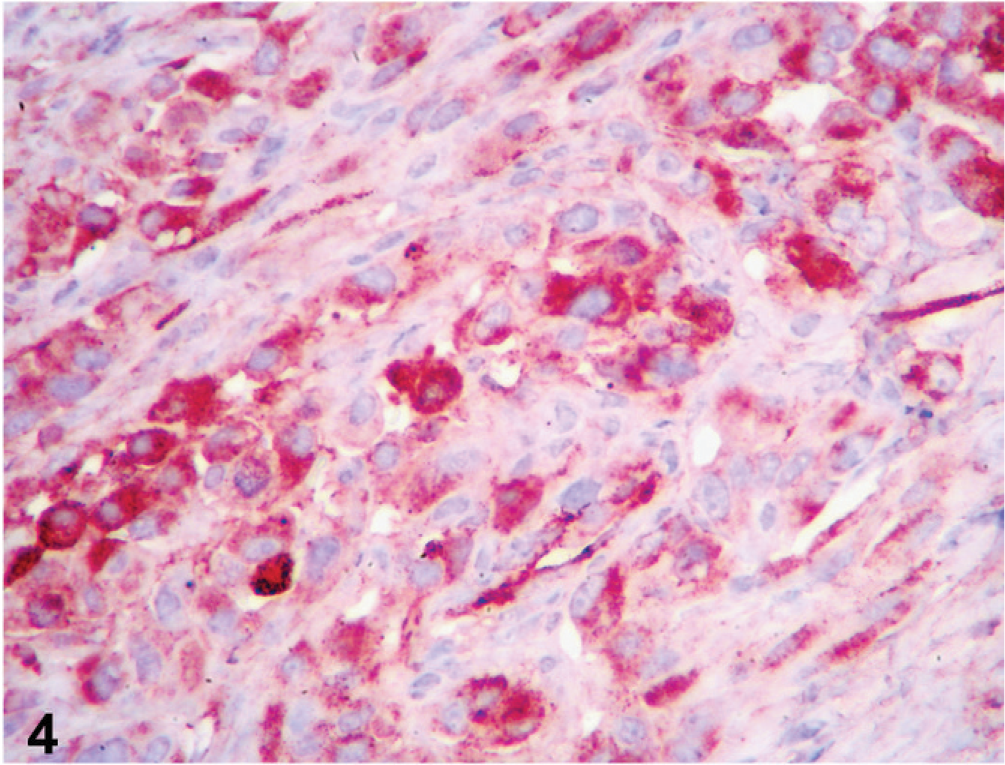
Prostate; dog. Epithelioid tumor cells show faint-to-strong cytoplasmic staining for vWF (aka factor VIII-related antigen). Labeled for vWF by immunohistochemistry.
Prostate; dog. Cytokeratin expression was not observed in epithelioid tumor cells, although the resident ductular epithelium is intensely stained. Labeled for AE1/AE3 by immunohistochemistry.
Diagnosis: Metastatic epithelioid hemangiosarcoma.
Discussion
Canine HSA is a malignant neoplasm of vascular endothelial origin that represents about 5% of all noncutaneous primary malignant tumors and 12% to 21% of all mesenchymal neoplasms in this species. 8, 9 HSA presents in 2 topographic distribution patterns: visceral and cutaneous. For visceral HSA, the spleen is the most common primary site, but it can occur as a primary tumor in many other organs. Typically, visceral HSAs are very aggressive, with rapid and widespread metastasis through the hematogenous route. 6, 8, 9 On histologic examination, visceral HSAs comprise vascular spaces lined by elongated, plump, anaplastic endothelial cells. Identification of vascular spaces in tumors with highly cellular stroma differentiates HSA from other sarcomas. 6 Confirmation of endothelial differentiation historically used staining of histologic specimens with various types of lectins. 1 The current gold standard used in veterinary anatomic pathology for assigning endothelial histogenesis to cells is positive expression of vWF (aka factor VIII-related antigen) and/or positive expression of CD31 (aka PECAM-1, platelet endothelial cell adhesion molecule) assessed through immunohistologic (IHC) techniques. 3, 10 Although we did not examine CD31 expression in the cells of this report, the ubiquitous vWF staining was highly supportive of an endothelial cell origin for the malignancy in this dog.
A histologic variant of HSA, epithelioid (histiocytoid) HSA, is rarely reported in the veterinary literature and may pose a diagnostic challenge on histopathologic and cytologic examination because of epithelial-like morphology of the cells that comprise this tumor. 4, 11 This variant was reported in the dog, horse, and cow. 2, 5, 11 Epithelioid vascular tumors in humans are positive for at least one of the commonly used vascular markers (i.e., vWF, CD31, CD34). 7 A percentage of these tumors in humans is positive for cytokeratin on immunohistochemistry and has been shown to have ultrastructural features of epithelial cells. 7 A recent immunohistologic study showed that epithelioid vascular tumors in domestic animals stain for at least one of two used vascular markers (vWF, CD31), whereas none stained for cytokeratin (AE1/AE3). 2 In most of the reported cases in the dog, the primary lesions were located in the dermis, subcutis, skeletal muscle, spleen, heart, or lung, and the tumors were locally aggressive, with metastasis chiefly confined to the regional lymph nodes without systemic dissemination. 2, 5, 11 In the dog of this report, there was no evidence of cutaneous involvement or history of the surgical removal of skin masses, and the epithelioid HSA was present and widely disseminated in numerous tissues, including some previously unreported locations (e.g., prostate, testis, kidney, liver, and brain). The clinical presentation (i.e., irregular prostatomegaly) and the widespread metastases of a malignancy with epithelial features mimicked prostatic carcinoma. The case reported here illustrates the potential for epithelioid HSA to present with a confusing constellation of clinical and pathologic findings and underscores the need to pursue IHC studies of anaplastic cells to confirm the histogenesis of epithelial HSA.
Footnotes
Acknowledgements
We thank Kay Fischer for excellent technical assistance.
