Abstract
A 30-year-old female Japanese macaque showed marked splenomegaly. The enlarged spleen consisted of neoplastic proliferation of anastomosing vascular channels resembling morphologic structures of red pulp sinuses; occasionally, papillary fronds were seen in dilated channels. Immunohistochemically, the lining cells reacted to both endothelial cell (von Willebrand factor) and macrophage (macrophage scavenger receptor class A) markers, indicating features of littoral cells of the spleen. Based on the pathologic characteristics, particularly the presence of neoplastic cells with macrophage/histiocyte-like attributes, this tumor was regarded as littoral cell angioma; this is a rare benign splenic vascular tumor.
Background
Besides degenerative and inflammatory lesions, primary vascular lesions usually found in the spleen include non-neoplastic hematoma and peliosis and neoplastic lesions, such as hemangioma and hemangiosarcoma. 1, 4, 19 In addition, in humans, littoral cell angioma and hemangiomatosis (angiomatosis) have been recorded as vasoformative disorders of the spleen. 4, 6, 12, 13, 16, 21 The littoral cell angioma, a benign vasoformative tumor, originates from the sinus endothelial cells, called littoral cells, of the splenic red pulp. 6 The littoral cells are considered to be unique lining cells possessing both vascular endothelial and macrophage-like properties. 3, 5, 6, 11, 21 Splenic hemangiomatosis consists of vascular endothelial cells forming cavernous and capillary vessels but lacks a macrophage-like nature of the neoplastic cells. 4, 16
Here, we describe a macaque case of a vasoformative lesion of the spleen, the pathologic characteristics of which bore a resemblance to those of littoral cell angiomas of the spleen in humans. 3, 5, 6, 21 In veterinary literature, there have been no reports of littoral cell angiomas in domestic and experimental animals. 7, 19 Splenic vasoformative disorders in humans are often accompanied by a disturbance of blood coagulation. 5, 21 Therefore, thrombotic and antithrombotic factors, as well as neovascularization factor (vascular endothelial growth factor-A [VEGF-A]), were investigated at the mRNA levels by the real-time polymerase chain reaction.
History
A 30-year-old female Japanese Macaque (Macaca fuscata) (7.2 kg body weight), named Wakasa 309, developed weight loss and anorexia and was euthanatized with an excessive injection of pentobarbital because of a poor prognosis; this macaque had been housed in an outdoor enclosure (Primate Research Institute, Kyoto University, Japan). She was fed a regular monkey chow supplemented with fruits and sweet potatoes and was supplied water ad libitum. Euthanasia and sample collections were approved by the Institutional Animal Care and Use Committee of the Primate Research Institute of Kyoto University, Japan. Age-matched Japanese macaques (n = 3; 23–31 years old) without splenic lesions served as controls.
Pathologic Findings
At necropsy, an enlarged spleen, measuring 6 cm in diameter, was found; the cut surface was reddish in color, and homogeneous in appearance (Fig. 1). No masses were detected in the liver, lungs, kidneys, heart, ovaries, adrenal glands, stomach, intestines, and brain. Those major organs, as well as the enlarged spleen, were fixed in 10% neutral buffered formalin, subsequently dehydrated, and embedded in paraffin. Deparaffinized sections were stained with HE and by the Watanabe's silver impregnation for reticulin. Sections were also labeled by immunohistochemistry with the following primary antibodies: mouse monoclonal antivimentin (1 : 200, clone V9, Dako, Carpinteria, CA, USA), mouse monoclonal anti-α-smooth muscle actin (α-SMA) (1 : 100, clone 1A4, Dako), mouse monoclonal antiproliferating cell nuclear antigen (anti-PCNA) (1 : 100, clone PC10, Dako), mouse monoclonal antimacrophage scavenger receptor A (MSR-A) (1 : 100, clone SRA-E5, Transgenic Inc., Kumamoto, Japan), and rabbit polyclonal anti-von Willebrand factor (vWF) (predilution, Dako). Before incubation with each primary antibody, deparaffinized sections were pretreated as follows: 1) microwave for 5 minutes in 0.01 M citrate buffer (pH 6.0) for vimentin, PCNA, and MSR-A immunohistochemistry; 2) 0.1% trypsin in phosphate-buffered saline at 37°C for 20 minutes for α-SMA immunohistochemistry; and 3) 10 μg/ml proteinase K in tris-buffered saline (pH 7.5) at room temperature for 10 minutes for vWF immunohistochemistry. Sections were reacted with each primary antibody at 4°C overnight and then incubated with horseradish peroxidase-conjugated antimouse or antirabbit secondary antibody for 45 minutes (Histofine Simplestain MAX-PO, Nichirei, Tokyo, Japan). Signals were visualized with 3,3′-diaminobenzidine tetrahydrochloride (DAB substrate kit, Vector Laboratories, Burlingame, CA, USA). Nonimmunized mouse or rabbit immunoglobulin G, which was used instead of the primary monoclonal and polyclonal antibodies, respectively, served as negative controls.
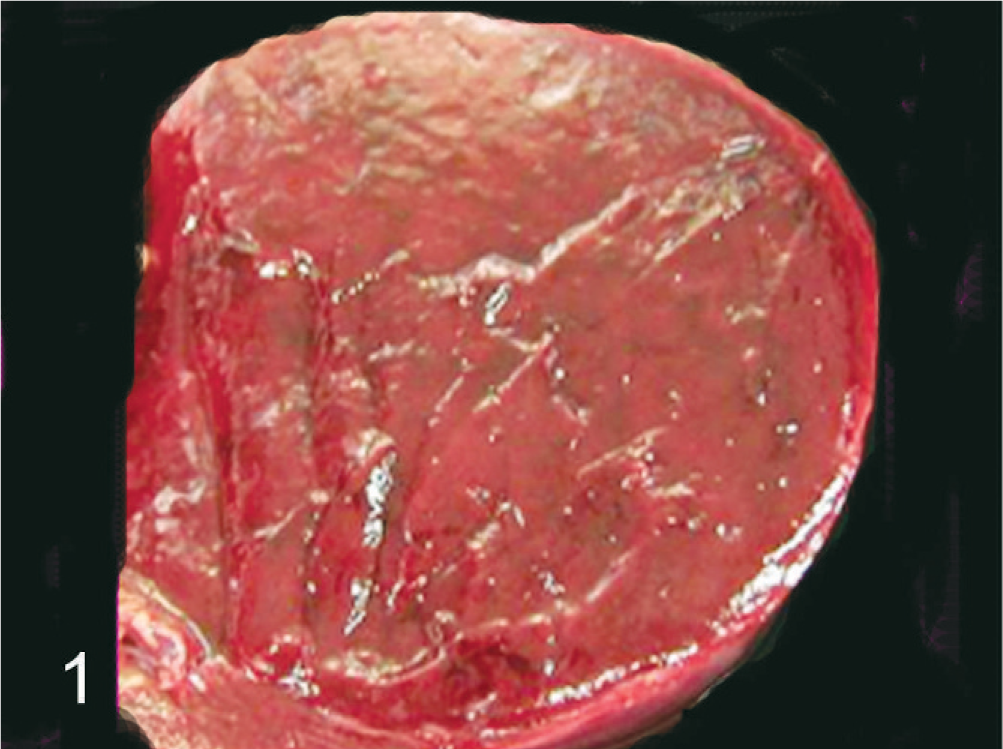
Spleen; monkey. Macroscopic view of the cut surface of enlarged spleen showing a homogeneous appearance.
Total RNA was extracted from fresh spleen using an RNeasy micro kit (Qiagen, Santa Clarita, CA, USA) according to the manufacturer's protocol in conjunction with on-column DNase treatment (RNase-Free DNase Set, Qiagen). The obtained RNA was then subjected to reverse transcription to yield cDNA using AMV RTasa XL (Takara Bio, Otsu, Japan) and oligo (dT)12-18 primer (Invitrogen, Carlsbad, CA, USA). Real-time polymerase chain reaction was performed using specific primers for tissue factor, plasminogen activator inhibitor-I, tissue factor pathway inhibitor, thrombomoduline, VEGF-A, and glyceraldehyde-3-phosphate dehydrogenase (a reference gene) with the ABI Prism 7700 Sequence Detector System (Applied Biosystems, Foster City, CA, USA) under the conditions described in a previous paper. 8 The quantitative assessment of mRNA was done by dividing by the expression level of the internal glyceraldehyde-3-phosphate dehydrogenase and expressed as a so-called relative expression to glyceraldehyde-3-phosphate dehydrogenase. 8
The capsule of the enlarged spleen was slightly roughened, but neither nodular lesion nor hematoma-like cyst filled with clots was detected (Fig. 1). Microscopic examination showed angiomatous lesions involving the entire spleen (Fig. 2), but residual white pulps were present. The lesions consisted of anastomosing, variably sized vascular channels filled with various amounts of erythrocytes (Fig. 2). The channels were lined by single- or multilayered, irregularly arranged neoplastic cells with a hyperchormatic nucleus; the lining cells showed slight cellular variability, such as elongated and cuboidal shapes, often protruding in the lumen (Figs. 2,3). Papillary fronds were occasionally seen in cystic dilated vascular lumina throughout tumor tissues (Fig. 2, inset). Some of the lining cells contained hemosiderin granules in the cytoplasm (Fig. 3, inset). These vasoformative structures occasionally appeared to be capillary vessels (Fig. 3). Although red pulp was present in some areas, their distinctive ring and mesh-like structures were lost; therefore, it was difficult to distinguish between red pulp sinuses and vasoformative structures (Fig. 2). Slightly thickened reticular fibers were demonstrable around each channel by the Watanabe's silver impregnation stain. Atypical cells or mitoses were rarely seen. Neither fibrinous nor organized thrombus was observed. Necrosis was not seen in tumor tissues.
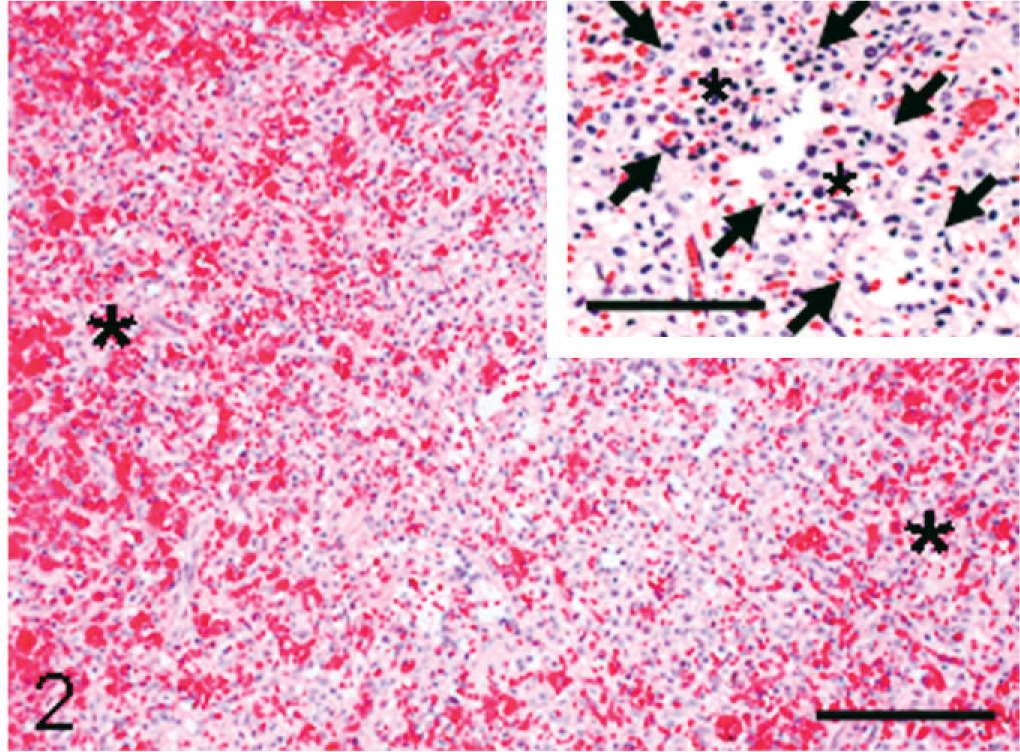
Spleen; monkey. Littoral cell angioma consisting of anastomosing channels filled with various amount of erythrocytes. Areas of red pulps (asterisks) are indistinguishable from the angiomatous lesion. HE. Bar = 120 μm. Inset: papillary fronds (asterisks) are seen in a dilated channel (arrows indicate verge of the dilated channel). HE. Bar = 60 μm.
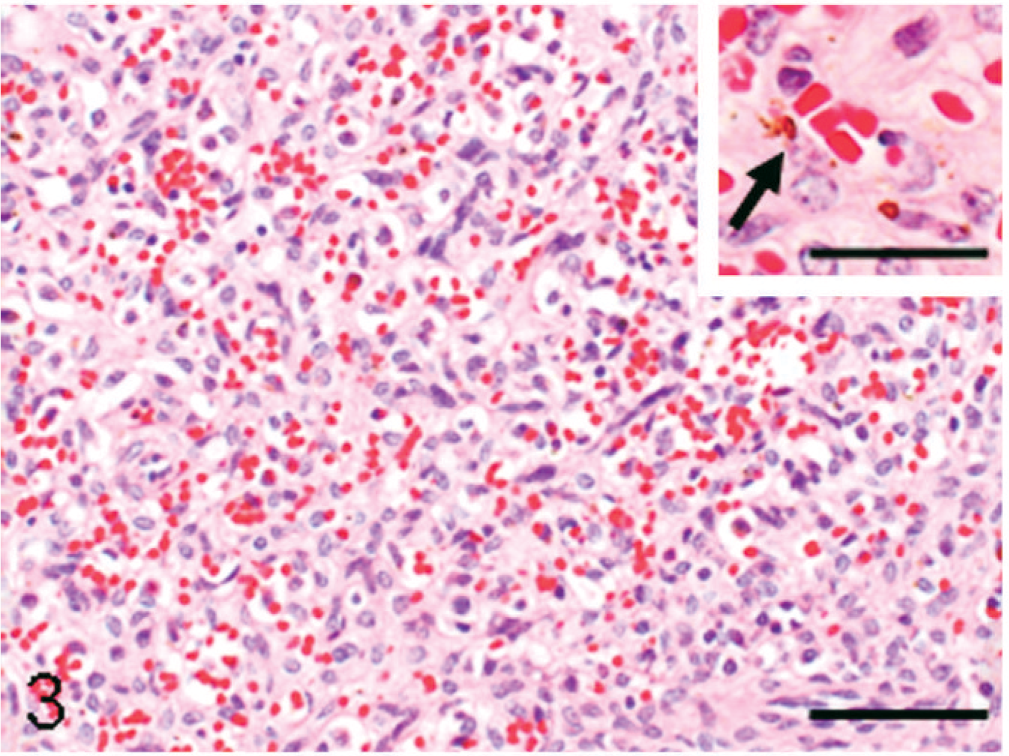
Spleen; monkey. The angiomatous lesion consisting of anastomosing channels lined by irregularly arranged elongated or cuboidal cells with a hyperchromatic nucleus; the vasoformative structures appear to be vague capillary vessels. HE. Bar = 50 μm. Inset; a neoplastic cell containing hemosiderin granules in the cytoplasm (arrow). HE. Bar = 15 μm
Immunohistochemically, the lining cells in the angiomatous lesions showed a positive reaction to vWF (Fig. 4), and occasional lining cells were reactive to vimentin. The lining cells also reacted to MSR-A; vascular lumina were rimmed clearly by MSR-A-positive cells (Fig. 5). Many cells consisting of papillary fronds reacted to MSR-A (Fig. 5, inset). Cells reacting to α-SMA were present around channels in varying degrees (Fig. 6). The vasoformative cells frequently gave a positive reaction to PCNA (Fig. 7).
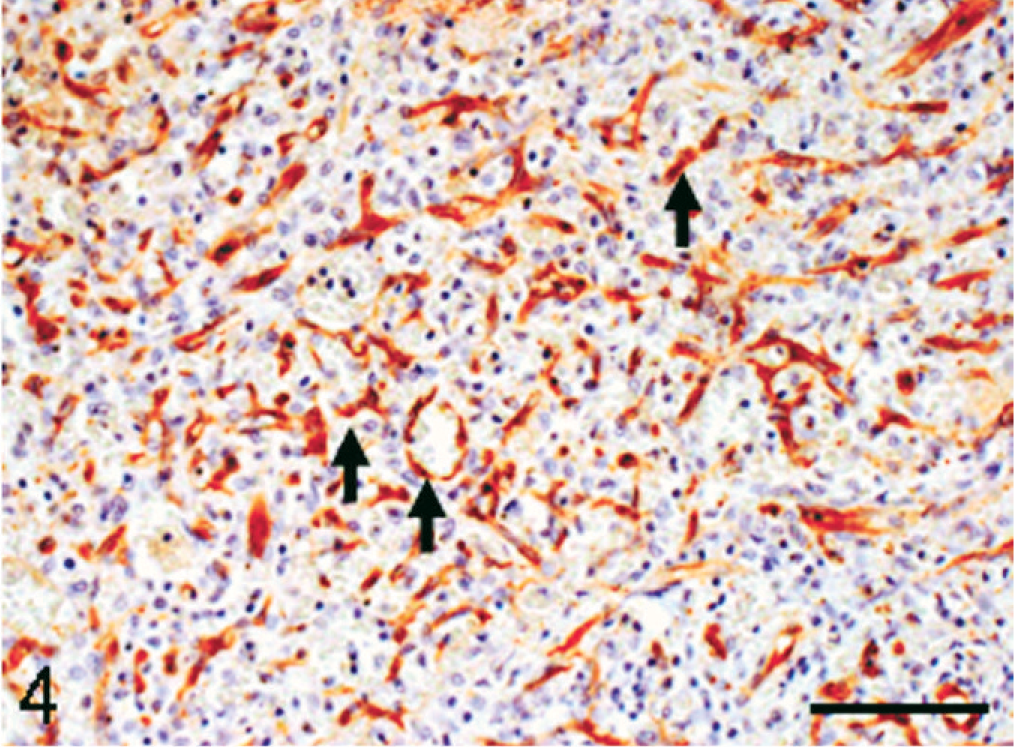
Spleen; monkey. Neoplastic cells in the angiomatous lesion react to von Willebrand factor (arrows). Immunohistochemistry, counterstained with hematoxylin. Bar = 60 μm.
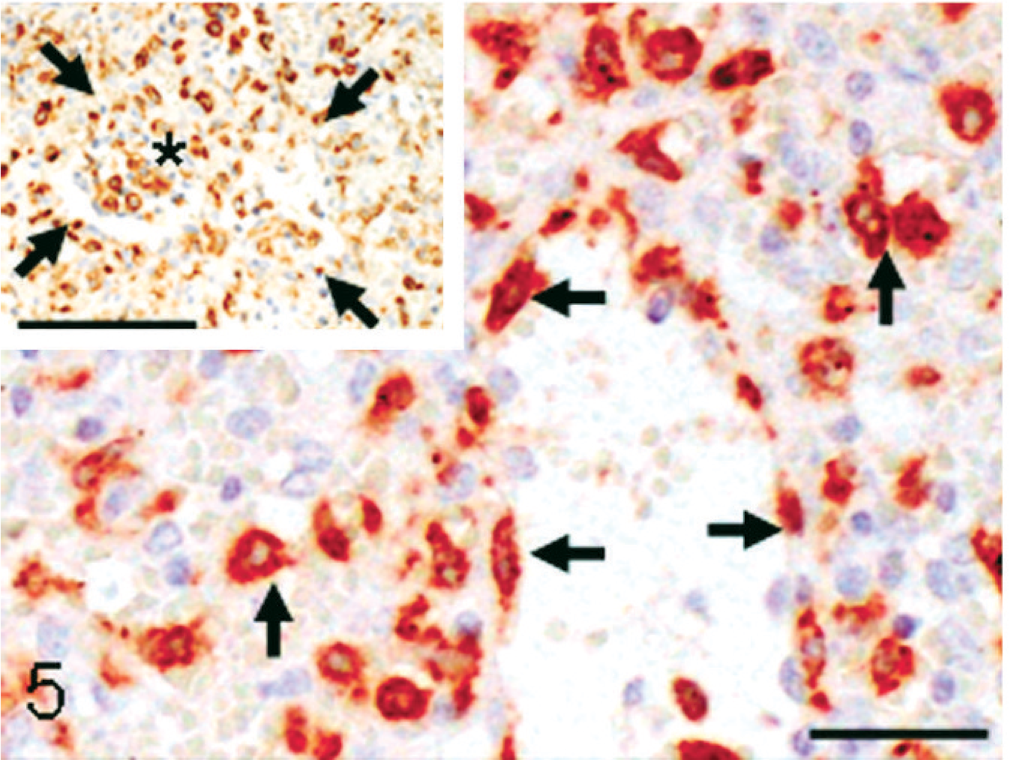
Spleen; monkey. Neoplastic cells rimming vascular channels in the angiomatous lesions give a positive reaction with an antibody against macrophage scavenger receptor class A (MSR-A) (arrows). Bar = 20 μm. Inset: neoplastic cells in the papillary frond also react to MSR-A (asterisk) (arrows indicate verge of a dilated channel). Bar = 60 μm. Immunohistochemistry, counterstained with hematoxylin.
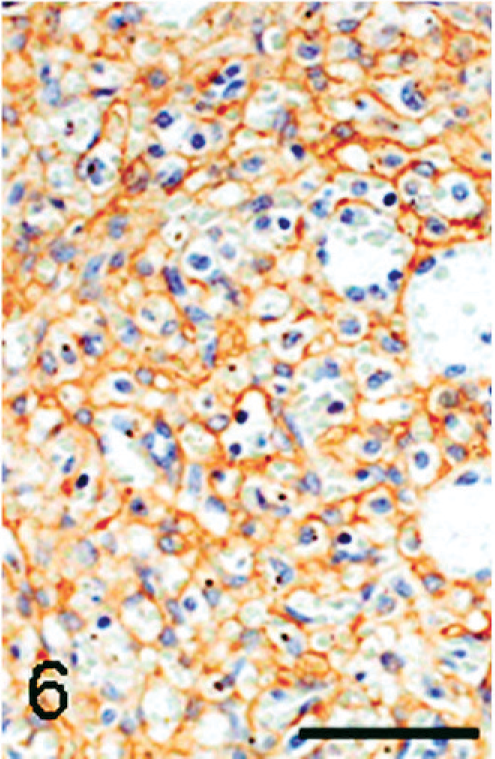
Spleen; monkey. Cells around vascular channels in the angiomatous lesion react with an antibody against α-smooth muscle actin, apparently indicating the presence of vascular smooth muscle. Immunohistochemistry, counterstained with hematoxylin. Bar = 50 μm.
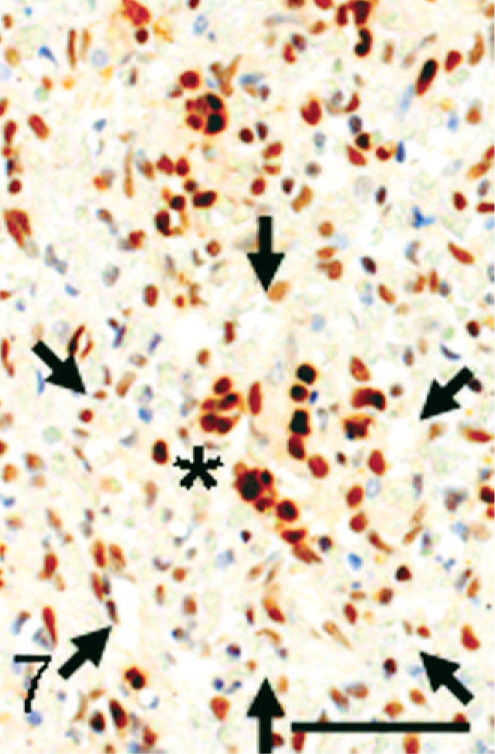
Spleen; monkey. Vasoformative cells in the angiomatous lesion give a positive reaction to proliferating cell nuclear antigen as a marker of proliferating index. Arrows indicate the verge of a dilated channel including a papillary frond (asterisk). Immunohistochemistry, counterstained with hematoxylin. Bar = 50 μm.
In other visceral organs examined, no neoplastic or vasoformative lesion was seen. Anthracosis in the lung and multifocal fibrosis and lipofuscin deposition in the myocardium were observed.
In thrombotic factors, the gene expression of tissue factor was upregulated, but that of plasminogen activator inhibitor-1 was suppressed; in antithrombotic factors, mRNA level of tissue factor pathway inhibitor was increased, but that of thrombomoduline did not change. Although these gene expressions in the affected spleen were upregulated, blood levels of tissue factor and tissue factor pathway inhibitor proteins were normal (data not shown). These findings indicated that blood coagulation state was normal in the monkey. Additionally, blood levels of thromobotic markers, thrombin-antithrombin III complex and D-dimer were normal (data not shown). The gene expression of VEGF-A was 2.6-fold upregulated, suggestive of an enhanced endothelial cell proliferation.
Diagnostic Criteria
Littoral cell angioma was introduced in 1991 as a new entity of primary benign vascular tumor in the spleen of humans. 6 The neoplasm generates from littoral cells of the red pulp sinuses; the cells have features that lie intermediately between vascular endothelial cells and macrophages/histiocytes, and the cells display higher lysosomal activity than vascular endothelial cells. 4, 21 The activity may be confirmed by hemosiderin deposition or hematophagocytosis. 21 Pathologic diagnosis is based on neoplastic proliferation of anastomosing vascular channels rimmed by cells reacting immunohistochemically to both endothelial cell and macrophage markers. 3, 5, 6, 21 The present macaque splenic lesion was composed of diffuse proliferation of vague vascular channels, whose structures closely resembled splenic sinuses. Therefore, there seemed to be gradual transition from normal-appearing red-pulp sinuses to vasformative lesions. Usually, lesions of human littoral cell angiomas range in size from small and isolated to large nodules that replace the entire splenic tissue. 6, 14, 21 The present case exhibited a homogenous growth without forming nodular lesions. In agreement with immunohistochemical findings in human littoral cell angiomas, the lining cells gave positive reactions to vascular endothelial cell (vWF) and macrophage (MSR-A) markers. The antibody to vWF has been widely used to detect endothelial cells of blood vessels not only in normal tissues but also in neoplastic tissues. 1, 4, 6, 15 The antibody to MSR-A was generated by immunizing MSR knockout mice with recombinant human type I MSR; the antibody is useful in identifying macrophages/histiocytes in human and monkey tissues. 15 Additionally, the lining cells reacted with the PCNA antibody, a marker of proliferating cells, but malignant features (e.g., cytological atypia, frequent mitotic figures) were rarely seen. Hemosiderin deposition, indicative of phagocytic activity, was seen in occasional neoplastic cells. Papillary fronds/projections in dilated channels, seen in the present tumor, are characteristic of human littoral cell angiomas. 3, 5, 21 Taking morphologic and immunohistochemical findings together, the present case was regarded as a littoral cell angioma arising in the spleen.
Differential Diagnosis
The differential diagnosis of splenic littoral cell angiomas must take into account non-neoplastic lesions (hematoma and peliosis) or other blood vessel–derived tumors, such as hemangioma, hemangiomatosis, and hemangiosarcoma. 1, 4, 6, 13, 21 Hematoma and peliosis consist of blood-filled cystic spaces lined by normal vascular endothelial cells. Therefore, the present splenic tumor was distinctly different from those non-neoplastic lesions in that neoplatic cells in the present case reacted to PCNA and mRNA of VEGF-A was increased. Hemangiosarcomas arising in the spleen develop marked nodular lesions protruding from the capsule. 4, 19 The malignant tumors are composed of highly variable neoplastic cells showing fusiform, polygonal, and oval configurations, indicating cytological atypia, and there are often areas consisting of unrecognizable vascular forms (hemangioendotheliomatous growth pattern). 4, 7, 19 Mitotic figures are frequent, and metastatic lesions may be found in the lungs and heart at advanced stages. 7, 19 No malignant features were seen in the present case. Hemangiomas originate from splenic blood vessels, probably veins, and are usually composed of capillary or cavernous vessels lined by a single layer of uniform vascular endothelial cells without cellular atypia. 1, 7, 13, 19 Hemangiomas often form an isolated nodular lesion within the spleen. 1, 4, 16 Human hemangiomatosis occurs as a manifestation of systemic angiomatosis involving the liver and spleen or, less commonly, confined to the spleen. 4, 12, 13, 16 The isolated splenic hemangiomatosis is characterized by diffuse proliferation of cavernous or cystic blood vessels rimmed by benign-appearing endothelial cells. 4, 13, 16 Splenic hemangiomatosis may be included in hemangiomas as a variant, in which the whole spleen is occupied by neoplastic vascular channels. 1 The histopathologic characteristics of splenic hemangiomas and hemangiomatosis seem to be similar to those in the present macaque case, and their endothelial cells may have the expression of vascular endothelial markers in common. 1, 4, 12, 13 However, splenic hemangiomas and hemangiomatosis were eliminated by the expression of macrophage/histiocytic markers. 4, 6, 12, 21 The papillary growth of neoplastic cells in dilated channels has not been described in hemangiomas and hemangiomatosis. 4, 16 Additionally, evidence for normal blood coagulation in the present case was different from that of hemangiomas and hemangiosarcomas, in which disseminated intravascular coagulation and its related disorders frequently occur. 5, 21 For further differential diagnosis between littoral cell angiomas and hemangiomatosis, it was reported that immunoreactivities for CD34 and CD21 might be useful, in that neoplastic cells of the former were negative for CD34 and positive for CD21. 1 However, immunoreactivities of littoral cell angiomas for these antibodies are not always consistent, 14, 21 and we notice that CD34 and CD21 antibodies are also makers for hematopoietic stem cells and B-cell maturation, respectively. 2, 9 The presence of α-SMA–positive cells around vascular channels was reported to be a characteristic of splenic hemangiomatosis. 1 However, the observation is difficult to interpret, because the positive cells might be either myofibroblasts corresponding to fibrosis within tumor tissues or collapsed endothelial cells reacting to α-SMA. 4 Because red pulp sinuses continue from simple arterial capillaries, vascular smooth muscle cells of the arterial capillaries might react to α-SMA, suggesting the close relationship of vascular endothelial cells with littoral cells. Although these immunohistochemical characteristics should be pursued further, the presence of MSR-A–positive histiocytic/macrophagic cells in variously sized vascular channels and papillary fronds supports a diagnosis of littoral cell angioma rather than splenic hemangiomatosis. The histiocytic/macrophage-like characteristics of the littoral cells in the red pulp sinuses may be enhanced by appropriate stimuli. 21 It would be interesting to investigate the mechanism of dual differentiation potential of littoral cell angioma cells.
In conclusion, the splenomegaly found in a geriatric female Japanese Macaque consisted of neoplastic proliferation of anastomosing vascular channels whose lining cells exhibited both vascular endothelial and histiocytic/macrophage-like features. The pathologic characteristics were in agreement with those of human littoral cell angiomas of the spleen. In human cases, the littoral cell angioma, a rare splenic benign vascular tumor, occurs at any age with no sex-based predilection. 6, 21 The Japanese macaque is a macaque native to Japan. The animals have been widely studied with regard to the biologic behavior and physiology. 10 It is important to establish the baseline data concerning the incidences of spontaneous tumors. In Japanese macaques, odontoameloblastoma, basal cell tumor, and peritoneal malignant mesothelioma have been recorded. 17, 18, 20 This case was the first report on littoral cell angioma of the spleen in a macaque.
Footnotes
Acknowledgements
This study was supported in part by the Cooperation Research Program (2006–2008) of Primate Research Institute, Kyoto University, Japan.
